Sidhu Gangadharan
![]() INDICATIONS/CONTRAINDICATIONS
INDICATIONS/CONTRAINDICATIONS
Indications
Tracheoplasty via posterior splinting of the airway is performed in cases of severe tracheomalacia. Often the malacia, or dynamic airway collapse, involves not only the trachea but also the bronchial tree as well. In these cases of tracheobronchomalacia, tracheobronchoplasty is utilized to achieve stabilization of the entirety of the abnormal airways. While very focal malacia may be detected, most cases of acquired dynamic airway collapse involve both the trachea and bronchial airways. For purposes of this chapter the terms tracheomalacia and tracheoplasty will be used to encompass the full spectrum of disease and treatment of the central airways, including the trachea and bronchial tree.
It is crucial that the indications for surgical intervention are well understood. If not, the surgeon may be treating the anatomic and radiographic findings, but not the patient. The simple presence of tracheomalacia is not an indication for intervention. In addition, some degree of collapsibility is normal, with nearly three-quarters of healthy volunteers exhibiting cross-sectional airway reduction greater than 50% during maneuvers designed to increase intrathoracic pressures such as forced expiration or Valsalva.
The symptoms that may result from tracheomalacia include dyspnea, cough, orthopnea, and retained secretions. In addition, tracheomalacia may lead to recurrent infections, and, in extreme cases, respiratory failure. The presence of tracheomalacia and mild symptoms would not warrant intervention. The indication for tracheoplasty would be severe malacia coupled with significant symptomatology. Previous experience shows that nearly all patients undergoing tracheoplasty (94%) will have dyspnea as one of their symptoms. Nearly three-quarters of patients will suffer intractable cough preoperatively, and half will report a history of recurrent respiratory infections.
A note of caution in interpreting the indications for surgery is that all of these symptoms and associated conditions are nonspecific.
Contraindications
Contraindications to surgery are relative and include previous airway interventions such as resection or tracheoesophageal fistula repair, esophagectomy, or left pneumonectomy. Caution must be taken regarding the tracheal blood supply in cases of previous airway surgery. Left pneumonectomy would impair the ability to perform a right thoracotomy approach for the tracheoplasty. Other conditions leading to impaired pulmonary function and inability to tolerate prolonged periods of single lung ventilation should also be taken into account. While the operation can be performed with two lung ventilation and a retractor system to help keep the inflated right lung out of the field, it is not ideal, and significant hypoxia or carbon dioxide retention preoperatively makes this elective surgical intervention less attractive.
Relapsing polychondritis is a condition that may manifest as tracheomalacia in addition to other stigmata of cartilaginous abnormality. However, the dynamic collapse in this condition tends to be concentric, and thus, the posterior stabilization afforded by tracheoplasty tends to be ineffective. It would also be important to search for other conditions that may be more straightforwardly treated and may be related to the patient’s nonspecific symptoms, for example, atypical reflux with cough, IgG deficiency and recurrent respiratory infections, allergic rhinitis and postnasal drip with cough, or vocal cord dysfunction with cough and dyspnea.
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
The preoperative planning for this operation can be thought of as three separate assessments.
1. Determining the degree of tracheomalacia
2. Eliminating confounding comorbidities
3. Predicting the likelihood of significant symptomatic relief from tracheoplasty
Establishing a diagnosis of tracheomalacia can be accomplished with dynamic airway computed tomography (CT) or functional bronchoscopy. Dynamic airway CT scanning has been shown to reliably identify airway collapse. The protocol for this involves scanning during a breath hold at total lung capacity and comparing the luminal patency to another series, which is obtained during a forced expiratory maneuver. The airway cross-sectional area is obtained by hand tracing the inner wall of the airway’s contour with an electronic tracing tool, and then the percentage of expiratory luminal collapse can be calculated (Fig. 40.1). Functional bronchoscopy involves fiberoptic endoscopy of the awake patient. Similar coached expiratory maneuvers are performed to estimate the degree of luminal collapse (Fig. 40.2). Concordance between these two diagnostic methods has been demonstrated. Nevertheless, both studies are utilized to assure that severe tracheomalacia is present. Air trapping, emphysema, or other parenchymal abnormalities may be detected additionally with CT scanning. Bronchoscopy may detect endobronchial abnormalities such as diverticula, tumor, or inflammation that might be missed radiographically.
