Guy J. Petruzzelli
INTRODUCTION
Due to the location and anatomic proximity to the central cranial–facial skeleton, the temporalis muscle and the associated fasciae have been studied extensively in the reconstruction of defects resulting from head and neck oncologic surgery, trauma, and skull base surgery. The flaps derived from this region include the temporal–parietal fascial flap, the temporalis muscle flap, the temporalis muscle calvarium osteomuscular flap, and the temporalis muscle pericranial flap. Each of the flaps has particular indications and utilities as they relate to reconstruction of malar, palatal–maxillary, auricular, temporal bone, intraoral, and cranial base defects. Although it lacks bulk, the thin and pliable temporal–parietal fascial flap can be used both as a pedicle flap and a microvascular free tissue transfer for limited soft tissue augmentation and as a scaffold for skin and/or cartilage grafts. Primarily used in auricular reconstruction, the temporal–parietal fascial flap has also been applied to the reconstruction of limited intraoral and pharyngeal mucosal defects, facial skin and contour defects, and orbital exenteration defects and obliteration of the mastoid cavity. The composite temporalis muscle-split calvarium flap has been used for the reconstruction of congenital, post-traumatic, and oncologic defects of the palate, orbital floor, and lateral orbital rim. The application of this flap is limited by the amount of bone that can be safely harvested and the availability of more reliable alloplastic material for orbital reconstruction. The temporalis muscle proper is broad and flat and fills the lateral (temporal) region of the head (Fig. 48.1). The consistently reliable bulk and arc of rotation of the temporalis muscle proper have led to its multiple applications in reconstruction of palatal, retromolar trigone, orbital exenteration, lateral temporal bone resection, and skull base defects. Additionally, the temporalis muscle has been used in the dynamic reanimation of the paralyzed face. This chapter will be limited to the applications of the temporalis muscle as a rotation flap for reconstruction of the central and paracentral skull base.
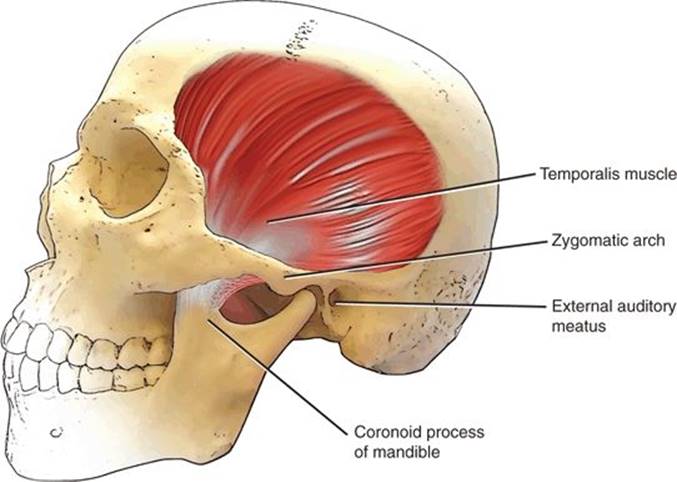
FIGURE 48.1 Diagram of the skull indicating the anatomic location of the temporalis muscle.
HISTORY
The temporalis muscle flap is not reliable in the setting of salvage cranial base surgery. The reduced muscle bulk and tenuous blood supply following prior surgery or radiation therapy make the muscle unsuitable in these situations. Therefore, patients should be carefully questioned regarding prior (1) surgical procedure for resection or attempted resection of cranial base tumors, (2) prior chemotherapy or radiation therapy, and (3) prior treatment for intractable epistaxis, which may have required embolization of the internal maxillary artery. In situations of reoperative or salvage surgery, reconstruction with microvascular free tissue transfer from another site should be considered.
Patients should also be questioned regarding prior trauma to the craniofacial skeleton, which may have altered the position of the zygoma or mandible.
PHYSICAL EXAMINATION
The physical examination should be directed at determining the size of the temporalis muscle with particular attention to temporal wasting indicating denervation atrophy. The temporal fossa should be palpated in the neutral position and with the teeth clenched to assess muscle bulk. The range of motion of the mandible should also be determined. Trismus may indicate fibrosis or scarring of the temporalis muscle further limiting its size or arc of rotation into the cranial base defect.
