Kathryn R. Matthias and Brian A. Potoski
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Explain the diagnostic criteria for significant bacteriuria.
2. Recognize the signs and symptoms of urinary tract infections (UTIs) and how they differ in upper versus lower urinary tract disease.
3. Identify the organism responsible for the majority of uncomplicated UTIs.
4. Assess the laboratory tests that help in diagnosing patients with UTI.
5. Determine appropriate drug, dose, and duration for uncomplicated and complicated UTI prophylaxis and empiric treatment.
6. Evaluate and select therapy for uncomplicated and complicated UTIs based on specific urine culture results and patient characteristics.
7. Formulate appropriate monitoring and education information for patients with UTIs.
KEY CONCEPTS
![]() Urinary tract infections (UTIs) are thought of as either uncomplicated or complicated. Generally this refers to absence or presence, respectively, of functional or structural abnormalities within the urinary tract.
Urinary tract infections (UTIs) are thought of as either uncomplicated or complicated. Generally this refers to absence or presence, respectively, of functional or structural abnormalities within the urinary tract.
![]() The majority (85%) of uncomplicated UTIs are caused by Escherichia coli. The majority of the remaining 15% are caused by Staphylococcus saprophyticus along with Klebsiella spp., Proteus spp., Pseudomonas spp., Enterobacter spp., and Enterococcus spp.
The majority (85%) of uncomplicated UTIs are caused by Escherichia coli. The majority of the remaining 15% are caused by Staphylococcus saprophyticus along with Klebsiella spp., Proteus spp., Pseudomonas spp., Enterobacter spp., and Enterococcus spp.
![]() Symptoms of lower UTIs include dysuria, gross hematuria, suprapubic heaviness, nocturia, increased urinary frequency, and urgency.
Symptoms of lower UTIs include dysuria, gross hematuria, suprapubic heaviness, nocturia, increased urinary frequency, and urgency.
![]() Symptoms of upper UTIs include fever, nausea, vomiting, malaise, and often severe flank pain.
Symptoms of upper UTIs include fever, nausea, vomiting, malaise, and often severe flank pain.
![]() The goals of treatment of UTIs are to eradicate the offending organism, to prevent or treat consequences of infection, and to prevent recurrence of infection.
The goals of treatment of UTIs are to eradicate the offending organism, to prevent or treat consequences of infection, and to prevent recurrence of infection.
![]() Uncomplicated UTIs may be managed with 3-day or even 1-day regimens, while complicated UTIs should be treated for at least 7 days and sometimes 2 weeks or longer.
Uncomplicated UTIs may be managed with 3-day or even 1-day regimens, while complicated UTIs should be treated for at least 7 days and sometimes 2 weeks or longer.
Urinary tract infections (UTIs) are comprised of a diverse array of syndromes depending on the location of the infection within the urinary tract. UTIs occur frequently and are responsible for approximately 8.3 million physician office and hospital outpatient visits annually.1–3 In simplest of terms, a UTI is bacteria in the urinary tract, that does not represent contamination.
Bacteriuria, or bacteria in the urine, does not always represent infection. For this reason a number of quantitative diagnostic criteria have been created to identify the amount of bacteria in the urine that most likely represents true infection (hence the term “significant bacteriuria”).4 These criteria are shown in Table 79–1. Furthermore, UTIs are classified as lower tract or upper tract disease. Patients can present differently with upper versus lower tract disease, and upper tract disease is thought of as a much more severe infection since patients are more likely to be admitted to the hospital with upper urinary tract disease than lower tract disease. An example of lower tract syndrome is cystitis which involves inflammation of the bladder and commonly causes symptoms such as dysuria, nocturia, gross hematuria, and occasional suprapubic tenderness. An example of upper urinary tract disease is pyelonephritis. Pyelonephritis is an inflammation of the kidney usually due to infection. Patients with uncomplicated UTI are more frequently treated as outpatients compared to those patients with complicated UTIs.
