Bengt Källén1
(1)
Tornblad Institute, Lund University, Lund, Sweden
Confounding is obtained if a factor affects both exposure risk and outcome risk.

A confounder will affect the strength of the effect of the exposure on the outcome or may even completely explain it. The effect of the confounder should therefore be removed as well as possible in the analysis. We will come back to how this can be done.
How do we know which confounders are involved? We often do not know all possible confounders, or we cannot measure them. It is therefore always a possibility that a noticed effect is caused by unidentified confounders or confounders which we have not been able to adequately take into consideration. In these discussions one has to apply some common sense. Is it reasonable that the factor in question affects the rate of the outcome and also the exposure rate in a way which will affect the risk estimates? Both effects are necessary for the identification of a confounder.
8.1 Mediating Factors Should Not Be Adjusted for
Quite often one sees that adjustments have been made for factors which are no confounders. Often this does not matter much, but a definite exception is adjustment for mediating factors.
A mediating factor is affected by the exposure and is then causing an outcome. So, for example, some drugs increase the risk for preterm birth, and preterm birth results in an increased risk of neonatal morbidity.
![]()
If this is the only way the exposure affects the outcome, adjustment for the mediating factor will eliminate the effect completely. In one study, an increased risk for childhood and young adult cancer was seen in children conceived by in vitro fertilization, IVF (Källén et al. 2010), and it was suggested that this was a consequence of the well-known increased risk for preterm birth, neonatal asphyxia, and low Apgar score after IVF, which would therefore act as mediating factors. In another study on partly the same material, one adjusted for preterm birth (and congenital malformations) and no increased risk of cancer was then found (Jerhamre Sundh et al. 2014). The conclusion to be drawn is not that there is no increased cancer risk among infants born after in vitro fertilization, but that it is mediated via an increased risk for preterm birth or factors associated with preterm birth, as previously suggested.
Adjustment for a mediating factor is useful if one wants to see if the effect of an exposure is completely or partly due to the mediating factor but should only be made for that purpose. It is then useful to make separate analyses with and without adjusting for the mediating factor. If one stratifies for preterm and term births and no effect remains in either stratum, one has shown that the effect of the exposure is completely due to preterm birth – if an effect remains within one or both strata, it seems likely that the effect does not (only) act via the studied mediator. In this example it should be remembered that preterm birth can mean anything from extreme preterm birth (e.g., <27 weeks) to late preterm birth (e.g., 35–36 weeks), so a remaining effect among preterm births could be due to different distributions of gestational length. In that situation one would have to adjust for actual pregnancy length.
8.2 Some Common and Sometimes Important Confounders
8.2.1 Year of Birth
When data are collected during a long period, one should consider if the registered rate of the studied outcome varies with year of delivery and if the use of the drug under investigation varies with year of delivery. This is exemplified in Fig. 8.1 which shows data for antidepressant use and the occurrence of septal heart defects in Sweden.
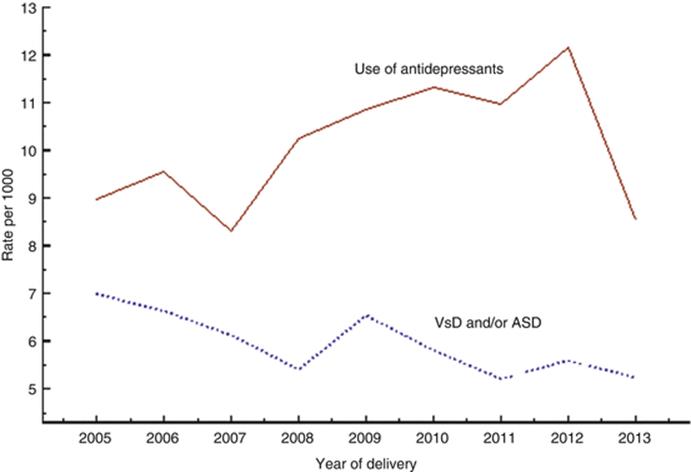
Fig. 8.1
Diagram showing rates of antidepressant use and of cardiac septum defects. Sweden, 2005–2013
One can see that the registration (and perhaps use) of antidepressants has been increasing up to 2012, followed by a drop during 2013, perhaps associated with an change in the administration of the register. The rate of diagnosed or registered cases with ventricular or atrium septum defects (without other heart defects) has declined during these years. If adjustment for year of delivery is not made, the association between the exposure and the outcome could be affected; in this case the result seemed to be a reduction of the estimate because the rate of the exposure increases and the rate of the outcome decreases. This graph is based on 9 873 infants exposed to antidepressants in early pregnancy among 971 287 infants studied – only 67 had a septum defect after antidepressant exposure. The crude odds ratio for a septum defect after antidepressant exposure was 1.09, and it increased to 1.15 if adjustment was made for year of birth, thus nearly a doubling.
The adjustment for year of delivery is obviously very important when long-term effects are studied because of the differences in follow-up time.
8.2.2 Maternal Age
Drug use during pregnancy varies much according to maternal age. Some examples are given in Fig. 8.2. For many drugs use increases with maternal age, but for some it decreases, e.g., antibiotics.
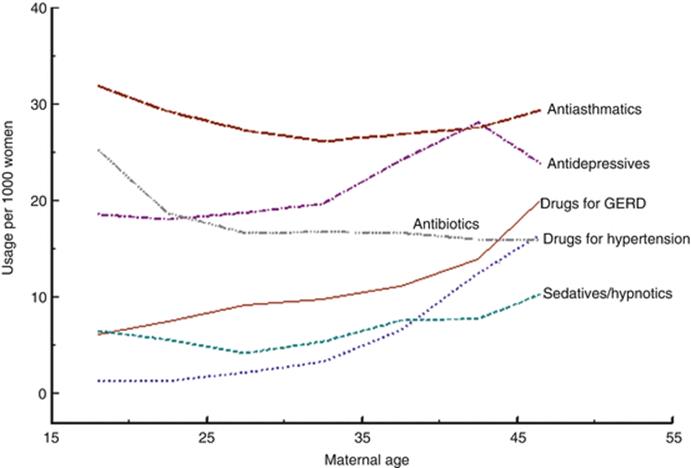
Fig. 8.2
Use of some drug groups at different maternal ages
For some congenital anomalies, notably chromosome anomalies, a strong relationship with a high maternal age is seen. For a few malformations, a high rate is associated with young maternal age (e.g., gastroschisis), while for the majority the age dependency is relatively weak (e.g., cardiovascular defects, Fig. 8.3). Note the markedly different maternal age dependencies of gastroschisis and omphalocele, both abdominal wall defects.
