Edward C. Li and James M. Hoffman
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Identify common causes of anemia.
2. Describe common signs and symptoms of anemia.
3. Discuss the appropriate diagnostic evaluation to determine anemia type and guide therapeutic decisions.
4. State the desired therapeutic outcomes for patients with anemia.
5. Compare and contrast the various oral and parenteral iron preparations.
6. Explain the optimal use of folic acid and vitamin B12 in patients with macrocytic anemia.
7. Evaluate the proper use of epoetin and darbepoetin in anemia patients with cancer and kidney disease.
8. Recommend a specific treatment regimen for anemia considering the underlying cause of anemia and patient-specific variables.
9. Develop a plan to monitor the outcomes of anemia pharmacotherapy.
KEY CONCEPTS
![]() Anemia is a reduction below normal in the concentration of hemoglobin (Hgb) in the body that results in a reduction of the oxygen-carrying capacity of the blood.
Anemia is a reduction below normal in the concentration of hemoglobin (Hgb) in the body that results in a reduction of the oxygen-carrying capacity of the blood.
![]() Common signs and symptoms of anemia include fatigue, lethargy, dizziness, shortness of breath, headache, edema, and tachycardia.
Common signs and symptoms of anemia include fatigue, lethargy, dizziness, shortness of breath, headache, edema, and tachycardia.
![]() A standard initial laboratory evaluation for anemia includes a CBC (evaluation of the serum Hgb and hematocrit [Hct] concentration, white blood cell count, platelets), measurement of the red blood cell (RBC) count and size, and review of peripheral smear.
A standard initial laboratory evaluation for anemia includes a CBC (evaluation of the serum Hgb and hematocrit [Hct] concentration, white blood cell count, platelets), measurement of the red blood cell (RBC) count and size, and review of peripheral smear.
![]() The goal of anemia therapy is to increase Hgb, which will improve red cell oxygen-carrying capacity, alleviate symptoms, and prevent anemia complications.
The goal of anemia therapy is to increase Hgb, which will improve red cell oxygen-carrying capacity, alleviate symptoms, and prevent anemia complications.
![]() The underlying cause of anemia (e.g., blood loss; iron, folic acid, or B12 deficiency; or chronic disease) must be determined and used to guide therapy.
The underlying cause of anemia (e.g., blood loss; iron, folic acid, or B12 deficiency; or chronic disease) must be determined and used to guide therapy.
![]() In patients with iron-deficiency anemia (IDA), appropriate oral iron therapy that delivers sufficient elemental iron should be attempted before giving parenteral iron.
In patients with iron-deficiency anemia (IDA), appropriate oral iron therapy that delivers sufficient elemental iron should be attempted before giving parenteral iron.
![]() Anemia from vitamin B12 or folic acid deficiency is treated effectively by replacing the missing nutrient.
Anemia from vitamin B12 or folic acid deficiency is treated effectively by replacing the missing nutrient.
![]() In cancer and kidney disease patients with anemia, therapy with epoetin or darbepoetin can increase Hgb, decrease transfusion requirements, and improve quality of life, but this therapy has safety risks and must be carefully monitored.
In cancer and kidney disease patients with anemia, therapy with epoetin or darbepoetin can increase Hgb, decrease transfusion requirements, and improve quality of life, but this therapy has safety risks and must be carefully monitored.
![]() After treatment, patients should be monitored for symptom resolution, Hgb concentration, and adverse effects.
After treatment, patients should be monitored for symptom resolution, Hgb concentration, and adverse effects.
INTRODUCTION
![]() Anemia is a reduction in the concentration of hemoglobin (Hgb) that results in a reduced oxygen-carrying capacity of the blood. Some patients with anemia may be asymptomatic initially, but eventually, the lack of oxygen to the tissues could result in fatigue, lethargy, shortness of breath, headache, edema, and tachycardia. Common causes of anemia include blood loss, decreased production of red blood cells (RBCs), increased destruction of RBCs, or some combination of these factors. Determination of the underlying cause of anemia is essential for successful management. Appropriate treatment of anemia will result in an increase in Hgb, with a corresponding increase in oxygen-carrying capacity and reduction in symptoms.
Anemia is a reduction in the concentration of hemoglobin (Hgb) that results in a reduced oxygen-carrying capacity of the blood. Some patients with anemia may be asymptomatic initially, but eventually, the lack of oxygen to the tissues could result in fatigue, lethargy, shortness of breath, headache, edema, and tachycardia. Common causes of anemia include blood loss, decreased production of red blood cells (RBCs), increased destruction of RBCs, or some combination of these factors. Determination of the underlying cause of anemia is essential for successful management. Appropriate treatment of anemia will result in an increase in Hgb, with a corresponding increase in oxygen-carrying capacity and reduction in symptoms.
EPIDEMIOLOGY AND ETIOLOGY
Anemia is a common condition, and the prevalence of anemia varies widely based on age, gender, race/ethnicity, and comorbid conditions. Studies have been done in the United States to describe differences in the prevalence of anemia in various populations.1 The prevalence of anemia in children (ages 1–16 years) was 6% to 9%, but the prevalence of anemia increases to approximately 11% in adults over age 65 years and to at least 20% in adults 85 years of age and older. Anemia is generally more common in women, particularly during their reproductive years (ages 17–49) occurring in 12% of this group. However, in the same age range, only 2% of men are anemic. In the population over age 65, non-Hispanic whites and Mexican Americans had similar prevalence of anemia (9% and 10.4%, respectively), but anemia was significantly more common in non-Hispanic blacks with a prevalence of 27.8%.