EPIDEMIOLOGY AND ETIOLOGY
The prevalence and type of UTIs generally varies by age and gender.5,6 UTIs may occur at any age, even in the very young. Premature infants, for example, have a higher rate than full-term infants, and neonatal boys are five to eight times more likely to have UTIs than neonatal girls. In young children 1 to 5 years of age, significant bacteriuria occurs more in girls than boys, 4.5% compared to 0.5%, respectively.7 Once adulthood is reached, bacteriuria increases in young, nonpregnant women (range, 1–3%), yet remains low in men (up to 0.1%).8 Symptomatic UTI affects 30% of women between 20 and 40 years of age, which represents a prevalence that is 30 times greater than men of the same age group. Upwards of 40% to 50% of the female population will experience a symptomatic UTI at sometime during their life.1
Table 79–1 Diagnostic Criteria for Significant Bacteriuria
|
• Greater than or equal to 102 CFU coliforms/mL or greater than or equal to 105 CFU noncoliforms/mL in a symptomatic female |
|
• Greater than or equal to 103 CFU organisms/mL in a symptomatic male |
|
• Greater than or equal to 105 CFU same organisms/mL in asymptomatic individuals on two consecutive specimens |
|
• Any growth of bacteria on suprapubic catheterization in a symptomatic patient |
|
• Greater than or equal to 102 CFU organisms/mL in a catheterized patient |
CFU, colony-forming unit.
The etiology of UTIs has remained relatively unchanged over the past several decades. ![]() UTIs are either uncomplicated or complicated. There is a lack of consensus regarding the definition of what makes a UTI complicated, but in general a complicated UTI refers to a structural or functional abnormality of the urinary tract. Patients with complicated UTIs are typically given longer treatment durations than those patients with uncomplicated infections. Those with complicated UTIs by definition are also prone to more frequent infections. It is important to note that an upper UTI does not necessarily imply complicated UTI, nor does lower UTI imply uncomplicated UTI.
UTIs are either uncomplicated or complicated. There is a lack of consensus regarding the definition of what makes a UTI complicated, but in general a complicated UTI refers to a structural or functional abnormality of the urinary tract. Patients with complicated UTIs are typically given longer treatment durations than those patients with uncomplicated infections. Those with complicated UTIs by definition are also prone to more frequent infections. It is important to note that an upper UTI does not necessarily imply complicated UTI, nor does lower UTI imply uncomplicated UTI.
While the frequency of causative organisms changes depending on the location of infection and patient characteristics, over 95% of uncomplicated UTIs are the result of a single causative organism. ![]() In 85% of the uncomplicated UTI cases, the causative organism is E. coli.9 A variety of other organisms may cause uncomplicated UTIs, but represent the minority of pathogens. Other organisms include gram-positives such as Staphylococcus saprophyticus and Enterococcus spp. and gram-negative bacteria such as Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus spp., and Enterobacter spp.10–12 The chances of isolating an organism other than E. coli are higher in patients that have recurrent UTIs, particularly those patients whose UTI is considered complicated. It is also more common for organisms other than E. coli to cause UTIs in the hospitalized population than in the general population.13,14
In 85% of the uncomplicated UTI cases, the causative organism is E. coli.9 A variety of other organisms may cause uncomplicated UTIs, but represent the minority of pathogens. Other organisms include gram-positives such as Staphylococcus saprophyticus and Enterococcus spp. and gram-negative bacteria such as Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus spp., and Enterobacter spp.10–12 The chances of isolating an organism other than E. coli are higher in patients that have recurrent UTIs, particularly those patients whose UTI is considered complicated. It is also more common for organisms other than E. coli to cause UTIs in the hospitalized population than in the general population.13,14
Patient Encounter, Part 1
VN is a 23-year-old female who presents to a local urgent care center with complaints of painful urination and frequent need to urinate especially at night which began 3 days ago. She denies vomiting, fever, nausea, or flank pain. Upon questioning she does admit that she is sexually active with only one partner and uses a diaphragm.
What symptoms are suggestive of urinary tract infection (UTI)?
Does she have risk factors for UTI?
What additional information do you need to know before creating a treatment plan for this patient?
PATHOPHYSIOLOGY
There are three potential ways for bacteria to enter into the urinary tract and cause infection: the ascending, hematogenous, and lymphatic pathways.
