Mahmoud Ismail, Marc Swierzy, and Jens C. Rückert
![]() INDICATIONS/CONTRAINDICATION
INDICATIONS/CONTRAINDICATION
Indications
Thymectomy is one of the rare indications in thoracic surgery. Thymectomy is most commonly considered to be a part of the complex treatment of myasthenia gravis (MG) and thymoma. Ectopic thymic tissue can be located at various locations throughout the anterior mediastinum; therefore, the radicality of thymectomy is crucial for tumor resection and complete remission of MG. Robotic thymectomy is the latest advancement in the surgery of the thymus gland. This is primarily due to the fact that it allows for a complete radical thymectomy, which improves the complete remission rate for MG when compared to the conventional thoracoscopic technique. The oncologic outcome in terms of overall survival and thymoma-related survival is promising, but a longer follow-up is needed to consider robotic thymectomy as a standard approach.
The first robotic thymectomy was published in 2001 though it was only a partial thymectomy for thymoma in this particular case. In 2003, a complete robotic thymectomy was described and after that many thoracic centers have started or switched to the robotic approach. Since that time, there were more than 3,500 robotic thymectomies registered by Intuitive Surgical.
MG is an antibody-mediated autoimmune disease of the neuromuscular junction. In the majority of patients (80% to 85%), autoantibodies to the muscle nicotinic acetylcholine receptor (AChR) are present. These antibodies lead to failure of neuromuscular transmission with muscle weakness. The minority of MG patients are seronegative for AChR antibodies. A second category of MG is due to autoantibodies against the muscle-specific kinase (MuSK) protein. These are present in 3% to 5% of MG patients. Thymectomy is considered for patients with seropositive AChR and negative for MuSK.
Other factor with relevance for the indication of thymectomy for MG is the clinical classification according to the Myasthenia Gravis Foundation of America (MGFA). According to the severity of the MG we distinguish two relevant groups, only ocular form (MGFA I) or generalized form (MGFA ≥II). According to the meta-analyses from Gronseth and Barohn, patients with moderate till severe form of generalized MG (MGFA ≥IIb) benefit from the thymectomy but not the ocular form. However, there is a controversy because 50% of MG patients initially have only an ocular form which in 50% to 70% of the cases change to generalized form in the first 2 to 3 years. In this case, a thymectomy is not possible in the first 2 years of the diagnoses of MG where benefit from thymectomy could be reached. Therefore, and according to retrospective studies, thymectomy is also indicated for patients with ocular MG.
Thymectomy is performed in all age categories according to many studies. Though many text books restrict the indication to “mainly patients between 8 and 60 years” there is no evidence on that. Our series contains successfully selected cases of patients down to the age of 4 years or beyond 60 years up to the age of 85 years.
Surgery is the mainstay of treatment for thymoma. When the tumor is at an early stage (Masaoka/Koga I through IIB), surgery is the first choice of treatment. If the tumor is apparently invasive and large (Masaoka/Koga II through III), preoperative (neoadjuvant) chemotherapy and/or radiotherapy may be used to decrease the size and improve resectability, before surgery is attempted.
Generally, thymectomy for MG without thymoma is relatively indicated when three of the following five factors are present: Generalized form of MG, seropositive AChR, suspicious thymic hyperplasia, initial diagnosis less than 2 years, and patient age between 8 and 60.
Contraindications
There is still doubt on the indication for thymectomy for patients with seronegative AChR antibodies, ocular MG, and MuSK positive antibodies. However, with the exception of MUSK-antibody-positive patients, there is some data that show benefit from thymectomy for these groups.
Due to unstable MG or patients in myasthenic crisis thymectomy always requires preparation with stabilization of MG by plasma exchange strategies or intravenous immunoglobulin treatment.
The principal contraindication to the performance of a thymectomy is advanced stage thymoma (Masaoka III through IVB). However, for Masaoka stage III, resectability should be judged upon the invaded organs.
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
All patients with MG should be evaluated by a neurologist, ideally in a center specialized for MG. All tests to diagnose MG should be reviewed. The antibody status for AChR and MuSK should be evaluated. All patients should undergo a contrast-enhanced CT scan of the thoracic cavity. Pulmonary function tests and arterial blood gas analyses are necessary. Beside these tests all patients have to be checked for operability due to other diseases. Optimally patients with a thymoma are discussed in a tumor board in large centers.
![]() SURGERY
SURGERY
The Left-Sided Robotic Thymectomy
Positioning
The left-sided minimally invasive thoracoscopic surgery employing the da Vinci telemetric robotic system is used. Surgery is performed under general anesthesia with one-lung ventilation. The patient is placed in a supine position on a vacuum mattress with the operating table slightly tilted to the right patient’s side. The left arm is positioned below the table level with flexion at the elbow. The initial position of the surgeon and his assistant is on the left side of the patient. The operation field is always prepared and draped for a conversion to median sternotomy or additional cervical approach or right-sided, thoracoscopy, respectively. The 12-mm trocar for the binocular camera is placed in the fourth intercostal space at the left anterior axillary line. A thoracoscopic 10-mm camera with a 30-degree optic is introduced to evaluate the operation field and help positioning the other two 8-mm trocars. The cranial trocar is introduced in the third intercostal space whereas the caudal trocar is located in the fifth intercostal space, both between the anterior axillary line and the midclavicular line. Thus, all three trocars are placed exactly along the submammary fold. The special da Vinci trocars are connected with the three robotic arms of the table cart. A harmonic scalpel is placed in the upper trocar whereas a bipolar forceps in the lower one.