Comorbid conditions can increase the risk of anemia substantially. Anemia is especially common in cancer patients receiving chemotherapy and patients with chronic kidney disease (CKD). The incidence of anemia in cancer patients varies based on tumor type and the level of myelosuppression of the chemotherapy regimen. For instance, severe anemia (Hgb 7.9 g/dL or less [79 g/L or 4.9 mmol/L]) occurs in at least 75% of patients who receive a common lymphoma chemotherapy regimen but severe anemia may occur in less than 10% of patients who receive common breast cancer chemotherapy regimen.2 Overall, retrospective reviews demonstrate that chemotherapy-induced anemia is most common in patients with lymphomas, lung tumors, and ovarian tumors, with an incidence of 50% to 60%. In addition, up to 60% of patients with serious kidney disease have anemia, which demonstrates how common anemia is in patients with CKD.3
The causes of anemia can be divided into three main categories: decreased production, increased destruction, and blood loss. Drug therapy is the mainstay of treatment for anemias caused by reduced production of erythrocytes and will be the focus of this chapter, and anemias due to destruction of erythrocytes will not be discussed.
A decrease in erythrocyte production can be multifactorial. Nutritional deficiencies (such as iron, vitamin B12, and folic acid) are common causes that are often easily treatable. In addition, patients with cancer and CKD are at risk for developing a hypoproductive anemia. Furthermore, patients with chronic immune-related diseases (such as rheumatoid arthritis and systemic lupus erythematosus) can develop anemia as a complication of their disease. Anemia related to chronic inflammatory conditions is typically termed anemia of chronic disease.
PATHOPHYSIOLOGY
Erythropoiesis
Erythropoiesis is a process that starts with a pluripotent stem cell in the bone marrow that differentiates into an erythroid colony-forming unit (CFU-E)4 (Fig. 66–1). The development of these cells depends on stimulation from the appropriate growth factors, primarily the renally produced hormone erythropoietin (EPO). Other cytokines involved include granulocyte-monocyte colony-stimulating factor (GM-CSF) and interleukin 3 (IL-3). These CFU-Es then differentiate into reticulocytes and cross from the bone marrow into the peripheral blood. Throughout this process, the cells gradually accumulate more Hgb and lose their nuclei.4 Finally, these reticulocytes mature into erythrocytes after 1 to 2 days in the bloodstream.
Patient Encounter 1, Part 1
A 74-year-old Caucasian woman with a past medical history significant for hypertension and type II diabetes mellitus presents to her primary care physician complaining of shortness of breath and fatigue for the past few days. She denies any recent bleeding manifestations, including bright red blood per rectum, hemoptysis, melena, or epistaxis. She denies any fevers, chills, nausea, vomiting, or recent weight loss. A CBC was taken and revealed Hgb of 9.3 g/dL (93 g/L or 5.8 mmol/L). Recent colonoscopy and endoscopy were normal. The patient weighs 54 kg (119 lb).
Is this patient anemic? If so, what are possible causes of anemia?
What additional laboratory assessments are required to make an appropriate therapeutic plan?
How would the requested laboratory parameter(s) aid your decision making?
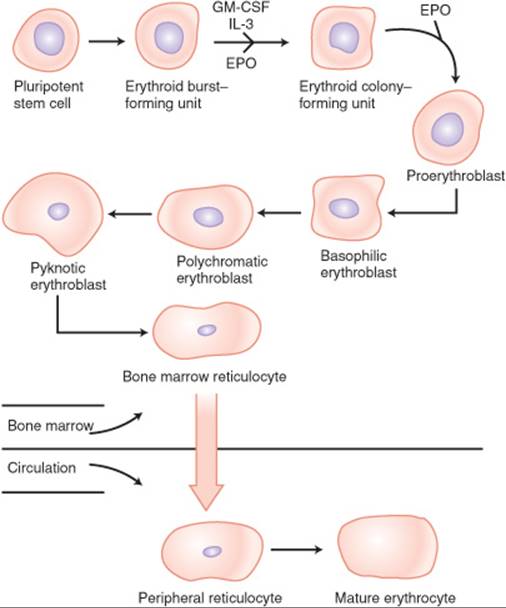
FIGURE 66–1. The process of erythropoiesis.
Hypoproliferative or Decreased-Production Anemias
Nutritional
Deficiencies in nutrients such as folic acid and vitamin B12 may hinder the process of erythrocyte maturation.4,5 Folic acid and vitamin B12 are required for the formation of DNA. When these nutrients are decreased, DNA synthesis is inhibited, and consequently, erythrocyte maturation also is inhibited.4,5 Poor diet can be a contributor to the deficiencies in these nutrients. Similarly, patients with a condition called pernicious anemia are unable to absorb B12 via their GI tract due to a lack in a glycoprotein called intrinsic factor. This glycoprotein binds to vitamin B12 and facilitates its absorption in the ileum. This condition results in B12 deficiency despite adequate dietary B intake.6
Iron is also a vital nutrient in the development of functioning erythrocytes as it is essential for the formation of Hgb. Lack of iron leads to a decrease in Hgb synthesis and ultimately RBCs. Normal homeostasis of iron transport and metabolism is depicted in Figure 66–2.7 Approximately 1 to 2 mg of iron is absorbed through the duodenum each day, and the same amount is lost via blood loss, desquamation of mucosal cells, or menstruation.
Since there is no true “excretion” of iron from the body, iron-deficiency anemia (IDA) typically occurs because of either inadequate absorption of iron or excess blood loss. Inadequate absorption may occur in patients who have congenital or acquired intestinal diseases, such as inflammatory bowel disease, celiac disease, or bowel resection. Achlorhydria and diets poor in iron also may contribute to iron deficiency states. In contrast, iron deficiency also may occur in patients who exhibit a higher rate of iron loss from the body. This is manifested in blood loss, either from the GI system, menstruation, cancer, or trauma.7
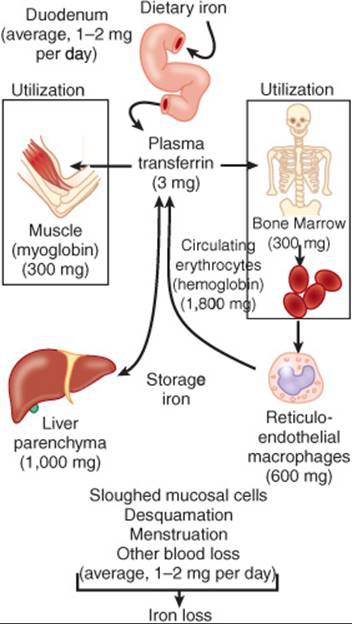
FIGURE 66–2. The distribution of iron use in adults. (From Ref. 7.)