Ascending Pathway
The ascending pathway occurs when bacteria colonizing the urethra subsequently travel upwards, or ascend, the urethra to the bladder and cause cystitis (Fig. 79–1). The ascending route may help to explain why UTIs occur more commonly in women than in men. Women have a shorter urethra than men, and colonization of the female urethra is likely due to its proximity to the perirectal area. The use of spermicidal agents increases the colonization of the vagina with uropathogens.15 Additionally, massage of the urethra in women as well as sexual intercourse may lead to bacteria gaining entrance into the bladder.16,17 Once in the bladder, bacteria are not limited to causing cystitis. These bacteria may continue to ascend the urinary tract via the ureters and cause more complicated infections, such as pyelonephritis.
Hematogenous Pathway
The hematogenous route occurs through the seeding of the urinary tract with pathogens carried by the blood supply. These pathogens represent an infection at some other primary site in the body. Staphylococcus aureus bacteremia, for example, can cause renal abscesses via the hematogenous route, and pyelonephritis can be experimentally produced by IV injection of Salmonella spp., Mycobacterium tuberculosis, or even yeast (Candida spp.) into rabbits.18However, experimentally creating this pathway has not been successful with all organisms. Experimental hematogenous seeding of the kidneys could not be created with the IV injection of large innocula of E. coli or P. aeruginosa in a mouse model.19
Lymphatic Pathway
The lymphatic system, also known as the secondary circulatory system, connects the bladder to the kidney and may represent a way for bacteria to be transported and subsequently cause infection. Although this pathway is classically included as a route of infection, there is a lack of data showing the lymphatic pathway as a significant mechanism for development of infection. As a result this pathway is not believed to be a significant host mechanism.
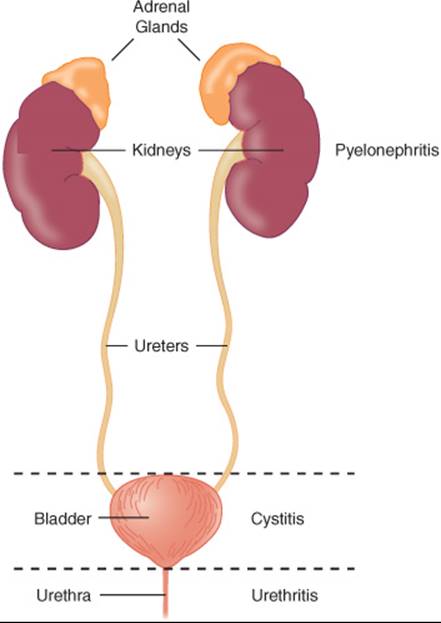
FIGURE 79–1. Anatomy and associated infections of the urinary tract. (From Sprandel KA, Lesch CA, Rodvold KA. Lower urinary tract infection. In: Schwinghammer TL (ed.) Pharmacotherapy Casebook: A Patient-Focused Approach, 6th ed. New York City: McGraw-Hill; 2005:315, with permission).
Host Defense Mechanisms
Urine, although not an antimicrobial itself, possesses characteristics that are less than ideal for bacterial growth. Some of these characteristics include low pH, significant urea concentration, and high osmolality. Also, bacteria in the bladder can stimulate an urge to urinate. Additionally, prostatic fluid secretions in men can inhibit bacterial growth while normal vaginal flora in women such as Lactobacillusspp. can secrete lactic acid which can decrease the pH of the enviroment.20–22
There are several other host factors that inhibit what are known as bacterial virulence factors. These virulence factors are mechanisms that bacteria utilize to cause infection and/or ensure their survival. The first is glycosaminoglycan, a compound produced by the body that coats the epithelial cells of the bladder. This compound essentially separates the bladder from the urine by forming a protective layer against bacterial adhesion.23 A second compound known as TammHorsfall protein is secreted into the urine, and prevents E. coli from binding to receptors present on the surface of the bladder. Other factors implicated in contributing to host defense mechanisms against UTIs include immunoglobulins, specifically IgA.
Clinical Presentation and Diagnosis of UTIs
General
• Most women present with hematuria; however, this is not a presentation restricted only to UTIs.
• Elderly patients frequently will not present with common signs and symptoms of UTI, but may present with altered mental status.