INDICATIONS
The principal indicator for the use of the temporalis muscle flap in cranial base surgery is the reconstruction of the floor of the central skull base following anterior–lateral skull base surgery with subtemporal craniectomy. Resection of neoplasms of the anterior and anterior–lateral cranial base, particularly those requiring a transcranial approach with subtemporal craniotomy, results in exposure of the temporal dura. The removal of bone and soft tissue required by either the surgical approach or the extirpation of the tumor may result in a surgical defect for which primary closure is not possible. Vascularized soft tissue augmentation of the resulting defect is necessary to (1) restore the anatomic separation and immune-competent barrier between the upper aerodigestive tract or paranasal sinuses and the epidural space to prevent meningitis and epidural abscess, (2) provide three-dimensional support and protection to the central nervous system, (3) reduce the probability of cerebrospinal fluid fistula, (4) provide vascular supply to dural reconstruction as needed, (5) enhance functional and aesthetic results by preventing contour deformities in the lateral orbit, and (6) maintain globe position and prevent enophthalmos or dystopia. Other indications for the temporalis muscle flap in extended head and neck and cranial base surgery include support for orbital contents following extended maxillectomy, obliteration of middle ear and mastoidectomy defects following temporal bone resection, and soft tissue augmentation of orbital exenteration defects.
CONTRAINDICATIONS
A common error in using the temporalis muscle is failing to realize the limitations of the size of the flap relative to the defect and not having satisfactory bulk to accomplish the reconstructive goals. Cranial base resection with either orbital exenteration or maxillectomy commonly results in a defect with significant exposure of the temporal lobe dura. This situation is frequently one in which the bulk of the temporalis flap is inadequate to match the defect, and a free tissue transfer is a more appropriate reconstructive option. Another error is not recognizing the need to sacrifice the blood supply to the temporalis muscle during the tumor resection.
PREOPERATIVE PLANNING
Minimal additional preoperative planning is needed when considering using the temporalis muscle for cranial base reconstruction. A detailed history should be obtained regarding previous surgical or nonsurgical treatments of head and neck or skull base malignancy. Prior radiation treatment to this area renders the temporalis muscle flap unreliable, and alternative reconstructive methods should be considered. Standard presurgical imaging of the skull base often includes both CT and MRI studies. The MRI will provide superior detail regarding the size, thickness, and bulk of the temporalis muscle and the potential size of the expected cranial base defect. Dedicated vascular or other specific studies are not necessary prior to using this flap.
SURGICAL TECHNIQUE (FIG. 48.2)
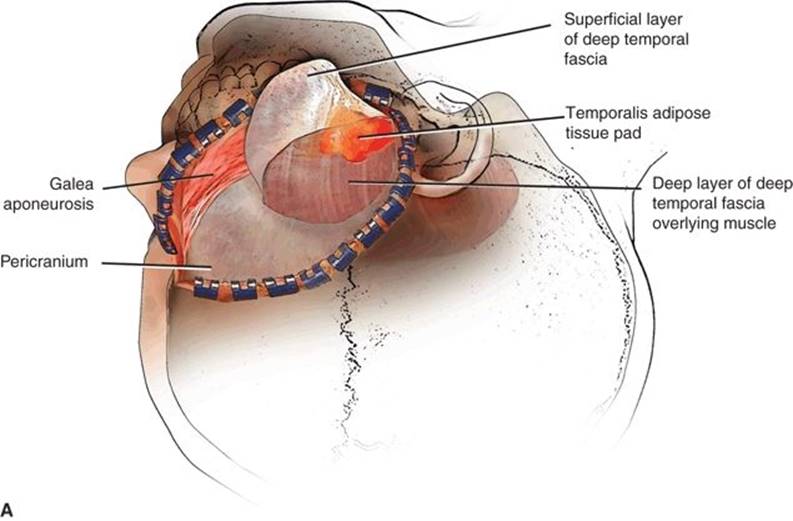

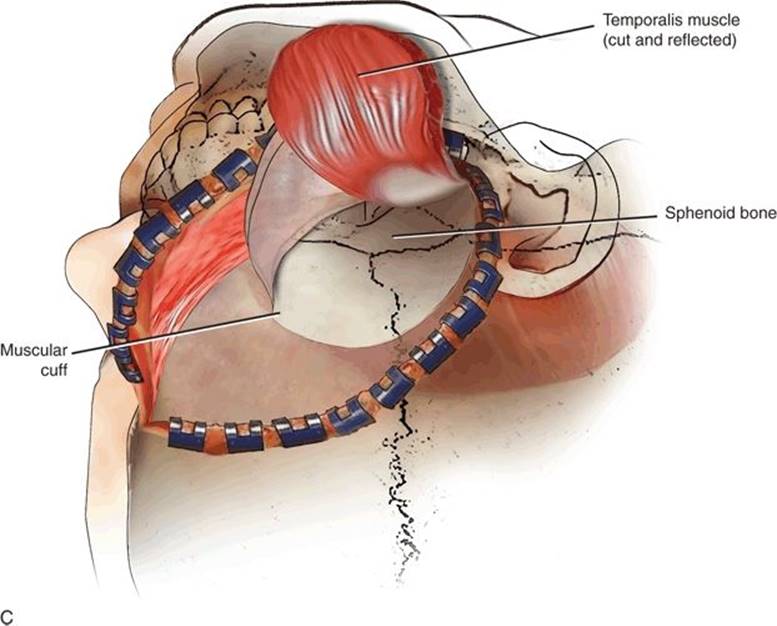
FIGURE 48.2 A-C Interfascial approach and harvest of temporalis muscle.
