Jill B. Whelan1 and Loryn S. Feinberg2
(1)
Beth Israel Deaconess Medical Center, Department of Medicine, Division of Cardiology, Boston, MA, USA
(2)
Women’s Cardiovascular Health Program, Beth Israel Deaconess Medical Center, Department of Medicine, Division of Cardiology, Boston, MA, USA
Jill B. Whelan
Email: jbwhelan@bidmc.harvard.edu
Loryn S. Feinberg (Corresponding author)
Email: lfeinber@bidmc.harvard.org
Keywords
PregnancyHeart diseaseCongenitalAnticoagulationArrhythmiaMyocardial infarctionHeart failure
Case 1: Congenital Heart Disease, Bicuspid Valve
A 27-year-old woman presents at 14-weeks gestation for evaluation of a heart murmur. She has no prior past medical history and is taking prenatal vitamins. She was recently diagnosed with a heart murmur at an annual exam with her new primary care provider three months prior to the visit. She underwent a transthoracic echocardiogram which demonstrated a mildly thickened bicuspid aortic valve, with evidence of moderate aortic regurgitation. The aortic root and ascending aorta were well visualized, with mild root dilation of 37 mm and high-normal ascending aortic dimension of 35 mm. She had normal left ventricular cavity size and systolic function. She denies symptoms of shortness of breath, chest pain, dyspnea, palpitations, or peripheral edema. She exercises daily, walking two miles on flat ground without limitation. On examination, her blood pressures were 126/56 mmHg in the right upper extremity and 131/54 mmHg in the left upper extremity. She has a 2/4 diastolic murmur at the left lower sternal border, and her lung sounds are clear bilaterally, without appreciable rales or wheezing. Her laboratory tests showed normal hematocrit and platelet counts, and chemistry panel demonstrated normal electrolytes and renal function. On review, her grandfather had an aortic valve replacement when he was 56 years old, and her father carries the diagnosis of aortic stenosis. She is thrilled about her first pregnancy but is worried about the consequences of her valve disorder. She undergoes dedicated MRA imaging of the thoracic aorta, which shows aortic root dimension of 38 mm, ascending aortic dimension of 35 mm, aortic arch dimension of 29 mm, and descending aortic dimension of 24 mm. There is no evidence of aortic coarctation, and there is no aortic flow reversal in the descending aorta. She was counseled regarding the presence of a congenital bicuspid aortic valve, mild aortic dilation, and the need to follow these findings over time. However, there was determined to be no contraindication to pregnancy. She underwent evaluation with a cardiologist in her second and third trimester, without development of clinical symptoms suggesting changes in valvular pathology. She delivered a healthy baby girl at 38 6/7 weeks without complication.
Aortic insufficiency (AI) or regurgitation is generally well tolerated in pregnancy due to a combination of reduced systemic vascular resistance and shortened diastole due to a rise in heart rate, which can reduce the severity of the regurgitant volume. In young women, AI may be due to a congenital bicuspid valve, rheumatic heart disease, infective endocarditis, an autoimmune disorder (i.e., rheumatoid arthritis), or a dilated aorta, such as in certain connective tissue disorders (Marfan syndrome, Ehlers-Danlos syndrome). Typically, symptoms are correlated with severity of regurgitation and the presence of left ventricular dilation and dysfunction. Severe aortic regurgitation with left ventricular dysfunction or acute onset regurgitation is poorly tolerated [1]. In this patient, the presence of a congenital bicuspid aortic valve has led to the development of moderate AI, and she has thus far remained asymptomatic.
Assessing maternal and fetal risk, ideally prior to conception, is central to the management of heart disease in pregnancy. Risk prediction scores of cardiac disease in pregnancy were studied by the CARPREG investigators, who examined four clinical features in the general population including prior arrhythmia or cardiac event, NYHA functional class > II or cyanosis, left heart obstruction, and systolic left ventricular dysfunction with LVEF < 40 %. They determined maternal cardiac event rates of 5 %, 27 %, and 75 % for the presence of 0, 1, and >1 of these clinical features (Tables 1 and 2) [2]. Adverse maternal cardiac events consisted of pulmonary edema, symptomatic arrhythmia, stroke, cardiac arrest, or death. More recently, the ZAHARA investigators expanded the clinical predictors of adverse maternal and fetal outcomes while studying a large population of congenital heart disease patients [3, 4]. In these risk models, severe aortic insufficiency with NYHA class III or IV symptoms was thought to be poorly prognostic of maternal and fetal outcomes. However, moderately regurgitant valves tended to be less symptomatic and typically did well with conservative medical management. According to the modified World Health Organization (WHO) risk classification, which integrates known maternal cardiovascular risk factors including conditions with contraindications to pregnancy (Tables 3 and 4) [5], moderate AI represents a modified WHO class II classification. Patients who are WHO class II have a small increased risk of maternal mortality or moderate increase in morbidity, and these patients are recommended to be evaluated by a cardiologist and high-risk obstetrician each trimester for clinical assessment. Modified WHO classification is a useful metric for assessing maternal and fetal risk of cardiac lesions that are established prior to pregnancy.