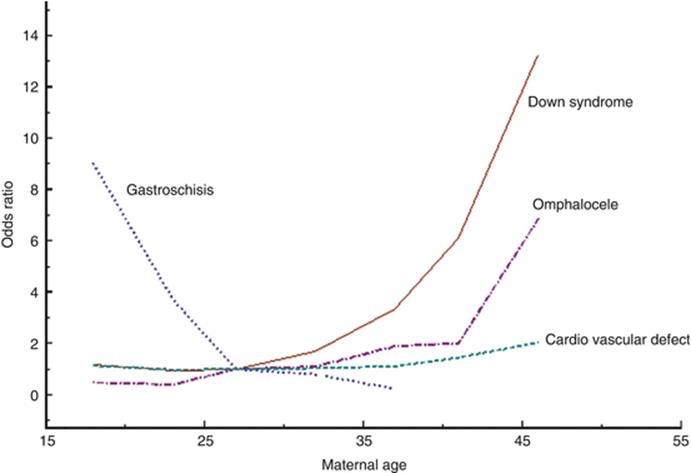
Fig. 8.3
Odds ratio for some malformations according to maternal age, adjusted for year of birth
For other outcomes than congenital malformations, maternal age plays a more important role, for instance, for preterm births (see Fig. 5.1).
When adjustments are made for maternal age, 5-year age groups are often used. When the risk is steeply changing with maternal age, for instance at Down syndrome or gastroschisis, adjustment for 1 year is recommended.
8.2.3 Parity and Gravidity
These two concepts – number of births and number of pregnancies, respectively – are of cause strongly connected. The difference is made up of spontaneous or induced abortions and of extrauterine pregnancies. Among these outcomes, perhaps previous spontaneous abortions are the most likely one as confounders for outcome of a later pregnancy (see below). Instead of working with the concept gravidity, I prefer to use parity and number of previous spontaneous abortions as separate variables.
Parity is the number of previously born infants. A pregnant woman expecting her first child is a nullipara (parity 0) during pregnancy, but after delivery she is of parity 1. This is a bit confusing, and it is useful to define what one means with parity. In developed countries rather few women are of higher parities than four. Therefore, in analyses four and higher parities are often grouped together.
Drug use is often higher at parity 1 than at higher parity, if adjustment for maternal age is made. The occurrence of most malformations is not influenced by parity, but for some, e.g., cardiovascular defects and hypospadias, the risk is higher at first parity than at higher parities after adjustment for age (Källén 2014).
8.2.4 Smoking
Information on smoking is usually obtained by interview or questionnaire. Such information may be somewhat uncertain, but repeated tests of the data in the Swedish Medical Birth Register indicate a reasonably good quality. Generally, one finds similar effects of smoking registered prospectively or retrospectively. Smoking is often stated as no/yes, but it is useful if some quantification can be made, notably in populations where smoking is relatively prevalent. In the Swedish Medical Birth Register and in some other sources of information, one has chosen to distinguish between smoking less than 10 cigarettes per day and 10 cigarettes or more per day. The latter group is often not very large but will also contain smokers of 20 cigarettes per day or more, and this subgroup may vary between different exposure groups.
Smoking is associated with a high use of some drugs (Fig. 8.4), notably psychotropic drugs,
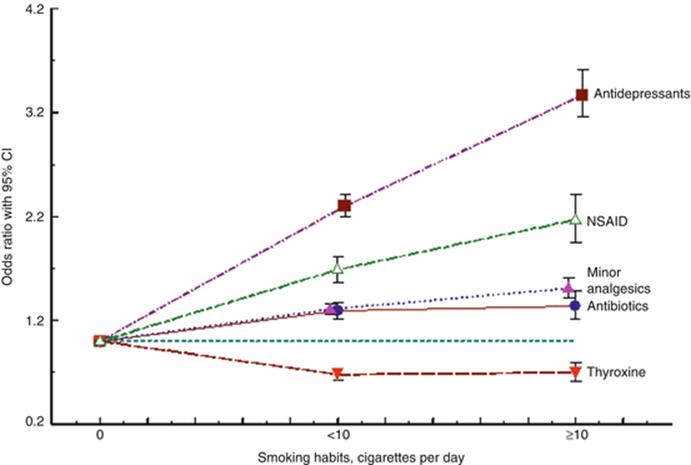
Fig. 8.4
Smoking habits among women using different drug groups in early pregnancy. ORs are adjusted for year of birth, maternal age, parity, and BMI
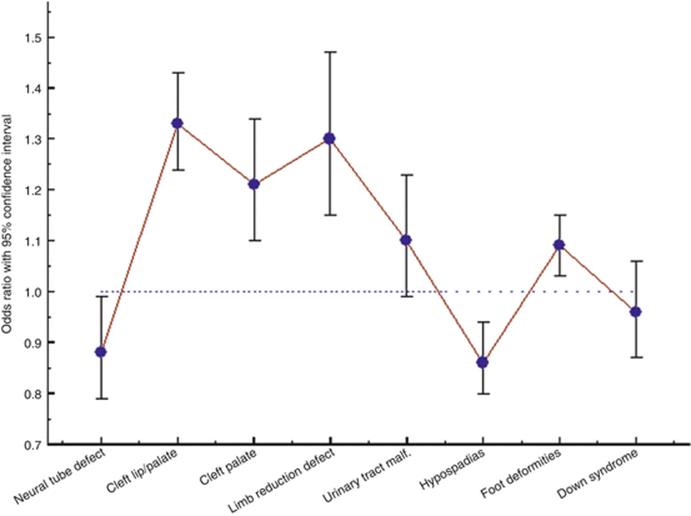
Fig. 8.5
Adjusted odds ratio for maternal smoking for some groups of congenital malformations (After Källén (2002))
The rate of some malformations is influenced by maternal smoking (Källén 2002; Hackshaw et al. 2011), and there is a statistically significant effect of maternal smoking on the occurrence of any major malformation (Källén 2014). Figure 8.4 shows the adjusted odds ratio for smoking at some groups of congenital malformations.