Hypoproliferative Marrow
Patients with chronic diseases exhibit a different patho-physiologic mechanism of disease. For example, patients with cancer may suffer from anemia because of chemotherapy and/or the tumor effects on the marrow itself. Chemotherapy may cause destruction of highly proliferating stem cells, thereby decreasing the production of mature erythrocytes.8 In addition, cancer can cause anemia via hemorrhage, replacing normal bone marrow with malignant cells, and releasing cytokines that lead to decreased EPO production. Both of these scenarios can lead to anemia from a hypoproliferative marrow.8
Decreased EPO Production or Response
Patients with CKD suffer from a decrease in erythropoietin production because EPO is produced mainly in the kidneys.4,5 In patients with anemia of chronic disease, there is a blunted EPO production as well as a diminished response to EPO.9 Anemia of chronic disease also affects iron homeostasis, causing iron sequestration into storage sites and decreasing the amount available to the rest of the body.9
TREATMENT
Desired Outcomes
![]() The goal of anemia therapy is to increase the Hgb level, which will improve red cell oxygen-carrying capacity, alleviate symptoms, and prevent complication from anemia. Normal Hgb values are 14 to 17.5 g/dL (140-175 g/L or 8.69-10.9 mmol/L) for males and 12. 3 to 15.3 g/dL (123-153 g/L or 7.63-9.5 mmol/L) for females. It is important to note that continuation of a patient’s therapy should be assessed primarily by resolution of clinical signs and symptoms. Patients who experience a resolution in their symptoms such as shortness of breath, tachycardia, fatigue, dizziness, and edema may not require aggressive therapy to maintain their Hgb values within normal limits. Ultimately, prevention of complications owing to anemia such as hypoxia and cardiovascular sequelae can be avoided if Hgb levels are greater than 7 g/dL (70 g/L or 4.34 mmol/L).10
The goal of anemia therapy is to increase the Hgb level, which will improve red cell oxygen-carrying capacity, alleviate symptoms, and prevent complication from anemia. Normal Hgb values are 14 to 17.5 g/dL (140-175 g/L or 8.69-10.9 mmol/L) for males and 12. 3 to 15.3 g/dL (123-153 g/L or 7.63-9.5 mmol/L) for females. It is important to note that continuation of a patient’s therapy should be assessed primarily by resolution of clinical signs and symptoms. Patients who experience a resolution in their symptoms such as shortness of breath, tachycardia, fatigue, dizziness, and edema may not require aggressive therapy to maintain their Hgb values within normal limits. Ultimately, prevention of complications owing to anemia such as hypoxia and cardiovascular sequelae can be avoided if Hgb levels are greater than 7 g/dL (70 g/L or 4.34 mmol/L).10
General Approach to the Anemic Patient
![]() The underlying cause of anemia (e.g., blood loss; iron, folic acid, or vitamin B12 deficiency; or chronic disease) must be determined and used to guide therapy (see Fig. 66–3). Subsequently, the appropriate pharmacologic treatment should be initiated based on the cause of anemia.
The underlying cause of anemia (e.g., blood loss; iron, folic acid, or vitamin B12 deficiency; or chronic disease) must be determined and used to guide therapy (see Fig. 66–3). Subsequently, the appropriate pharmacologic treatment should be initiated based on the cause of anemia.
Clinical Presentation and Diagnosis of Anemia
Signs and Symptoms
![]() Generally, the signs and symptoms of anemia are nonspecific and may include:
Generally, the signs and symptoms of anemia are nonspecific and may include:
• Fatigue, lethargy, dizziness
• Shortness of breath
• Headache
• Edema
• Tachycardia
Other findings that may be present in some patients include:
• Dry skin, chapped lips
• Nail brittleness
• Hunger for ice, starch, or clay (termed pica)
Past Medical History
Inquire about the following conditions:
• History of blood loss, such as hemorrhoids, melena, or menorrhagia (IDA)
• Malnourished or recent weight loss (vitamin B12 or folate deficiency)
• Alcoholism (folate deficiency)
• Cancer or chronic kidney disease (CKD)
• Chronic autoimmune disorders or infections, such as HIV infection or rheumatoid arthritis (anemia of chronic disease)
Physical Examination
These findings aid the clinician in determining the severity of the anemia:
• Orthostatic hypotension and tachycardia secondary to volume depletion
• Mental status changes and confusion
• Cutaneous changes such as pallor, jaundice, and nail brittleness
Laboratory Evaluation
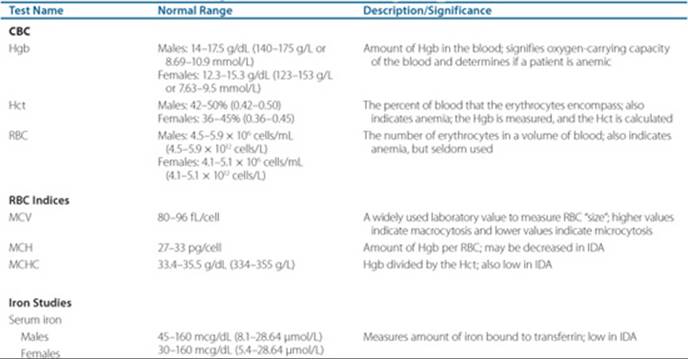
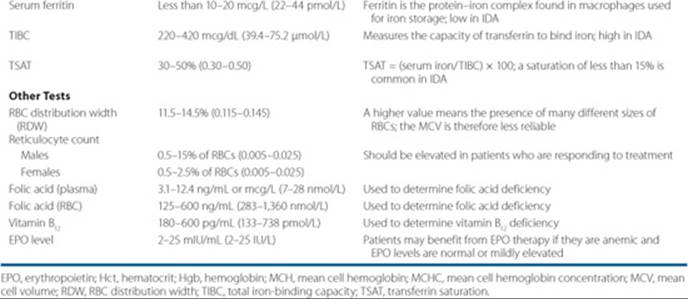
Table 66–1 describes common tests used to determine the etiology of anemia. A diagnostic and treatment algorithm for anemia is outlined in Figure 66–3.
