Spyridon Gennatas and Sanjay Popat
Case history
A 67-year-old South Asian woman who had never smoked presented with a 3-month history of back pain, dry cough, and increasing malaise. Her ECOG performance status was 2. A CT scan demonstrated a right upper lobe primary lung tumour, bilateral mediastinal and ipsilateral supraclavicular nodal involvement, bilateral lung metastases involving all lung lobes, and left adrenal and widespread bony metastases. Spinal MRI confirmed multiple vertebral metastases with threatened spinal cord compression at multiple levels. She received 20Gy in five fractions of radiotherapy to C7–T3 and T11–L1. Ultrasound-guided biopsy of an involved right supraclavicular fossa node confirmed adenocarcinoma positive for cytokeratin 7 (CK7), TTF-1, Napsin A, and carcinoembryonic antigen (CEA) and negative for cytokeratin 20 (CK20), caudal type homeobox 2 (CDX2), and GCDFP-15 by immunohistochemistry. These features were consistent with lung adenocarcinoma.
Questions
1. What is the stage of the patient’s disease?
2. What other investigations need to be undertaken prior to deciding on a treatment plan?
3. What is the significance of a history of never smoking in patients with lung cancer?
Answers
1. What is the stage of the patient’s disease?
T4 N3 M1b:
|
T4 |
– |
separate tumour nodule(s) within an ipsilateral lobe |
|
N3 |
– |
contralateral mediastinal and ipsilateral supraclavicular lymph nodes |
|
M1b |
– |
distant metastases (bone and adrenal). |
2. What other investigations need to be undertaken prior to deciding on a treatment plan?
The tumour needs to be tested for EGFR mutations.
3. What is the significance of a history of never smoking in patients with lung cancer?
Lung cancer is generally associated with tobacco exposure. However, occurrence in patients who have never smoked is well recognized. In thoracic oncology, a ‘never-smoker’ has arbitrarily been defined as smoking 100 or fewer cigarettes over a lifetime. The WHO estimates that never-smokers account for 25% of new cases of lung cancer worldwide. Whether the incidence of never-smokers with lung cancer is increasing or whether a decrease in smoking-related lung cancers has been observed remains unclear, since tobacco exposure is poorly documented in most registries.
There is no conclusive evidence that mean age at presentation differs between never-smokers and smokers with lung cancer, although most studies indicate peak incidence is perhaps at a younger age in never-smokers. Lung cancer in never-smokers is wholly associated with adenocarcinoma (or variants thereof), whilst squamous carcinomas and small cell lung cancer are typically associated with tobacco. Indeed, pathological review is recommended in the case of such tumours diagnosed without tobacco exposure. A number of risk factors are recognized that are important not only in never-smokers but in smokers too. These include hereditary risks, exposure to environmental pollution—including second-hand tobacco smoke and cooking and heating fumes—and ionizing radiation. An increased incidence in women is observed, and this might reflect hormonal factors, with ER receptors expressed more commonly in lung cancer tissue than normal lung tissue and potentially correlating with poorer outcomes. Other risk factors include infections, particularly viral, low socio-economic status, immunosuppression, and diabetes mellitus.
Recently, a number of somatic molecular aberrations that are principally observed in never-smokers have been shown in a type of lung cancer that appears to be a distinct clinical entity—it is composed of different subtypes of carcinoma driven by different molecular aberrations. These will be discussed in more detail in the following sections.
EGFR mutation analysis was performed and a mutation was detected. This was an EGFR exon 19 deletion.
Questions
4. How common are EGFR mutations in lung cancer? Are there any particular subgroups more likely to harbour such mutations?
5. What EGFR mutations have been identified and which ones are associated with response to treatment with tyrosine kinase inhibitors (TKIs)?
6. Which TKIs are licensed for the first-line treatment of EGFR-mutant adenocarcinomas and what evidence supports their use?
7. What other molecular markers have been identified that could guide treatment decisions in lung cancer and what is their relationship to smoking status?
8. Are there any drugs that are currently licensed for any of these indications?
Answers
4. How common are EGFR mutations in lung cancer? Are there any particular subgroups more likely to harbour such mutations?
