Wendy L. Wahl
Presentation
A 57-year-old man with multiple medical problems presents to the emergency department feeling lightheaded with hematemesis and melena. His medical history includes end-stage renal disease and bladder cancer, for which he recently underwent cystectomy with an ileal conduit. He is hypotensive, and placement of a nasogastric tube yielded bright red blood clots. He is currently on hemodialysis three times each week and has no prior history of ulcers. He does not take nonsteroidals or aspirin on a regular basis. Large-bore intravenous access is established and his blood pressure improves with fluid resuscitation. His abdomen is mildly distended but nontender on palpation. He has a healing midline abdominal incision with a pink ileal conduit with minimal dark-appearing urine. Digital rectal examination reveals dark, tarry stool, which is guaiac positive.
Differential Diagnosis
His recent major surgery, and the attendant lack of oral intake, place him at risk for stress gastritis or peptic ulcer disease. His history of renal failure may also make him more likely to have arterial-venous malformations or bleeding from gastritis from platelet dysfunction. He could also have esophageal disease such as a Mallory-Weiss tear from vomiting and, less likely, variceal hemorrhage since he provides no history of cirrhosis. But with his history of hemodialysis and renal failure, he does have a higher risk of hepatitis. Less likely in this scenario, given the acute onset, would be upper gastrointestinal tract neoplasms such as esophageal, gastric, or duodenal tumors.
Workup
At this point, if a nasogastric tube were not in place, one should be placed for gastric lavage and evacuation of the stomach. Given his history of hematemesis, an esophagogastroduodenoscopy (EGD) would be the next diagnostic and potentially therapeutic procedure of choice. If there is any concern over perforation in addition to bleeding, an abdominal radiograph should be performed prior to EGD. Laboratory tests that should be drawn to establish baseline values would be a complete CBC with platelets, PT with INR, PTT, type and screen, a comprehensive metabolic panel to assess for electrolyte abnormalities. An arterial blood gas should be drawn to evaluate for metabolic acidosis due to underresuscitation.
Diagnosis and Treatment
In severely ill patients, endotracheal intubation should be considered prior to EGD, especially in patients who are in shock or unable to protect their airway. This patient is awake and alert and has normal vital signs after fluid resuscitation. An EGD is performed, which reveals a large clot and active bleeding in the first portion of the posterior duodenum (see Figure 1).

FIGURE 1 • Endoscopic view of bleeding duodenal ulcer.
This patient is at high risk for rebleeding (active bleeding during endoscopy—90% chance of recurrence, visible vessel—50% chance of rebleeding, adherent clot—25% to 30% chance of recurrence). Despite this risk, attempts at endoscopic control of the bleeding should be made. Recent experience suggests that removing the clot in order to treat the underlying ulcer can reduce the risk of rebleeding. Studies show that epinephrine injection alone is inferior to combined therapies with epinephrine injection and thermal coagulation or placement of a hemoclip.
Administration of proton pump inhibitors (PPIs) has been shown to reduce the risk of rebleeding. Most of these studies used intravenous omeprazole, although other PPIs can serve as reasonable alternatives. No studies have shown that high-dose infusions are more effective than routine intravenous doses of PPIs. In addition to PPIs, any potentially precipitating medications such as aspirin or nonsteroidal anti-inflammatory agents should be discontinued. The patient should also be evaluated with gastric biopsies for the presence of Helicobacter pylori as a precipitating factor for ulcer formation.
Presentation Continued
The patient stabilizes after a total of four units of PRBCs. Three days after his initial EGD he develops recurrent hematemesis with an associated drop in his blood pressure. Blood transfusion and fluids are started.
Discussion
Based on a controlled trial that assessed efficacy of endoscopic therapy compared to surgery for recurrent bleeding, patients who had a second therapeutic endoscopy had fewer complications than those who underwent operative intervention, although mortality was equivalent. Given that this patient has had a recent midline incision and has not failed a second endoscopic attempt, a repeat EGD is indicated. In our case, at his second EGD, the endoscopist is able to slow the bleeding down with epinephrine injections but cannot reliably use cautery or other methods for hemostasis.
Although endoscopy is the best initial diagnostic and therapeutic procedure, failure to achieve hemostasis leaves surgery and transcatheter arterial intervention (TAI) as possible therapeutic modalities. At this point, indications for surgery for active rebleeding from a duodenal ulcer include hemodynamic instability or shock despite blood transfusion, rebleeding after two attempts at endoscopic therapy, or continued slow bleeding requiring blood transfusion exceeding more than three units per day. In our case, after discussion with the family and patient, they do not wish to pursue surgery at this time and opt for TAI. Active bleeding is identified (Figure 2A), and successful coil embolization of the gastroduodenal artery (GDA) is performed and the bleeding has stopped (Figure 2B). On the day of discharge, the patient has recurrent, large-volume hematemesis with hemodynamic instability. At this point, the patient has failed two endoscopic therapies and angiographic embolization. Surgical intervention is warranted. Depending on institutional resources, this may have been the selected therapeutic option after the second failed endoscopy.
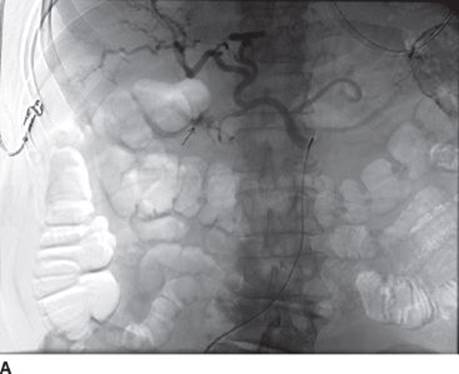
FIGURE 2A • Active bleeding during angiography.