The successful application of the temporalis muscle flap begins with a clear understanding of the regional anatomy, various investing fasciae, and the vascular supply to the muscle. The temporalis muscle is usually approached through the same incision as the ablative procedure with either a hemi- or bicoronal scalp incision. With a hemicoronal incision, the apex of the incision should be taken to the vertex to expose the ipsilateral pericranium in the event additional tissue is needed in the reconstruction. The inferior aspect of the incision should be carried 1 cm below the tragus to facilitate safe exposure of the zygomatic root and eventually the zygomatic arch. The skin and subcutaneous tissue of the scalp are incised parallel to the hair follicles, and the subgaleal plane is developed anteriorly and posteriorly. Injury to the hair follicles and subsequent permanent alopecia can be avoided by maintaining a plane of dissection deep to the galea or superficial temporal fascia. Bipolar cautery should be used judiciously for hemostasis. The anterior limit of the exposure is the orbital process of the frontal bone and posteriorly the vertical plane of the mastoid tip. The posterior dissection is necessary to mobilize the entire temporalis muscle and gain the maximum arc of rotation.
The temporoparietal fascia (TPF) is attached superiorly to the galea aponeurotic and inferiorly to the superficial muscular aponeurotic system. The TPF is supplied by the frontal and parietal branches of the superficial temporal artery and has been extensively used as both a pedicle flap and a microvascular free tissue transfer for the reconstruction of limited-size head and neck defects. The TPF will need to be divided to fully mobilize the temporalis muscle; therefore, the identification and preservation are critical to maintain the integrity of this flap.
With the scalp reflected anteriorly, the surgeon will encounter the pale yellow temporal pad of adipose tissue deep to the TPF. The TPF is incised with a curvilinear incision approximately midway between the superior temporal line and the zygomatic arch preserving the superficial temporal artery system. A broad sharp periosteal elevator is used to mobilize the TPF inferiorly to the level of the temporal pad of adipose tissue. The pad of adipose tissue is located between the layers of the investing fascia of the temporalis muscle (deep temporal fascia). The deep temporal fascia is continuous with the pericranium (periosteum of the skull) superiorly and the parotid-masseteric fascia inferiorly. The more superficial layer of the deep temporal fascia that may be inseparable from the TPF is mobilized with the adipose tissue pad to preserve its blood supply. Mobilization of the pad of adipose tissue in this plane provides for protection of the frontal branches of the facial nerve. The deep layer of the deep temporal fascia should remain intact and be attached on the surface of the muscle. The thickness and strength of the deep temporal fascia are critical to hold sutures when securing the flap in the defect.
