Charles A. Peloquin and Rocsanna Namdar
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Compare the risk for active tuberculosis (TB) disease among patients based on their age, immune status, place of birth, and time since exposure to an active case.
2. Design, evaluate, and assess an appropriate therapeutic plan for an immunocompetent, immunocompromised, pregnant, and pediatric patient with pulmonary TB.
3. Assess the effectiveness of therapy in TB patients.
4. Describe the common and important adverse drug effects caused by TB drugs.
5. Select patients for whom therapeutic drug monitoring (TDM) may be valuable and identify the necessary laboratory monitoring parameters for patients on antituberculosis medications.
6. Design, evaluate, and assess appropriate regimens for the treatment of latent TB infection (LTBI) in all patient populations.
7. Design a therapeutic plan for a patient with TB meningitis or TB osteomyelitis.
KEY CONCEPTS
![]() Tuberculosis (TB) is the most prevalent communicable infectious disease on earth and remains out of control in many developing nations. These nations require medical and financial assistance from developed nations in order to control the spread of TB globally.
Tuberculosis (TB) is the most prevalent communicable infectious disease on earth and remains out of control in many developing nations. These nations require medical and financial assistance from developed nations in order to control the spread of TB globally.
![]() In the United States, TB disproportionately affects ethnic minorities as compared with whites, reflecting greater ongoing transmission in ethnic minority communities. Additional TB surveillance and preventive treatment are required within these communities.
In the United States, TB disproportionately affects ethnic minorities as compared with whites, reflecting greater ongoing transmission in ethnic minority communities. Additional TB surveillance and preventive treatment are required within these communities.
![]() Coinfection with HIV and TB accelerates the progression of both diseases, thus requiring rapid diagnosis and treatment of both diseases.
Coinfection with HIV and TB accelerates the progression of both diseases, thus requiring rapid diagnosis and treatment of both diseases.
![]() Mycobacteria are slow-growing organisms; in the laboratory, they require special stains, special growth media, and long periods of incubation to isolate and identify.
Mycobacteria are slow-growing organisms; in the laboratory, they require special stains, special growth media, and long periods of incubation to isolate and identify.
![]() TB can produce atypical signs and symptoms in infants, the elderly, and immunocompromised hosts, and it can progress rapidly in these patients.
TB can produce atypical signs and symptoms in infants, the elderly, and immunocompromised hosts, and it can progress rapidly in these patients.
![]() Latent tuberculosis infection (LTBI) can lead to reactivation disease years after the primary infection occurred.
Latent tuberculosis infection (LTBI) can lead to reactivation disease years after the primary infection occurred.
![]() The patient suspected of having active TB disease must be isolated until the diagnosis is confirmed and he or she is no longer contagious. Often, isolation takes place in specialized “negative pressure” hospital rooms to prevent the spread of TB.
The patient suspected of having active TB disease must be isolated until the diagnosis is confirmed and he or she is no longer contagious. Often, isolation takes place in specialized “negative pressure” hospital rooms to prevent the spread of TB.
![]() Isoniazid and rifampin are the two most important TB drugs; organisms resistant to both these drugs (multidrug resistant tuberculosis [MDR-TB]) are much more difficult to treat.
Isoniazid and rifampin are the two most important TB drugs; organisms resistant to both these drugs (multidrug resistant tuberculosis [MDR-TB]) are much more difficult to treat.
![]() Never add only a single antituberculosis drug to a failing regimen for active TB!
Never add only a single antituberculosis drug to a failing regimen for active TB!
![]() Directly observed therapy (DOT) should be used whenever possible to reduce treatment failures and the selection of drug-resistant isolates.
Directly observed therapy (DOT) should be used whenever possible to reduce treatment failures and the selection of drug-resistant isolates.
Worldwide, tuberculosis (TB) kills about 1.5 million people each year, more than any other infectious organism. TB is caused by Mycobacterium tuberculosis, it presents either as latent TB infection (LTBI) or as progressive active disease.1 The latter typically causes progressive destruction of the lungs, leading to death in most patients who do not receive treatment. Currently, one-third of the world’s population is infected, and drug resistance is increasing in many areas.1
EPIDEMIOLOGY
![]() Roughly one of every three people on earth is infected by Mycobacterium tuberculosis.1–3 The distribution is uneven, with the highest incidences found in southern Asia and sub-Saharan Africa. In the United States, about 13 million people have LTBI, evidenced by a positive skin test (purified protein derivative ([PPD]) but no signs or symptoms of disease. Such patients have roughly a 1 in 10 chance of active disease during their lives, with the greatest risk in the first 2 years after infection. Active disease occurs in over 13,000 Americans each year, resulting in about 1,500 deaths.4 (For detailed data analysis, visit the Centers for Disease Control and Prevention [CDC] website at www.cdc.gov/nchstp/tb.)
Roughly one of every three people on earth is infected by Mycobacterium tuberculosis.1–3 The distribution is uneven, with the highest incidences found in southern Asia and sub-Saharan Africa. In the United States, about 13 million people have LTBI, evidenced by a positive skin test (purified protein derivative ([PPD]) but no signs or symptoms of disease. Such patients have roughly a 1 in 10 chance of active disease during their lives, with the greatest risk in the first 2 years after infection. Active disease occurs in over 13,000 Americans each year, resulting in about 1,500 deaths.4 (For detailed data analysis, visit the Centers for Disease Control and Prevention [CDC] website at www.cdc.gov/nchstp/tb.)
M. tuberculosis is transmitted from person to person by coughing or sneezing.2,6,18 This produces small particles known as droplet nuclei that float in the air for long periods of time. Each droplet contains one to three organisms. Thirty percent of individuals with prolonged contact with an infectious TB patient become infected.
Risk Factors for Infection
Location and Place of Birth
California, New York, Florida, and Texas accounted for 48% of all TB cases in 2006, reflecting the high immigration rates into these states.4 TB is most prevalent in large urban areas, exacerbated by crowding in poor immigrant neighborhoods, where 57% of all U.S. cases were found in 2006.3,4 Mexico, the Philippines, Vietnam, India, China, Haiti, Guatemala, and South Korea account for the largest numbers of these immigrants.4 Those in close contact with patients with active pulmonary TB are most likely to become infected.2,3 These include family members, coworkers, or coresidents in places such as prisons, shelters, and nursing homes.