The degree and the extent of malacia are determined by dynamic airway CT and functional bronchoscopy and are reported in standardized fashion as a percentage of luminal collapse during expiration at the proximal tracheal, distal tracheal, right mainstem bronchial, bronchus intermedius, and left mainstem bronchial levels. Additional information about the condition of the cervical trachea or the lobar and segmental bronchi is also reported. In general only patients with excessive expiratory collapse (>80% to 90%) would be considered for surgical stabilization.
While the etiology of most cases of acquired severe diffuse tracheomalacia remains unknown there are conditions that may confound the understanding of the impact that tracheomalacia may have on a patient’s symptomatology. Vocal cord dysfunction or paradoxical vocal fold motion is evaluated by endoscopic examination and if found may be treated with voice therapy or medications such as proton pump inhibitors, anxiolytics, gabapentin, or even botulinum toxin injection. In some cases patients who have symptoms of cough or breathlessness may have sufficient improvement to avoid the need for surgical airway stabilization. Gastroesophageal reflux disease (GERD) appears to be highly prevalent in the tracheomalacia patient population. Nearly 50% of patients who underwent tracheoplasty in the largest published series were also found to have GERD. While a causal link between GERD and microaspiration and the development or propagation of tracheomalacia has not been established, the possibility of GERD causing chronic cough is enough of a confounder that a formal pH study to evaluate for GERD is performed prior to intervention for the tracheomalacia. Some patients will undergo an antireflux procedure based on the degree of reflux detected, and in some cases tracheoplasty is rendered unnecessarily following this.
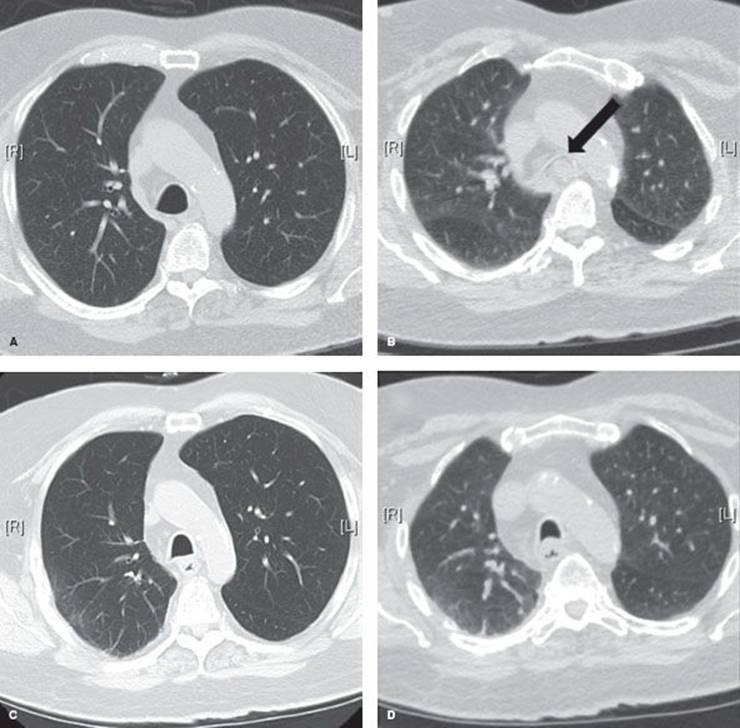
Figure 40.1 Dynamic airway CT images. A: Preoperative end-inspiration at distal trachea. B: Preoperative dynamic expiration at distal trachea. Arrow points to 100% collapsed airway lumen. C: Posttracheoplasty end-inspiration image at distal trachea. D: Posttracheoplasty dynamic expiration image at distal trachea.
Once a diagnosis of severe, diffuse tracheomalacia is established, an attempt is made to understand the potential salutary effects of airway stabilization. A stent trial is performed utilizing a Y-shaped silicone tracheobronchial stent, which is placed via rigid bronchoscopy and will achieve internal stabilization of the thoracic trachea and bilateral mainstem bronchi. After a 2-week trial, patients are seen in clinic to review the degree of improvement in symptomatology if any that might be attributed to stabilization of the central airways. Patients who reported marked symptomatic improvement would be considered for definitive surgical intervention.