There is an effect of maternal smoking on preterm birth and notably on intrauterine growth retardation. Also other ill effects of maternal smoking have been described (Källén 2001).
8.2.5 Use of Alcohol
Maternal alcoholism or high use of alcohol is certainly a risk factor for many pregnancy outcomes, including congenital malformations. The result of maternal alcoholism is a typical condition, the fetal alcohol syndrome (FAS), characterized by intrauterine growth retardation, certain dysmorphic features, an effect on cognitive development, and an increased risk for some specific malformations, notably cardiovascular defects (Jones et al. 1973). The use of moderate amounts of alcohol during pregnancy should be avoided but no clear-cut effects are seen, not even on preterm births (Smith et al. 2015). The use of alcohol and the presence of alcoholism differ between different societies.
The use of alcohol during pregnancy is sometimes difficult to ascertain. Interview data are often unreliable because women may be concerned about stating their alcohol habits. In Sweden women coming to the first prenatal visit are interviewed by a midwife with a standard form which also includes questions on alcohol use. Very few women have then spontaneously admitted the use of alcohol during pregnancy. It is important to identify women with severe alcohol problems, but a simple question is hardly useful. Other methods are needed for their identification.
8.2.6 Overweight and Obesity
Overweight and obesity are identified and registered as prepregnancy BMI. If the woman comes early in pregnancy to prenatal care, BMI may be based on present body weight and height; otherwise, one has to rely on reported prepregnancy weight which may be uncertain.
The usual division of BMI is into <18.5 (underweight), 18.5–24.9 (normal), 25–29.9 (overweight), 30–34.9 (moderate obesity), 35–39.9 (severe obesity), and ≥40 (morbid obesity).
Overweight and obesity are associated with the use of some drug categories. Figure 8.6 shows the effect of maternal BMI on the use of three drug groups. The effect of BMI on the use of antibiotics is rather weak (although statistically significant), while the effect on the use of minor analgesics and antidepressants is marked. For most drug groups, usage increases with BMI.
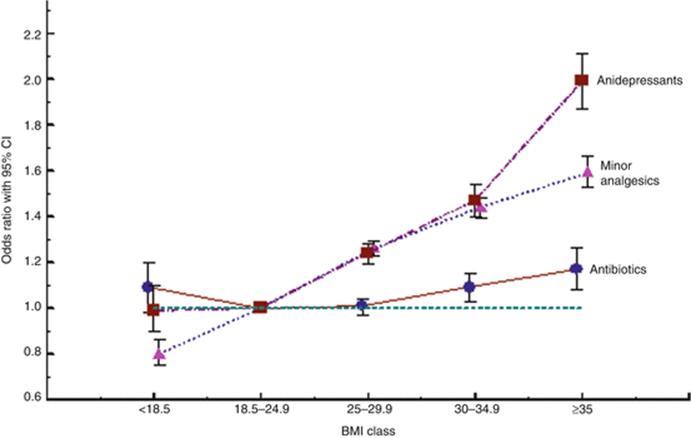
Fig. 8.6
Maternal use in early pregnancy of three drug groups according to maternal BMI. Adjustment for year of birth, maternal age, parity, and smoking
Maternal obesity has been shown to have important effects on various pregnancy outcomes, including many congenital malformations (Blomberg and Källén 2010). Figure 8.7 summarizes the effect of maternal obesity on the risk for a number of congenital malformations.
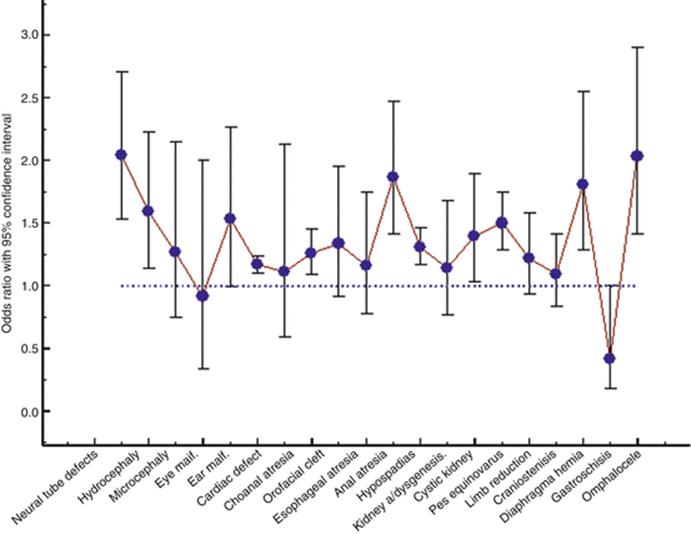
Fig. 8.7
Adjusted odds ratio for maternal obesity at different infant congenital malformations (Blomberg and Källén 2010)
Also other pregnancy complications are associated with maternal obesity (Cedergren 2006; Mission et al. 2015).
8.2.7 Subfertility
Subfertility means difficulty to get pregnant. The time it takes to achieve a pregnancy with unprotected intercourse is measured as the “time to pregnancy,” TTP. A common definition of subfertility is that the couple should have tried to achieve a pregnancy at least 1 year without success. Even then, a considerable proportion of the couples will have a spontaneous pregnancy after further time, but perhaps 5 % will not (Gnoth et al. 2005). They can then be regarded as infertile, and their only chance to get a pregnancy is by medical treatment, including in vitro fertilization (IVF).