Race, Ethnicity, Age, and Gender
![]() In the United States, the incidence of TB is more concentrated in nonwhite individuals. In 2007, non-Hispanic blacks accounted for 26% of all TB cases, followed by Hispanics at 29%.5 Asians and Pacific Islanders accounted for 26%, whereas non-Hispanic whites accounted for only 17% of the new TB cases.5 TB is most common among people 25 to 44 years of age (32% of all cases), followed by those 45 to 64 years of age (30%) and 65 or more years of age (19%).
In the United States, the incidence of TB is more concentrated in nonwhite individuals. In 2007, non-Hispanic blacks accounted for 26% of all TB cases, followed by Hispanics at 29%.5 Asians and Pacific Islanders accounted for 26%, whereas non-Hispanic whites accounted for only 17% of the new TB cases.5 TB is most common among people 25 to 44 years of age (32% of all cases), followed by those 45 to 64 years of age (30%) and 65 or more years of age (19%).
Coinfection With HIV
![]() HIV is the most important risk factor for active TB because the immune deficit prevents patients from containing the initial infection.2,3,5,6 Roughly 10% of TB patients in the United States are coinfected with HIV, and roughly 20% of TB patients ages 25 to 44 years are coinfected with HIV.4,5 Consistent with HIV in general, HIV-associated TB is most common among 25 to 44 year olds. Substance abuse and other risk factors are shared among some TB and HIV-infected individuals, promoting the spread of both diseases.2,7,8
HIV is the most important risk factor for active TB because the immune deficit prevents patients from containing the initial infection.2,3,5,6 Roughly 10% of TB patients in the United States are coinfected with HIV, and roughly 20% of TB patients ages 25 to 44 years are coinfected with HIV.4,5 Consistent with HIV in general, HIV-associated TB is most common among 25 to 44 year olds. Substance abuse and other risk factors are shared among some TB and HIV-infected individuals, promoting the spread of both diseases.2,7,8
Patient Encounter 1
HPI: AF is a 56-year-old man who presents to the medical clinic complaining of a 1-month history of a persistent cough that has become productive over the past 2 weeks. He also complains of malaise, fever, night sweats, and a 6-kg (13-lb) weight loss over the past 2 months.
PMH: Type II diabetes mellitus (NIDDM)—well controlled; hypertension (HTN) × 5 years—well controlled
FH: Mother and father died in an MVA 10 years ago; one brother, age 54, is HIV positive and lives with the patient; one sister, age 50, is alive and has had breast cancer
SH: Single, one daughter. He works as an undercover agent and just returned from an operation in Cambodia. He denies smoking or IV drug use. He had a 20-year history of alcohol abuse but has been sober for 10 years.
Meds: Lisinopril 20 mg daily; amlodipine 5 mg daily; metformin 500 mg twice daily. Patient reports that he tries to be compliant with his therapies and takes them regularly except when he is unable to get his refills; over the past 2 months, he has gone 3 to 4 days without medication.
Allergies: NKDA
What information is suggestive of TB?
What factors place this patient at increased risk for acquiring TB?
Risk Factors for Disease
Once infected with M. tuberculosis, a person’s lifetime risk of active TB is about 10%, with about half this risk evident during the first 2 years after infection.2,3,6 Young children, the elderly, and immunocompromised patients have greater risks. HIV-infected patients with M. tuberculosis infection are roughly 100 times more likely to develop active TB than normal hosts owing to the lack of normal cellular immunity.3,9
ETIOLOGY
![]() Microscopic examination of infected material (“smear”) detects about 8 to 10 × 103 mm3/organisms of specimen using the older AFB (acid-fast bacillus) stain. The newer auraminerhodamine fluorsecent technique is one-third more sensitive. A smear-negative patient still can grow M tuberculosis on culture, which is more sensitive than either staining technique. Unfortunately, culture is much slower than staining due to the doubling time of the bacilli of about 20 hours. Further, microscopic examination cannot determine which of over 90 mycobacterial species is present. The usual practice is to assume the worst (TB) until confirmed by genetic probe or positive culture.
Microscopic examination of infected material (“smear”) detects about 8 to 10 × 103 mm3/organisms of specimen using the older AFB (acid-fast bacillus) stain. The newer auraminerhodamine fluorsecent technique is one-third more sensitive. A smear-negative patient still can grow M tuberculosis on culture, which is more sensitive than either staining technique. Unfortunately, culture is much slower than staining due to the doubling time of the bacilli of about 20 hours. Further, microscopic examination cannot determine which of over 90 mycobacterial species is present. The usual practice is to assume the worst (TB) until confirmed by genetic probe or positive culture.
Culture and Susceptibility Testing
Susceptibility testing is essential for directing proper treatment. The most common agar method, known as the proportion method, takes many weeks to produce results. The Bactec and newer mycobacterial growth indicator tube (MGIT) systems use liquid media and detect live mycobacteria in about 2 weeks.1,10,11 Rapid-identification tests include nucleic acid probes and DNA fingerprinting using restriction fragment length polymorphism (RFLP) analysis, and polymerase chain reaction (PCR).1,6,10,12–14 These tests differentiate among mycobacterial species but currently cannot provide susceptibility data. New tests looking for specific mutations associated with drug resistance may facilitate rapid drug therapy decisions in the future. Nitrate reductase assays and porous ceramic support systems are among other rapid drug susceptibility testing techniques currently being investigated.15,16
PATHOPHYSIOLOGY
Primary Infection
Primary infection usually results from inhaling droplet nuclei that contain M. tuberculosis.2,6,17 The progression to clinical disease depends on three factors: (a) the number of M. tuberculosis organisms inhaled (infecting dose), (b) the virulence of these organisms, and (c) the host’s cell-mediated immune response.2,4,6,12,18,19 If pulmonary macrophages inhibit or kill the bacilli, the infection is aborted.18 If not, M. tuberculosis eventually spreads throughout the body through the bloodstream.2,6,18 M. tuberculosis most commonly infects the posterior apical region of the lungs, where conditions are most favorable for its survival.