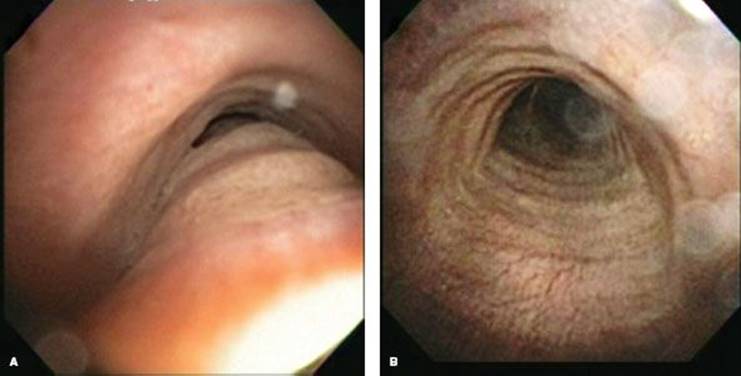
Figure 40.2 Functional bronchoscopy. A: Preoperative inspiratory image of the same patient as Fig. 40.1. B: Preoperative forced expiratory image. Note the near-complete obliteration of distal tracheal lumen.
Of the possible symptoms that internal stabilization with a stent might ameliorate, alleviation of dyspnea is the most consistent and obvious measure of treatment effect, partially because dyspnea is the most common presenting complaint, and partially because the stent has an immediate effect on expiratory airflow. The impact of stent stabilization on intractable cough may be more equivocal. The stent may alleviate the paroxysmal barking cough caused by severe expiratory airway collapse; however, the effect of a foreign body (stent) in the airway may in and of itself cause an aggravating cough and therefore limit the “readout.” The stent trial also may be limited because of a stent-related complication such as mucous plugging, granulation tissue formation, or infection. Judging the impact on symptoms may be difficult because of the negative symptoms generated by the stent itself or because the duration of stenting prior to its removal for adverse events is too short. The impact of a stent on the risk of recurrent respiratory infections is impossible to truly judge in a short duration as well.
A stent trial may not be possible to complete in patients with unusually large or small airways if correct stent sizing is not available, or in patients whose anatomy precludes safe rigid bronchoscopy. Given these limitations, it is important to attempt to tease out the beneficial outcomes of stent placement and consider them separately from the stent-related complications and adverse symptoms that might affect a majority of patients, peaking at the 3-week mark poststent placement. It is important to assess for the potential “placebo effect” of a stent. In addition, it is important that significant improvement is the benchmark. Slight improvement of symptoms should not constitute a positive stent trial. Despite the limitations, a planned short-term (<2 weeks) stent trial usually yields useful information about the likelihood of the patient with tracheomalacia to respond to airway stabilization. Between 60% and 75% of patients with tracheomalacia will respond to a stent trial in positive fashion and improvement in quality of life following definitive surgical airway stabilization with tracheoplasty is seen in 80% of these carefully selected patients.
Finally, the usual preoperative stratification of risk from cardiac or other comorbidities would be performed. Pulmonary function testing is performed and may reveal obstructive disease though this is a variable finding. Similarly flow-volume loop analysis may reveal normal patterns or a varying set of abnormal patterns. Moreover, the degree of expiratory tracheal collapse does not correlate with airflow limitation in patients with tracheomalacia. In general, though pulmonary function tests are obtained on all patients prior to intervention, a primary use for the results is to predict the likelihood of an arduous or complicated recovery from invasive intervention due to pulmonary comorbidity. Six-minute walk duration is measured preoperatively and poststabilization of the airway to assess the impact on functional status. In addition, determination of Karnofsky performance status, American Thoracic Society dyspnea score, and respiratory-affected quality of life based on the St. George’s Respiratory Questionnaire are done to ensure that significant impairment of a patient’s well-being exists prior to considering intervention for tracheomalacia.
![]() SURGERY
SURGERY
Tracheoplasty was initially described by Nissen and Herzog in the 1950s and the objective is to reconstitute a D-shaped trachea in cross section and to prevent intrusion of the membranous wall into the lumen of the airway (Fig. 40.3). As opposed to stenting, which has increasing complication rates the longer the stent is in place, tracheoplasty is designed to be a permanent solution to tracheomalacia (TBM).
Anesthetic Considerations
Patients will have an epidural placed for analgesia in the preoperative holding area. Standard antibiotic prophylaxis for skin flora is given immediately before incision. The airway management is accomplished by using a modified double-lumen left-sided endobronchial tube. The tracheal lumen is shaved off with a scalpel before placement. This allows minimal deformation of the trachea during suturing, and the smaller balloon of the endobronchial tube distends the airway less than a comparable small single-lumen tube placed in the bronchial position. This is useful both to accurately reshape the airway and to avoid inadvertent full-thickness suturing that might also deflate the balloon. In some cases a single-lumen tube with a bronchial blocker may be used, but this comes with the dual disadvantages of a longer balloon distending the trachea as well as a balloon, which may be easily dislodged during lung retraction, within the right mainstem bronchus. Close communication with the anesthesia team is necessary during the surgery as periods of apnea or deflation of the balloon and movement of the tube away from areas of active suturing are common, especially around the carina and left mainstem bronchus.