FIGURE 2B • Cessation of bleeding after embolization coils.
Surgical Approach
Much of the literature surrounding choices for operative management for complicated peptic ulcer disease heralds from the era that predated the use of H2-blockers and PPI therapy. Controversy exists as to whether a definitive ulcer operation is now required, particularly for perforated peptic ulcers. The priority for patients undergoing operation for a bleeding duodenal ulcer is to control the bleeding. If the exact site of the bleeding is not known, a longitudinal pyloro-duodenotomy allows for inspection of the duodenal bulb and the gastric antrum. Otherwise, the proximal duodenum can be incised directly over the ulcer site. Suture of the bleeding vessel in the base of the ulcer can be performed with sutures placed superiorly and inferiorly to the ulcer crater (Figure 3). If bleeding continues, the GDA can be ligated superiorly to the duodenum. While placing the sutures, the surgeon must be aware that the common bile duct courses posteriorly, and if the location cannot be determined, a Fogarty or similar biliary catheter should be inserted into the duct to avoid injury. After achieving hemostasis, attempts to approximate the ulcer crater should be made. The longitudinal pyloroduodenotomy should then be closed transversely to avoid narrowing of the pylorus and duodenum (Heineke-Mikulicz pyloroplasty).

FIGURE 3 • Duodenotomy incision.
After control of hemorrhage from the GDA, an acid-reduction procedure is generally recommended for patients with a history of refractory peptic ulcer disease (i.e., a history of long-standing PPI use or noncompliance). The type of procedure is dependent on patient stability and surgeon experience. The most expeditious procedure is a truncal vagotomy; however, if the patient has stabilized and the surgeon is experienced, a parietal cell vagotomy (highly selective vagotomy) is a reasonable alternative. Maneuvers for truncal vagotomy include retracting the left lobe of the liver for exposure. The serosa overlying the esophagus is opened and using a combination of blunt and sharp dissection, the esophagus is encircled with a Penrose drain. By retracting the esophagus caudally and palpating, the firm “violin string” of the vagus nerve is felt. The anterior vagus is easier to find and larger. If the posterior vagus is not felt, it may not be included in the Penrose drain contents, and the search should continue. After identification, the nerves should be clipped proximally and distally and a small segment excised.
Case Conclusion
The patient undergoes exploratory laparotomy, pyloroduodenotomy, and oversewing of a posterior duodenal bulb ulcer (Table 1). Given his hemodynamic instability, and the fact that he has not previously been treated with PPIs, an acid reduction procedure is not performed. He recovers well from surgery without further bleeding. Gastric biopsies from his EGD return H. pylori positive. He is sent home on PPIs and H. pylori treatment.
TABLE 1. Key Technical Steps to Maneuvers in Oversewing a Bleeding Duodenal Ulcer

TAKE HOME POINTS
· Ensure adequate intravenous access and an adequate airway prior to any procedure.
· EGD is the most important assessment tool since it can be both diagnostic and therapeutic.
· Treat patients with a PPI and evaluate for H. pylori infection.
· EGD is a reasonable approach for patients who rebleed and are not profoundly unstable.
· Angiographic embolization is an option for patients who fail endoscopic therapy or have contraindications to surgery, but this option must be readily available.
· The operative approach should focus on cessation of bleeding followed by an acid reduction procedure, if appropriate.
SELECTED READINGS
Bleau BL, Gostout CJ, Sherman KE, et al. Recurrent bleeding from peptic ulcer associated with adherent clot: a randomized study comparing endoscopic treatment with medical therapy. Gastrointest Endosc. 2002;56:1–6.
Elmunzer BJ, Young SD, Inadomi JM, et al. Systematic review of the predictors of recurrent hemorrhage after endoscopic hemostatic therapy for bleeding peptic ulcers. Am J Gastroenterol. 2008;103:2625–2632.
Katschinski B, Logan R, Davies J, et al. Prognostic factors in upper gastrointestinal bleeding. Dig Dis Sci. 1994;39:706–712.
Lau JY, Sung JJ, Lam YH, et al. Endoscopic retreatment compared with surgery in patients with recurrent bleeding after initial endoscopic control of bleeding ulcers. N Engl J Med. 1999;340:751–756.
Lau JY, Sung JJ, Lee KK, et al. Effect of intravenous omeprazole on recurrent bleeding after endoscopic treatment of bleeding peptic ulcers. N Engl J Med. 2000;343:310–316.
Leontiadis GI, Sharma VK, Howden CW. Systematic review and meta-analysis of proton pump inhibitor therapy in peptic ulcer bleeding. BMJ. 2005;330:568.
Marmo R, Rotondano G, Piscopo R, et al. Dual therapy versus monotherapy in the endoscopic treatment of high-risk bleeding ulcers: a meta-analysis of controlled trials. Am J Gastroenterol. 2007;102:279–289.
Vergara M, Calvet X, Gisbert JP. Epinephrine injection versus epinephrine injection and a second endoscopic method in high risk bleeding ulcers. Cochrane Database Syst Rev. 2007;CD005584.
Zittel TT, Jehle EC, Becker HD. Surgical management of peptic ulcer disease today–Indication, technique and outcome. Langenbecks Arch Surg. 2000;385:84–96.