T lymphocytes become activated over the course of 3 to 4 weeks, producing interferon-γ (IFN-γ) and other cytokines. These stimulate microbicidal macrophages to surround the tuberculous foci and form granulomas to prevent further extension.18 At this point, the infection is largely under control, and bacillary replication falls off dramatically. Any remaining mycobacteria are believed to reside primarily within granulomas or within macrophages that have avoided detection and lysis. Over 1 to 3 months, tissue hypersensitivity occurs, resulting in a positive tuberculin skin test.2,6,17 ![]() Progressive primary diseaseoccurs in roughly 5% of patients, especially children, the elderly, and immunocompromised patients.20,21 This presents as a progressive pneumonia and frequently spreads, leading to meningitis and other severe forms of TB, even before their skin tests become positive.20
Progressive primary diseaseoccurs in roughly 5% of patients, especially children, the elderly, and immunocompromised patients.20,21 This presents as a progressive pneumonia and frequently spreads, leading to meningitis and other severe forms of TB, even before their skin tests become positive.20
Reactivation Disease
![]() About 10% of infected patients develop reactivation TB, with half occurring in the first 2 years after infection.2,6,12 Upper lobe pulmonary disease is the most common (85% of cases).2 Caseating granulomas result from the vigorous immune response, and liquefaction leads to local spread. Eventually, a pulmonary cavity results, and this provides a portal to the outside that allows for person-to-person spread. Bacterial counts in the cavities can be as high as 1011/L of cavitary fluid (108/mL).2,18 Prior to the chemotherapy era, pulmonary TB usually was associated with hypoxia, respiratory acidosis, and eventually death.
About 10% of infected patients develop reactivation TB, with half occurring in the first 2 years after infection.2,6,12 Upper lobe pulmonary disease is the most common (85% of cases).2 Caseating granulomas result from the vigorous immune response, and liquefaction leads to local spread. Eventually, a pulmonary cavity results, and this provides a portal to the outside that allows for person-to-person spread. Bacterial counts in the cavities can be as high as 1011/L of cavitary fluid (108/mL).2,18 Prior to the chemotherapy era, pulmonary TB usually was associated with hypoxia, respiratory acidosis, and eventually death.
Extrapulmonary and Miliary Tuberculosis
Caseating granulomas, regardless of location, can undergo liquefaction, spread tubercle bacilli and cause symptoms.2,6 Because of muted or altered symptoms, the diagnosis of TB is difficult and often delayed in immunocompromised hosts.2,3,6 HIV-infected patients may present with only extrapulmonary TB, which is uncommon in HIV-negative persons. A widely disseminated form of the disease called miliary TBcan occur, particularly in children and immunocompromised hosts, and it can be rapidly fatal.17 Immediate treatment is required.
Influence of HIV Infection on Pathogenesis
HIV infection is the most important risk factor for active TB.2,6,17 As CD4+ lymphocytes multiply in response to the mycobacterial infection, HIV multiplies within these cells and selectively destroys them, gradually eliminating the TB-fighting lymphocytes.17 HIV-infected patients coinfected with TB are at a substantially higher risk of early mortality compared with HIV-negative TB patients.22,23 Because of the large pill burden, overlapping toxicities, and paradoxical worsening of the TB when TB and HIV treatments are initiated simultaneously, most clinicians elect to begin TB treatment first.12,24 A reasonable time to begin HIV treatment is after 2 months of TB treatment, although individual circumstances often dictate the exact timing.
CLINICAL PRESENTATION
Fever, night sweats, weight loss, fatigue, and a productive cough are the classic symptoms of TB.1,2,6,19 Onset may be gradual, and the diagnosis is easily missed if the symptoms are muted, such as in the elderly.2,6,19 Progressive pulmonary disease leads to cavitation visible on x-ray. Physical examination is nonspecific but may be consistent with pneumonia. Dullness to chest percussion, rales, and increased vocal fremitus may be observed on examination. Laboratory data often are uninformative, but a modest increase in the white blood cell (WBC) count with a lymphocyte predominance can be seen.
Atypical presentations are common in patients coinfected with HIV.1,2,6,19,25 HIV-positive patients often have negative skin tests and fail to produce cavitary lesions, and fever may be absent. Symptoms for these patients range from classic pulmonary to muted and nonspecific. Extrapulmonary TB typically presents as a slowly progressive decline in organ function, and lymphadenopathy is relatively common.26,18,19 Abnormal behavior, headaches, or convulsions suggest tuberculous meningitis, although other acute CNS infections must be exlcuded.6,19
The Elderly
![]() Many clinical findings are muted in the elderly or absent altogether, so there can be considerable diagnostic uncertainty. Positive skin tests, fevers, night sweats, sputum production, or hemoptysis may be absent, making TB hard to distinguish from other bacterial or viral infections or chronic lung diseases.2,19,26,27 In contrast, mental status changes are twice as common in the elderly, and CNS disease must be considered when TB is entertained. Mortality is six times higher in the elderly in part owing to delays in diagnosis.2,19,26 Ethnic distributions of disease are different in the elderly and include more white patients because these patients often were infected decades ago, when TB was more prevalent in the United States.
Many clinical findings are muted in the elderly or absent altogether, so there can be considerable diagnostic uncertainty. Positive skin tests, fevers, night sweats, sputum production, or hemoptysis may be absent, making TB hard to distinguish from other bacterial or viral infections or chronic lung diseases.2,19,26,27 In contrast, mental status changes are twice as common in the elderly, and CNS disease must be considered when TB is entertained. Mortality is six times higher in the elderly in part owing to delays in diagnosis.2,19,26 Ethnic distributions of disease are different in the elderly and include more white patients because these patients often were infected decades ago, when TB was more prevalent in the United States.
Children
![]() Because very young children (less than 5 years old) have immature cellular immunity, TB can be particularly dangerous in this population. TB in children may present as a typical bacterial pneumonia, called progressive primary TB, and often involves the lower and middle lobes.17,19–21 Dissemination to the lymph nodes, GI and genitourinary tracts, bone marrow, and meninges is fairly common. For these reasons, bacille Calmette-Guérin (BCG)vaccinations are administered in countries where TB remains common. BCG appears to stimulate the children’s immune systems just enough to ward off the most serious forms of the disease. However, BCG does not block infection, and these same children often experience reactivation TB as young adults. Because cavitary lung lesions are uncommon, children do not spread TB readily. From the public health perspective, pediatric TB is the clearest indication of recent spread of TB.
Because very young children (less than 5 years old) have immature cellular immunity, TB can be particularly dangerous in this population. TB in children may present as a typical bacterial pneumonia, called progressive primary TB, and often involves the lower and middle lobes.17,19–21 Dissemination to the lymph nodes, GI and genitourinary tracts, bone marrow, and meninges is fairly common. For these reasons, bacille Calmette-Guérin (BCG)vaccinations are administered in countries where TB remains common. BCG appears to stimulate the children’s immune systems just enough to ward off the most serious forms of the disease. However, BCG does not block infection, and these same children often experience reactivation TB as young adults. Because cavitary lung lesions are uncommon, children do not spread TB readily. From the public health perspective, pediatric TB is the clearest indication of recent spread of TB.