• More than 95% of UTIs are caused by a single organism.
• Patients may present with urosepsis.
![]() Signs and Symptoms of Lower UTI
Signs and Symptoms of Lower UTI
• Dysuria, gross hematuria, suprapubic heaviness, nocturia, increased urinary frequency and urgency
![]() Signs and Symptoms of Upper UTI
Signs and Symptoms of Upper UTI
• Fever, nausea, vomiting, malaise, and often severe flank pain
Laboratory Tests
Urinalysis should show:
• Pyuria typically greater than 10 white blood cells/mm3 urine
• Bacteriuria, usually greater than 105 CFU organisms/mL
• Nitrites present
• Leukocyte esterase present
Other Diagnostic Tests
• Bacterial urine culture
• Upper UTI: presence of costovertebral tenderness
Risk Factors
There are several risk factors for development of UTIs.8 Common risk factors for UTIs in women include sexual intercourse, use of a diaphragm, use of spermicidal jellies, diabetes, and pregnancy.24–26 In men, the risks primarily relate to lack of circumcision and at an older age include prostatic hyperplasia. Common risk factors for both men and women include urologic instrumentation, renal transplantation, neurogenic bladder, and urinary tract obstruction.27,28
TREATMENT
Desired Outcomes
![]() The goals of treatment are to eradicate the invading organism, to prevent or treat consequences of infection, and to prevent, if possible, recurrence of infection. Therapy is directed at microbiologic eradication of the offending organism through antibiotics.
The goals of treatment are to eradicate the invading organism, to prevent or treat consequences of infection, and to prevent, if possible, recurrence of infection. Therapy is directed at microbiologic eradication of the offending organism through antibiotics.
Patient Encounter, Part 2: Medical History, Physical Exam, and Diagnostic Tests
PMH: Asthma (controlled)
FH: Father living with diabetes mellitus, type 2 (controlled) and chronic obstructive pulmonary disease; mother living with hypertension (controlled)
SH: Second year pharmacy student. Works as a pharmacy intern at a local hospital
Allergies: Penicillin (patient states she was admitted to a hospital at the age of 7 years with hives and throat swelling after receiving amoxicillin)
Meds: Albuterol (salbutamol) inhaler as needed
ROS: (+) dysuria, urinary frequency; (−) fever, nausea, vomiting, flank pain
PE:
VS: BP 122/64 mm Hg, P 62 bpm, RR 16 per minute, afebrile
CV: RRR, normal S1, S2; normal findings
Abd: Soft, nontender, nondistended; (+) bowel sounds, no hepatosplenomegaly, heme (−) stool
Labs: Within normal limits; (−) pregnancy test
Given this additional information, what is your assessment of the patient’s condition?
Identify your treatment goals for the patient.
What nonpharmacologic pharmacologic alternatives are available for the patient?
General Approach to Treatment
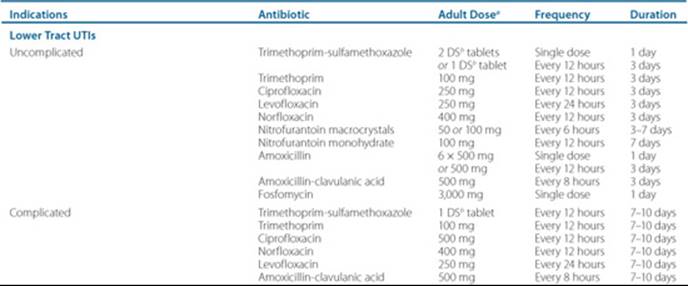
Antimicrobial therapy is the cornerstone of treatment in UTIs. Antimicrobials should ideally be well tolerated, narrow in antimicrobial spectrum, lend itself to patient compliance (low total number of doses), have adequate concentrations at the site of the infection, and have good oral bioavailability. Table 79–2 reviews oral and IV antibiotics frequently used to treat UTIs with comments on their use, and Table 79–3 reviews frequency, duration, and doses of oral antibiotics used commonly for outpatient treatment of UTIs.