Table 1
New York Heart Association functional classification
|
NYHA class |
Symptoms |
|
I |
Cardiac disease present, but no symptoms and no limitation in ordinary physical activity, e.g., no shortness of breath with walking (>3–4 city blocks), climbing stairs (two flights), etc. |
|
II |
Mild symptoms (mild shortness of breath and/or angina) and slight limitation during ordinary activity |
|
III |
Marked limitation in activity due to symptoms, even during less-than-ordinary activity, e.g., walking short distances (<100 m). Comfortable only at rest |
|
IV |
Severe limitations. Experiences symptoms even while at rest. Mostly bedbound patients |
Table 2
Predictors of maternal cardiovascular events and risk score based on the CARPREG study
|
Cardiovascular event prior to pregnancy (heart failure, transient ischemic attack, stroke, or arrhythmia) |
|
Baseline NYHA functional class > II or cyanosis |
|
Obstructive left ventricular lesions (by echocardiography: mitral valve area <2 cm2, aortic valve area <1.5 cm2, peak LV outflow tract gradient > 30 mmHg) |
|
Left ventricular systolic dysfunction (ejection fraction <40 %) |
Adapted from Regitz-Zagrosek et al. [1] and Thorne et al. [5]
One point assigned per predictor. Risk estimation of cardiovascular maternal complications: 0 points, 5 %; 1 point, 27 %; >1 point, 75 %
LV left ventricle, NYHA New York Heart Association
Table 3
Modified World Health Organization (WHO) classification of maternal cardiovascular risk principles
|
Risk class |
Risk of pregnancy by medical condition |
|
I |
No or mild increase in maternal morbidity, and no discernable increased risk of maternal mortality |
|
II |
Moderate increase in maternal morbidity and small increased risk of mortality |
|
III |
Potential for severe maternal morbidity and significantly increased risk of mortality. Expert counseling regarding risk in pregnancy required. If pregnancy is decided upon, multidisciplinary cardiac and obstetric monitoring needed throughout pregnancy, childbirth, and puerperium |
|
IV |
Extremely high risk of severe maternal morbidity or mortality; pregnancy contraindicated. If pregnancy occurs, termination should be discussed. If pregnancy continues, care as class III with multidisciplinary team |
Adapted from Regitz-Zagrosek et al. [1] and Thorne et al. [5]
Table 4
Modified WHO classification of maternal risk by cardiovascular disease
|
Conditions in which pregnancy risk is WHO I |
|
Mild pulmonic stenosis, patent ductus arteriosus, or mitral valve prolapse, if uncomplicated |
|
Repaired atrial or ventricular septal defect, patent ductus arteriosus, anomalous pulmonary venous drainage |
|
Isolated atrial or ventricular premature contractions |
|
Conditions in which pregnancy risk is WHO II to III |
|
WHO II (if otherwise asymptomatic and uncomplicated) |
|
Uncorrected atrial or ventricular septal defect |
|
Tetralogy of Fallot, repaired |
|
Majority of arrhythmias |
|
WHO II–III (depending on individual) |
|
Mild left ventricular systolic dysfunction |
|
Hypertrophic cardiomyopathy |
|
Valvular regurgitation or stenosis (native or prosthetic) not considered WHO I or IV |
|
Marfan syndrome in the absence of aortic dilation |
|
Aortic dilation (<45 mm) associated with bicuspid aortic valve |
|
Repaired aortic coarctation |
|
WHO III |
|
Prosthetic mechanical valve |
|
Systemic right ventricle |
|
Fontan circulation |
|
Cyanotic congenital heart disease (unrepaired) |
|
Complex congenital heart disease |
|
Marfan syndrome with aortic dilation of 40–45 mm |
|
Bicuspid aortic valve with aortic dilation of 45–50 mm |
|
Conditions in which pregnancy risk is WHO IV (pregnancy contraindicated) |
|
Pulmonary arterial hypertension (of any cause) |
|
Severe left ventricular systolic dysfunction (LVEF <30 %, NYHA III–IV) |
|
Prior peripartum cardiomyopathy, with any residual left ventricular systolic dysfunction |
|
Severe mitral stenosis, severe symptomatic aortic stenosis |
|
Marfan syndrome with aortic dilation of >45 mm |
|
Bicuspid aortic valve with aortic dilation of >50 mm |
|
Severe aortic coarctation |
Adapted from Regitz-Zagrosek et al. [1] and Thorne et al. [5]
WHO World Health Organization, LVEF left ventricular ejection fraction, NYHA New York Heart Association
The evaluation of regurgitant valvular lesions should include a complete evaluation of symptoms, echocardiographic evaluation of regurgitation severity, left ventricular dimensions, and systolic function [6]. Ideally, valvular dysfunction should be assessed prior to pregnancy to establish the stability of the regurgitation over time, to counsel on maternal and fetal risk, and to initiate therapy appropriate for pregnancy. Patients with severely regurgitant valves are recommended against pregnancy prior to valve repair or replacement. In moderate-to-severe regurgitant valves, exercise testing is recommended to fully characterize the patient’s functional capacity, the hemodynamic consequences of the lesion, and evaluate for inducible pulmonary hypertension. Symptoms during pregnancy can typically be managed conservatively with judicious use of diuretics for congestive symptoms and afterload-reducing agents, such as hydralazine. Angiotensin Converting Enzyme (ACE) inhibitors and angiotensin receptor blockers, which are also afterload-reducing agents, are strictly contraindicated in pregnancy due to their teratogenicity.
In acute severe regurgitation refractory to therapy, surgery may be unavoidable. If the fetus is mature enough, delivery should occur prior to surgery. In patients with any valvular regurgitation, vaginal delivery is preferable, and symptomatic patients should receive regional anesthesia with epidural or spinal to minimize the cardiovascular consequences of catecholamine surges. In some cases, an assisted second stage of labor may be appropriate [1].
Congenital bicuspid aortic valves are often associated with an underlying aortopathy, and it is important to screen for concomitant aortic dilation and aneurysm. Likewise, there is an important link between bicuspid aortic valves and coarctation of the aorta, and individuals with bicuspid aortic valves should be screened for this condition as well. Approximately 50 % of patients with bicuspid aortic valve and aortic valve pathology (aortic stenosis or aortic regurgitation) have dilation of the ascending aorta [7]. Also, dilation is often maximal at the distal end of the ascending aorta, which cannot be adequately visualized with transthoracic echocardiography, and Magnetic Resonance Imaging (MRI) or Compted Tomography (CT) of the chest should be performed prior to conception. Pre-pregnancy surgery should be considered when the thoracic aortic diameter reaches 50 mm [8]. Immer et al. [9] found that increased aortic root dilation during pregnancy in patients with bicuspid aortic valve carries a significant risk of type A aortic dissection.
Our patient underwent a transthoracic echocardiogram, with visualization of the aortic root, which was mildly dilated. The entire ascending aorta, aortic arch, and descending aorta were not fully visualized. Dedicated MRI of the aorta without contrast administration (an optimal choice to decrease radiation exposure) showed stable dilation of the aortic root and no evidence of coarctation, thoracic aneurysm, or dissection. She underwent a normal pregnancy, with regularly scheduled follow-up with her cardiologist. In the setting of congenital bicuspid aortic valve disease, screening of first-degree relatives, including children, siblings, and parents, is recommended [8].