As the dissection proceeds inferior to the temporal pad of adipose tissue, the zygoma is palpated. The zygoma will be exposed by sharp dissection as a subperiosteal plane entered posteriorly on the arch. Beginning the dissection here and maintaining the subperiosteal plane will further protect the temporal and frontal branches of the facial nerve. The lateral dissection in the plane of the parotideomasseteric fascia proceeds until the superior most fibers of the masseter muscle are identified where they attach to the zygoma. The zygoma is skeletonized as far as the zygomatic root posteriorly, to the zygomatico-orbital suture anteriorly, and to the malar eminence inferiorly. Submillimeter holes for microplates or wires are drilled with the zygoma in situ. Osteotomies of the zygoma are carried out with a high-speed reciprocating saw and can either be left attached to the masseter muscle or be removed and replaced as a nonvascular autograft. Keeping the zygoma attached to the masseter muscle facilitates inferior retraction of the muscle and identification of the tendinous insertion of the temporalis muscle onto the coronoid process. The zygomaticofacial foramen is a consistent anatomic reference for the lateral extent of the maxillary antrum; placing the osteotomy posterior to it on the malar eminence will prevent entrance into the maxillary sinus. Anterior–inferior exposure can be safely facilitated by using the Cushing subtemporal blunt retractors, thereby avoiding direct injury to distal branches of the facial nerve.
Mobilization of the zygoma allows for complete visualization of the temporal fossa and the size and position of the temporalis muscle. The muscle originates from the squamous portion of the temporal bone at the level of the superior temporal line and the greater wing of the sphenoid bone; therefore, there is no fascia on the deep surface of the muscle. The only superior fascial attachments are those to the pericranium. Inferiorly, the muscle fibers condense into a dense tendon that inserts onto the medial and lateral surfaces of the coronoid processes and the anterior aspect of the ramus of the mandible. The vertical height of the muscle is up to 12 cm from the superior origin to the insertion at the coronoid. The broad belly of the muscle is up to 2 cm thick and ranges from 12 to 14 cm in the anterior–posterior dimension. The motor and proprioceptive innervations of the muscle are from the anterior and posterior deep temporal nerve branches of the anterior division of the mandibular branch of the trigeminal nerve (V3). These nerves enter the deep aspect of the muscle after passing superiorly to the lateral pterygoid muscle.
Once fully exposed, the muscle is now completely elevated from the temporal and sphenoid bones. If a pericranial extension is needed, the pericranium is marked and divided with electrocautery to access the subperiosteal plane, and sharp dissection proceeds broadly in a superior-to-inferior direction. Caution is needed when dissecting the attachments of the muscle to the superior temporal line to prevent shearing of the pericranium from the distant end of the temporalis muscle. If a composite osseous–muscle flap is to be used, the calvarium attached to the muscle is harvested at this point, preserving multiple microscopic perforating vessels to the bone. The dissection proceeds inferiorly separating the muscle from the greater wing of the sphenoid and lateral pterygoid plates. The deep temporal nerve will be encountered just medial to the lateral pterygoid plate. It is not necessary to routinely divide these nerves as their preservation results in some maintained muscle bulk. Extreme caution is needed when elevating the most inferior aspects of the muscle as to not injure the vascular supply. The muscle is supplied by the paired anterior and posterior deep temporal arteries and branches of the second or pterygoid division of the internal maxillary artery, and venous drainage is from the associated venae comitantes draining into the pterygoid plexus. The second (pterygoid or muscular) division of the internal maxillary artery courses anteriorly and superiorly for a short distance between the heads of the lateral pterygoid muscle and gives off branches to the temporalis, pterygoid, masseter, and buccinator muscles before becoming the third (pterygopalatine) segment as it enters the pterygopalatine canal. The anterior and posterior deep temporal arteries enter the temporalis muscle on the deep surface supplying the muscle and pericranium in a consistent pattern allowing for the safe partitioning of the muscles into discrete anterior and posterior components. To do this safely, the muscle should be divided sharply in its midportion in an anterior–posterior direction using meticulous bipolar electrocautery for hemostasis. To reliably preserve the vascular pedicle, the elevation of the muscle should terminate when the root of the lateral pterygoid is identified. Unless the muscle is to be harvested and used as a free tissue transfer, the routine identification of the vascular pedicle is not indicated (Fig. 48.3).
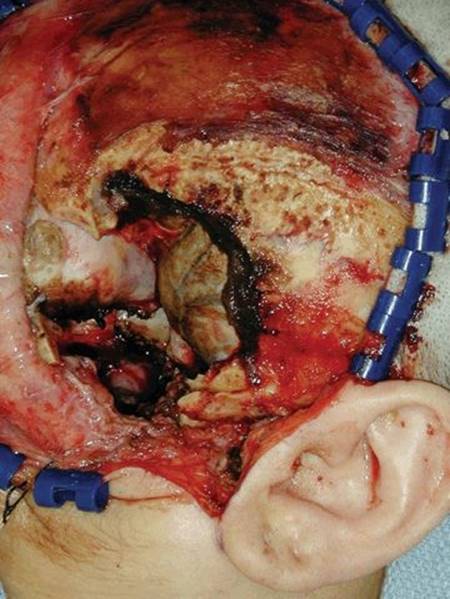
FIGURE 48.3 Surgical defect prior to inset of temporalis muscle.