Figure 26.1 Operation field after mobilization of the thymic gland along the left phrenic nerve.
Technique
Step 1 (Fig. 26.1): The left phrenic nerve is the point of reference for the dissection of the thymic gland. The dissection starts caudally in the middle of the pericardium along the left phrenic nerve. This area is very often completely free of fatty tissue, and the left phrenic nerve can be well recognized. In some cases, the thymic gland extends below or over the phrenic nerve. In these cases it is necessary to isolate the phrenic bundle and mobilize the tissue in the aortopulmonary window completely.
Step 2 (Fig. 26.2): Further dissection is performed cranially along the phrenic nerve till identifying and opening the cervical pleura at the entrance of the left innominate vein. The incision of the cervical pleural fold is extended till the median retrosternal line. The dissection is continued to the right side until the subxiphoid pleural fold is reached. With blunt gentle dissection the right lung is made visible and is covered only by the right pleura parietalis. Whenever possible the right pleural cavity should stay closed at that stage of the operation. This allows for extension of the operation field by insufflation of CO2.

Figure 26.2 Incision of the pleural fold to localize the innominate vein as a guiding structure for the robotic thymectomy.

Figure 26.3 Opening of the cervical compartment of the anterior mediastinum. With the exception of anatomical deviations the upper poles of the thymic gland are mobilized behind the sternum and in front of the innominate vein. Exceptionally, the hook is used in this case. The use of harmonic scalpel is recommended.
Step 3 (Fig. 26.3): The upper poles, mobilized after careful dissection of their capsule, are gently grasped and then pulled down. At the cranial end, the thyrothymic ligament becomes clearly visible. Under tension of the completely exposed upper thymic pole, this ligament is severed by ultrasonic dissection or between clip ligatures.
Step 4 (Fig. 26.4): In the majority of cases there are two to four thymic veins. These veins have to be dissected without tension and injury to the innominate vein. The veins are divided between clip ligatures while severing seems also to be possible by using the ultrasonic scalpel. Atypical locations of thymic veins have to be noticed, but they are rare.
Step 5 (Fig. 26.5): The whole median retrosternal tissue portion is mobilized. Often, the right main thymic lobe may be demarcated from the surrounding fatty tissue. The aortocaval groove is dissected free and the right lung, only covered by the mediastinal pleura, is exposed.
Step 6 (Fig. 26.6): Preparation of the right thymic lobe follows under CO2 insufflation. In most cases, the right phrenic nerve can be identified. After opening the mediastinal pleura of the right lung, the right phrenic nerve can be well recognized.

Figure 26.4 The thymic veins are dissected by the harmonic scalpel, for large vessels a clip may be necessary.
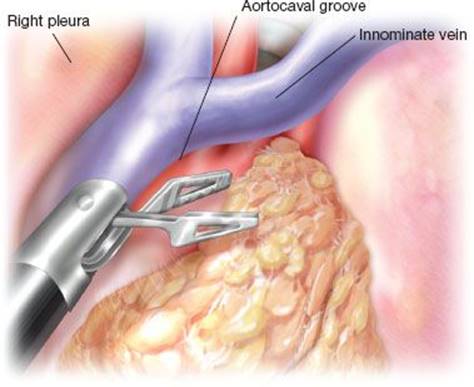
Figure 26.5 Dissection of the thymic tissue portion inside the aorto-caval groove, where sometimes a prominent right main thymic lobe may be found.
The en bloc resected specimen of the thymus with all surrounding fatty tissue is placed in an endobag and then removed through the middle trocar incisions. The operation field with the venous confluence, the supra-aortal arteries, and parts of the anterior tracheal wall is examined for the presence of residual tissue and hemostasis. A chest tube is placed in the left pleural cavity. Reinflation of both lungs is followed by closure of the trocar incisions.
Technical Note
Thoracoscopic operation technique has been reported in bilateral or unilateral modifications. Actually the preferred side of unilateral thoracoscopic thymectomy is most frequently discussed. Both left- and right-sided unilateral robotic thymectomy are feasible. Radical thoracoscopic thymectomy seems to be better accomplished by the left-sided technique. In the learning curve and with less experience many surgeons prefer the right-sided technique. Surgical school, habits, and traditions influence the choice of the procedure. Optimal long-term results require the ability to perform either left- or right-sided technique. Robotic assistance is the adequate technical refinement for thymectomy.