NSCLC can be divided into subgroups based solely on genetically discrete subsets according to the activating mutations they harbour. Somatic EGFR mutations were first identified in NSCLC in 2004 and were associated with a dramatic response to gefitinib. Since then considerable research has defined the relationship between genotype and clinical demographics. Mutations are more prevalent in East Asians, women, and never-smokers. They are predominantly observed in adenocarcinomas that tend to be TTF-1 positive. The exact prevalence of mutations is difficult to define since studies have used different denominators. However, prevalence varies by ethnicity, ranging from 5% in American current smokers, to 28% in American never-smokers, and as high as 68% in East Asian never-smokers. Prevalence seems to be inversely proportional to tobacco pack-years exposure.
5. What EGFR mutations have been identified and which ones are associated with response to treatment with tyrosine kinase inhibitors (TKIs)?
Activating EGFR mutations are restricted to exons 18–21 which encode the tyrosine kinase domain and result in constitutional activation of the kinase. Exon 19 mutations and the exon 21 L858R missense mutation account for about 90% of mutations. The remaining 10% are therefore rare, and are a mixture of missense, insertional, and deletional mutations.
Patients harbouring EGFR exon 19 deletions and L858R point mutations have impressively good outcomes from EGFR TKI-directed therapy. Exon 20 insertions are uncommon but are well recognized at presentation, and such tumours are typically resistant to EGFR TKI therapy.
6. Which TKIs are licensed for the first-line treatment of EGFR-mutant adenocarcinomas and what evidence supports their use?
In 2005 two trials first demonstrated the superiority of two EGFR TKIs (gefitinib and erlotinib) over placebo in certain patient subsets. The BR.21 phase III trial demonstrated that erlotinib prolonged survival in relapsed NSCLC following progression on first- or second-line chemotherapy compared with placebo (6.7 versus 4.7 months) (Shepherd et al. 2005). High response rates were observed in East Asian patients with the EGFR mutation who had never smoked. The ISEL phase III study investigating gefitinib in a similar population to the BR.21 trial did not show an overall survival advantage over placebo in the intention-to-treat population, but a benefit was identified in never-smokers and East Asian patients (Thatcher et al. 2005).
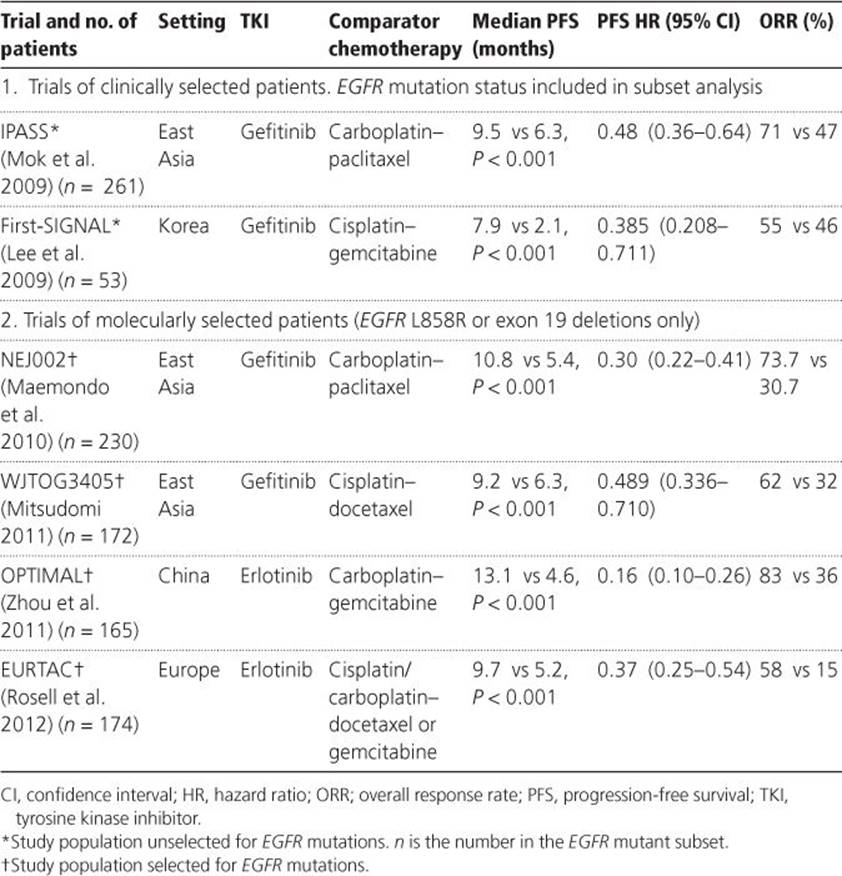
Since then, seven randomized phase III trials have since shown TKIs to be superior to platinum-doublet chemotherapy for progression-free survival (PFS), overall response rate (ORR), and quality of life in first-line patients with EGFR mutation. The first was the IPASS study, which compared gefitinib versus carboplatin and paclitaxel in never-smokers or ex-light smokers from East Asia with adenocarcinoma, a population enriched in patients with EGFR mutation. Despite this, only 59.7% of these adenocarcinomas harboured EGFR mutations, indicating that clinical demographics alone are inadequate in predicting somatic mutation and that molecular screening is required. In the subgroup with EGFR mutation a significantly increased PFS was seen with gefitinib [median 9.5 versus 6.3 months; hazard ratio (HR) 0.48 (95% confidence interval 0.36–0.64); P < 0.001]. The ORR favoured gefitinib (71.2 versus 47.3%), and underpinned significant improvements in quality of life domains in the gefitinib group. Similar outcomes were observed in the First-SIGNAL trial investigating a similar group of clinically selected Korean patients likely to have EGFR mutation. A benefit on overall survival (OS) was not observed in either trial due to post-progression crossover therapy.