With the muscle completely elevated, it can be transposed medially into the defect in the paracentral skull base region. Although not always necessary, an additional 1 to 2 cm of rotation can be obtained by resecting the coronoid process of the mandible. The coronoid is approached from its anterior–lateral aspect distant from the deep temporal arteries. A sharp periosteal elevator is used to dissect the attachments of the temporalis from the mandible and expose the coronoid from the tip to the base at the level of the sigmoid notch of the mandible. A small reciprocating saw or rongeur is used to remove the bone using malleable retractors medially to protect the vessels. The muscle is then free to maximally rotate on the vascular pedicle into the defect (Figs. 48.4 and 48.5).
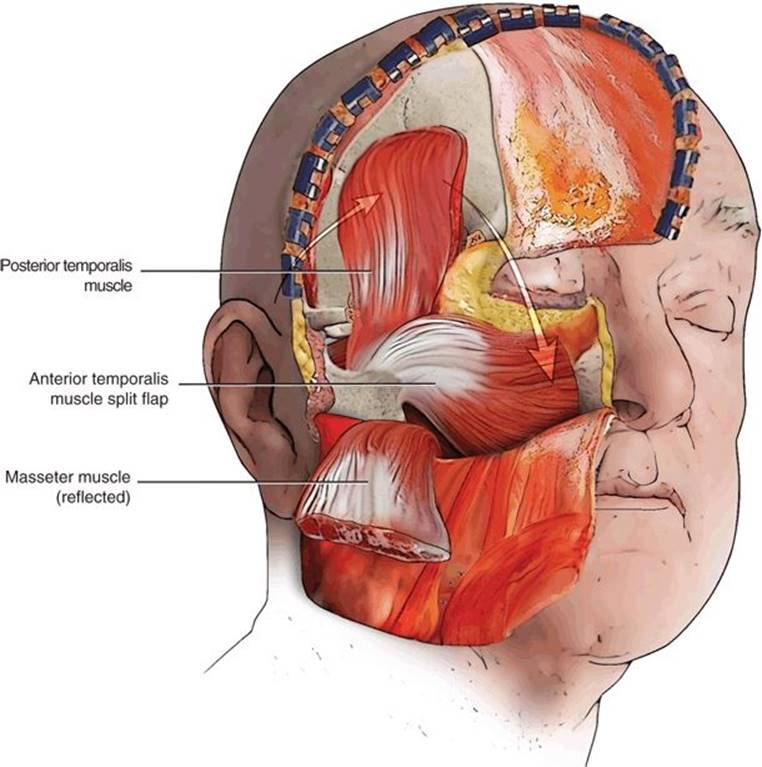
FIGURE 48.4 Schematic representation of split temporalis muscle reconstruction with anterior segment into skull base defect and posterior segment rotated anteriorly.
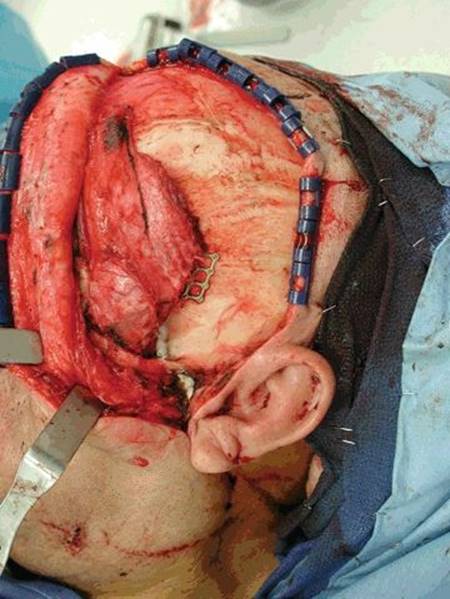
FIGURE 48.5 Split temporalis muscle in reconstruction. Anterior segment has been rotated anteriorly and inferiorly and cannot be seen while the posterior segment is rotated anteriorly and is visible in the temporal defect.