Figure 26.6 For demonstration of the right phrenic nerve the wristed tips of the instruments, CO2-insufflation, and 3-dimensional view is required.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
After the operation, the patient is extubated immediately and put on patient-controlled analgesia. Peridural analgesia is not required. A chest drain is only necessary for a short postoperative period and may be removed after normal x-ray and with adequate clinical findings. The hospital stay usually is about 2 to 3 days but may be reduced to 1 day in selected cases.
![]() COMPLICATIONS
COMPLICATIONS
The robotic thymectomy has been proven to be feasible and safe. The perioperative morbidity rate in our series is less than 2%. The general risks of robotic thymectomy include incomplete removal of the thymic gland, nerve injuries (phrenic nerves, laryngeal recurrent nerves, and intercostal nerves), and bleeding (innominate vein and tributaries, aortic arch, mammarian vessels). For complete thymic resection, every single step of the technique needs to be controlled. If it cannot be completed satisfactorily, an extension of the technique is required. This does not necessarily mean a conversion as the first step, but the effective use of extra incisions or trocars. A rapid conversion to sternotomy, however, should be possible at any time during robotic thymectomy.
![]() RESULTS
RESULTS
The cosmetic results are very acceptable and highly appreciated by the patients. Moreover, there is no interference with immunosuppression. After robotic thymectomy, a sternotomy could be easier if necessary in later life. The impairment of the patient by this operation is minimal. According to our experience the development of chronic pain at the trocar sites is a very rare condition. Until 2002 we have performed 80 thoracoscopic thymectomies without robotic assistance. Between 2003 and 03/2013 we performed 335 robotic thymectomies (Tables 26.1 and 26.2). There are approximately 3,500 robotic thymectomies registered worldwide between 2001 and 2012. The real number of these operations might be even larger and is growing rapidly. A literature summary of all published robotic thymectomy series including more than 20 cases is presented in Table 26.3. A special advantage of robotic assistance is its practicability even for demanding indications such as small children, obese patients, and older patients with large amounts of tissue inside the mediastinal area. The main outcome parameter after thymectomy for MG is the cumulative complete stable remission rate of myasthenia symptoms. This is prospectively estimated according to the recommendations of the MGFA. After robotic thymectomy, the cumulative complete stable remission rate was 58%. This is comparable to the results published by the group around Jaretzki et al.
TABLE 26.1 Patient Characteristics and Degree of Severity of Myasthenia Gravis according to MGFA—Classification

TABLE 26.2 Results of Robotic Thymectomy, Thymoma-classification, and Preoperative Morbidity

TABLE 26.3 Literature Summary of Robotic Thymectomy Series Including More Than 20 Cases