Case 2: Arrhythmia in Pregnancy
A 31-year-old woman presents to the emergency department at 35-weeks gestation with acute shortness of breath, chest pressure, and sudden onset of palpitations for the preceding 30 minutes. Her medical history is notable for a history of hyperthyroidism for which she received radioactive iodine ablation. She takes levothyroxine 75 mcg by mouth daily, which was recently increased in the first trimester. Six years ago, she had presented to the emergency department with thyrotoxicosis and a supraventricular tachycardia. Her initial vital signs demonstrate a heart rate of 164 beats per minute, blood pressure of 109/56 mmHg, and respiratory rate of 26 respirations per minute. An electrocardiogram is shown in Fig. 1. Laboratory values are not yet obtained. Carotid sinus pressure is applied at the bedside, which decreases the rate to 146 beats per minute (bpm) briefly. Adenosine 6 mg IV once is administered, with a two-second pause and termination of the arrhythmia with conversion to sinus rhythm at 83 bpm. Her shortness of breath and chest discomfort resolve. She is admitted for further monitoring on telemetry, but she remains in sinus rhythm for 24 hours (h) and is discharged home without medication changes. Twelve days later, she re-presents with recurrent symptoms and is found to have a supraventricular tachycardia at 157 bpm. She is given adenosine 6 mg IV once, with resolution of the tachycardia and reestablishment of sinus rhythm. During the preceding 10 days, she noted intermittent palpitations for 1–2 min before resolving either on their own or with vagal maneuvers. She is admitted for monitoring and started on digoxin 0.125 mg daily. She undergoes induction of labor on hospital day three, with placement of an epidural, and delivery of a healthy baby boy at 37 0/7 weeks gestation. On hospital day six, she undergoes an electrophysiology study and atrioventricular nodal reentrant tachycardia (AVNRT) ablation. She is discharged on hospital day seven in stable condition.
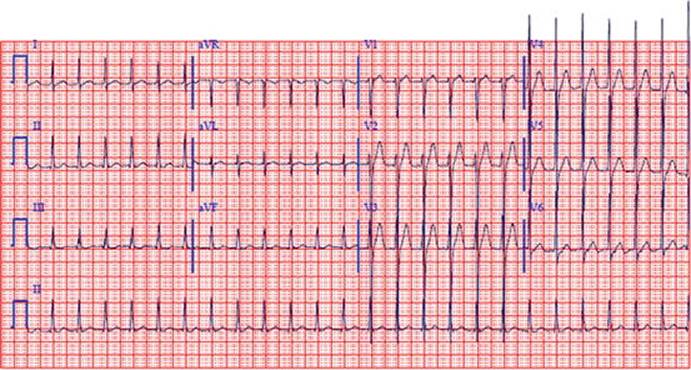
Fig. 1
ECG of supraventricular tachycardia
Premature extra beats and atrial tachyarrhythmias are more frequent and may even manifest for the first time, during pregnancy. The sensation of palpitations is common in pregnancy, with sinus tachycardia, premature atrial contractions, and premature ventricular complexes representing the most common findings [10, 11]. Premature beats manifest most frequently during the second trimester. They are not associated with adverse maternal or fetal outcomes and require treatment only if symptoms are intolerable to the mother. The occurrence of atrial fibrillation or atrial flutter is relatively rare during pregnancy and is usually associated with hyperthyroidism or structural cardiac disease [1], such as underlying valvular disease, congestive heart failure, or congenital heart disease.
AVNRT is the most common supraventricular arrhythmia in pregnant and nonpregnant women. In patients with a preexisting history of supraventricular tachycardia, the incidence of recurrence during pregnancy is as high as 20–44 % in case studies [12, 13]. Previously highly symptomatic tachyarrhythmias should be treated with catheter ablation prior to pregnancy, when possible. During pregnancy, as in our patient, most supraventricular arrhythmias represent a WHO class II risk category, with a small increase in maternal mortality and a moderate increase in morbidity.
Typically, supraventricular tachyarrhythmias are not well tolerated in pregnancy, if rapid and sustained. Thus, the general preference is to restore sinus rhythm. AVNRT or atrioventricular reentrant tachycardia (AVRT) involving an accessory pathway can often be terminated by successful vagal maneuvers, such as having the patient perform the Valsalva maneuver and cough vigorously or by applying carotid sinus pressure. If vagal maneuvers fail, adenosine is the first drug of choice and can be administered safely and intravenously in pregnancy for diagnostic and therapeutic purposes [13, 14]. Adenosine interrupts conduction down the accessory atrioventricular nodal pathway by prolonging the refractory period of the atrioventricular (AV) node and revealing the underlying atrial arrhythmia. Intravenous metoprolol can be used if adenosine fails and serves to slow the ventricular rate to control symptoms but will not typically convert the rhythm to sinus. Direct current synchronized cardioversion is also safe in all stages of pregnancy [13] and is used typically in situations of hemodynamic instability or arrhythmias refractory to medical therapy.
Digoxin is considered safe in pregnancy and can be used to control the ventricular rate but is not an effective medication to use in the acute setting [13]. Other agents, such as specific medications acting on the AV node (beta-blockers, non-dihydropyridine calcium channel blockers; most class C agents) (Table 5), may also be safe and tolerated during pregnancy to treat the symptoms of the arrhythmia. Antiarrhythmic drug therapies can be toxic to the developing fetus and should be discontinued prior to conception [13]. Major controlled studies of antiarrhythmic drugs during pregnancy are lacking. Typically, antiarrhythmic therapy is reserved for hemodynamic compromise or refractory or recurrent arrhythmia. Specific antiarrhythmic medications are considered safer to use in pregnancy, such as sotalol (class B), flecainide (class C), and ibutilide (class C) [13]. They have not demonstrated any adverse fetal effects, but experience with these medications in pregnancy is limited. Amiodarone is usually recommended against in pregnancy. It is a pregnancy class D medication and its use in pregnancy should be restricted to arrhythmias that are resistant to other drugs or are life threatening [15]. Bypass tract or atrioventricular nodal ablation is possible during pregnancy if necessary and is best performed during the second trimester when the fetus has undergone initial development and the mother and fetus can still be adequately shielded from radiation exposure.