DIAGNOSIS
Skin Testing
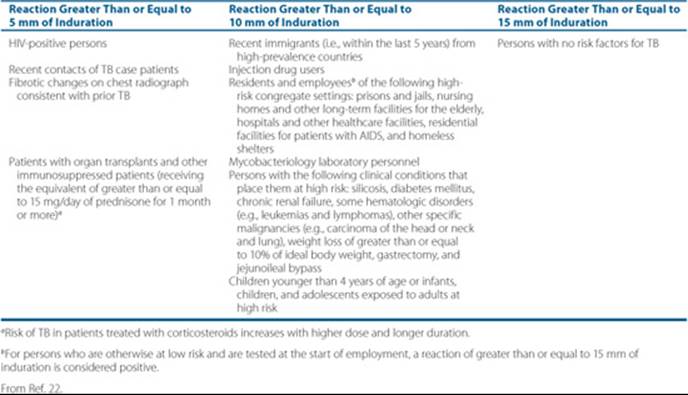
TB skin testing with the 5-TU strength of Tubersol PPD, also known as the Mantoux test, is the preferred method for skin testing.2,19,22 The product is injected into the skin (not subcutaneously) with a fine (27-gauge) needle and produces a small, raised, blanched wheal to be read by an experienced professional in 48 to 72 hours. Criteria for interpretation are listed in Table 75–1.1,2,6,19,22 The CDC does not recommend the routine use of anergy panels.22,28The “booster effect” occurs in patients who do not respond to an initial skin test but show a positive reaction if retested about a week later.19,28
Additional Tests
Morning sputum collections have the highest yield of organisms.2,10,19 Daily sputum collections over three consecutive days improve the yield of positive results. Sputum induction with aerosolized hypertonic saline may produce a diagnostic sample in patients unable to produce sputum. Bronchoscopy or aspiration of gastric fluid via a nasogastric tube may be attempted in selected patients, the latter being used more often in children.19 For patients with suspected extrapulmonary TB, samples of draining fluid, biopsies of the infected site, or both may be attempted. Blood cultures are positive occasionally, especially in AIDS patients who have low CD4 counts.19,25,29
Table 75–1 Criteria for Tuberculin Positivity by Risk Group
Patient Encounter 2
PE:
Gen: Thin, emaciated man.
VS: BP 126/78, P 90 bpm, RR 18, T 39.3°C (102.7°F), O2 sat 82% on room air, wt 51 kg (112 lb)
HEENT: PERRLA; EOMI
Neck: Supple; no lymphadenopathy, bruits, or JVD; no thyromegaly
Chest: Diffuse rhonchi, decreased breath sounds on left
CV: RRR; no murmurs, rubs, gallops
Abd: (+) BS; nontender, nondistended
Neuro: A&O × 3
Laboratory Values (U.S. Units):
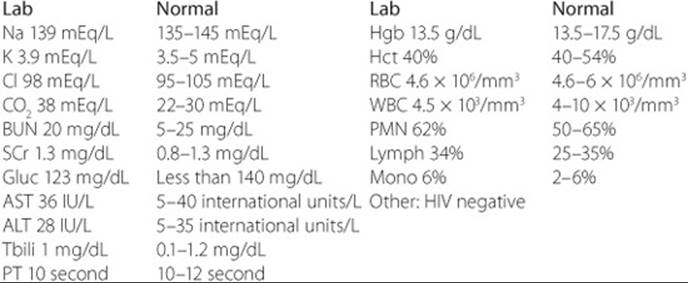
Laboratory Values (SI Units)

CXR: Profound bilateral upper lobe infiltrates with cavitation on left; small left pneumothorax
Clinical Course: The patient was admitted and placed on respiratory isolation. Three separate sputum AFB stain specimens were reported to contain 3+ AFB. A PPD tuberculin skin test was placed. Sputum samples were sent for AFB, fungi, and bacterial cultures and sensitivities. After 48 hours, the PPD skin test was read as a 12-mm area of induration.
Assessment: Active pulmonary TB; pneumothorax; HTN; type II diabetes mellitus
Which signs, symptoms, and other findings are consistent with active TB infection?
Advances in TB diagnosis include methods for rapid identification of patients with suspected TB. Improved smear microscopy, automated liquid cultures, nucleic acid amplification tests, antibody detection tests, antigen detection tests are under development.30
Interferon-gamma release assay (IGRA) is a new method for the diagnosis of LTBIs. The main advantage of this assay with respect to tuberculin skin test is the lack of cross-reaction with BCG and most nontuberculous mycobacteria. It also eliminates the need for the patient to return for test reading in 48 to 72 hours. The IGRAs cannot distinguish between latent and active TB and data are lacking in children and HIV-infected individuals.31–33
TREATMENT
General Approaches to Treatment
Monotherapy can be used only for infected patients who do not have active TB (LTBI, as shown by a positive skin test in the absence of signs or symptoms of disease). Once active disease is present, a minimum of two drugs and typically three or four drugs must be used simultaneously from the outset of treatment.2,6,12,34 For most patients, the shortest duration of treatment is 6 months, and 2 to 3 years of treatment may be necessary for advanced cases of MDR-TB.2,6,12,35 DOT is a method used to insure compliance. Patients are directly observed by a health care worker while taking their antituberculosis medication. This is also a cost-effective way to ensure completion of treatment.2,6,12,34–36
Nonpharmacologic Therapy
![]() Steps should be taken to: (a) prevent the spread of TB (respiratory isolation); (b) find where TB has already spread (contact investigation); and (c) return the patient to a state of normal weight and well-being. The older term for TB is consumption because wasting was a primary symptom of disease progression in the prechemotherapy era and remains descriptive today. Items 1 and 2 are performed by public health departments. Clinicians involved in the treatment of TB should verify that the local health department has been notified of all new cases of TB. Surgery may be needed to remove destroyed lung tissue, space-occupying infected lesions (tuberculomas), and certain extrapulmonary lesions.2,12,34
Steps should be taken to: (a) prevent the spread of TB (respiratory isolation); (b) find where TB has already spread (contact investigation); and (c) return the patient to a state of normal weight and well-being. The older term for TB is consumption because wasting was a primary symptom of disease progression in the prechemotherapy era and remains descriptive today. Items 1 and 2 are performed by public health departments. Clinicians involved in the treatment of TB should verify that the local health department has been notified of all new cases of TB. Surgery may be needed to remove destroyed lung tissue, space-occupying infected lesions (tuberculomas), and certain extrapulmonary lesions.2,12,34
Pharmacologic Therapy
Treating Latent Tuberculosis Infection (LTBI)
Isoniazid is used for treating LTBI.2,6,12,34 Typically, isoniazid 300 mg daily (5–10 mg/kg of body weight) is given alone for 9 months. Lower doses usually are less effective.2,37 The treatment of LTBI reduces a person’s lifetime risk of active TB from about 10% to about 1%22 (Table 75–2). Rifampin 600 mg daily for 4 months can be used when isoniazid resistance is suspected or when the patient cannot tolerate isoniazid.2,21,37,38 Rifabutin 300 mg daily might be substituted for rifampin in patients at high risk of drug interactions. A 12-dose, once weekly regimen of isoniazid and rifapentine, a long half-life cyclopentyl-rifampin derivative, is under study. The combination of pyrazinamide and rifampin is no longer recommended because of unacceptable rates of hepatotoxicity.39 When resistance to isoniazid and rifampin is suspected in the isolate causing infection, there is no regimen proven to be effective.2,34
Treating Active Disease
In the United States, all patients diagnosed with TB can receive treatment free of charge through the local health department, and this is encouraged because local health departments generally have the greatest expertise. Treating active TB disease requires combination chemotherapy. Generally, four drugs are given at the onset of treatment. ![]() Isoniazid and rifampin should be used together for most cases because they are the best drugs for preventing drug resistance.2,6,34,40,41 Drug susceptibility testing should be done on the initial isolate for all patients with active TB and should be used to guide the selection of drugs over the course of treatment.2,6,12,34 Susceptibility testing may be repeated in cases where the patient remains culture-positive 8 weeks or more into therapy.