Nonpharmacologic Therapy
Although there is limited data in the literature and sometimes conflicting results, several nonpharmacologic therapies have been proposed for prevention of UTIs. The intake of large volumes of cranberry juice can decrease the number of UTIs over a year period in patients with recurrent UTIs but uncertain efficacy in the general population.29 Probiotics such as Lactobacillus spp. have been used to decrease vaginal pH in women which may decrease the growth of certain pathogenic bacteria.21–22 Topical estrogen replacement therapy significantly decreases the incidence of UTIs in postmenopausal women compared to placebo.30 Methenamine hippurate and methenamine mandalate have no antimicrobial properties but decrease the incidence of UTIs when used for prophylaxis. Patient education of common risk factors is important.
Pharmacologic Therapy
Uncomplicated Cystitis
Uncomplicated cystitis represents the most common of UTIs, is frequently managed in the outpatient setting, and occurs in women of childbearing age.3 E. coli is the most frequent causal organisms in this setting, but in a minority of cases may be caused by S. saprophyticus, K. pneumoniae, P. mirabilis, Enterococcus spp., and a small percentage of other organsims.9–12 As such, treatment in the outpatient setting is frequently relegated to a urinalysis and empiric therapy without a urine culture.31,32 Patients are subsequently followed up for resolution of signs and symptoms. ![]() One significant benefit of treatment in the setting of uncomplicated cystitis is that treatment duration can be less than 7 days, and often may be 3 days or even 1 day.
One significant benefit of treatment in the setting of uncomplicated cystitis is that treatment duration can be less than 7 days, and often may be 3 days or even 1 day.
Although treatment duration of 1 day is advantageous because it strictly limits adverse events and drug interactions, and increases compliance, health care providers should know that 3-day courses of fluoroquinolones and trimetho prim-sulfamethoxazole are superior to single-doses in terms of cure rates in uncomplicated UTIs.10,33 For acute uncomplicated UTIs, it is reasonable to pursue a 1-day course of therapy. Which agent to choose empirically partly hinges on known resistance rates in the geographic region, particularly E. coli resistance to trimethoprim-sulfamethoxazole.9,34,35 No consensus has been reached on what percentage of E. coli isolates resistant to trimethoprim-sulfamethoxazole should preclude its use; however, a model has been created that suggests that this threshold resistant rate is between 19% and 21% in the empiric setting.36
Acute Pyelonephritis
In contrast to patients that present with lower UTIs, those that present with pyelonephritis usually have high-grade fever (greater than 38.3°C [100.9°F]) and severe flank pain. Select patients with pyelonephritis may be treated in the outpatient setting; however, patients whose infection is severe enough to cause vomiting, decreased food intake, and dehydration may need to be treated in an inpatient hospitalized setting. These patients will usually receive IV antibiotics at first before being switched to oral therapy depending on susceptibility testing.
Table 79–2 Commonly Used Antimicrobial Agents for the Treatment of UTIs



Table 79–3 Overview of Outpatient, Oral Antimicrobial Therapy for Lower and Upper Tract UTIs

Patients with pyelonephritis are traditionally given 14 days of therapy; however, there are limited data showing success in treating acute uncomplicated pyelonephritis for 7 to 10 days. More studies need to be conducted on treating for these shorter durations.3 Gram stain and culture are important in ensuring that appropriate antimicrobial coverage is selected. Stratification is used to manage patients with acute pyelonephritis. Women who present with mild cases of pyelonephritis (defined as low-grade fever and a normal to slightly elevated peripheral white blood count, without nausea or vomiting) may be treated as outpatients.3 Those women who exhibit more severe signs and symptoms will need to be admitted to an acute care setting for appropriate treatment. The same holds true for antibiotic selection in these patients. Those who are treated in an outpatient setting can be treated with trimethoprim-sulfamethoxazole, fluoroquinolones, or even β-lactam/β-lactamase inhibitors, such as amoxicillin-clavulanic acid. In those patients that are admitted to the hospital, antibiotic therapy is usually broader in nature, especially in patients suspected of having bacteremia or urosepsis. These patients will typically receive IV therapy such as a fluoroquinolone, or a β-lactam plus an aminoglycoside.3,37,38
Special Populations
Pregnant Women
Changes to the urinary tract in pregnant women predispose them to an increased incidence of bacteriuria, and subsequent UTIs that may follow. These changes include alterations in amino acid and other nutrient concentrations in the urine along with physiologic changes such as reduced bladder tone and dilation of the renal pelvis and ureters.39,40
An association exists between maternal UTI during pregnancy and fetal death, mental retardation, and developmental delay.41 Due to this known association and since up to 7% of pregnant women will develop an asymptomatic bacteriuria that may progress to pyelonephritis, screening for UTI is necessary.24,42 In pregnant patients with significant bacteriuria, whether symptomatic or asymptomatic, treatment is recommended to avoid the complications discussed previously. In the majority of patients, a sulfonamide (not in the third trimester due to concerns for hyperbilirubinemia), amoxicillin-clavulanic acid, cephalexin, or nitrofurantoin are effective treatment options. Tetracyclines and fluoroquinolones should be avoided due to risk of teratogenicity and ability to inhibit cartilage and bone development, respectively. Follow-up usually consists of a urine culture 1 to 2 weeks after completion of therapy, and afterwards monthly until birth.