1. ![]() A CBC is a necessary first step in evaluating a patient with anemia. If the Hgb and Hct are less than the normal range, the patient is anemic. Subsequent evaluations of RBC indices and the peripheral smear often are necessary to determine the etiology (and ultimately, the treatment) of the anemia.
A CBC is a necessary first step in evaluating a patient with anemia. If the Hgb and Hct are less than the normal range, the patient is anemic. Subsequent evaluations of RBC indices and the peripheral smear often are necessary to determine the etiology (and ultimately, the treatment) of the anemia.
2. Evaluating the mean corpuscular volume (MCV) is the next step in an anemia workup. It is classified as microcytic, normocytic, or macrocytic if the MCV is below, within, or above the normal range of 80 to 96 fL/cell, respectively.
Microcytic Anemia and Iron Evaluation
Iron studies (see Table 66–1) should be evaluated in the setting of a low MCV. These include:
• Serum iron
• Serum ferritin—the best indirect determinant of body iron stores. It is commonly decreased in patients with IDA
• Total iron-binding capacity (TIBC)—quantifies the iron-binding capacity of transferrin and is increased in IDA
• Transferrin saturation (TSAT) (serum iron/TIBC)—indicates the amount of transferrin that is bound with iron; it is lower in IDA
Macrocytic Anemia
• Evaluate folic acid and vitamin B12 levels in the setting of an elevated MCV
• Further investigation by administering radiolabeled B12 (i.e., Schilling test) to determine if lack of intrinsic factor
• Consider obtaining homocysteine and methylmalonic acid levels
Normocytic Anemia
• Evaluate reticulocytes and CBC
• High reticulocyte counts may indicate RBCs loss via acute blood loss, hemolysis, or splenic sequestration
• Low reticulocyte counts may indicate a diseased bone marrow (e.g., aplastic anemia, myelodysplasia, or leukemia), especially if the WBC and platelets are low
• High WBC/platelets may be from anemia of chronic disease, malignancy, or CKD
Nonpharmacologic Therapy
The most important nonpharmacologic treatment of anemia is the transfusion of RBCs. However, because of the risk of infection, immunosuppression, and microcirculatory complications and the high cost of the procedure, the threshold for transfusion has been debated.11 Generally, only patients requiring immediate correction such as those with acute symptoms, receive blood transfusions. Determining which patient requires immediate correction is left to the health care provider. Usually, symptomatic patients who present with a Hgb concentration of 7 to 8 g/dL (70–80 g/L or 4.34–4.96 mmol/L) are candidates for transfusion.12
Other than transfusion, nonpharmacologic therapy plays a limited role in the management of anemia. Certainly, some causes of anemia can be attributed to diets poor in iron, folic acid, or vitamin B12. However, in the United States, nutrient-poor diets are rarely the sole cause of anemia in a patient. Therefore, ingesting a diet that is rich in iron, folic acid, or vitamin B12 should be encouraged but it is rarely the sole modality of treatment. Food sources of iron, folic acid, and vitamin B12 are listed in Table 66–2. 5
Table 66–1 Pertinent Laboratory Tests in the Evaluation of Anemia
Pharmacologic Therapy
Iron-Deficiency Anemia
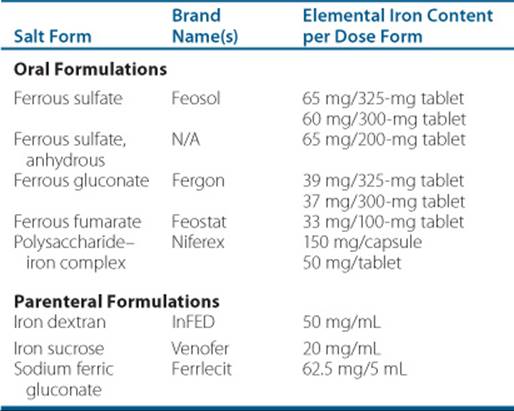
![]() The initial treatment of IDA is oral iron therapy with a goal of 200 mg of elemental iron daily for those who are able to tolerate the oral route. Many different iron products and salt forms are available. Table 66–3 lists the various salt forms of oral iron available, the amount of elemental iron in each product, and the approximate daily dose of the salt to attain 200 mg of elemental iron daily.
The initial treatment of IDA is oral iron therapy with a goal of 200 mg of elemental iron daily for those who are able to tolerate the oral route. Many different iron products and salt forms are available. Table 66–3 lists the various salt forms of oral iron available, the amount of elemental iron in each product, and the approximate daily dose of the salt to attain 200 mg of elemental iron daily.
Iron supplementation resolves anemia by replacing iron stores in the body that are necessary for RBC production and maturation. If treated properly, a response (via the presence of reticulocytosis) should be seen in 7 to 10 days, and Hgb values should rise by about 1 g/dL (10 g/L or 0.62 mmol/L) per week. Patients should be reassessed if Hgb does not increase by 2 g/dL (20 g/L or 1.24 mmol/L) in 3 weeks.