Isoniazid and rifampin should be used together for most cases because they are the best drugs for preventing drug resistance.2,6,34,40,41 Drug susceptibility testing should be done on the initial isolate for all patients with active TB and should be used to guide the selection of drugs over the course of treatment.2,6,12,34 Susceptibility testing may be repeated in cases where the patient remains culture-positive 8 weeks or more into therapy.
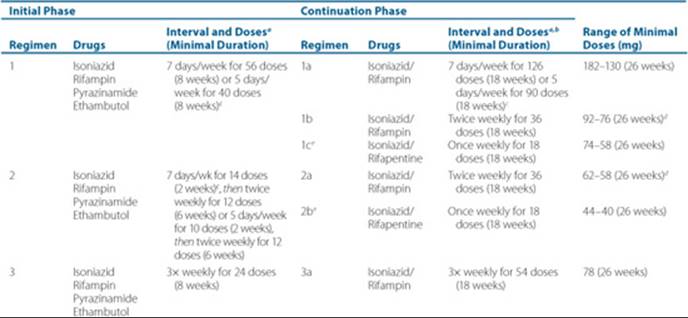
![]() The standard TB treatment regimen is isoniazid, rifampin, pyrazinamide, and ethambutol for 2 months, followed by isoniazid and rifampin for 4 months, for a total of 6 months of treatment.2,12,34 Extending treatment to 9 months of isoniazid and rifampin treatment is recommended for patients at greater risk of failure and relapse, including those with cavitation on initial chest radiograph or positive cultures at the completion of the initial 2-month phase of treatment, as well as for patients treated initially without pyrazinamide. Treatment should be continued for at least 6 months from the time that patients convert to a negative smear and culture.2,6,12,34 Some authors recommend TDM for such patients because one of the proven reasons for treatment failure is malabsorption of orally administered drugs.2,34,41,42 Table 75–3 shows the recommended treatment regimens for TB. When intermittent therapy is used, DOT is essential. Doses missed during an intermittent TB regimen decrease the efficacy of the regimen and increase the relapse rate. Further, outcomes appear to be worse for immunocompromised patients when intermittent treatment, especially twice-weekly treatment, is used. Therefore, HIV-positive TB patients should receive TB drugs at least three times weekly. When the patients’ sputum smears convert to negative, the risk of them infecting others is greatly reduced, but it is not zero.2,15,34 Such patients can be removed from respiratory isolation, but they must be careful not to cough on others and should meet only in well-ventilated places.
The standard TB treatment regimen is isoniazid, rifampin, pyrazinamide, and ethambutol for 2 months, followed by isoniazid and rifampin for 4 months, for a total of 6 months of treatment.2,12,34 Extending treatment to 9 months of isoniazid and rifampin treatment is recommended for patients at greater risk of failure and relapse, including those with cavitation on initial chest radiograph or positive cultures at the completion of the initial 2-month phase of treatment, as well as for patients treated initially without pyrazinamide. Treatment should be continued for at least 6 months from the time that patients convert to a negative smear and culture.2,6,12,34 Some authors recommend TDM for such patients because one of the proven reasons for treatment failure is malabsorption of orally administered drugs.2,34,41,42 Table 75–3 shows the recommended treatment regimens for TB. When intermittent therapy is used, DOT is essential. Doses missed during an intermittent TB regimen decrease the efficacy of the regimen and increase the relapse rate. Further, outcomes appear to be worse for immunocompromised patients when intermittent treatment, especially twice-weekly treatment, is used. Therefore, HIV-positive TB patients should receive TB drugs at least three times weekly. When the patients’ sputum smears convert to negative, the risk of them infecting others is greatly reduced, but it is not zero.2,15,34 Such patients can be removed from respiratory isolation, but they must be careful not to cough on others and should meet only in well-ventilated places.
Adjustments to the regimen should be made once the susceptibility data are available.2,12,34 Drug resistance should be expected in patients who have been treated previously for TB. Two or more drugs with in vitro activity against the patient’s isolate that were not used before should be added to the regimen as needed.2,12,34 When isoniazid and rifampin cannot be used, treatment durations typically become 2 years or more, regardless of immune status.2,12,34,41TB specialists should be consulted regarding cases of drug-resistant TB or in any setting where there is uncertainty regarding appropriate treatment.2,12,34 ![]() It is critical to avoid monotherapy, and it is critical to avoid adding only a single drug to a failing regimen.2,12,34
It is critical to avoid monotherapy, and it is critical to avoid adding only a single drug to a failing regimen.2,12,34
Table 75–2 Recommended Drug Regimens for Treatment of LTBI in Adults


Special Populations
Patients with CNS TB usually are treated for longer periods (9–12 months instead of 6 months) because the consequences of undertreatment are severe.2,12,34 TB of the bone typically is treated for 6 to 9 months, occasionally with surgical débridement.2,12,34 Drug selection is the same as for pulmonary disease. Extrapulmonary TB of the soft tissues can be treated with conventional regimens.2,12,34 TB in children may be treated with regimens similar to those used in adults, although some physicians extend treatment to 9 months.2,12,19,20,34,38,43 Pediatric doses of isoniazid and rifampin on a milligram per kilogram basis are higher than those used in adults34 (Table 75–4).