Patient Encounter, Part 3: Creating a Care Plan
Based on the information presented, create a care plan for this patient’s UTI. Your plan should include:
(a) a statement of the drug-related needs and/or problems,
(b) a patient-specific detailed therapeutic plan, and (c) monitoring parameters to assess efficacy and safety.
Catheterized Patients
An indwelling catheter is commonly used in various health care settings and is associated with UTIs.27 Bacteria may be introduced into the bladder via the catheter in several ways including direct infection introduction during catheterization (via colonization and subsequently traveling the length of the catheter through bacterial motility or capillary action). UTIs as a result of an indwelling catheter are common and occur at a rate of 5% per day of catheter presence.43
The approach in the setting of a patient with bacteriuria and an indwelling urinary catheter follows two paths. The first, in asymptomatic patients with catheterization, is to hold antibiotics and remove the catheter if possible. The second, in symptomatic patients with catheterization, is to initiate antibiotic therapy and removal of the catheter if possible. In both of the above situations, if discontinuation of the catheter is not possible, the patient should be recatheterized with a new urinary catheter if the previous catheter is greater than 2 weeks old.
UTIs in Men
Although UTIs in men are not always complicated by definition, due to the relative infrequency of UTIs in men compared to women, an abnormality (structural or functional) should be suspected and therefore treated as a probable complicated infection until proven otherwise.44 For this reason, men should not be treated with a single dose or short course of therapy if diagnosed with a UTI. Typically these patients will receive 2 weeks of therapy, and in situations of failure may be treated up to 6 weeks, particularly if a prostatic source of infection is suspected. Prostatic enlargement, as previously mentioned, is a risk factor in men, and the prevalence of benign prostatic hyperplasia in the elderly population may predispose this population to UTIs.
OUTCOME EVALUATION
• Monitor the patient for resolution of symptoms with a goal of 48 to 72 hours to resolution after start of antimicrobial therapy
• If possible, follow-up on susceptibilities of the infecting organism (urine culture)
• Repeat culture is necessary only if symptoms do not acutely abate or reinfection or recurrence occurs
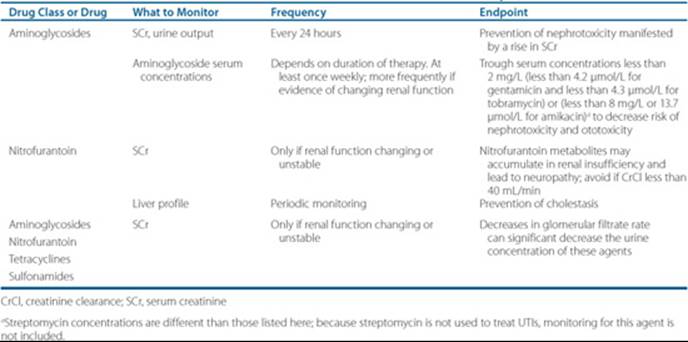
• Depending on chosen antibiotic therapy, evaluate patient based on drug therapy monitoring parameters including those presented in Table 79–4 to optimize therapy and decrease incidence of adverse drug events.