Information on TTP relies on information from the couple and will be sensitive for various biases including recall bias. For epidemiologic purpose, one will have to do with that information, imperfect as it may be. Many studies have shown that subfertility is associated with an increased risk for pregnancy and delivery complications, and this seems to be the major cause of abnormalities (including congenital malformations) seen after IVF. Subfertility is obviously associated with the use of fertility drugs but may have an effect also on the use of other groups of drugs, either an increased usage (e.g., thyroxin) or a reduced usage (e.g., sedatives/hypnotics and antidepressants) (Källén 2009).
8.2.8 Race/Nationality
In many populations, racial differences in pregnancy outcome are important, partly because of associations with socioeconomic conditions, partly because of genetics. This is true also for some congenital malformations, e.g., neural tube defects which are much more frequent in the US population among whites than among blacks. In other countries, e.g., the Scandinavian, the race issue is much less important – and furthermore in some countries it is illegal to register race in official registers. A proxy for race in the Scandinavian countries is the country of birth of the parents. In an analysis of the years 1978–1993 (Källén 1998), it was shown that the major anomaly in delivery outcome in Sweden according to maternal country of birth was a possible worse outcome for women who came from sub-Saharan Africa. At that time the proportion of these women was relatively low, less than 0.1 % in 1978 and about 1 % 1993. For the period 2005–2012 (Fig. 8.8), the proportion of women who were born in sub-Saharan Africa increased from 1.5 to 3 %. The majority of these women were probably black. The proportion of women born in East Asia increased from 0.2 % in 1978 to 1.4 % in 1993 and from 2.1 to 2.7 % in the period 2005–2012. The percentage of Swedish-born women decreased from 87.6 % in 1978 to 83.2 % in 1993 and from 80.7 % in 2005 to 74.9 % in 2012. Even though at present 25 % of women who gave birth were not born in Sweden, the vast majority of these were what would be called “Caucasian” in the USA, and only a small part were likely to be “Black” or “Asian.”
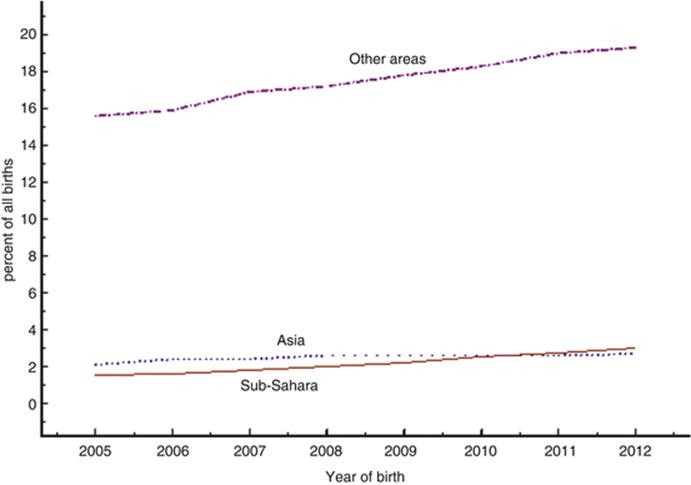
Fig. 8.8
Proportion of women who gave birth and were born outside Sweden, 2005–2012
That the mother was not born in Sweden may mean different things. Some were adopted as children and had grown up in Sweden, and the main difference from Swedish-born women would be the genetic setup. Many were immigrants and may have other lifestyle and socioeconomic conditions than Swedish women. There is information on the year of immigration, but it is not routinely included in the Swedish Medical Birth Register.
There are sometimes definite differences in drug use (or registration) in immigrant women compared to Swedish-born women. Some examples can be mentioned taken from Källén (2009). The use of antihypertensive drugs is roughly the same among Swedish-born women and women born in other Nordic countries but clearly lower among other immigrant women. The same is seen for thyroxine and, less clearly, for antibiotics and anticonvulsants. On the other hand, the use of antipsychotic drugs is higher in non-Nordic immigrant women, while antidepressants are used less often than by Swedish-born women. The same is true for anti-asthmatics.
On the other hand the malformation risk is rarely affected by maternal country of birth. Exceptions exist, so for instance the risks for microcephaly and hypospadias are increased and the risk of a cardiovascular malformation or an orofacial cleft is decreased in infants born of immigrant women (Källén 2014).
8.2.9 Socioeconomic Level
For many pregnancy outcomes, socioeconomic variables are of importance, e.g., for preterm birth. The significance of socioeconomic variables varies according to the nature of the society. In some countries, large differences and extreme poverty exist – in other countries like the Scandinavian ones, differences are much less marked, and extreme poverty is rare due to the existing social security system. The significance of socioeconomic level for pregnancy outcome may also depend on the health system. In countries where prenatal and delivery care is free and drugs to a large extent are paid by society, the economic aspects are of less importance than in countries where the patients themselves have to pay for care. There may still be socioeconomic differences of significance. In Sweden low socioeconomic level is associated with smoking during pregnancy and obesity, and if adjustment for these factors is made, only a moderate effect of socioeconomy remains. Remaining effects could, for instance, act via nutrition or occupational hazards.
There are different ways to measure socioeconomic level. Sometimes a socioeconomic index has been defined, valid individually or for the area where the woman lives. Such indexes are often built on income, occupation, type of housing, etc. For individual evaluation, the educational level is often used, notably when registers on education exist in the country.
In the Swedish school system, 9 years are compulsory, and most children continue for another 3 years in so-called gymnasium. For many drugs, one can see no variation in usage according to maternal education level, but in some a variation is seen (Fig. 8.9). The use of sedatives and hypnotics is clearly associated with a short education – so is also use of antidepressants (not shown in the Figure). A marked variation in the use of ophthalmologicals is also seen, but then low education level is associated with a low usage and high educational level with a high usage.