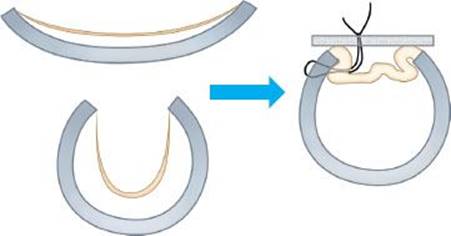
Figure 40.3 Diagram of tracheoplasty. Both airways with weakened cartilage as well as those with excessive membranous intrusion into the lumen benefit from posterior stabilization with mesh.
Positioning
Standard left lateral decubitus positioning is used. Given the long duration of the operation care is taken to avoid unpadded pressure points. In addition, it is important to have assurance that multiple degrees of table rotation will not lead to any instability of the patient as these are often necessary to perform the left-sided portion of the dissection and suturing from the right chest.
Surgical Technique
A standard right posterolateral thoracotomy is performed, with division of the latissimus dorsi muscle and preservation of the serratus anterior muscle. The fourth interspace is entered, and for additional exposure, posterior shingling or resection of the fourth or fifth rib is performed. To reduce postthoracotomy pain a periosteal elevator may be used to free the intercostal muscle and neurovascular bundle from the inferior aspect of the fourth rib prior to placement of the retractor. The dissection commences with ligation of the azygos vein, which provides the initial exposure to the distal trachea. The pleura overlying the posterior aspect of the trachea is then incised carefully, avoiding the right vagus nerve, which is dissected free and preserved. Branches of the vagus to both the lung and esophagus may be divided to allow mobility of the nerve. Care is taken not to entrap the nerve with the mesh or sutures once the posterior splinting is begun. Traction on the vagus nerve at its most superior thoracic extent is avoided so as not to damage the right recurrent laryngeal nerve. When working on the left aspect of the trachea, awareness of the location of the left recurrent laryngeal nerve is also important. At the level of the superior thoracic trachea the occasional aberrant right subclavian artery may be encountered and is best planned for by careful analysis of the preoperative CT scan.
The entirety of the thoracic trachea is exposed and the posterior membrane is dissected free of any adventitial attachments, so that suturing will occur directly onto the airway wall (Fig. 40.4). This dissection is carried all the way to the cartilaginous–membranous junctions bilaterally. To avoid ischemia, dissection onto the lateral walls of the trachea is minimized. The left mainstem bronchus is dissected free down to the level of the secondary or lobar carina if possible, utilizing gentle posterior retraction of the esophagus with a malleable retractor and rightward retraction of the right lung to deliver the left-sided airway into the field. Care is taken to avoid damage to the thoracic duct. The right-sided airways are dissected to the level of the distal bronchus intermedius, stopping at the level of the origin of the superior segmental bronchus. The subcarinal lymph node packet is left in situ, but dissected away from the cartilaginous edges of the bilateral mainstem bronchi to allow precise suture placement.
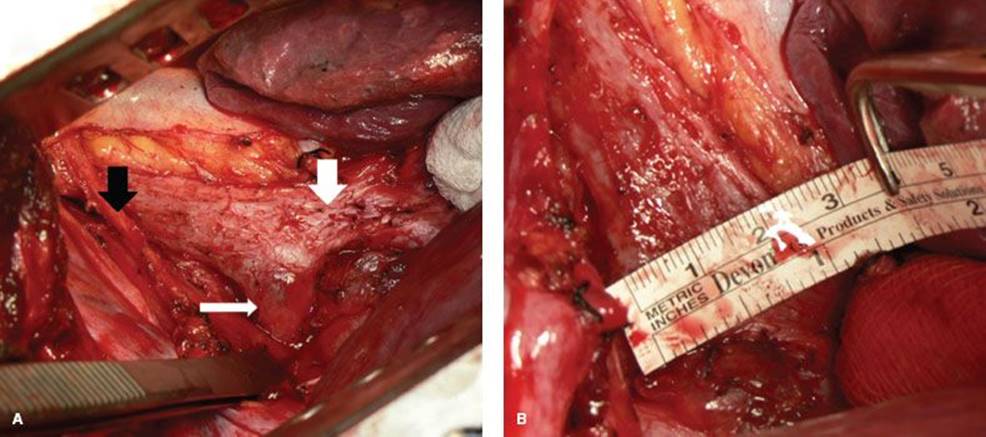
Figure 40.4 Airway dissection and measurement. A: Dissection of trachea and bronchi. Black arrow indicates vagus nerve. Thick white arrow indicates right mainstem bronchus. Thin white arrow indicates left mainstem bronchus. B: Measurement of transverse airway diameter of distal trachea.