Pregnant women receive the usual treatment of isoniazid, rifampin, and ethambutol for 9 months.2,34,38,41,43 Pyrazinamide has not been studied in large numbers of pregnant women, but anecdotal data suggest that it may be safe.34B vitamins should be provided. Streptomycin, other aminoglycosides, capreomycin, and ethionamide generally are avoided because they have been associated with toxic effects on the fetus.34,44 Para-aminosalicylic acid and cycloserine are used sparingly.44 Quinolones generally are avoided in pregnancy because of concern about adverse effects on cartilage development.34,44 Although most antituberculosis drugs are excreted in breast milk, the amount of drug received by the infant through nursing is insufficient to cause toxicity. Quinolones should be avoided in nursing mothers, if possible, for the same reason as above.
Patient Encounter 3: Creating a Care Plan
Based on the information provided, what are the goals of therapy for this patient? Select and recommend a therapeutic plan for treatment of this patient’s TB infection. What drugs, dose, schedule, and duration of therapy are best for this patient? How should any contacts infected by this patient be evaluated and treated? What drugs, dose, and schedule of therapy are best for his close contacts?
Table 75–3 Drug Regimens for Culture-Positive Pulmonary Tuberculosis Caused by Drug-Susceptible Organisms

Human Immunodeficiency Virus
Patients with AIDS and other immunocompromised hosts may be managed with chemotherapeutic regimens similar to those used in immunocompetent individuals, although treatment is often extended to 9 months2,12,34 (Table 75–3). The precise duration to recommend remains a matter of debate. Highly intermittent regimens (twice or once weekly) are not recommended for HIV-positive TB patients.34 Prognosis has been particularly poor for HIV-infected patients infected with MDR-TB. Some patients with AIDS malabsorb their oral medications, and drug interactions are common.2,34,41,42 It is advisable that such patients are managed by TB-HIV experts because the challenges are many.
Renal Failure
Because they are primarily hepatically cleared, isoniazid and rifampin usually do not require dose modification in renal failure.41,44,45 Pyrazinamide and ethambutol typically are reduced to three times weekly to avoid accumulation of the parent drug (ethambutol) or metabolites (pyrazinamide).34,45 Renally cleared TB drugs include the aminoglycosides (e.g., amikacin, kanamycin, and streptomycin), capreomycin, ethambutol, cycloserine, and levofloxacin.34,35,44,45 Dosing intervals need to be extended for these drugs. Serum concentration monitoring must be performed for cycloserine to avoid dose-related toxicities in renal failure patients.37,41,42
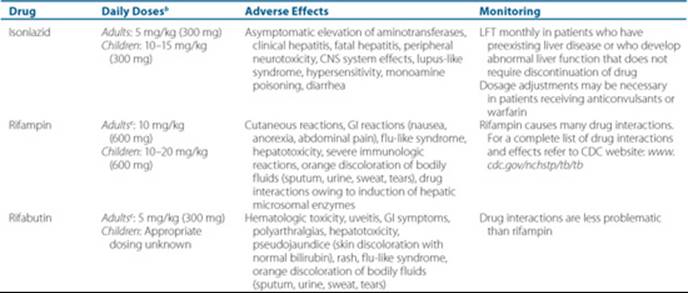
Table 75–4 Antituberculosis Drugs for Adults and Childrena

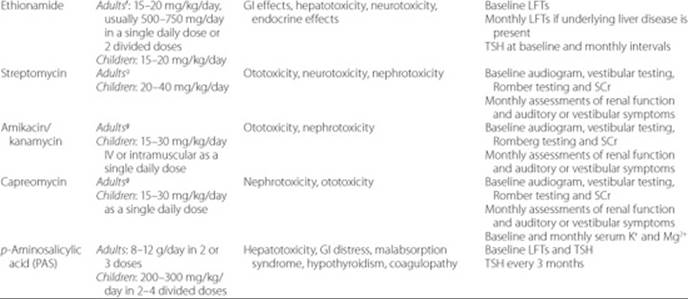


Hepatic Failure
Elevations of serum transaminase concentrations generally are not correlated with the residual capacity of the liver to metabolize drugs, so these markers cannot be used directly as guides for residual metabolic capacity. Hepatically cleared TB drugs include isoniazid, rifampin, pyrazinamide, ethionamide, and p-aminosalicylic acid.44 Ciprofloxacin and moxifloxacin are about 50% cleared by the liver. Further, isoniazid, rifampin, pyrazinamide, and to a lesser degree ethionamide, p-aminosalicylic acid, and rarely ethambutol may cause hepatotoxicity.34,41,44 These patients require close monitoring, and serum concentration monitoring may be the most accurate way to dose them.
The TB Drugs
The interested reader is referred to several other publications for more detailed information regarding these drugs.2,11,34,39–42,44–47 A summary of daily doses, adverse effects, and monitoring parameters of first- and second-line antituberculosis drugs is provided in Table 75–4.34 Isoniazid and rifampin are considered the two key drugs for the treatment of active TB, followed by pyrazinamide, which has a special role in the first 2 months of treatment. Other drugs are used to suppress the emergence of drug resistance in conjunction with the first-line drugs or for pre-existing drug-resistant TB. In general, the most important toxicity with first-line drugs is hepatotoxicity, whereas various organs may be affected by each of the second-line drugs. Recent research is placing emphasis on the potential role of quinolones such as moxifloxacin in the treatment of TB. The role of these agents in the first 2-month intensive phase of therapy is currently being evaluated. It is possible that future regimens may consider these agents part of the first-line drugs.48 Other new therapies include investigational vaccines, and investigational drugs such as PA-824, OPC67683, TMC207, and SQ109 which are in clinical trials.49,50
EVALUATION OF OUTCOMES
Effectiveness of TB therapy is determined by AFB smears and cultures. Sputum samples should be sent for AFB staining and microscopic examination (smears) every 1 to 2 weeks until two consecutive smears are negative. This provides early evidence of a response to treatment.34 Once on maintenance therapy, sputum cultures can be performed monthly until two consecutive cultures are negative, which generally occurs over 2 to 3 months. If sputum cultures continue to be positive after 2 months, drug susceptibility testing should be repeated, and serum concentrations of the drugs should be checked.
![]() The most serious problem with TB therapy is patient nonadherence to the prescribed regimens.51,52 Unfortunately, there is no reliable way to identify such patients a priori. The most effective way to achieve this end is with DOT.2,11,34 The use of DOT in noncompliant patients will be of benefit.53 DOT also provides increased opportunities to observe the patient for any apparent toxicities, thus improving overall care.