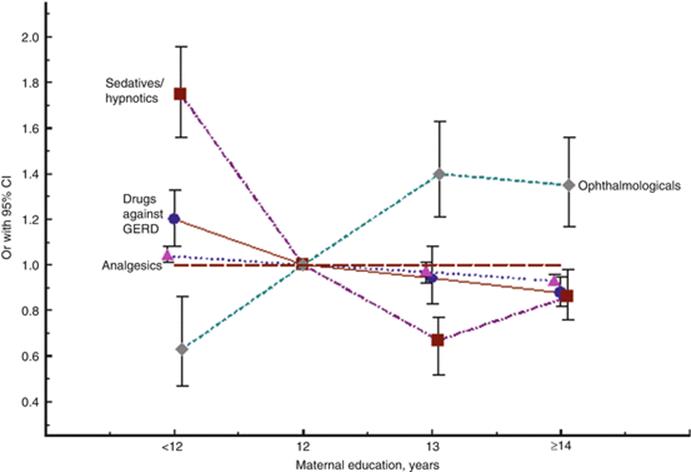
Fig. 8.9
Odds ratio (OR) for the use of some drugs according to length of maternal education
The impact of maternal education (adjusted for smoking and BMI) on the risk for congenital malformation is relatively weak (Källén 2014). A classical relationship exists between a low socioeconomic level and infant spina bifida (Elwood et al. 1992), at least in the UK and some parts of the USA. Early studies in Sweden found no relationship between maternal occupation and the risk for neural tube defects in the infants (Ericson et al. 1988).
More evident effects are seen of the parental socioeconomic level on preterm birth, infant birth weight, and intrauterine growth. A large part of this effect is due to smoking and high BMI.
8.2.10 Geography
There may be geographical variations in the recording of drug use, either because of different prevalence of underlying disease, different therapeutic tradition, or different completeness in the ascertainment of drug use. There may also be geographical differences in the registered rates of outcome, for instance, congenital malformations. If both factors are under-ascertained in some areas and completely ascertained in other, it will give a false association between exposure and outcome. The highest risk to get this is if the source of information is the same, for instance, delivery records. If data are obtained from different sources, the risk is reduced but may still exist.
Obviously true differences may also exist in drug usage in different geographical areas, and in true differences in malformation rates and even if the actual risk with the drug is lacking, an apparent effect may appear.
8.2.11 Previous Reproductive History
The previous reproductive history may affect the risk that a new pregnancy results in a malformed infant or some other negative outcome. Such an association may be due to parental genetic or nongenetic factors which increase the risk. To act as confounders, the previous reproductive outcome must also affect the probability of the exposure to drugs.
Previous miscarriages are sometimes associated with an increased risk for a congenital malformation, e.g., a cardiovascular defect, hypospadias, or a multimalformed infant (Källén 2014). For most malformations, the association is weak or absent. The effect is hardly a direct one but more a sign of a common cause, e.g., a chronic condition like preexisting diabetes or a genetic burden, sometimes causing embryonic death, sometimes malformation.
For all three drugs in Fig. 8.10, an increased use is associated with an increased number of previous miscarriages, but the explanation to this may vary.
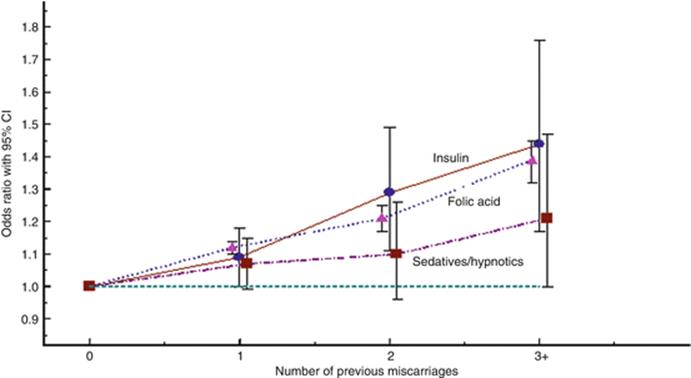
Fig. 8.10
The effect (estimated as odds ratios adjusted for year of birth, maternal age, parity, smoking, and BMI) of the number of previous miscarriages on the use of three drug categories: insulin, folic acid, and sedatives or hypnotics
In case of preexisting diabetes (which is the reason for the administration of insulin), it is likely that the disease increases the risk for a miscarriage so the use of insulin in the present pregnancy indicates a previous risk for miscarriage because most likely the woman was diabetic also during the previous pregnancy.

Folic acid may be taken at an increased rate after previous miscarriages. They will then be a probably weak confounder in an analysis of the effect of folic acid on congenital malformations by increasing the estimate for at least some malformations.

A weak similar confounding may exist for sedatives/hypnotics where previous miscarriages may increase the use of psychoactive drugs, including sedatives/hypnotics. This would result in a slight increase of the malformation risk (which incidentally is very weak for most such drugs). Some careful considerations are needed before adjustment for previous miscarriages is made.
A similar situation exists when an earlier child was malformed as is seen in Fig. 8.11. The presence of a previous infant with malformations is associated with an increased risk for some drugs which indicates the occurrence of chronic diseases with a potential to teratogenesis, e.g., insulin and drugs for chronic hypertension. An increased use of folic acid is also seen (still stronger if the previous malformation was a neural tube defect (OR = 6.0)). It is clinical practice to give high folic acid doses after a previous neural tube defect fetus which explains the strong effect. An increased use of psychoactive drugs can be noted, perhaps from a psychological distress caused by the birth of the damaged infant. In this situation, the birth of a previously malformed infant will be a confounder, notably if a strong heredity for the malformation exists.
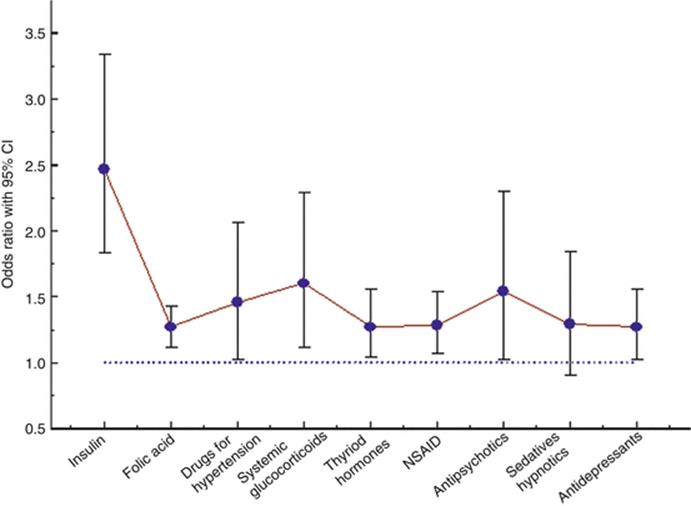
Fig. 8.11
Diagram showing the odds ratios for a number of drug groups, comparing usage among women with and without a previous malformed infant
There are rather few signs that the occurrence of a malformation in a previous pregnancy would decrease drug use in the following pregnancy. The OR estimate is low for migraine drugs (OR = 0.57, 95 % CI 0.29–1.32), but statistical significance is not reached.