Dosing for iron should be divided equally into two to three doses daily. An empty stomach (1 hour before or 2 hours after a meal) is preferred for maximal absorption. After absorption, iron binds to transferrin in the plasma and is transported to the muscles (for myoglobin), liver (for storage), or bone marrow (for red cell production). Iron is not actively excreted from the body but is “lost” through other measures already described.7 Some studies suggest that iron absorption may be increased by adding ascorbic acid (vitamin C) to the drug regimen.13 However, administration of iron on an empty stomach and with ascorbic acid may increase the incidence of GI side effects, such as abdominal pain, nausea, and heartburn. Patients who cannot tolerate iron on an empty stomach can take it with food, but iron absorption is reduced when it is taken with food.
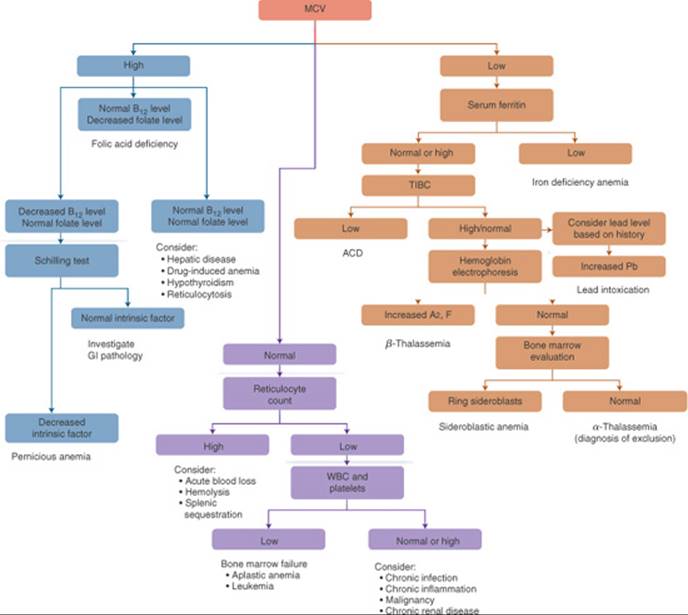
FIGURE 66–3. The anemia evaluation process. (ACD, anemia of chronic disease; MCV, mean corpuscular volume; Pb, lead; TIBC, total iron-binding capacity.)
Common toxicities associated with oral iron products include abdominal pain, nausea, heartburn, constipation, and dark stools. Drug interactions may occur with iron products, predominantly owing to iron-drug binding, resulting in decreased absorption of the interacting drug. Examples include fluoroquinolones, tetracyclines, and phenytoin. To avoid this interaction, doses of iron and the interacting drug should be separated by 2 to 4 hours.
Parenteral iron therapy may be appropriate in cases where patients are unable to tolerate the oral formulation because of toxicities or compliance. In addition, those who have IDA that has not responded to oral iron therapy (e.g., because of malabsorption) are also candidates for parenteral iron therapy.
Parenteral iron therapy currently is available in three different formulations, which are listed in Table 66–3. Iron dextran was the first parenteral iron formulation to be approved, followed by ferric gluconate, and then iron sucrose. Although these newer agents are only approved by the FDA to treat anemia associated with CKD in patients receiving EPO products, they are effective in treating IDA as well. Iron dextran is FDA approved for treating documented iron deficiency in patients who are unable to tolerate the oral formulation.
Patient Encounter 1, Part 2
Additional laboratory parameters are ordered, and the following is observed:
CBC
• WBC: 5.50 × 103/µL (5.50 × 109/L)
• Hgb: 9.3 g/dL (93 g/L or 5.77 mmol/L)
• Hct: 27.8% (0.278)
• Plt: 170 × 103/µL (170 × 109/L)
RBC
• MCV: 78 fL/cell
• MCH: 26 pg/cell
• MCHC: 33.5 g/dL (335 g/L)
Others
• RBC count: 3 M/µL (3 × 1012/L)
• RDW: 15% (0.15)
• Retic: 3% (0.03)
Iron Studies
• Serum iron: 45 mcg/dL (8.1 µmol/L)
• Serum ferritin: 7 mcg/L (15.7 pmol/L)
• TIBC: 462 mcg/dL (82.7 µmol/L)
• TSAT: 10% (0.10)
B12 and Folate
• Serum folate: 20 mcg/dL (45 nmol/L)
• Serum B12: 675 pg/mL (498 pmol/L)
Is this a macrocytic or microcytic anemia?
Specifically, what is the etiology of the anemia?
What therapy should the patient receive?
Patient Encounter 1, Part 3
The patient is diagnosed with IDA and is started on ferrous sulfate 325 mg orally three times daily to be taken on an empty stomach. Follow-up CBC 1 month later reveals a Hgb of 10 g/dL (100 g/L or 6.2 mmol/L), previously 9.3 g/dL (93 g/L or 5.77 mmol/L). The patient complains of shortness of breath on exertion and constipation. She also admits to taking only one tablet a day because of nausea.
What Hgb level would constitute a therapeutic response in this patient?
How can nausea secondary to ferrous sulfate be reduced in this patient?
What changes to this patient’s iron therapy do you recommend?