Table 5
Federal drug administration pregnancy categories
|
Risk class |
Risk of medication in pregnancy |
|
A |
Adequate and well-controlled studies have failed to demonstrate a risk to the fetus in the first trimester of pregnancy (and there is no evidence of risk in later trimesters) |
|
B |
Animal reproduction studies have failed to demonstrate a risk to the fetus, and there are no adequate and well-controlled studies in pregnant women |
|
C |
Animal reproduction studies have shown an adverse effect on the fetus, and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks |
|
D |
There is positive evidence of human fetal risk based on adverse reaction data from investigational or marketing experience or studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks |
|
X |
Studies in animals or humans have demonstrated fetal abnormalities, and/or there is positive evidence of human fetal risk based on adverse reaction data from investigational or marketing experience, and the risks involved in use of the drug in pregnant women clearly outweigh potential benefits |
Case 3: Valvular Stenosis, Mitral Stenosis
A 28-year-old Indian American woman presents for evaluation at 8-weeks gestation with a recent diagnosis of rheumatic mitral stenosis (MS). This is her second pregnancy, and the first was an uncomplicated pregnancy with vaginal delivery and home birth three years ago with a midwife while living in India. She was noted to have a murmur on exam by her current midwife and referred for further evaluation. She underwent a transthoracic echocardiogram one week ago which demonstrated mild dilation of the left atrium and a characteristic rheumatic deformity of the mitral valve with severe mitral stenosis and a calculated mitral valve area of 1.3 cm2. Prior to her pregnancy, she did not experience dyspnea on exertion, orthopnea, paroxysmal nocturnal dyspnea, or peripheral edema. She does note occasional palpitations that she describes as “skipped beats” for the last four weeks, occurring approximately every other day. She does not exercise regularly and is able to care for her three-year-old son, cook, and clean the home without limitations.
On examination, her blood pressure is 110/64 mmHg and heart rate 64 beats per minute and regular. She is noted to have jugular venous distension at 10 cm of H2O and a 2/6 apical systolic murmur with a soft opening snap, followed by a 2/4 diastolic rumble. Her lung exam is clear, without rales. She has no evidence of peripheral edema. Her electrocardiogram shows normal sinus rhythm with no abnormalities. Her other medical problems include a history of hypothyroidism, for which she takes levothyroxine 37.5 mcg daily.
Four months later, at 24-weeks gestation, she presents to the emergency room with one week of dyspnea on minimal exertion. She is now experiencing mild shortness of breath at rest. Her vital signs show a blood pressure of 96/54 mmHg and pulse rate of 116 beats per minute. A chest x-ray shows bilateral perihilar infiltrates consistent with mild-to-moderate pulmonary congestion. Duplex of the lower extremity shows no evidence of deep venous thrombosis. Her laboratory values are notable for normal chemistry values, including a potassium of 4.6 mEq/L, a blood urea nitrogen of 11 mg/dL, a creatinine of 0.7 mg/dL, a troponin I of 0.01 ng/mL, and a N-Terminal pro-brain natrideretic peptude (NTproBNP) level of 2675 pg/mL. Her electrocardiogram shows an irregularly irregular narrow complex rhythm without organized atrial activity consistent with atrial fibrillation with a rapid ventricular response. She is admitted to the hospital and started on furosemide 40 mg IV daily with improvement in her symptoms. She is started on therapeutic low molecular weight heparin twice daily and metoprolol 25 mg orally twice daily and spontaneously converts to sinus rhythm on hospital day two. She is discharged from the hospital on day four on furosemide 20 mg oral daily, metoprolol succinate 25 mg once daily, and therapeutic low molecular weight heparin. At 37-week gestation, she undergoes a planned induction of labor with an interdisciplinary team. Low molecular weight heparin is discontinued 12 h prior to induction and anesthesia. An epidural is placed, and she receives an assisted second stage of labor with low forceps extraction and delivers a healthy baby girl. In the immediate postpartum period, she is restarted on therapeutic low molecular weight heparin 12 h after delivery. She notes increased shortness of breath at 18 h after delivery, and a chest radiograph reveals mild pulmonary vascular congestion. An EKG shows atrial fibrillation at 108 bpm. She is restarted on furosemide 20 mg IV daily with resolution of her symptoms.
Rheumatic heart disease is the most common etiology of mitral stenosis in reproductive-aged women. It remains a major problem in developing countries and is still seen in developed countries, especially in immigrant populations. In general, stenotic valves confer a higher risk to the pregnant patient than regurgitant valves, and left-sided valve diseases involving the mitral and aortic valves have a higher complication rate than right-sided valve lesions of the tricuspid and pulmonic valves [1–3, 16]. Physiologic changes during pregnancy can cause previously asymptomatic patients to become symptomatic, as the heart rate may increase by 25 % and blood volume can expand by 40–50 % during pregnancy, leading to a marked increase in stroke volume and cardiac output. In stenotic valve disease, these hemodynamic changes increase the transvalvular gradient and decrease the filling time of the left ventricle and, thus, increase left atrial pressures, resulting in pulmonary venous congestion, pulmonary edema, and congestive heart failure. The presence of atrial fibrillation with a rapid ventricular rate often causes further clinical deterioration. Additionally, the rapid increase in venous return during labor and delivery, partially related to autotransfusion of maternal blood into the circulation from uterine contractions and from relief of inferior vena cava compression from the gravid uterus, may cause a further surge in preload to the left atrium, which can stress the heart further.
Management of MS is related to the severity of stenosis, the presence of symptoms, and timing of diagnosis. Moderate or severe MS is typically poorly tolerated during pregnancy [16], even when the patient is previously asymptomatic. Two main predictors of adverse outcomes during pregnancy include mitral valve area < 1.5 cm2 and abnormal functional class prior to pregnancy. If diagnosed prior to pregnancy, patients with severe MS regardless of symptoms (valve area < 1 cm2) or moderate symptomatic stenosis (valve area 1–1.5 cm2) should be considered for percutaneous mitral balloon valvuloplasty (PMBV) or mitral valve replacement if PMBV is not appropriate. If a patient is pregnant and congestive heart failure cannot be managed with medical therapy, valvuloplasty can be performed resulting in a significant hemodynamic benefit and usually favorable pregnancy outcome [16, 17]. However, valvuloplasty-related complications including cardiac tamponade, systemic embolization, maternal arrhythmias, initiation of uterine contractions, fetal distress, and fetal loss have been reported, in addition to radiation exposure to the fetus [16]. Optimal management of the pregnant patient with MS is focused on reducing left atrial pressures with careful diuresis and improving left ventricular filling with heart rate control, typically using beta-blockers. Judicious diuresis is crucial to avoid hypotension and volume depletion which may result in decreased uteroplacental perfusion and intrauterine growth restriction.