8.2.12 Infant Sex
Many congenital malformations and other outcomes differ in rates between males and females, but infant sex is a confounder only if it also affects the rate of drug exposure. This is rare, but it does happen: nausea and vomiting in pregnancy (NVP) occur more often in pregnancies carrying a female than a male embryo, and if one studies a malformation which has a deviating sex distribution, infant sex will be a confounder. It is possible that the teratogenic effect of a drug differs on male and female embryos. This can be studied by stratifying for infant sex (but not by adjusting for infant sex).
8.2.13 Concomitant Maternal Disease and Drug Use
Some chronic diseases in the mother can affect infant outcome, including congenital malformations. The most well-known one is preexisting maternal diabetes, but also other diseases may affect embryonic or fetal development, e.g., other endocrine diseases and chronic hypertension. In rare cases also acute diseases may be relevant – a classical example is maternal rubella which carries a high risk for damage of the embryo if occurring in early pregnancy.
Maternal diseases will be relevant for the discussion of confounding by indication (see below) but may also complicate analyses by the effect of other drugs than those used for the disease. So, for examples, diabetes may be associated with chronic hypertension. Usually numbers are not very high but may motivate an exclusion of women with preexisting diabetes in the analysis of the effects of drugs for hypertension.
An expression of comorbidity is the use of other drugs in excess when the effect of a specific drug is studied. This is exemplified in Fig. 8.12.
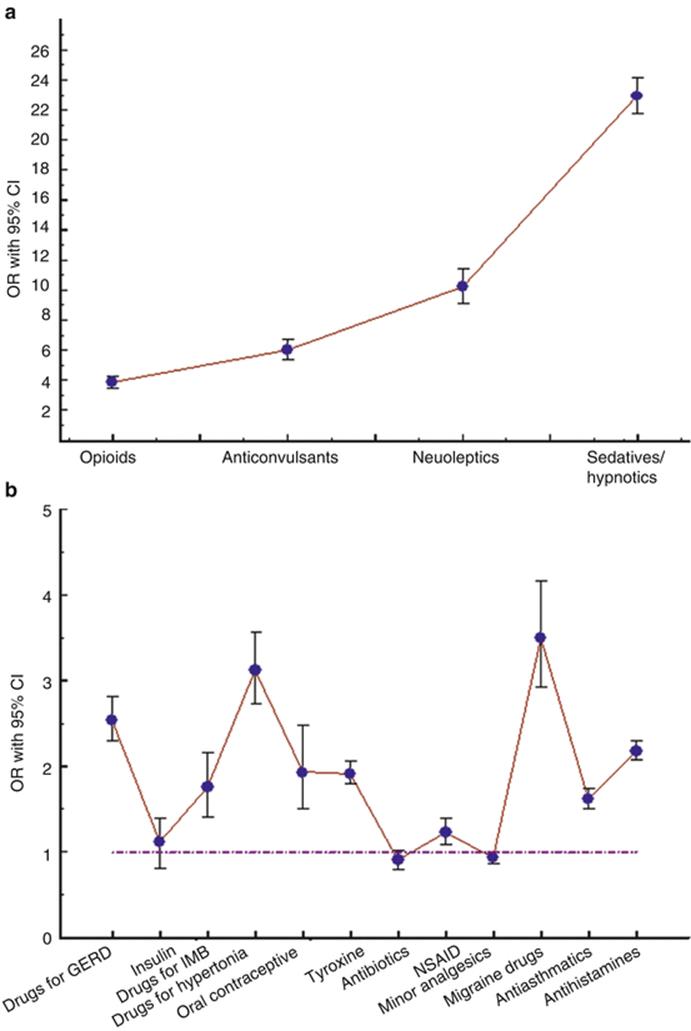
Fig. 8.12
Odds ratio for women having used various groups together with antidepressants versus women who did not use antidepressants, adjusted for year of birth, maternal age, parity, smoking, and BMI. (a) shows some drugs with an effect on CNS, (b) some other drugs
If drugs used in excess together with the drug under study have a teratogenic effect, this has to be adjusted for or individuals with such exposure should be excluded from the analysis. Obviously such exclusion should then be made also from the non-exposed comparison group. A typical example is the use of anticonvulsants as mode stabilizers with antidepressants. If this is a frequent event, a falsely increased effect of the antidepressant may be obtained. As seen in Fig. 8.12, there is also an excess use, for instance, of drugs for migraines (mainly triptans) but as these have no obvious teratogenic effect they cannot affect the observed effect of antidepressants.
This phenomenon should not be mixed up with the possibility that two drug categories (e.g., antidepressants and sedatives) could act synergistically.
8.2.14 Confounding by Indication
Confounding by indication means that an effect of a drug is really due to underlying disease that was the reason for the drug use. A classic example is diabetes and insulin. There is no difficulty to show that women who took insulin in early pregnancy have an increased risk for having a baby with a congenital malformation. Such infants have a nearly doubled risk to have a major malformation and a three times increased risk of a cardiovascular defect (Källén 2009). The risk for many other types of congenital malformations is also increased. Insulin is reasonably not the cause of this, but the underlying disease, diabetes, is. In order to prove that this is the case one would need to have a large group of pregnant women who got insulin without having diabetes (which is impossible to find) and pregnant women with diabetes type 1 who did not get insulin (which is equally impossible). In practice, insulin is only given to patients with diabetes, and all pregnant type 1 diabetic patients get insulin.