![]() CONCLUSIONS
CONCLUSIONS
Thymectomy is a rare operation in thoracic surgery. The main indication is MG and thymoma. The indication for thymectomy should be discussed in a certified interdisciplinary center for MG and in tumor board for thymoma. The robotic approach seems to have greater potential for new technical developments than other nonrobotic minimally invasive procedures. Due to the promising results of the robotic thymectomy, many thoracic surgeons and neurologists are more convinced of this procedure.
Recommended References and Readings
Masaoka A, Maeda M, Monden Y, et al. [Distribution of the thymic tissue in the anterior mediastinum–studies on the methods of thymectomy]. Nihon Kyobu Geka Gakkai Zasshi. 1975;23(8):1016–1021.
Jaretzki A 3rd, Penn AS, Younger DS, et al. “Maximal” thymectomy for myasthenia gravis. Results. J Thorac Cardiovasc Surg. 1988; 95(5):747–757.
Marulli G, Schiavon M, Perissinotto E, et al. Surgical and neurologic outcomes after robotic thymectomy in 100 consecutive patients with myasthenia gravis. J Thorac Cardiovasc Surg. 2013;145(3):730–735; discussion 5–6.
Ruckert JC, Swierzy M, Ismail M. Comparison of robotic and nonrobotic thoracoscopic thymectomy: A cohort study. J Thorac Cardiovasc Surg. 2011;141(3):673–677.
Yoshino I, Hashizume M, Shimada M, et al. Thoracoscopic thymomectomy with the da Vinci computer-enhanced surgical system. J Thorac Cardiovasc Surg. 2001;122(4):783–785.
Ashton RC Jr, McGinnis KM, Connery CP, et al. Totally endoscopic robotic thymectomy for myasthenia gravis. Ann Thorac Surg. 2003;75(2):569–571.
Bodner J, Wykypiel H, Wetscher G, et al. First experiences with the da Vinci operating robot in thoracic surgery. Eur J Cardiothorac Surg. 2004;25(5):844–851.
Ismail M, Swierzy M, Ruckert JC. State of the art of robotic thymectomy. World J Surg. 2013;37(12):2740–2746.
Drachman DB. Myasthenia gravis. N Engl J Med. 1994;330(25):1797–1810.
Silvestri NJ, Wolfe GI. Myasthenia gravis. Semin Neurol. 2012;32(3):215–226.
Gronseth GS, Barohn RJ. Practice parameter: Thymectomy for autoimmune myasthenia gravis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology.2000;55(1):7–15.
Grob D, Brunner N, Namba T, et al. Lifetime course of myasthenia gravis. Muscle Nerve. 2008;37(2):141–149.
Mineo TC, Ambrogi V. Outcomes after thymectomy in class I myasthenia gravis. J Thorac Cardiovasc Surg. 2013;145(5):1319–1324.
Hohlfeld R, Goebels N, Engel AG. Cellular mechanisms in inflammatory myopathies. Baillieres Clin Neurol. 1993;2(3):617–635.
Toker A, Sonett J, Zielinski M, et al. Standard terms, definitions, and policies for minimally invasive resection of thymoma. J Thorac Oncol. 2011;6(7 suppl 3):S1739–S1742.
Eng TY, Thomas CR Jr. Radiation therapy in the management of thymic tumors. Semin Thorac Cardiovasc Surg. 2005;17(1):32–40.
Yuan HK, Huang BS, Kung SY, et al. The effectiveness of thymectomy on seronegative generalized myasthenia gravis: Comparing with seropositive cases. Acta Neurol Scand. 2007;115(3):181–184.
Ruckert JC, Ismail M, Swierzy M, et al. Thoracoscopic thymectomy with the da Vinci robotic system for myasthenia gravis. Ann N Y Acad Sci. 2008;1132:329–335.
Cea G, Benatar M, Verdugo RJ, et al. Thymectomy for non-thymomatous myasthenia gravis. Cochrane Database Syst Rev. 2013;10:CD008111.
Keijzers M, Dingemans AM, Blaauwgeers H, et al. 8 years’ experience with robotic thymectomy for thymomas. Surg Endosc. 2013. [Epub ahead of print].
Freeman RK, Ascioti AJ, Van Woerkom JM, et al. Long-term follow-up after robotic thymectomy for nonthymomatous myasthenia gravis. Ann Thorac Surg. 2011;92(3):1018–1022; discussion 22–23.
Melfi F, Fanucchi O, Davini F, et al. Ten-year experience of mediastinal robotic surgery in a single referral centre. Eur J Cardiothorac Surg. 2012;41(4):847–851.
Augustin F, Schmid T, Sieb M, et al. Video-assisted thoracoscopic surgery versus robotic-assisted thoracoscopic surgery thymectomy. Ann Thorac Surg. 2008;85(2):S768–S771.
Cerfolio RJ, Bryant AS, Minnich DJ. Starting a robotic program in general thoracic surgery: Why, how, and lessons learned. Ann Thorac Surg. 2011;91(6):1729–1736; discussion 36–37.
Castle SL, Kernstine KH. Robotic-assisted thymectomy. Semin Thorac Cardiovasc Surg. 2008;20(4):326–331.
Goldstein SD, Yang SC. Assessment of robotic thymectomy using the Myasthenia Gravis Foundation of America Guidelines. Ann Thorac Surg. 2010;89(4):1080–1085; discussion 5–6.
Tomulescu V, Stanciulea O, Balescu I, et al. First year experience of robotic-assisted laparoscopic surgery with 153 cases in a general surgery department: Indications, technique and results. Chirurgia (Bucur).2009;104(2):141–150.
Seong YW, Kang CH, Choi JW, et al. Early clinical outcomes of robot-assisted surgery for anterior mediastinal mass: Its superiority over a conventional sternotomy approach evaluated by propensity score matching. Eur J Cardiothorac Surg. 2013;45(3):e68–e73.
Schneiter D, Tomaszek S, Kestenholz P, et al. Minimally invasive resection of thymomas with the da Vinci(R) Surgical System. Eur J Cardiothorac Surg. 2013;43(2):288–292.