Subsequently, four phase III trials have confirmed the superiority of gefitinib or erlotinib over platinum-doublet chemotherapy in first-line patients proven to harbour EGFR L858R or exon 19 deletions (Table 5.1), and as a result both are licensed for this indication. EGFR mutation testing should now therefore be performed on all patients eligible for TKI treatment and on mutation-positive patients treated with first-line EGFR TKI.
Table 5.1 Phase III Trials of gefitinib/erlotinib versus chemotherapy in first-line patients with EGFR mutant non-small-cell lung cancer
Acquired resistance to EGFR TKI has been shown to be mediated in about 40% of patients through a second EGFR mutation, T790M (exon 20). Such resistance can be overcome in vitro by irreversible EGFR inhibitors, which specifically inhibit T790M (e.g. afatinib or dacomitinib) and therefore have the potential to delay acquired resistance.
Afatinib (BIBW2992) is one such pan-ErbB-family (EGFR, HER2, ErbB3, and ErbB4) TKI, and has a proven advantage for PFS over placebo in relapsed EGFR-mutant NSCLC with acquired gefitinib/erlotinib resistance. Most recently, first-line afatinib has been shown to be superior to cisplatin–pemetrexed in patients with EGFR mutant adenocarcinoma [median PFS 13.61 versus 6.9 months for L858R/exon 19 deletion; HR 0.58 (0.43–0.78), P = 0.0004; objective response rate 56 versus 23%, P < 0.0001]. However, toxicities were frequent (afatinib toxicity, grade 3/4 diarrhoea in 14.4%).
7. What other molecular markers have been identified that could guide treatment decisions in lung cancer and what is their relationship to smoking status?
(a) Anaplastic lymphoma kinase (ALK).
In 2007 ALK-EML4 gene rearrangements were identified in NSCLC. Since then a number of rarer non-ALK fusion partners have been identified. ALK rearrangements can be identified by a number of methods including fluorescence in situ hybridization (FISH), reverse transcriptase polymerase chain reaction, and immunohistochemistry, since the fusion protein is not normally expressed. ALK rearrangements are primarily identified in adenocarcinomas and are uncommon (with a prevalence of 2.4–5.6%). They are more frequent in younger patients (median 50 years) and never-smokers or light smokers. Paik et al. (2012) demonstrated prevalence in 12% of never-smokers versus 2% of former/current smokers. Although some studies have suggested an intrinsically poorer prognosis for patients with ALK rearrangement several biases may have confounded these results, and further work in this area is ongoing. However, most studies have shown that ALK-positive adenocarcinomas do not respond to anti-EGFR TKIs.
(b) KRAS
In Western countries, KRAS mutations have been identified in about 25% of lung adenocarcinomas. They are associated with resistance to gefitinib or erlotinib, and have a poor prognosis. They are less prevalent in East Asian populations. Ninety-five per cent of KRAS mutations occur in codons 12–13 and tend to be observed in smokers. A study of 481 lung adenocarcinomas identified KRAS mutations in 15% of never-smokers and 47% of ex-/current smokers. No clinical demographic differences were observed compared with patients with wild-type KRAS. In never-smokers, KRAS mutations tend to be observed in mucinous invasive adenocarcinomas with a lepidic pattern (formerly called bronchioloalveolar carcinomas). KRAS and EGFR mutations tend to be mutually exclusive.