Table 66–2 Food Sources of Iron, Folic Acid, or Vitamin B12

Table 66–3 Iron Products (IV and PO) and Elemental Iron Content
The dose of iron dextran can be calculated by the following equation: dose (mL) = 0.0442 (desired Hgb–observed Hgb) × body weight + (0.26 × body weight). The body weight that should be used is lean body weight for adults and children weighing more than 15 kg and actual body weight for children weighing 5 to 15 kg. The dose in milligrams can be calculated based on a standard concentration of 50 mg elemental iron per milliliter.14 The prescribing information recommends administering iron dextran in 100-mg aliquots daily until the total dose is achieved. However, anecdotal evidence reports that the total calculated dose can be administered safely over 4 to 6 hours in 1 day. It is important to note that a test dose of iron dextran (0.5 mL over at least 30 seconds) must be administered to patients who are about to receive their first dose of iron dextran because of the risk of anaphylaxis. Patients should be monitored for signs of anaphylaxis for at least 1 hour after the test dose before administering the total dose. Other adverse effects include arthralgias, arrhythmias, hypotension, flushing, and pruritus.
Patient Encounter 1, Part 4
Because the patient is not able to tolerate oral iron therapy (constipation and nausea), changing to IV iron therapy is appropriate.
What formulation of IV iron therapy should the patient receive?
What adverse effects are associated with parenteral iron?
The use of the newer parenteral iron products, iron sucrose and ferric gluconate, for the treatment of IDA is controversial; they are currently only FDA approved for the treatment of anemia associated with CKD. However, since the newer agents are relatively safe and have a lower risk of anaphylaxis, their use is attractive in other causes of anemia. However, the high cost and lack of reimbursement for these agents may preclude their routine use in IDA patients without kidney disorders.12,13
Vitamin B12 and Folic Acid Anemia
![]() Anemia from vitamin B12 or folic acid deficiency is treated effectively by replacing the missing nutrient. Both folic acid and vitamin B12 are essential for erythrocyte production and maturation. Replacing these factors allows for normal DNA synthesis and, consequently, normal erythropoiesis.
Anemia from vitamin B12 or folic acid deficiency is treated effectively by replacing the missing nutrient. Both folic acid and vitamin B12 are essential for erythrocyte production and maturation. Replacing these factors allows for normal DNA synthesis and, consequently, normal erythropoiesis.
Vitamin B12 (cyanocobalamin) administered both orally and parenterally is equally effective in treating anemia from vitamin B12 deficiency. However, use of parenteral cyanocobalamin is the most common method of vitamin B12replacement because it may be more reliable and practical. Vitamin B12 is absorbed completely following parenteral administration, whereas oral vitamin B12 is absorbed poorly via the GI tract. Furthermore, use of parenteral vitamin B12 to treat this type of anemia may circumvent the need to perform a testing to diagnose a deficiency of intrinsic factor.
A typical cyanocobalamin dosing regimen is 800 to 1,000 mcg/day for 1 to 2 weeks, followed by 100 to 1,000 mcg/day every week until the Hgb/Hct normalizes and with subsequent maintenance of 100 to 1,000 mcg monthly for life. A common oral dosing regimen is from 1,000 to 2,000 mcg/day. If parenteral cyanocobalamin is used initially, oral vitamin B12 can be useful as maintenance therapy. Typically, the response to therapy is quick. Neurologic symptoms and megaloblastic cells disappear within a few days, and Hgb levels increase after a week of therapy.
Vitamin B12 generally is well tolerated and exhibits minimal adverse effects. Injection-site pain, pruritus, rash, and diarrhea have been reported. Drug interactions have been observed with omeprazole and ascorbic acid that decrease oral absorption.
When treating folic acid deficiency, an initial daily dose of 1 mg/day by mouth typically is effective. Absorption of folic acid generally is rapid and complete. However, patients with malabsorption syndromes may require larger doses (up to 5 mg/day). Similar to vitamin B12 deficiency, resolution of symptoms and reticulocytosis is prompt, occurring within days of commencing therapy. Hgb will start to rise after 2 weeks of therapy and may take from 2 to 4 months to resolve the deficiency completely. Afterwards, if the underlying deficiency is corrected, folic acid replacement can be discontinued. However, in cases where folic acid is consumed rapidly or absorbed poorly, chronic replacement may be required.
Folic acid is also well tolerated. Some nonspecific adverse effects include allergic reactions, flushing, malaise, and rash. Folic acid has been reported to decrease phenytoin levels by inducing its metabolism.
Anemia of Chronic Disease
Anemia of chronic disease is a term given those with underlying conditions that contribute to or cause anemia in a patient. These chronic diseases can include cancer, CKD, and other ![]() inflammatory disorders. In patients with anemia owing to cancer and CKD, therapy with epoetin or darbepoetin can increase Hgb, decrease transfusion requirements, and improve quality of life, but this therapy has safety risks and monitoring requirements that are discussed further below.
inflammatory disorders. In patients with anemia owing to cancer and CKD, therapy with epoetin or darbepoetin can increase Hgb, decrease transfusion requirements, and improve quality of life, but this therapy has safety risks and monitoring requirements that are discussed further below.
Chemotherapy-Induced Anemia
Studies have shown that in patients with chemotherapy-related anemia, therapy with the erythropoietin stimulating agents (ESA), epoetin-alfa and darbepoetin, can increase Hgb, and decrease transfusion requirements.14 Epoetin is recombinant human EPO, and darbepoetin is structurally similar to endogenous EPO. Both bind to the same receptor to stimulate RBC production. Darbepoetin differs from epoetin in that it is a glycosylated form and exhibits a longer half-life in the body, allowing for a longer dosing interval. The half-lives of a single subcutaneous injection of epoetin or darbepoetin in patients are roughly 27 and 43 hours, respectively.