In other circumstances it is less obvious if the drug or the disease caused the outcome. An example is anticonvulsants. Women with epilepsy who use anticonvulsants generally have an increased risk to get a malformed infant. When this was initially detected, the question came if it was the drug or the disease (notably the occurrence of seizures in early pregnancy) that caused the malformations. It is possible to identify women with epilepsy who did not use anticonvulsant drugs in pregnancy (and they have no increased risk to have a malformed infant), but they reasonably have another or milder form of epilepsy than the women who used anticonvulsants so they are no perfect controls.
Other efforts have been made to separate the effect of drugs from the effect of underlying disease. One has tried to quantify the underlying disease by scoring (propensity scores), often based on disease history before pregnancy, and then in one way or another adjust for disease score. This, for instance, has been made for depression and antidepressants (Oberlander et al. 2006). These authors found an effect on neonatal outcome of the disease but an added effect of the drug use. Other authors found no effect of depression when studying gestational age at birth as outcome (e.g., Suri et al. 2007).
Another way to separate drug effect and disease effect is to compare different drugs, used for the same condition. This can be exemplified with anticonvulsants where the teratogenic effect varies between different drugs – but it is also possible that this is due to the use of different drugs at different forms of epilepsy. In some studies, most SSRI drugs show no signs of teratogenicity, but an association between paroxetine use and cardiovascular defects may exist. Again the use of the specific SSRI drugs may vary between different underlying conditions, e.g., depression and anxiety, so it is still possible that the noted differences between the SSRI drugs are explained by underlying disease.
A further example refers to chronic hypertension and drugs used for that condition. Cooper et al. (2006) found an increased risk for congenital malformations after maternal use of ACE inhibitors but not after the use of other antihypertensives. This would speak for a specific drug effect. A later study of Lennestål et al. (2009) verified the risk increase after ACE inhibitors but found a similar effect also of beta-blockers when used for hypertension. The effect could therefore be a result of the underlying disease, chronic hypertension.
It should be observed that many studies which are quoted as evidence of an effect of underlying disease (e.g., depression and stress) have not distinguished between the disease and the drugs used.
8.3 Interaction Between Confounders
One confounder may interact with another. Above it was shown that the risk for preterm birth varied with maternal age, but that the graphs showing this relationship differed markedly between parity (Fig. 5.1). Furthermore, parity and BMI increase and smoking decreases with maternal age.
In order to study the impact of a specific confounder, adjustment for other confounders should be made. Often tables are produced which show the distribution of for instance maternal age and parity – it is more informative to calculate the odds ratio for each maternal age adjusting for parity and for each parity adjusting for maternal age.
Table 8.1 shows the two ways to tabulate characteristics of women using antidepressants compared with other women. The adjusted ORs thus show the effect of each variable irrespective of the other variables in the Table. This makes it possible, for instance, to evaluate if the maternal age affects drug use irrespective of parity and if parity does so irrespective of age.
Table 8.1
Characteristics of women reporting the use of antidepressants (AD) in early pregnancy and of all women who gave birth, Sweden 2005–2013
|
Variable |
AD, number |
AD, percent |
Population, number |
Population, percent |
OR |
95 % CI |
|
Year of birth |
||||||
|
2005 |
881 |
9.1 |
91,046 |
10.3 |
0.93 |
0.55–1.02 |
|
2006 |
986 |
10.1 |
95,021 |
10.7 |
0.96 |
0.88–1.04 |
|
2007 |
868 |
8.9 |
96,299 |
10.9 |
0.85 |
0.78–0.93 |
|
2008 |
1087 |
11.2 |
97,594 |
11.0 |
1.00 |
Reference |
|
2009 |
1171 |
12.0 |
99,077 |
11.2 |
1.06 |
0.98–1.16 |
|
2010 |
1274 |
13.1 |
104,354 |
11.8 |
1.10 |
1.01–1.19 |
|
2011 |
1188 |
12.2 |
99,418 |
11.2 |
1.08 |
0.99–1.17 |
|
2012 |
1318 |
13.6 |
99,494 |
11.2 |
1.21 |
1.11–1.30 |
|
2013 |
948 |
9.8 |
102,295 |
11.6 |
0.84 |
0.77–0.92 |
|
Maternal age |
||||||
|
<20 |
144 |
1.5 |
14,872 |
1.7 |
0.74 |
0.62–0.88 |
|
20–24 |
1131 |
11.6 |
115,179 |
13.0 |
0.85 |
0.79–0.91 |
|
25–29 |
2584 |
26.1 |
254,911 |
28.8 |
1.00 |
Reference |
|
30–34 |
3281 |
33.8 |
306,239 |
34.6 |
1.14 |
1.08–1.20 |
|
35–39 |
2072 |
21.3 |
158,615 |
17.9 |
1.40 |
1.32–1.49 |
|
40–44 |
487 |
5.0 |
33,048 |
3.7 |
1.52 |
1.37–1.69 |
|
≥45 |
22 |
0.2 |
1736 |
0.2 |
1.27 |
0.82–1.95 |
|
Parity |
||||||
|
1 |
4592 |
47.2 |
396,970 |
44.0 |
1.00 |
Reference |
|
2 |
3060 |
31.5 |
323,944 |
36.6 |
0.75 |
0.71–0.78 |
|
3 |
1424 |
14.6 |
115,178 |
13.0 |
0.86 |
0.81–0.92 |
|
≥4 |
645 |
1.6 |
48,506 |
5.5 |
0.80 |
0.73–0.88 |
|
Smoking |
||||||
|
Unknown |
74 |
0.8 |
45,563 |
5.2 |
– |
|
|
None |
8215 |
84.5 |
782,311 |
88.4 |
1.00 |
Reference |
|
<10 cigs/day |
1003 |
10.3 |
43,695 |
4.9 |
2.20 |
2.12–2.41 |
|
≥10 cigs/day |
429 |
4.6 |
13,029 |
1.5 |
3.15 |
2.86–3.46 |
|
BMI |
||||||
|
Unknown |
426 |
4.3 |
74,930 |
8.5 |
– |
|
|
<18.5 |
198 |
2.0 |
19,778 |
2.2 |
1.00 |
0.86–1.15 |
|
18.5–24.9 |
4809 |
50.3 |
487,427 |
55.1 |
1.00 |
Reference |
|
25–29.9 |
2528 |
26.0 |
203,520 |
23.0 |
1.20 |
1.14–1.26 |
|
30–34.9 |
1056 |
10.9 |
69,425 |
7.8 |
1.44 |
1.35–1.54 |
|
≥35 |
624 |
6.4 |
29,518 |
3.3 |
1.94 |
1.78–2.11 |
|
Total number |
9721 |
– |
884,598 |
– |
– |
– |
Odds ratios (OR) with 95 % confidence intervals (95 % CI) adjusted for all other variables in the table
8.4 Residual Confounding
Even when extensive efforts have been made to adjust for confounding, residual confounding may remain. Either important confounders exist which have not been identified or it has not been possible to include them in the adjustment, or adjustment has been made for a factor but in an incomplete way. If adjustment for maternal smoking has been based on three levels: none, <10 cigarettes per day, and ≥10 cigarettes per day, no complete correction has been made for smoking 20 cigarettes per day. If in a group of women more smoke ≥10 cigarettes per day than another group, it is likely that the proportion smoking ≥20 cigarettes a day are also overrepresented and that has not been possible to adjust for. If adjustment for the two known levels of smoking results in a reduction of the risk estimate, it is likely that a further reduction had been obtained if adjustment could have been made for ≥20 cigarettes per day.