8. Are there any drugs that are currently licensed for any of these indications?
Crizotinib is an ATP-competitive inhibitor of ALK, MET, RON, and ROS1 kinases. It was being developed in phase I trials as a MET inhibitor when NSCLC ALK rearrangements were identified and development was subsequently restricted to ALK-rearranged NSCLC. It achieved accelerated Food and Drug Administration (FDA) approval for treatment of ALK-positive locally advanced or metastatic NSCLC after phase I and II trials demonstrated impressive response rates of 54–61% in ALK-positive NSCLC, disease control rates of 90%, and PFS of 10 months. Phase III trials comparing crizotinib with pemetrexed or docetaxel in the second-line setting, and with cisplatin–pemetrexed in the first-line setting will quantify the magnitude of benefit from crizotinib.
The patient was commenced on treatment with gefitinib at the standard daily dose of 250mg with the addition of zolendronic acid every 4 weeks. Her tumour was sent for further molecular analysis as part of an on-going study. No codon 12–13 KRAS mutations, exon 15 BRAF mutations, or ALK rearrangements were detected.
Treatment and follow-up
The patient was last reviewed 4 months into treatment with gefitinib. She attained an excellent clinical and radiological response. Her only current symptom is grade 1 anorexia. She is pain free and off opiates. Her cough has completely resolved and her ECOG performance status is 1. An up-to-date staging CT scan shows an ongoing partial remission across all disease sites.
Learning points
In NSCLC:
♦ testing for EGFR mutations is critical in therapeutic decision-making;
♦ smoking history is critical to determining likelihood of EGFR mutation present;
♦ patients whose tumours have known EGFR mutations should have first-line treatment with a TKI (gefitinib or erlotinib) over platinum-doublet chemotherapy;
♦ crizotinib is FDA approved for ALK-rearranged relapsed NSCLC; European Medicines Agency approval is awaited;
♦ KRAS mutations are well recognized in never-smokers and are associated with a poor prognosis and lack of response to gefitinib or erlotinib;
♦ KRAS and EGFR mutations are relatively mutually exclusive.
Further reading
Couraud S, Zalcman G, Milleron B, Morin F, Souquet P-J. Lung cancer in never smokers—a review. European Journal of Cancer 2012; 48: 1299–1311.
Lee JS, Park K, Kim S-W, et al. A randomized phase III study of gefitinib (IRESSA®) versus standard chemotherapy (gemcitabine plus cisplatin) as a first-line treatment for never-smokers with advanced or metastatic adenocarcinoma of the lung. Journal of Thoracic Oncology 2009; 4 (9 Suppl 1): 283.
Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non–small-cell lung cancer with mutated EGFR. New England Journal of Medicine 2010; 362: 2380–2388.
Mitsudomi T. Erlotinib, gefitinib, or chemotherapy for EGFR mutation-positive lung cancer? Lancet Oncology 2011; 12: 710–711.
Mok TS, Wu Y-L, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. New England Journal of Medicine 2009; 361: 947–957.
Paik PK, Johnson ML, D’Angelo SP, et al. Driver mutations determine survival in smokers and never-smokers with stage IIIB/IV lung adenocarcinomas. Cancer 2012; 118: 5840–5847.
Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncology 2012; 13: 239–246.
Rudin CM, Avila-Tang E, Harris CC, et al. Lung cancer in never smokers: molecular profiles and therapeutic implications. Clinical Cancer Research 2009; 15: 5646–5661.
Scagliotti G, Stahel RA, Rosell R, Thatcher N, Soria J-C. ALK translocation and crizotinib in non-small cell lung cancer: an evolving paradigm in oncology drug development. European Journal of Cancer 2012; 48: 961–973.
Shepherd FA, Rodrigues Pereira J, Ciuleanu T, et al. Erlotinib in previously treated non-small-cell lung cancer. New England Journal of Medicine 2005; 353: 123–132.
Thatcher N, Chang A, Parikh P, et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). The Lancet 2005; 366: 1527–1537.
Yang JC-H, Shih J-Y, Su W-C, et al. Afatinib for patients with lung adenocarcinoma and epidermal growth factor receptor mutations (LUX-Lung 2): a phase 2 trial. Lancet Oncology 2012; 13: 539–548.
Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncology 2011; 12: 735–742.