The most serious problem with TB therapy is patient nonadherence to the prescribed regimens.51,52 Unfortunately, there is no reliable way to identify such patients a priori. The most effective way to achieve this end is with DOT.2,11,34 The use of DOT in noncompliant patients will be of benefit.53 DOT also provides increased opportunities to observe the patient for any apparent toxicities, thus improving overall care.
Serum chemistries, including blood urea nitrogen (BUN), creatinine, aspartate transaminase (AST), and alanine transaminase (ALT), and a complete blood count with platelets should be performed at baseline and periodically thereafter depending on the presence of other factors that may increase the likelihood of toxicity (e.g., advanced age, alcohol abuse, and pregnancy).2,34 Hepatotoxicity should be suspected in patients whose transaminases exceed five times the upper limit of normal or whose total bilirubin exceeds 3 mg/dL (51 μmol/L) and in patients with symptoms such as nausea, vomiting, and jaundice. At this point, the offending agent(s) should be discontinued. Sequential reintroduction of the drugs with frequent testing of liver enzymes is often successful in identifying the offending agent; other agents may be continued34 (Table 75–4).
Therapeutic Drug Monitoring
TDM or applied pharmacokinetics is the use of serum drug concentrations to optimize therapy.34,41,42 Non-AIDS patients with drug-susceptible TB generally do well. TDM may be used if patients are failing appropriate DOT (no clinical improvement after 2–4 weeks or smear-positive after 4–6 weeks). On the other hand, patients with AIDS, diabetes, and various GI disorders often fail to absorb these drugs properly and are candidates for TDM. Also, patients with hepatic or renal disease should be monitored, given their potential for overdoses. In the treatment of MDR-TB, TDM may be particularly useful.44,46Finally, TDM of the TB and HIV drugs is perhaps the most logical way to untangle the complex drug interactions that take place. For a complete list of drug interactions visit the CDC website at www.cdc.gov/nchstp/tb/tb_hiv_drugs/toc.htm.54 In particular, interactions between the rifamycins (e.g., rifampin, rifapentine, and rifabutin) and the HIV protease inhibitors and NRTIs are common and require dose and frequency modifications in many cases. Since these are constantly being updated, the preceding link is an excellent way to keep current.
Patient Encounter 4: Creating a Care Plan
Based on the information provided, which clinical and laboratory parameters should be monitored in this patient to determine efficacy and avoid toxicity?
Is this patient a candidate for therapeutic drug monitoring? Why or why not?
Patient Care and Monitoring
1. Rapidly identify a new TB case.
2. Assess the patient’s risk factors and signs and symptoms to determine if the patient might be infected with TB.
3. Isolate the patient with active disease to prevent the spread of the disease.
4. Collect appropriate samples for smears and cultures.
5. Obtain a thorough medication history.
6. Select and recommend appropriate antituberculosis treatment. Consider HIV status, pregnancy, type of TB infection, renal function, liver function etc.
7. Ensure adherence to the treatment regimen by the patient.
8. Obtain AFB stains to evaluate the effectiveness of treatment.
9. Consider TDM if no clinical improvement.
10. Secondary goals are identification of the index case that infected the patient, identification of all persons infected by both the index case and the new case of TB, and the completion of appropriate treatments for those individuals.
Abbreviations Introduced in This Chapter

Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. World Health Organization Report on the Global Tuberculosis Epidemic 1998.
2. Iseman MD. A Clinician’s Guide to Tuberculosis. Philadelphia, Lippincott Williams & Wilkins, 2000.
3. McCray E, Weinbaum CM, Braden CR, et al. The epidemiology of tuberculosis in the United States. Clin Chest Med 1997;18:99–113.
4. Centers for Disease Control and Prevention. Trends in Tuberculosis Morbidity–United States, 1992–2002. MMWR 2003;52:222–224.
5. CDC. Reported tuberculosis in the United States, 2007. Atlanta, GA: U.S. Department of Health and Human Services, CDC, September, 2008.
6. Haas DW. Mycobacterium tuberculosis. In: Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases, 5th ed. New York, Churchill Livingstone, 2000:2576–2607.
7. Small PM, Shafer RW, Hopewell PC, et al. Exogenous reinfection with multidrug-resistant Mycobacterium tuberculosis in patients with advanced HIV infection. N Engl J Med 1993:328;1137–1144.
8. Beck-Sague C, Dooley SW, Hutton MD, et al. Hospital outbreak of multidrug-resistant Mycobacterium tuberculosis infections: Factors in transmission to staff and HIV-infected patients. JAMA 1992:268;1280–1286.
9. Centers for Disease Control and Prevention. Meeting the challenge of multidrug-resistant tuberculosis: Summary of a conference. MMWR 1992;41(RR-11):51–57.
10. Heifets L. Mycobacteriology laboratory. Clin Chest Med 1997;18: 35–53.
11. Heifets LB. Drug susceptibility tests in the management of chemotherapy of tuberculosis. In: Heifets LB, ed. Drug Susceptibility in the Chemotherapy of Mycobacterial Infections. Boca Raton, FL: CRC Press, 1991:89–122.
12. Daley CL, Chambers HF. Mycobacterium tuberculosis complex. In: Yu VL, Weber R, Raoult D, eds. Antimicrobial therapy and vaccines, Volume I: Microbes, 2nd ed. New York, Apple Trees Productions, LLC, 2002:841–865.
13. Roberts GD, Böttger EC, Stockman L. Methods for the rapid identification of mycobacterial species. Clin Lab Med 1996;16: 603–615.
14. Sandin RL. Polymerase chain reaction and other amplification techniques in mycobacteriology. Clin Lab Med 1996;16:617–639.
15. Ingham CJ, Ayad AB, Nolsen K, Mulder B. Rapid drug susceptibility testing of mycobacteria by culture on a highly porous ceramic support. Int J Tuberc Lung Dis 2008;12(6):645–650.
16. Martin A, Panaiotov S, Portaels F, Hoffner S, Palomino JC, Angeby K. The nitrate reductase assay for the rapid detection of isoniazid and rifampicin resistance in Mycobacterium tuberculosis: A systematic review and meta-analysis. Journal of Antimicrobial Chemotherapy 2008;62(1):56–64.
17. Daniel TM, Boom WH, Ellner JJ. Immunology of Tuberculosis. In: Reichman LB, Hershfield ES. Tuberculosis. A Comprehensive International Approach, 2nd ed. New York, Marcel Dekker, 2000:157–185.