In nearly every study, it is possible that residual confounding exists. The larger effect one has obtained by the adjustments applied, the more likely it is that the association is sensitive for residual confounding.
References
Blomberg M, Källén B (2010) Maternal obesity and morbid obesity: the risk for birth defects in the offspring. Birth Def Res (Part A) 88:35–40
Cedergren M (2006) Effects of gestational weight gain and body mass index on delivery outcome in Sweden. Int J Gyneacol Obstet 93:269–274CrossRef
Cooper WO, Hernandez-Dias S, Arbogast PG, Dudley JA, Gideon PS, Hall K et al (2006) Major congenital malformations after first-trimester exposure to ACE-inhibitors. N Engl J Med 354:2443–2451CrossRefPubMed
Elwood JM, Little J, Elwood JH (1992) Epidemiology and control of neural tube defects. Monographs in epidemiology and biostatistics, vol 20. Oxford University Press, Oxford, New York, Tokyo
Ericson A, Källén B, Löfqvist E (1988) Environmental factors in the etiology of neural tube defects. A negative study. Enviroment Res 45:38–47CrossRef
Gnoth C, Godehardt E, Frank-Herrmann P, Friol K, Tigges J, Freundl G (2005) Definition and prevalence of subfertility and infertility. Hum Reprod 20:1144–1147CrossRefPubMed
Hackshaw A, Rodeck C, Boniface S (2011) Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update 17:589–604CrossRefPubMedPubMedCentral
Sundh KJ, Henningsen A-J, Källén K, Bergh C, Romundstad LB, Gissler M et al (2014) Cancer in children and young adults born after assisted reproductive technology: a Nordic cohort study from the Committee of Nordic ART and Safety (CoNARTaS). Hum Reprod 29:2050–2057CrossRefPubMed
Jones KL, Smith DW, Streissguth AP, Myrianthopoulus N (1973) Outcome in offspring of chronic alcoholic women. Lancet i:1076–1078
Källén B (2009) Drugs during pregnancy. Nova Biomedical Books, New York
Källén B (2014) Epidemiology of human congenital malformations. Springer, Heidelberg, New York, Dordrecht, London
Källén B, Finnström O, Lindam A, Nilsson E, Nygren K-G, Otterblad OP (2010) Cancer risk in children and young adults conceived by in vitro fertilization. Pediatrics 126:e270–e276. doi:10.1542/peds.2009-3225CrossRef
Källén K (1998) Förlossningsresultat bland invandrarkvinnor i Sverige. (Delivery results in immigrant women in Sweden). EpC Report 1998:1. Socialstyrelsen
Källén K (2001) The impact of maternal smoking during pregnancy on delivery outcome. Eur J Pub Health 11:329–335CrossRef
Källén K (2002) Maternal smoking and congenital malformations. Rev Fetal Maternal Rev 13:63–86
Lennestål R, Otterblad Olausson P, Källén B (2009) Maternal use of antihypertensive drugs in early pregnancy and delivery outcome, notably the presence of congenital heart defects in the infants. Eur J Clin Pharmacol 65:615–625CrossRefPubMed
Mission JF, Marshall NE, Caughey AB (2015) Pregnancy risk associated with obesity. Obst Gynecol Clin North Am 48:335–353CrossRef
Oberlander TF, Warburton W, Misri S, Aghajanian J, Herzman C (2006) Neonatal outcome after prenatal exposure to selective serotonin reuptake inhibitor antidepressants and maternal depression using population-based linked health data. Arch Gen Psychiatry 63:898–906CrossRefPubMed
Oberlander TF, Warburton W, Misri S, Riggs W, Aghajanian J, Herzman C (2008) Major congenital malformations following prenatal exposure to serotonin reuptake inhibitors and benzodiazepines using population-based health data. Birth Def Res (Part B) 83:68–76CrossRef
Reis M, Källén B (2013) Combined used of selective serotonin reuptake inhibitors and sedatives/hypnotics during pregnancy; risk of relatively severe congenital malformations or cardiac defects. A register study. BMJ Open 3:e002166. doi:10.1136/bmjopen-2012-002166CrossRefPubMedPubMedCentral
Smith LK, Draper ES, Evans TA, Field DI, Johnson SJ, Manktelow BN et al (2015) Associations between late and moderately late preterm birth and smoking, alcohol, drug use and diet: a population-based case-cohort study. Arch Dis Child Fetal Neonatal Ed 0:F1–F6. doi: 10.1136/archdischild-2014-307265
Suri R, Altschuler L, Hellenmann G, Burt VK, Aquino A, Mintz J (2007) Effects of antenatal depression and antidepressant treatment on gestational age at birth and risk of preterm birth. Am J Psychiatry 164:1206–1211CrossRefPubMed