Table 79–4 Monitoring Parameters for Select Antibiotics Used in the Treatment of UTIs
Patient Care and Monitoring
1. Assess the patient’s symptoms to determine response to the antimicrobial regimen you have chosen.
2. Review any microbiologic data:
• Based on urinalysis and gram stain (if available), is your empiric selection reasonable?
• Based on culture and susceptibility data (if available), are there any changes that need to be made from your initial empiric antimicrobial selection (i.e., resistance to the regimen initially selected)?
3. Determine if the patient may benefit from prophylactic therapy (i.e., recurrent UTIs secondary to chronic urinary catheterization due to paraplegia).
4. Evaluate the patient for the presence of adverse drug reactions, drug allergies, and potential drug interactions.
5. Stress the importance of complying with the prescribed antimicrobial regimen and to follow-up with the health care provider if signs and symptoms recur.
Abbreviations Introduced in This Chapter


Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Foxman B. Epidemiology of urinary tract infections: Incidence, morbidity, and economic costs. Am J Med 2002;113(Suppl 1A): 5S–13S.
2. Fihn SD. Clinical practice. Acute uncomplicated urinary tract infection in women. N Engl J Med 2003;349:259–266.
3. Warren JW, Abrutyn E, Hebel JR, et al. Guidelines for antimicrobial treatment of uncomplicated acute bacterial cystitis and acute pyelonephritis. Clin Infect Dis 1999;29:745–758.
4. Bent S, Nallamothu BK, Simel DL, et al. Does this woman have an acute, uncomplicated urinary tract infection? JAMA 2002;287: 2701–2710.
5. Alper BS, Curry SH. Urinary tract infection in children. Am Fam Physician 2005;72:2483–2488.
6. Shortliffe LM, McCue JD. Urinary tract infections at the age extremes: Pediatrics and geriatrics. Am J Med 2002;113(Suppl 1A):55S–66S.
7. Smellie JM, Prescod NP, Shaw PJ, et al. Childhood reflux and urinary infection: A follow-up of 10–41 years in 225 adults. Pediatr Nephrol 1998;12:727–736.
8. Ronald AR, Pattullo AL. The natural history of urinary tract infection in adults. Med Clin North Am 1991;75:299–312.
9. Zhanel GG, Hisanaga TL, Laing NM, et al. Antibiotic resistance in Escherichia coli outpatient urinary isolates: Final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA). Int J Antimicrob Agents 2006;27:468–475.
10. Stamm WE, Hooton TM. Management of urinary tract infections in adults. N Engl J Med 1993;329:1328–1334.
11. Ronald A. The etiology of urinary tract infection: Traditional and emerging pathogens. Am J Med 2002;113(Suppl 1A):14S–19S.
12. Raz R, Colodner R, Kunin CM. Who are you–Staphylococcus saprophyticus? Clin Infect Dis 2005;40:896–898.
13. Wagenlehner FM, Naber KG. Hospital-acquired urinary tract infections. J Hosp Infect 2000;46:171–181.
14. Lundstrom T, Sobel J. Nosocomial candiduria: A review. Clin Infect Dis 2001;32:1602–1607.
15. Hooten TM, Hillier S, Johnson C, et al. Escherichia coli bacteriuria and contraceptive method. JAMA 1991;265:64–69.
16. Stamatiou C, Bovis C, Panaguopoulos P, et al. Sex-induced cystitis–patient burden and other epidemiological features. Clin Exp Obstet Gynecol 2005;32:180–182.
17. Bran JL, Levison ME, Kaye D. Entrance of bacteria into the female urinary bladder. N Engl J Med 1972;286:626–629.
18. Freedman LR. Experimental pyelonephritis. VI. Observation on susceptibility of the rabbit kidney to infection by a virulent strain of Staphylococcus aureus. Yale J Biol Med 1960;32:272–279.
19. Gorrill RH, DeNavasquez SJ. Experimental pyelonephritis in the mouse produced by Escherichia coli, Pseudomonas aeruginosa, and Proteus mirabilis. J Pathol Bacteriol 1964;87:79–87.