The muscle is sutured into the defect with absorbable sutures placed through the deep temporal fascia. Ideally, anchoring sutures in the defect are placed in small drill holes into the skull base. Even if the defect extends into the nasopharynx or paranasal sinuses, there is no need for a skin graft on the exposed muscle surface. Both the deep and lateral surfaces of the muscle tolerate exposure to the pharynx and will support the growth of the mucosa. The key principles in positioning the flap are to avoid tension, excessive angular rotation, and twisting and to ensure coverage of exposed major vessels and bone. If the defect is small, then the temporalis muscle can be split in the anterior–posterior dimension. The anterior component can be rotated inferiorly and medially into the defect in the paracentral skull base and the posterior component rotated anteriorly into the anterior portion of the donor site defect in the temporal fossa. The flap is secured anteriorly to the zygoma and orbital process of the frontal bone with absorbable sutures to small drill holes or the periosteum (Fig. 48.6).
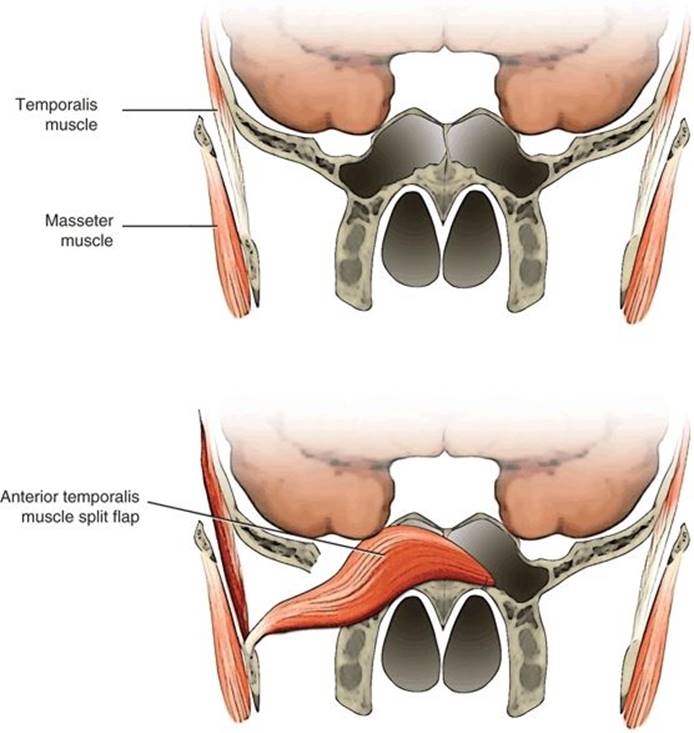
FIGURE 48.6 Schematic representation of medial inset of the temporalis muscle flap across a defect in the middle cranial base obliterating the sphenoid sinus and secured to the floor of the contralateral sphenoid sinus.
Transposition of the temporalis muscle will leave a depression at the donor site. Adipose tissue grafts have been used to eliminate this defect as have alloplastic materials. Women can minimize the cosmetic impact of this depression with appropriate hairstyling.
POSTOPERATIVE MANAGEMENT
At the completion of the procedure, the surgeon must be certain that there is complete separation between the upper aerodigestive tract and the (infra)temporal fossa. Following replacement of the zygoma, a medium-caliber suction drain (7-mm Jackson-Pratt) is inserted into the temporal fossa defect and placed to continuous wall suction. A tight circumferential dressing is to be avoided to prevent external compression of the flap. The eye should be carefully examined and lid function evaluated. If there is paresis of the upper divisions of the facial nerve, corneal protection protocols should be instituted. Temporary large-bore nasal airways can be used to maintain patency of the nasal cavity and nasopharynx and to support the medial portions of the temporalis flap. The nasal airway should be placed under direct vision with a sinus telescope to prevent disruption of the inset of the temporalis muscle flap.
COMPLICATIONS
Complications of the temporalis muscle flap occur in greater frequency in patients who have received prior radiation therapy to the temporal region and skull base; therefore, serious consideration should be given to reconstruction using free tissue transfer. The surgeons should always be cognizant of the blood supply to the muscle (see Surgical Technique section) and avoid excessive cautery on the deep surface of the muscle. A very generous tunnel should be created to avoid constricting the flap when transposing the muscle medially into the infratemporal fossa to reach the paracentral skull base, sphenoid, or carotid artery regions. Necrosis and loss of the flap can result in potentially devastating infectious complications such as epidural abscess, brain abscess, or meningitis. Loss of protection of the dura or dural reconstruction can lead to recurrent CSF leak.