The transverse airway diameter is measured at the proximal and distal trachea, right mainstem bronchus, bronchus intermedius, and left mainstem bronchus (Fig. 40.4). A polypropylene mesh is then cut with scissors into a Y-shaped posterior splint based on these measurements. Large airways typically can undergo more of a transverse downsizing by reducing the width of the mesh. A small airway with primarily membranous intrusion into the lumen as opposed to a lateral splaying of the cartilage may need to have a mesh sized nearly identical to its native measurements. A 0.5 cm margin is left on the edge of the mesh to allow a secure, unfrayed material through which to suture. While the original description of surgical stabilization was performed with a splint made of bone, and other published and unpublished researchers have utilized polytetrafluoroethylene (PTFE), cartilage, acellular dermis, or other materials, the most studied material remains the polypropylene mesh.
The mesh is sutured into place to splint the posterior airway walls using 4-0 polypropylene. Other groups prefer absorbable material such as polyglactin, but the longevity of polypropylene seems useful in these cases where remodeling forces may continue to exist postsurgery. The risk of mesh infection from inadvertently full-thickness sutures has not been borne out. Partial-thickness U-stitches are placed in a row across the distal trachea, a row across the bilateral mainstem bronchial origins, as well as a single suture, which is centered in the middle of this “carinal triangle” of sutures. Each row comprises four sutures. The first suture is placed through the mesh, then in mattress fashion at the cartilaginous–membranous junction of the airway, making sure that the cartilaginous ring and not simply an interspace is engaged, and then the suture is passed back up through the mesh. The next two mattress sutures are placed to gather up the laxity in the membranous tracheal wall. These gather 2 to 3 mm of posterior membrane each (Fig. 40.5). Previously these were placed one-third and then two-thirds across the width of the trachea. As a modification to further tighten the plication, the sutures are spaced in those positions in the native tracheal wall, but the placement in the mesh is moved laterally toward each edge. In this way the excess laxity of the membrane is tightened toward the cartilage–membranous junctions (Fig. 40.6). The fourth suture is a cartilaginous–membranous suture on the contralateral side.
After the distal tracheal row is placed, the right mainstem row of sutures is placed, and then a central membranous wall suture is usually placed before the left mainstem row is performed. Sutures are managed by ordering them using hemostats to avoid tangling. The mesh is then parachuted into place and then the sutures are tied, with the cartilaginous sutures tied first, then the membranous wall sutures, to avoid avulsing the sutures from the less substantial membranous wall tissue.
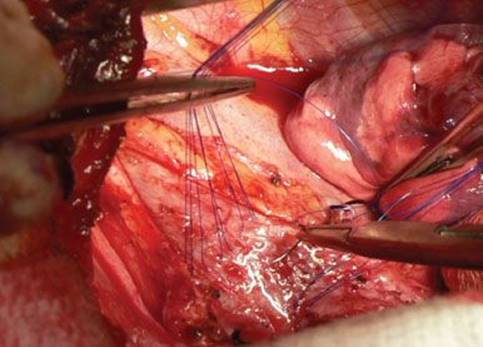
Figure 40.5 Suture placement at the carina. Distal tracheal and right mainstem bronchial rows are completed. Central triangle suture is being placed.
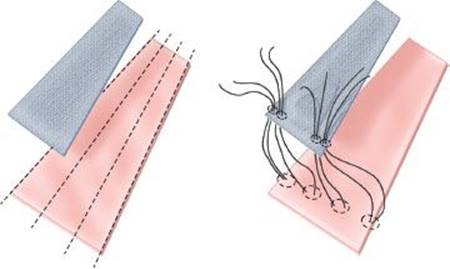
Figure 40.6 Modification to suture placement. The mesh is blue; native trachea is pink. The rows of sutures are placed evenly spaced across the trachea but are gathered toward the lateral edge of the mesh. This creates tension on the membranous wall in addition to affixing the mesh to the airway wall for support.