With pressure and volume overload, mitral stenosis can lead to left atrial stretch and dilatation and place the patient at increased risk of atrial arrhythmias, the most common of which is atrial fibrillation. Patients with valvular atrial fibrillation or atrial flutter require systemic anticoagulation as they are more prone to thromboembolic events than the general population with atrial fibrillation. With any episode of atrial fibrillation, continuous anticoagulation is recommended throughout pregnancy and up to 12 weeks postpartum, when the hypercoagulable state of pregnancy resolves. It is important to screen patients with mitral stenosis for symptoms of atrial arrhythmias, which includes sensations of palpitations, skipped beats, or rapid heart rates. This patient is presenting in the second trimester when intravascular volume is highest with congestive heart failure and atrial fibrillation. Mitral stenosis represents at least a WHO class III risk classification, with significant increased risk of maternal mortality and severe morbidity. She requires immediate treatment with intravenous diuretics, medications for heart rate control, and admission for monitoring. She should be started on unfractionated heparin (UFH) continuously for therapeutic anticoagulation of valvular atrial fibrillation, due to her risk of intracardiac thrombus and systemic embolism. UFH does not cross the placenta and does not expose the fetus to anticoagulation, as does warfarin. Unlike warfarin, heparin does not confer teratogenic effects and is, therefore, considered safer for the fetus. Low molecular weight heparin (LMWH) may also be utilized, using a twice daily weight-based dosing schedule. During pregnancy the volume of distribution for LMWH is variable, and it is essential to monitor anti-factor Xa levels drawn four hours after administration to establish an appropriate dose. Appropriate levels of anti-Xa should range between 0.7 and 1.2 U/mL.
Most patients with MS with NYHA class I/II symptoms can safely undergo vaginal delivery. Patients with class III/IV symptoms or with pulmonary hypertension who do not undergo valvuloplasty should be considered for cesarean delivery [1]. In patients with symptomatic moderate-to-severe mitral stenosis, anesthesia with regional epidural is recommended with an assisted second stage of delivery to decrease pain, abrupt hemodynamic changes, and the adverse cardiovascular effects of repeated, prolonged Valsalva maneuver. Additionally, patients need close monitoring during the immediate postpartum period (12–24 h) when uterocaval venous obstruction is relieved, and sudden increased venous flow to the heart can result in pulmonary edema. Patients with rheumatic valvular disease who traditionally require antibiotic prophylaxis for dental procedures do not routinely require antibiotic prophylaxis for vaginal or cesarean delivery, as long as infection is not suspected and aseptic measures are followed [6, 18].
Case 4: Prosthetic Valve
A 26-year-old woman with a history of intravenous drug abuse and infective aortic valve endocarditis requiring bioprosthetic aortic valve replacement six years prior presents at 7-weeks gestation with her second child. She denies intravenous drugs abuse since prior to the surgery. Her most recent transthoracic echocardiogram three months ago demonstrated a well-seated and normally functioning bioprosthetic aortic valve with mild perivalvular regurgitation and normal transvalvular gradients and otherwise normal left ventricular function and left ventricular cavity size. She denies symptoms of dyspnea, orthopnea, paroxysmal nocturnal dyspnea, palpitations, or chest pain. She is reassured that she does not need to start any new medications, and she is told to continue aspirin 81 mg daily that she is taking for prevention of bioprosthetic valve thrombosis. She has an uneventful pregnancy course and follows up during her second and third trimester with her cardiologist without concerns for clinical deterioration. She delivers a healthy baby boy at 39 and 3/7 weeks.
The selection of artificial valves in women of childbearing age is controversial. Typically, most agree that selection of bioprosthetic valves is safer for mother and child during this period, although their use is associated with an increased risk of structural degeneration in younger patients, which also may be accelerated by pregnancy [19]. In general, patient with bioprosthetic valves will, on average, require repeat valve replacement in 10–15 years after initial placement due to degeneration. This exposes patients to repeated surgical risk associated with open-heart surgery versus the advantage of valve longevity attributed to mechanical valves. However, mechanical valves and their complicated anticoagulation requirements confer increased maternal morbidity and mortality with the possibility of valve dysfunction, valve thrombosis and heart failure, and a higher risk of fetal complications, including fetal demise. The risk of valvular complications during pregnancy depends on the type and age of the valve, position of the valve (i.e., mitral vs. aortic), left ventricular function, and presence of atrial arrhythmias. In patients with well-functioning bioprosthetic valves, the management is similar to that in patients with native valves. There is no indication for systemic anticoagulation in patients with bioprosthetic valves, but aspirin, which is recommended for all prosthetic valves, may be continued in the setting of pregnancy [6]. Patients with bioprosthetic valves should be educated about the possibility of valve dysfunction and should be monitored for signs and symptoms of this. A baseline echocardiogram prior to pregnancy is important to assess the function of the prosthetic valve, including transvalvular gradient; the presence of valvular regurgitation, in addition to evaluating for pulmonary hypertension; ventricular cavity sizes and systolic function; and hemodynamics. In this patient, she has evidence of mild valve degeneration, with mild thickening and regurgitation of the valve. With mild valve dysfunction and normal left ventricular function and no symptoms of congestive heart failure, she is at minimal (WHO class I) risk during this pregnancy. Additionally, according to the American Heart Association, antibiotic prophylaxis in patients with a prior history of endocarditis or with biologic or mechanical prosthetic valves who are undergoing uncomplicated vaginal delivery or cesarean section is considered optional, as these patients are considered high risk. Many practitioners have a tendency to give antibiotics prior to delivery in high-risk patients [18].