A number of clinical trials and a recent meta-analysis suggest that epoetin and darbepoetin may be detrimental to cancer patients.15 This may be related to increased thrombotic events, increased tumor progression, or a combination of the two. In the meta-analysis, a total of 13,933 cancer patients from 53 trials were analyzed. ESAs increased on study mortality (combined hazard ratio [cHR] 1.17; 95% CI 1.06-1.30) and worsened overall survival (cHR 1.06; 95% CI 1.00-1.12). This corresponds to a 17% increased risk of mortality for patients treated with ESAs while on study and a 6% increase overall. Based on these findings, the use of ESAs is restricted to patients with chemotherapy-induced anemia without a curative intent. ESAs should only be used to prevent a transfusion, and should not be initiated unless the hemoglobin is less than or equal to 10 g/dL (10 g/L, 6.1 mmol/L). Patients should be monitored every 4 to 6 weeks. If the Hg has not increased by 1 g/dL (1 g/L, 0.61 mmol/L) in this time period and remains less than 10 g/dL (10 g/L, 6.1 mmol/L), a one-time dose escalation of 25% may be performed. If the Hg increases by more than 1 g/dL (1 g/L, 0.61 mmol/L) or is more than 10 g/dL (10 g/L, 6.1 mmol/L), the ESA should be discontinued. ESAs should also be discontinued within 6 weeks of completing chemotherapy, whether the anemia has resolved or not.14
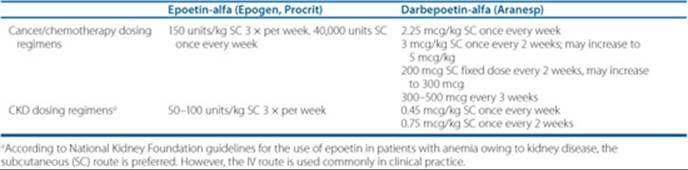
Table 66–4 EPO Products and Usual Doses for Anemia from Cancer/Chemotherapy and CKD
Cancer patients also may have concurrent iron deficiency secondary to EPO use (“functional” iron deficiency) or to cancer. Therefore, it is imperative that these patients have iron studies done to assess adequate iron stores needed to drive hematopoiesis. If the patient is determined to have suboptimal iron stores or is iron deficient, then replacement either orally or intravenously may be necessary, in addition to the use of EPO products. The use of iron in these patients is the same as discussed previously under iron-deficiency anemia (IDA).
Chronic Kidney Disease (CKD)
Patients with CKD progress through five stages of disease based on the glomerular filtration rate (GFR).16,17 Anemia is a common development in patients with CKD, and anemia evaluation and treatment should be initiated in patients with stage 3 CKD patients (GFR less than 60 mg/dL). Early treatment of anemia in patients with CKD has been associated with slower disease progression and a lower risk of death in dialysis patients.18,19 Therefore, it is essential to evaluate and treat anemia in patients before they progress to stage 5 CKD, which is a GFR of less than 15 mg/dL or patients that require dialysis.
To rule out other causes of anemia, a thorough anemia evaluation should be completed in patients with CKD.20 CKD anemia typically is a normocytic, normochromic anemia that is due to EPO deficiency. Therefore, therapy with epoetin and darbepoetin is effective in treating CKD anemia. The target Hgb value in patients with CKD is 11 to 12 g/dL (110–120 g/L or 6.82–7.44 mmol/L), but the epoetin and darbepoetin doses required for CKD anemia typically are lower than the doses for anemia from cancer/chemotherapy, and subcutaneous administration is the preferred route of administration (Table 66–4). According to the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKFK/DOQI) guidelines, epoetin doses should be increased by 50% if patients do not have an adequate response (less than 2% increase in Hct) after 2 to 4 weeks of therapy and decreased by 25% if the absolute increase in Hgb is greater than 3 g/dL (30 g/L or 1.9 mmol/L) or the target Hgb is surpassed.
Although EPO deficiency is the primary cause of CKD anemia, iron def iciency is often present as well, and it is essential to assess and monitor the CKD patient’s iron status (NKFK/DOQI guidelines). Iron stores in patients with CKD should be maintained so that TSAT is greater than 20% and serum ferritin is greater than 100 ng/mL (100 mcg/L or 225 pmol/L). If iron stores are not maintained appropriately, epoetin or darbepoetin will not be effective, and most CKD patients will require iron supplementation. Oral iron therapy can be used, but it is often ineffective, particularly in CKD patients on dialysis. Therefore, IV iron therapy is used extensively in these patients. Details of the pharmacology, pharmacokinetics, adverse effects, interactions, dose, and administration of EPO and iron products have been discussed previously.
Other Chronic Diseases
Besides anemia associated with cancer and CKD, anemia of chronic disease can result from inflammatory processes and occurs commonly in autoimmune disorders such as rheumatoid arthritis and systemic lupus erythematosus. In treating these types of anemia of chronic disease, the most important principle is treating the underlying disease. These patients also may have iron deficiency and should be treated in the manner already discussed. EPO therapy such as epoetin-alfa therapy at a dose of 150 units/kg three times a week may also be used in these patients.
OUTCOME EVALUATION
![]() After treatment, patients should be monitored for symptom and laboratory value resolution, Hgb concentration, and adverse effects. The goal of anemia therapy is to correct the underlying source of the anemia, normalize the Hgb, and alleviate associated symptoms.
After treatment, patients should be monitored for symptom and laboratory value resolution, Hgb concentration, and adverse effects. The goal of anemia therapy is to correct the underlying source of the anemia, normalize the Hgb, and alleviate associated symptoms.
Patient Encounter 2
A 65-year-old male with a diagnosis of metastatic colon cancer being treated with irinotecan presents with chemotherapy-induced anemia, with a Hgb of 8.3 g/dL (83 g/L or 5.14 mmol/L).
What discussion must be presented to the patient before ESA therapy can be initiated?
What laboratory parameters and symptoms should be assessed prior to initiating ESA therapy?
If after 4 weeks of ESA therapy, the Hgb is 9.3 g/dL (93 g/L or 5.3 mmol/L), what action should be taken?
When should ESA therapy be stopped?
• Monitor the CBC to ensure the correct Hgb titration.