The trachea is then splinted by placing successive rows moving from distal to proximal on the trachea. Another modification of the technique, which is especially useful in cases where tension cannot be created on the membrane through the use of a downsized mesh, is by spacing the rows on the mesh every 4 mm apart while spacing each successive row on the native trachea every 7 mm apart. Thus there is axially generated tension on the lax membrane wall without additional narrowing of the tracheal diameter. Attempt is made to place the highest row of sutures up to the level of the thoracic inlet.
The right mainstem bronchus and bronchus intermedius bronchoplasty is then completed, utilizing the right limb of the Y-shaped mesh. Around the right upper lobe bronchial origin care is taken not to create a kink in the airway, which narrows that orifice. The left-sided bronchoplasty is created in similar fashion. In addition to backing out the endobronchial tube from the area of dissection as mentioned above, placement of the most distal sutures in the left may be guided by bronchoscopic assessment. The distal airway bilaterally is often too small to accommodate four sutures, so two cartilaginous sutures and one membranous wall suture may be used once the airway begins to narrow. At the completion of the tracheoplasty, the entirety of the thoracic airways to the level just before the lobar bronchi should be stabilized posteriorly.
Once the posterior splinting is completed, the pleural space is irrigated and a chest drain is placed. Bronchoscopy is performed through the modified endobronchial tube after it is pulled back into the tracheal lumen to assure that the airway looks well stabilized and that no lobar or segmental orifices appear compromised. If necessary, individual sutures may be removed and replaced. After the thoracotomy is closed and the patient is repositioned supine, the endotracheal tube may be exchanged over a bougie for a standard endotracheal tube through which aspiration bronchoscopy can be performed. The average duration of tracheoplasty is approximately 6 hours, with an additional 1 to 2 hours in the operating room for pre- and postincision activities such as skin preparation, bronchoscopy, positioning, etc.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
Patients will recover initially in an intensive care unit. Pulmonary toilet remains the most significant issue in the initial postoperative period. The patient’s management of secretions may be impaired by pain from the thoracotomy as well as edema in the airway and lack of effective cough. Usually the secretions and congestion will begin to subside after 2 to 3 days. Nevertheless, many patients will require aspiration bronchoscopy in the postoperative period. Routine laboratory studies are sent postoperatively. In addition, consideration may be given to sending serial creatine kinase levels as with prolonged immobilization for the duration of the operation many patients will have a rise in levels, some over 10,000 unit/L. Generally intravenous fluid administration is kept fairly liberal during the operation and immediate postoperative period. Subsequent gentle diuresis is sometimes necessary.
After an average of 3 days in the ICU, patients will spend 5 to 6 days on the ward. The pleural drain is managed using standard criteria for removal. Typically air leak is not seen after tracheoplasty unless lung parenchyma was lacerated during dissection of adhesions or with aggressive retraction. Nearly half of patients will discharge directly home, with the remaining half split nearly evenly between discharging home with visiting nurse services and discharging to a rehabilitation facility.
Pain is managed initially via the epidural catheter and is transitioned to oral narcotic medication. In general nonsteroidal anti-inflammatory medications are avoided given the risk of renal failure as well as some theoretical risk of impairment of scarring or fibrosis of the mesh and posterior airway membrane that facilitates stabilization.
Respiratory medications such as bronchodilators and mucolytics are continued in the postoperative period. Steroids are avoided if possible though patients on chronic low doses will maintain this after a brief pulse of stress-dose steroids. Adjunctive devices such as oxygen humidification units, incentive spirometers, and flutter valves to promote secretion mobilization may be used. Patients who require noninvasive positive airway pressure for sleep apnea may continue these devices in the postoperative period. If noninvasive positive airway pressure solely had been used to pneumatically stent the airway open due to tracheomalacia this may not be required to be continued postoperatively.
The patient’s diet is progressed cautiously in the postoperative period. Generally due to manipulation of the vagus nerve and esophagus during dissection and retraction, sips are reserved for the first postoperative day and a slow progression of diet proceeds from then on. Aspiration precautions and a formal swallowing evaluation may be necessary if hoarseness and recurrent laryngeal nerve injury is found.
Assessment of the results of tracheoplasty should be performed 3 months postoperatively, including dynamic airway CT and functional bronchoscopy (Fig. 40.1).
![]() COMPLICATIONS
COMPLICATIONS
The most common complication in the postoperative period is a respiratory infection, which occurred in 20% to 25% of patients in the largest series of tracheoplasty. This is not entirely unexpected given the high percentage (74%) of tracheoplasty patients who had a preoperative history of recurrent respiratory infections. Often these patients are colonized with more virulent or resistant bacterial strains such as Pseudomonas aeruginosa. An antecedent stent trial complicated by a stent-related infection may also increase this risk of postoperative infection. If significant purulence is encountered during intraoperative bronchoscopy, an aspirate or lavage should be taken to help guide antibiotic therapy postoperatively if necessary.