Over the next 5–8 years, however, this patient may become symptomatic from aortic valve dysfunction. At that point, she may require a repeat sternotomy and valve replacement, and she will still be of reproductive age. Prior to this time, it will be important to establish with this patient her desire for future childbearing. She should be counseled regarding the surgical risk of repeat valve replacement, and there should be consideration for replacement with a mechanical aortic valve during her next surgery due to its longevity. Pregnancy is generally considered high risk in patients with mechanical valves, as management of adequate therapeutic anticoagulation is challenging, and it puts the mother and fetus at risk of complications. One of the reasons for this complication rate includes the fact that pregnancy is a prothrombotic state, and patients with prosthetic mechanical valves are at a higher risk of valve thrombosis throughout pregnancy. In a large review, this risk was found to be 3.9 % with warfarin throughout pregnancy, 9.2 % when unfractionated heparin was used in the first trimester and warfarin in the second and third trimester, and 33 % when unfractionated heparin was used throughout pregnancy. Maternal death complicated these groups in 2 %, 4 %, and 15 % and was usually related to valve thrombosis [10]. According to the American College of Cardiology (ACC) American Heart Associtaion (AHA) guidelines, all pregnant patients with mechanical prosthetic valves must receive continuous therapeutic anticoagulation throughout pregnancy [6].
The need for anticoagulation must be weighed with the risk of fetal complications related to anticoagulation. Warfarin is the anticoagulant of choice for mechanical valves, but it freely crosses the placental barrier. It can adversely affect fetal development, particularly in early pregnancy, and increases the risk of serious bleeding in later stages of development. Fetal embryopathy syndrome, characterized by cognitive impairment, optic atrophy, and fetal bone and cartilage malformations, is highest during the 6th through 12th weeks of gestation [1]. Some patients may elect to hold warfarin between these developmental weeks with transition to dose-adjusted UFH or LMWH, which does not cross the blood-placental barrier. Others may elect to use heparin throughout the pregnancy, decreasing fetal risk but increasing maternal risk of prosthetic valve thrombosis, systemic embolism, and heparin-induced thrombocytopenia. After 36 weeks, however, warfarin should be stopped in all pregnant patients, with transition to UFH or LMWH prior to planned delivery and regional anesthesia.
Case 5: Peripartum Cardiomyopathy
A 33-year-old woman of Ghanaian descent presents in labor at 37 and 4/7 weeks with a twin gestation. At 42 h, she is taken to cesarean section delivery for failure to progress. Cesarean section is complicated by uterine atony and postpartum hemorrhage requiring three units packed red blood cells and two liters of crystalloid for hemodynamic stabilization with application of Bakri balloon and eventually emergent uterine artery embolization. Hematocrit drops to 21.3 % from 37.6 % and stabilizes at 28.2 % after transfusion. The twin baby boys are healthy with Apgar scores of 8 at 5 min after delivery. At 36 h after delivery, she develops increasing shortness of breath at rest and a new oxygen requirement. Her physical exam reveals bilateral rales at the lung bases, without wheezing, and 2+ bilateral lower extremity edema to just below the knees. A chest x-ray demonstrates bilateral perihilar infiltrates consistent with mild pulmonary edema and small left-sided pleural effusion. A proBNP level is 2,579 pg/mL. A transthoracic echocardiogram is performed on postpartum day 2 and shows left ventricular cavity dilation of 5.9 cm (normal < 5.3 cm) and reduced global left ventricular systolic function with an ejection fraction of 35 %. She is admitted to the Intersive Care Unit (ICU) and started on IV furosemide boluses with clinical improvement in her symptoms.
Peripartum cardiomyopathy (PPCM) is an idiopathic cardiomyopathy that can occur from the last month of pregnancy up until five months postpartum and is characterized by left ventricular dysfunction developing during this time frame in the absence of any identifiable or preexisting cause of heart failure. The incidence of PPCM occurs in up to one in 3000 to 4000 births, with variation based on genetic and cultural factors [20]. It is suspected to be the consequence of an imbalance of oxidative stress in which infections, inflammation, and autoimmune processes play a role. Predisposing factors include maternal age greater than 30 years; multiparity and multiple childbirths; history of preeclampsia, eclampsia, or postpartum hypertension; ethnicity (particularly of African descent); low socioeconomic status; or tocolytic therapy with beta-agonists [1]. Heart failure in PPCM is manifested by symptoms such as fatigue, dyspnea, nonspecific chest pain, abdominal distension, symmetric peripheral edema, orthopnea, or paroxysmal nocturnal dyspnea. Transthoracic echocardiography is the appropriate diagnostic test of choice for assessing left ventricular function when PPCM is suspected.
Standard medical management of pregnant patients with decompensated heart failure includes oxygen, diuretics, and vasodilators, such as nitroglycerin or hydralazine. Diuretics should be used judiciously to manage symptomatic shortness of breath and pulmonary congestion, as they can cause placental hypoperfusion. Angiotensin-converting-enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs), which are a standard of care in systolic heart failure, are contraindicated in pregnant patients due to fetal teratogenicity (class D) and should not be initiated until after delivery. When ACE inhibitors are needed during breastfeeding, benazepril, captopril, and enalapril are preferred due to reduced excretion in breast milk compared to other agents [21]. Beta-1 selective beta-blockers (most class C) are indicated in patients with heart failure but should not be initiated until the patient is hemodynamically stabilized and her volume status has been optimized. Anticoagulation should be considered in patients with very low ejection fraction <30 % and is recommended in patients with intracardiac thrombus or evidence of systemic embolism.
The prognosis after PPCM is variable; when compared to other etiologies of cardiomyopathy, PPCM generally has a more favorable survival rate [22]. Approximately 50 % of women completely recover normal heart size and function, usually within six months of delivery, but ejection fraction can continue to improve up to two years postpartum. Prognosis is more favorable when the ejection fraction at initial diagnosis is >30 %. The remainder of patients either experience persistent stable left ventricular dysfunction or continue to experience clinical deterioration over time. Predictors of poor prognosis include those with an initial left ventricular ejection fraction <30 % at diagnosis, echocardiographic left ventricular cavity dilation >6 cm, or elevated troponin at diagnosis [23].
Women with a history of peripartum cardiomyopathy should be risk stratified by a cardiologist prior to planning future pregnancies. Subsequent pregnancies carry a recurrence risk of peripartum cardiomyopathy up to 30–50 % [1]. As in this patient, women with a history of peripartum cardiomyopathy and a persistently low ejection fraction are considered modified WHO criteria class IV with respect to future pregnancies. If pregnancy occurs in patients with LVEF less than 40 %, this is a predictor of high-risk maternal and fetal complications, and these patients should consider termination, especially if symptomatic at baseline assessment. If asymptomatic, the pregnancy is still considered high risk, and these patients should be monitored monthly with a tertiary care interdisciplinary team of high-risk obstetricians, anesthesiology, and cardiologists trained in caring for pregnant patients. If LVEF is less than 20 %, maternal risk, including that of death, may be high, and termination of pregnancy should be advised.