• A 1 g/dL (10 g/L or 0.62 mmol/L) per week titration is desirable in patients with IDA. Reevaluate patients with an increase of less than 2 g/dL (20 g/L or 1.24 mmol/L) in 3 weeks.
• In patients with folic acid deficiency, methylmalonic acid may be normal, and homocysteine may be high. Monitor Hgb periodically, and reevaluate patients who fail to normalize Hgb levels after 2 months of therapy.
• In patients with vitamin B12 deficiency, methylmalonic acid and homocysteine levels may be high. Monitor for resolution of neurologic symptoms (i.e., confusion and paresthesias), if applicable, and Hgb levels periodically until the levels normalize.
• Do not exceed more than 1 g/dL (10 g/L or 0.62 mmol/L) every 2 weeks when using ESAs to increase Hgb. Decrease the dose of the ESA if this occurs.
• When using ESAs, do not exceed Hgb of greater than 10 g/dL (10 g/L or 6.2 mmol/L) in cancer patients because of an increased risk of death and adverse effects.
• Monitor other laboratory tests, such as mean cell volume (MCV), iron studies, and presence of reticulocytosis.
Abbreviations Introduced in This Chapter

Patient Care and Monitoring
1. Identify and treat the underlying cause of anemia.
2. Determine if immediate correction of anemia is required with a transfusion or if chronic therapy can be initiated.
3. Monitor symptoms such as fatigue, shortness of breath, lethargy, headache, edema, and tachycardia for resolution.
4. Monitor the CBC monthly.
5. When initiating ESAs, assess the patient’s iron status.
6. Monitor side effects of therapy, such as
• Oral iron: nausea, vomiting, abdominal pain, heartburn, constipation, and dark stools
• Parenteral iron: anaphylaxis (test dose required for iron dextran and observe for 1 hour after), injection-site pain/irritation, arthralgias, myalgias, flushing, malaise, and fever
• Folic acid: bad taste and nausea, rash, and allergic reactions
• Vitamin B12: hyperuricemia, hypokalemia, and sodium retention (rare)
• ESAs: hypertension (monitor blood pressure), thrombosis (e.g., DVT/PE, MI, CVA, and TIA), arthralgias, and headache

 Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Guralnik JM, Eisenstaedt RS, Ferrucci L, Klein HG, Woodman RC. Prevalence of anemia in persons 65 years and older in the United States: Evidence for a high rate of unexplained anemia. Blood 2004;104(8):2263–2268.
2. Martin M, Pienkowski T, Mackey J, et al. Adjuvant docetaxel for node-positive breast cancer. N Engl J Med 2005;352:2302–2313.
3. Xue JL, St PW, Ebben JP, Everson SE, Collins AJ. Anemia treatment in the pre-ESRD period and associated mortality in elderly patients. Am J Kidney Dis 2002;40(6):1153–1161.
4. Guyton AC, Hall JE. Red blood cells, anemia, and polycythemia. In: Guyton AC, Hall JE, eds. Textbook of Medical Physiology, 11th ed. Philadelphia: WB Saunders, 2006:419–428.
5. Kaushansky K, Kipps TJ. Hemapoietic Agents: Growth Factors, Minerals, and Vitamins. In: Brunton LL, Laza LS, Parker KL, eds. Goodman & Gilman’s the Pharmacological Basis of Therapeutics, 11th ed. New York: McGraw-Hill, 2006:1433–1466.
6. Toh BH, van Driel IR, Gleeson PA. Pernicious anemia. N Engl J Med 1997;337(20):1441–1448.
7. Andrews NC. Disorders of iron metabolism. N Engl J Med 1999;341(26):1986–1995.
8. Spivak JL. The anaemia of cancer: Death by a thousand cuts. Nat Rev Cancer 2005;5(7):543–555.
9. Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med 2005;352(10):1011–1023.
10. Aird William C. Anemia. In: Furie Bruce, Cassileth Peter A, Atkins Michael B, Mayer Robert J, eds. Clinical Hematology and Oncology, 1st ed. Philadelphia: Churchill Livingstone, 2003:232–240.
11. Petrides M. Red cell transfusion “trigger”: A review. South Med J 2003;96(7):664–667.
12. Infed (Iron Dextran) Prescribing Information. Morristown, NJ: Watson Pharma, March 2006.
13. Silverstein SB, Rodgers GM. Parenteral iron therapy options. Am J Hematol 2004;76(1):74–78.
14. National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology™—Cancer and Chemotherapy-Induced Anemia V.3. 2009. National Comprehensive Cancer Network 2009, URL: http://www.nccn.org/professionals/physician_gls/PDF/anemia.pdf.
15. Bohlius J, Schmidlin K, Brillant C, et al. Erythropoieten or darbepoieten for patients with cancer—meta-analysis based on individual patient data. Cochrane Database Syst Rev 2009;8:CD007303.
16. Levey AS, Coresh J, Balk E, et al. National Kidney Foundation practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Ann Intern Med 2003;139(2):137–147.
17. Fink J, Blahut S, Reddy M, Light P. Use of erythropoietin before the initiation of dialysis and its impact on mortality. Am J Kidney Dis 2001;37(2):348–355.
18. Jungers P, Choukroun G, Oualim Z, Robino C, Nguyen AT, Man NK. Beneficial influence of recombinant human erythropoietin therapy on the rate of progression of chronic renal failure in predialysis patients. Nephrol Dial Transplant 2001;16(2):307–312.
19. Eschbach J, DeOreo PB, Adamson J, et al. NKF-K/DOQI Clinical Practice Guidelines. National Kidney Foundation—Kidney Disease Outcomes Quality Initiative (NKF K/DOQI) 2000, Available from: URL: http://www.kidney.org/professionals/kdoqi/guidelines_updates/doqi_uptoc.html#an.
20. KDOQ1; National Kidney Foundation. Am J Kid Dis 2006;47:S109–S116.