18. Piessens WF, Nardell EA. Pathogenesis of Tuberculosis. In: Reichman LB, Hershfield ES. Tuberculosis. A Comprehensive International Approach, 2nd ed. New York, Marcel Dekker, 2000:241–260.
19. American Thoracic Society / Centers for Disease Control and Prevention. Diagnostic standards and classification of tuberculosis in adults and children. Am J Respir Crit Care Med 2000;161:1376–1395.
20. Peloquin CA, Berning SE. Tuberculosis and multi-drug resistant tuberculosis in children. Pediatr Nurs 1995;21:566–572.
21. Correa AG. Unique aspects of tuberculosis in the pediatric population. Clin Chest Med 1997;18:89–98.
22. American Thoracic Society / Centers for Disease Control and Prevention. Targeted tuberculin skin testing and treatment of latent tuberculosis infection. Am J Respir Crit Care Med 2000;161: S221–S247.
23. Pape JW, Jean SS, Ho JL, et al. Effect of isoniazid prophylaxis on incidence of active tuberculosis and progression of HIV infection. Lancet 1992;342:268–272.
24. Narita M, Ashkin D, Hollender ES, Pitchenik AE. Paradoxical worsening of tuberculosis following antiretroviral therapy in patients with AIDS. Am J Respir Crit Care Med 1998;158:157–161.
25. Barnes PF, Bloch AB, Davidson PT, Snider DE. Tuberculosis in patients with human immunodeficiency virus infection. N Engl J Med 1991;324:1644–1650.
26. Alvarez S, Shell C, Berk SL. Pulmonary tuberculosis in elderly men. Am J Med 1987;82:602–606.
27. Umeki S. Comparison of younger and elderly patients with pulmonary tuberculosis. Respiration 1989;55:75–83.
28. Centers for Disease Control and Prevention. Anergy skin testing and preventive therapy for HIV-infected persons: revised recommendations. MMWR 1997; 46 (RR-15):1–10.
29. Bouza E, Diaz-Lopez MD, Moreno S, et al. Mycobacterium tuberculosis bacteremia in patients with and without human immunodeficiency virus infection. Arch Intern Med 1993;153:496–500.
30. Pai M, O’Brien R. New Diagnostics for latent and active tuberculosis: State of the art and future prospects. Semin Respir Crit Care Med 2008;29:560–568.
31. Nienhaus A, Schablon A, Diel R. Interferon-gamma release assay for the diagnosis of latent TB infection—Analysis of discordant results, when compared to the tuberculin skin test. PLoS ONE 2008;3(7):e2665.
32. Pai M, Zwerling A, Menzies D. Systematic review: T-cell based assays for the diagnosis for latent tuberculosis infection. Ann Intern Med 2008;149:177–184.
33. Pai M, Dheda K, Cunningham J, Scano F, O’Brien R. T-cell assays for the diagnosis of latent tuberculosis infection: moving the research agenda forward. Lance Infect Dis 2007;7:428–438.
34. American Thoracic Society / Centers for Disease Control / Infectious Disease Society of America. Treatment of tuberculosis. Am J Respir Crit Care Med 2003;167:603–662.
35. Fujiwara PI, Larkin C, Frieden TR. Directly observed therapy in New York City. Clin Chest Med 1997;18:135–148.
36. Weis SE. Universal directly observed therapy. Clin Chest Med 1997;18:155–163.
37. Malone RS, Fish DN, Spiegel DM, Childs JM, Peloquin CA. The effect of hemodialysis on cycloserine, ethionamide, para-aminosalicylate, and clofazimine. Chest 1999;116:984–990.
38. Vallejo JG, Starke JR. Tuberculosis and pregnancy. Clin Chest Med 1992;13:693–707.
39. Centers for Disease Control and Prevention. Update: Fatal and severe liver injuries associated with rifampin and pyrazinamide for latent tuberculosis infection, and revisions in the American Thoracic Society/CDC recommendations. MMWR 2001;50(34):733–735.
40. Mitchison DA. Basic mechanisms of chemotherapy. Chest 1979;76 (Suppl):771–781.
41. Peloquin CA. Pharmacological Issues in the Treatment of Tuberculosis. Ann NY Acad Sci 2001;953:157–164.
42. Peloquin CA. Therapeutic Drug Monitoring in the Treatment of Tuberculosis. Drugs 2002;62:2169–2183.
43. Hamadeh MA, Glassroth J. Tuberculosis and pregnancy. Chest 1992; 101:1114–1120.
44. Peloquin CA. Antituberculosis drugs: Pharmacokinetics. In: Heifets LB, ed. Drug Susceptibility in the Chemotherapy of Mycobacterial Infections. Boca Raton, FL: CRC Press, 1991:59–88.
45. Malone RS, Fish DN, Spiegel DM, Childs JM, Peloquin CA. The effect of hemodialysis on isoniazid, rifampin, pyrazinamide, and ethambutol. Am J Respir Crit Care Med 1999;159:1580–1584.
46. Holdiness MR. Clinical pharmacokinetics of the antituberculosis drugs. Clin Pharmacokinet 1984;9:511–544.
47. Girling DJ. Adverse effects of antituberculous drugs. Drugs 1982;23: 56–74.
48. Burman WJ. Moxifloxacin versus ethambutol in the first 2 months of treatment for pulmonary tuberculosis. Am J Respir Crit Care Med 2006;174:331–338.
49. Johnson JL. Early and extended early bactericidal activity of levofloxacin, gatifloxacin and moxifloxacin in pulmonary tuberculosis. Int J. Tuberc Lund Dis 2006;10:605–612.
50. Zhang Y. Advances in the treatment of tuberculosis. Clinical Pharmacology and Therapeutics 2007;82:595–600.
51. Brudney K, Dobkin J. Resurgent tuberculosis in New York City: Human immunodeficiency virus, homelessness, and the decline of tuberculosis control programs. Am Rev Resp Dis 1991;144:745–749.
52. Mahmoudi A, Iseman MD. Pitfalls in the care of patients with tuberculosis: Common errors and their association with the acquisition of drug resistance. JAMA 1993;270:65–68.
53. Chaulk CP, Bartlett JG, Chaisson RE. 15 years of directly observed therapy for TB. Program and Abstracts, 32nd Annual Meeting, Infectious Diseases Society of America, Orlando, FL, October 7–9, 1994. Abstract 181.
54. Namdar R, Ebert S, Peloquin CA. Drugs for Tuberculosis. In: Piscitelli SC, Rodvold KA, eds. Drug Interactions in Infectious Diseases, 2d., Totowa, NJ, Humana Press, 2000,191–214.