Case 6: Myocardial Infarction in Pregnancy
A 42-year-old woman presents to the emergency department at 34 and 1/7 weeks gestation with severe substernal chest pressure associated with nausea. She has a medical history significant for hypercholesterolemia and type I (juvenile onset) diabetes mellitus. She is an executive at a public relations firm and has recently been under stress preparing for a quarterly review. She was in a board meeting today when she had sudden onset chest discomfort at an intensity of 4/10 for the last hour that escalated to 6/10 and was associated with pain radiating down her left arm. A co-worker accompanied her to the emergency department due to ongoing pain. She also has noted contractions for the last hour that are increasing in frequency and severity. Nitroglycerin is administered sublingually, and the chest pain decreases from 7/10 to 3/10. Her initial electrocardiogram (EC6) is shown in Fig. 2. An ST-elevation myocardial infarction (STEMI) is suspected, and she is taken emergently to the catheterization lab. She is given an intravenous heparin bolus and clopidogrel 600 mg once. An acute 99 % proximal plaque thrombosis of the left anterior descending artery is diagnosed during coronary angiography, and a bare metal stent is placed with good angiographic result. Her chest pain post-procedure has resolved, and ST segment elevations have returned almost to baseline on a repeat electrocardiogram. Her uterine contractions have become less frequent. She is admitted to the critical care unit for monitoring for 48 h and eventually discharged in stable condition on aspirin 81 mg daily and clopidogrel 75 mg daily. Three weeks later, at 37-weeks gestation, she is instructed to discontinue clopidogrel in anticipation of delivery. She is induced at 37 and 5/7 weeks gestation with placement of an epidural and delivers a healthy baby girl via vaginal delivery after 15 h of labor.
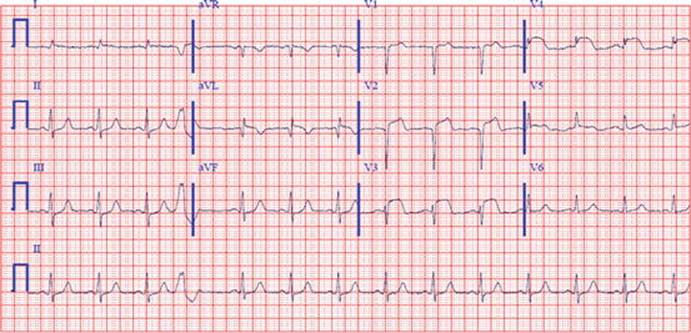
Fig. 2
ECG of supraventricular tachycardia infarction
The incidence of acute myocardial infarction (MI) in pregnancy is rare, complicating 3–6 out of every 100,000 pregnancies [24, 25]. While this is an uncommon event in childbearing women, the risk increases three to four times in pregnancy due to increased hypercoagulability, higher myocardial oxygen demand related to increased cardiac output, and decreased oxygen supply with anemia and decreased diastolic blood pressures. Additionally, as women become pregnant later in life with increasing frequency and comorbid conditions such as diabetes mellitus and tobacco abuse, the risk of coronary artery disease increases. The risk of myocardial infarction is also strongly related to major cardiovascular risk factors, such as hypertension, hyperlipidemia, older age, and a positive family history of extensive or early coronary artery disease. Though relatively uncommon, ischemic heart disease is the most common cause of cardiac disease leading to maternal death [26].
Ischemic heart disease can be caused by many different pathologic processes. Coronary artery vasospasm and coronary dissection more frequently result in acute coronary syndrome during pregnancy than the general population; however, classic obstructive atherosclerosis with plaque rupture or fissure with overlying thrombosis is still the leading cause [24]. Pregnant women at higher risk include older patients in their third trimester, who have had multiple prior pregnancies. Other conditions that contribute risk include the presence of preeclampsia or eclampsia, thrombophilia, postpartum infections, and severe postpartum hemorrhage [24, 25]. The diagnosis of acute coronary syndrome is the same in pregnant patients as nonpregnant patients and consists of the clinical history, electrocardiographic changes, and cardiac biomarkers. The diagnosis of a myocardial infarction is often delayed, as more subtle complaints can be attributed to pregnancy-related symptoms. For non-ST-elevation MI (NSTEMI), troponin levels are superior for diagnosis during labor and delivery, since CK and CK-MB enzymes are increased twofold within 30 min of delivery. However, severe postpartum hemorrhage with shock may also lead to elevated troponin levels with ischemic ECG changes and transient left ventricular wall motion abnormalities. STEMI is a clinical diagnosis based on symptoms and electrocardiographic changes and should not be delayed to wait for cardiac biomarkers to be processed, which can be normal in the acute setting.
Treatment of acute coronary syndrome may be modified in pregnancy to avoid added risk. In general, aspirin, nitroglycerin, beta-blockers, and heparin are considered safe in pregnancy. Statins, ACE inhibitors, and ARBs are contraindicated during pregnancy, and there is no established data for P2Y12 (i.e., clopidogrel, prasugrel, ticagrelor) or glycoprotein IIb/IIIa inhibitors (i.e., abciximab, eptifibatide, tirofiban), though P2Y12 inhibitors are used if necessary (e.g., the presence of an aspirin allergy or if stenting occurs). Emergent coronary angiography should be performed if primary percutaneous coronary intervention is anticipated, such as during an STEMI or if there is refractory ischemia despite optimal medical therapy. Alternatively, medical management may be considered in appropriate patients with an NSTEMI or unstable angina who remain asymptomatic and stable following initiation of therapy. Ideally, the myocardium should have two to three weeks to recover after myocardial injury prior to labor and delivery. In addition, if used, P2Y12 inhibitors need to be continued uninterrupted for at least three weeks after stent implantation and need to be discontinued five days prior to delivery in order to safely administer regional anesthesia, which assists with minimizing oxygen demand on the heart muscle during labor and delivery.
References
1.
Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C, Cifkova R, Ferreira R, Foidart JM, et al. ESC Guidelines on the management of cardiovascular diseases during pregnancy: the Task Force on the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur Heart J. 2011;32(24):3147–97.CrossRefPubMed
2.