Alopecia can develop when raising the scalp flap in too superficial a plane placing the hair follicles at risk. Injury to the frontal or temporal branches of the facial nerve may develop from incorrect placement of the incisions between the layers of the temporalis fascia (interfascial incisions), incomplete mobilization of the temporal pad of adipose tissue, or excessive retraction of poor placement of retractors on the anterior scalp and skin flaps.
The consequences of the temporalis muscle transfer include a mild insignificant mandibular drift to the contralateral side and the donor site deformity or temporal concavity. Patients will accommodate to the mild temporary mandibular crossbite, and no secondary procedures are indicated. Regarding the donor site deformity, autologous nonvascularized adipose grafts, alloplasts, and vascularized free tissue transfers have all been used in this situation. The use of nonvascularized adipose tissue and alloplasts carries a significant risk of infection and extrusion particularly when there is contamination from the oral cavity or paranasal sinuses. I prefer a delayed reconstitution of the temporal defect to allow for satisfactory healing of the primary cranial base defect, the timely initiation of adjuvant treatment as needed, and time for the definitive three-dimensional geometry of the defect to stabilize. Secondary reconstruction of the temporal defect can then be tailored to more specifically accommodate the final defect.
RESULTS
The temporalis flap, taken from a donor site not previously operated, traumatized, or radiated is a reliable flap for reconstruction of a defect in the floor of the central skull base. The bulk and arc of rotation of the temporalis muscle make it a reliable flap for a variety of reconstructive applications, including reconstruction of the palate, retromolar trigone, and buccal mucosa, orbital exenteration, and lateral temporal bone resections.
PEARLS
· Due to anatomic proximity, the temporalis flap has been useful in reconstructing defects of the central skull base.
· A complete history and physical examination are important to rule out previous trauma, surgery, or radiation to the donor site.
· The surgeon must have a thorough understanding of the surgical anatomy of the temporalis muscle, particularly the blood supply and the proximity to the frontalis branch of the facial nerve.
· The patient should be counseled preoperatively about the possible aesthetic changes, due primarily to the concavity of the donor site, which follows transposition of the temporalis muscle.
· It is important to maintain the dissection in the proper plane to avoid injury to the frontalis branch of the facial nerve.
· Dividing the insertion of the temporal muscle to the coronoid process of the mandible will increase its reach and arc of rotation.
PITFALLS
· Not recognizing the sacrifice of the blood supply to the temporalis flap during tumor resection may lead to flap necrosis.
· Preoperative embolization of the muscular branches of the internal maxillary artery will render the flap nonviable.
· Previous surgery, trauma, or radiation to the donor site may result in flap necrosis due to fibrosis or injury to the blood supply.
· Not dissecting in the proper plane may result in paresis or paralysis of the frontal branch of the facial nerve.
· Failure to counsel the patient about the possible aesthetic consequences, such as concavity of the temporalis fossa, may result in a dissatisfied patient.
INSTRUMENTS TO HAVE AVAILABLE
· Broad sharp periosteal elevator
· Narrow sharp elevator (Lempert type)
· Bipolar electrocautery
· Cushing subtemporal retractor
· Reciprocating saw with small blade
· Malleable retractors
SUGGESTED READING
Hollinshead WH. Anatomy for surgeons. In: The head and neck, 3rd ed. Vol. 1. Philadelphia, PA: Harper and Row, 1982.
Nuss DW, Russavage JM, Janecka IP. The temporalis muscle flap for cranial base reconstruction. Prob Plast Reconstr Surg 1993;3:193–206.
Hanasono MM, Utley DS, Goode RL. The temporalis muscle flap for reconstruction after head and neck oncologic surgery. Laryngoscope 2001;111:1719–1725.
Youssef AS, Ahmadian A, Ramosi E, et al. Combined subgaleal/myocutaneous technique for temporal muscle dissection. J Neurol Surg B Skull Base 2012;73(6):387–393.
Hoffman TK, El Hindy N, Muller OM, et al. Vascularized local and free flaps in anterior skull base reconstruction. Eur Arch Otorhinolaryngol 2013;270(3):899–907.