Atrial arrhythmia occurred in nearly 10% of patients. Patients are maintained on cardiac telemetry in the ward after transfer from the ICU to monitor for this. Respiratory failure requiring reintubation also occurred in 10% of patients. Preoperative pulmonary comorbidities, which may have influenced the likelihood of respiratory failure included Chronic Obstructive Pulmonary Disease (COPD) (37%), asthma (23%), and interstitial lung disease (6%). The preoperative forced expiratory volume in 1 second (FEV1) averaged 1.62 L. Tracheotomy was placed in 14% of the patients postoperatively though one-third of these tubes were placed preemptively immediately at the completion of tracheoplasty to facilitate aspiration bronchoscopy and suctioning in the postoperative period. Other morbidities included renal failure (6%), pulmonary embolus (3%), and myocardial infarction (2%). The mortality rate in this series of 63 patients was 3%.
The lack of long-term performance data regarding the implanted mesh is a caveat that warrants frank discussion with patients preoperatively. While erosion of mesh into the airway has been seen anecdotally in the immediate postoperative period and is thought to be a result of dissection and ischemic factors, it is unknown what if any risk exists for erosion of the mesh over the longer term.
![]() RESULTS
RESULTS
In the three largest series of tracheoplasty for severe acquired tracheomalacia subjective improvement was reported by patients postoperatively. While substantial long-term follow-up data are lacking, significant increases in the initial postoperative period are well substantiated. Karnofsky performance scale improved from 62 ± 12 (mean ± SD) to 76 ± 14 (p < 0.001). The American Thoracic Society dyspnea score decreased from 3.06 ± 0.9 to 1.65 ± 1.01 (p< 0.001), a clinically significant improvement in a validated measurement of dyspnea. In addition, the St. George’s Respiratory Quality of Life score demonstrated a significant improvement of 74 ± 13 to 46 ± 21 (p < 0.001). Analysis using the Transitional Dyspnea Index demonstrated that nearly 80% of patients reported improvement in the dyspnea following tracheoplasty. From a functional standpoint, the 6-minute walk distance also significantly increased postoperatively (987 ± 502 ft vs. 1,187 ± 347, p < 0.005). This represents a clinically meaningful increase in functionality.
There is some debate regarding the impact of surgical stabilization on pulmonary function testing. One smaller study reported that in 14 patients the mean percent predicted FEV1 improved from 51.2% to 73.5% after tracheoplasty (p < 0.0009) and the mean percent predicted forced vital capacity (FVC) improved from 68% to 79.8% (p < 0.01). The percent predicted peak expiratory flow rate improved from 49% to 70% after surgery as well (p < 0.0001). However, in a series of 63 patients, 37 of whom had complete sets of preoperative and postoperative measurements of FEV1, no significant difference was found (preoperative FEV1 1.62 ± 0.76 mL, postoperative FEV1 1.66 ± 0.67 mL; p = 0.29). Interestingly, in this group, 22/37 patients had improved FEV1, with a mean increase of 234 mL (range 20 to 870 mL), but 15/37 patients demonstrated no improvement or frank worsening of their postoperative FEV1, with a mean decrease of 235 mL (range 10 to 1,160 mL; one patient also had the exact same FEV1 values pre- and postoperatively). In addition, in a report on stent stabilization of the airway there was no difference in pre- and poststenting FEV1 values. This seems to corroborate the finding that PFTs are largely unreliable in quantifying or describing symptomatic tracheomalacia. It is possible that this improvement in airflow may be assessed more reliably with newer modalities such as negative expiratory pressure measurements, which have shown that stent stabilization of the malacic airway leads to elimination of tidal expiratory flow limitation.
Overall patients report satisfaction with tracheoplasty. Half of patients reported greater than 75% subjective improvement in symptoms. Nearly half of patients stated that they had an excellent result from surgery with a third as many stating that the results were good.
Progression of comorbidities, progression of malacia into untreated airways of the cervical trachea or distal bronchial tree, and recurrence of the central tracheomalacia are all possibilities that might limit the long-term benefit of tracheoplasty. This has not been studied adequately to date. Better understanding of the morphologic characteristics of tracheomalacia that might predict significant or durable benefit from tracheoplasty is another needed analysis. Finally, novel techniques in achieving airway stabilization need continued investigation, whether from the standpoint of materials, tissue engineering, surgical technique, or biologic modification.