Siu SC, Sermer M, Colman JM, Alvarez AN, Mercier LA, Morton BC, et al. Prospective multicenter study of pregnancy outcomes in women with heart disease. Circulation. 2001;104(5):515–21.CrossRefPubMed
3.
Drenthen W, Boersma E, Balci A, Moons P, Roos-Hesselink JW, Mulder BJ, et al. Predictors of pregnancy complications in women with congenital heart disease. Eur Heart J. 2010;31(17):2124–32.CrossRefPubMed
4.
Balci A, Sollie-Szarynska KM, van der Bijl AG, Ruys TP, Mulder BJ, Roos-Hesselink JW, et al. Prospective validation and assessment of cardiovascular and offspring risk models for pregnant women with congenital heart disease. Heart. 2014;100(17):1373–81.CrossRefPubMed
5.
Thorne S, MacGregor A, Nelson-Piercy C. Risks of contraception and pregnancy in heart disease. Heart. 2006;92(10):1520–5.CrossRefPubMedPubMedCentral
6.
Vahanian A, Baumgartner H, Bax J, Butchart E, Dion R, Filippatos G, et al. Guidelines on the management of valvular heart disease: The Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology. Eur Heart J. 2007;28(2):230–68.PubMed
7.
Fedak PW, de Sa MP, Verma S, Nili N, Kazemian P, Butany J, et al. Vascular matrix remodeling in patients with bicuspid aortic valve malformations: implications for aortic dilatation. J Thorac Cardiovasc Surg. 2003;126(3):797–806.CrossRefPubMed
8.
Baumgartner H, Bonhoeffer P, De Groot NM, de Haan F, Deanfield JE, Galie N, et al. ESC Guidelines for the management of grown-up congenital heart disease (new version 2010). Eur Heart J. 2010;31(23):2915–57.CrossRefPubMed
9.
Immer FF, Bansi AG, Immer-Bansi AS, McDougall J, Zehr KJ, Schaff HV, et al. Aortic dissection in pregnancy: analysis of risk factors and outcome. Ann Thorac Surg. 2003;76(1):309–14.CrossRefPubMed
10.
Chan WS, Anand S, Ginsberg JS. Anticoagulation of pregnant women with mechanical heart valves: a systematic review of the literature. Arch Intern Med. 2000;160(2):191–6.CrossRefPubMed
11.
Cruz MO, Hibbard JU, Alexander T, Briller J. Ambulatory arrhythmia monitoring in pregnant patients with palpitations. Am J Perinatol. 2013;30(1):53–8.PubMed
12.
Silversides CK, Harris L, Haberer K, Sermer M, Colman JM, Siu SC. Recurrence rates of arrhythmias during pregnancy in women with previous tachyarrhythmia and impact on fetal and neonatal outcomes. Am J Cardiol. 2006;97(8):1206–12.CrossRefPubMed
13.
Blomstrom-Lundqvist C, Scheinman MM, Aliot EM, Alpert JS, Calkins H, Camm AJ, et al. ACC/AHA/ESC guidelines for the management of patients with supraventricular arrhythmias – executive summary. a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients with Supraventricular Arrhythmias) developed in collaboration with NASPE-Heart Rhythm Society. J Am Coll Cardiol. 2003;42(8):1493–531.CrossRefPubMed
14.
Elkayam U, Goodwin TM. Adenosine therapy for supraventricular tachycardia during pregnancy. Am J Cardiol. 1995;75(7):521–3.CrossRefPubMed
15.
Bartalena L, Bogazzi F, Braverman LE, Martino E. Effects of amiodarone administration during pregnancy on neonatal thyroid function and subsequent neurodevelopment. J Endocrinol Invest. 2001;24(2):116–30.CrossRefPubMed
16.
Hameed A, Karaalp IS, Tummala PP, Wani OR, Canetti M, Akhter MW, et al. The effect of valvular heart disease on maternal and fetal outcome of pregnancy. J Am Coll Cardiol. 2001;37(3):893–9.CrossRefPubMed
17.
Ben Farhat M, Gamra H, Betbout F, Maatouk F, Jarrar M, Addad F, et al. Percutaneous balloon mitral commissurotomy during pregnancy. Heart. 1997;77(6):564–7.CrossRefPubMedPubMedCentral
18.
Wilson W, Taubert KA, Gewitz M, Lockhart PB, Baddour LM, Levison M, et al. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007;116(15):1736–54.CrossRefPubMed
19.
Elkayam U, Bitar F. Valvular heart disease and pregnancy: part II: prosthetic valves. J Am Coll Cardiol. 2005;46(3):403–10.CrossRefPubMed
20.
Sliwa K, Hilfiker-Kleiner D, Petrie MC, Mebazaa A, Pieske B, Buchmann E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of peripartum cardiomyopathy: a position statement from the Heart Failure Association of the European Society of Cardiology Working Group on peripartum cardiomyopathy. Eur J Heart Fail. 2010;12(8):767–78.CrossRefPubMed
21.
Beardmore KS, Morris JM, Gallery ED. Excretion of antihypertensive medication into human breast milk: a systematic review. Hypertens Pregnancy. 2002;21(1):85–95.CrossRefPubMed
22.
Felker GM, Thompson RE, Hare JM, Hruban RH, Clemetson DE, Howard DL, et al. Underlying causes and long-term survival in patients with initially unexplained cardiomyopathy. N Engl J Med. 2000;342(15):1077–84.CrossRefPubMed
23.
Fett JD, Christie LG, Carraway RD, Murphy JG. Five-year prospective study of the incidence and prognosis of peripartum cardiomyopathy at a single institution. Mayo Clin Proc. 2005;80(12):1602–6.CrossRefPubMed
24.
Roth A, Elkayam U. Acute myocardial infarction associated with pregnancy. J Am Coll Cardiol. 2008;52(3):171–80.CrossRefPubMed
25.
James AH, Jamison MG, Biswas MS, Brancazio LR, Swamy GK, Myers ER. Acute myocardial infarction in pregnancy: a United States population-based study. Circulation. 2006;113(12):1564–71.CrossRefPubMed
26.
Roos-Hesselink JW, Duvekot JJ, Thorne SA. Pregnancy in high risk cardiac conditions. Heart. 2009;95(8):680–6.CrossRefPubMed