![]() CONCLUSIONS
CONCLUSIONS
Tracheoplasty is an arduous procedure, which has significant benefit for carefully selected patients. Polypropylene mesh sutured to the posterior membrane is the most established method of achieving stabilization of the malacic airway. Future work should be directed at refining the technical aspects of the operation and improving patient selection. In addition, better objective metrics of the benefit of treatment of tracheomalacia need to be validated.
The ideal care of these complex patients involves close collaboration of thoracic surgeons, interventional pulmonologists, and chest radiologists. From the endoscopic and radiographic confirmation of diagnosis, to the trial of airway stabilization with silicone stents, to the postoperative interventions and maintenance of airway patency in the short and long term, a multidisciplinary team provides the most comprehensive care. For patients in whom tracheomalacia is the cause of significant decrement in their quality of life, tracheoplasty should markedly improve their well-being.
The evaluation and treatment of tracheomalacia is summarized as follows:
![]() Patients with symptoms or histories of significant dyspnea, intractable cough, retained secretions, or recurrent infections may have tracheomalacia
Patients with symptoms or histories of significant dyspnea, intractable cough, retained secretions, or recurrent infections may have tracheomalacia
![]() Dynamic CT scanning and functional bronchoscopy are used to confirm a diagnosis of severe tracheomalacia
Dynamic CT scanning and functional bronchoscopy are used to confirm a diagnosis of severe tracheomalacia
![]() Confounding medical conditions should be treated
Confounding medical conditions should be treated
![]() Silicone stenting is used to assess the potential benefit of permanent stabilization of the malacic airway
Silicone stenting is used to assess the potential benefit of permanent stabilization of the malacic airway
![]() Tracheoplasty is the definitive technique for stabilization of tracheomalacia
Tracheoplasty is the definitive technique for stabilization of tracheomalacia
Recommended References and Readings
Boiselle PM, O’Donnell CR, Bankier AA, et al. Tracheal collapsibility in healthy volunteers during forced expiration: Assessment with multidetector CT. Radiology. 2009;252(1):255–262.
Ernst A, Majid A, Feller-Kopman D, et al. Airway stabilization with silicone stents for treating adult tracheobronchomalacia: A prospective observational study. Chest. 2007;132(2):609–616.
Gangadharan SP, Bakhos CT, Majid A, et al. Technical aspects and outcomes of tracheobronchoplasty for severe tracheobronchomalacia. Ann Thorac Surg. 2011;91(5):1574–1581.
Gonfiotti A, Jaus MO, Barale D, et al. The first tissue-engineered airway transplantation: 5-year follow-up results. Lancet. 2014; 383(9913):238–244.
Handa H, Miyazawa T, Murgu SD, et al. Novel multimodality imaging and physiologic assessments clarify choke-point physiology and airway wall structure in expiratory central airway collapse. Respir Care.2012;57(4):634–641.
Lee KS, Ashiku SK, Ernst A, et al. Comparison of expiratory CT airway abnormalities before and after tracheoplasty surgery for tracheobronchomalacia. J Thorac Imaging. 2008;23(2):121–126.
Lee KS, Sun MR, Ernst A, et al. Comparison of dynamic expiratory CT with bronchoscopy for diagnosing airway malacia: A pilot evaluation. Chest. 2007;131(3):758–764.
Loring SH, O’Donnell CR, Feller-Kopman DJ, et al. Central airway mechanics and flow limitation in acquired tracheobronchomalacia. Chest. 2007;131(4):1118–1124.
Majid A, Sosa AF, Ernst A, et al. Pulmonary function and flow-volume loop patterns in patients with tracheobronchomalacia. Respir Care. 2013;58(9):1521–1526.
Murgu SD, Colt HG. Tracheobronchomalacia and excessive dynamic airway collapse. Respirology. 2006;11(4):388–406.
Murgu SD, Colt HG. Description of a multidimensional classification system for patients with expiratory central airway collapse. Respirology. 2007;12(4):543–550.
Wright CD, Grillo HC, Hammoud ZT, et al. Tracheoplasty for expiratory collapse of central airways. Ann Thorac Surg. 2005;80(1):259–266.
Zopf DA, Flanagan CL, Wheeler M, et al. Treatment of severe porcine tracheomalacia with a 3-dimensionally printed, bioresorbable, external airway splint. JAMA Otolaryngol Head Neck Surg.2014;140(1):66–71.