20. Stamey TA, Fair WR, Timothy MM, et al. Antibacterial nature of prostatic fluid. Nature 1968;218:444–447.
21. Kwok L, Staphleton AE, Stamm WE, et al. Adherence of Lactobacillus crispatus to vaginal epithelial cells from women with or without a history of recurrent urinary tract infection. J Urol 2006;176; 2050–2054.
22. Gupta K, Stapleton AE, Hooton TM, et al. Inverse association of H2O2-producing lactobacilli and vaginal Escherichia coli colonization in women with recurrent urinary tract infections. J Infect Dis 1998;178:446–450.
23. Parsons CL, Schrom SH, Hanno P, et al. Bladder surface mucin: Examination of possible mechanisms for its antibacterial effect. Invest Urol 1978;6:196–200.
24. U.S. Preventive Services Task Force. Screening for asymptomatic bacteriuria in adults: U.S. Preventive Services Task Force reaffirmation recommendation statement. Ann Intern Med 2008;149:43–47.
25. Nicolle LE. Urinary tract infection in diabetes. Curr Opin Infect Dis 2005;18:49–53.
26. Harding GK, Zhanel GG, Nicolle LE, et al. Antimicrobial treatment in diabetic women with asymptomatic bacteriuria. N Engl J Med 2002;347:1576–1583.
27. Niël-Weise BS, van den Broek PJ. Urinary catheter policies for long-term bladder drainage. Cochrane Database Syst Rev 2005;1:CD004201.
28. Sobel JD, Kaye D. Urinary tract infection. In: Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases, 6th ed. Philadelphia: Elsevier, 2005:875.
29. Jepson RG, Craig JC. Cranberries for preventing urinary tract infection. Cochrane Database Syst Rev 2008;1:CD001321.
30. Raz R, Stamm WE. A controlled trial of intravaginal estriol in post-menopausal women with recurrent urinary tract infections. N Engl J Med 1993;329:753–756.
31. Carson C, Naber KG. Role of fluoroquinolones in the treatment of serious bacterial urinary tract infections. Drugs 2004;64:1359–1373.
32. Miller LG, Tang AW. Treatment of uncomplicated urinary tract infections in an era of increasing antimicrobial resistance. Mayo Clin Proc 2004;79:1048–1054.
33. Wong ES, McKevitt M, Running K, et al. Management of recurrent urinary tract infections with patients-administered single-dose therapy. Ann Intern Med 1985;102:302–307.
34. Wagenlehner FME, Naber KG. Treatment of bacterial urinary tract infections: Presence and future. Eur Urol 2006;49:235–244.
35. Gupta K, Sahm DF, Mayfield D, et al. Antimicrobial resistance among uropathogens that cause community-acquired urinary tract infections in women: A nationwide analysis. Clin Infect Dis 2001;33:89–94.
36. Perfetto EM, Gondek EK. Escherichia coli resistance in uncomplicated urinary tract infection: A model for determining when to change first-line empirical antibiotic choice. Manag Care Interface 2002;6: 35–42.
37. Wagenlehner FM, Pilatz A, Naber KG, et al. Anti-infective treatment of bacterial urinary tract infections. Curr Med Chem 2008;15: 1412–1427.
38. Rubenstein JN, Schaeffer AJ. Managing complicated urinary tract infections: The urologic view. Infect Dis Clin North Am 2003;17: 333–351.
39. Ovalle A, Levancini M. Urinary tract infections in pregnancy. Curr Opin Urol 2001;11:55–59.
40. Christensen B. Which antibiotics are appropriate for treating bacteriuria in pregnancy? J Antimicrob Chemother 2000;46(suppl S1): 29–34.
41. McDermott S, Daguise V, Mann H, et al. Perinatal risk for mortality and mental retardation associated with maternal urinary tract infections. J Fam Pract 2001;50:433–437.
42. Nicolle LE, Bradley S, Colgan R, et al. Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults. Clin Infect Dis 2005;40:643–654.
43. Warren JW. The catheter and urinary tract infection. Med Clin North Am 1991;75:481–493.
44. Naber KG, Bergman B, Bishop MC, et al. EAU guidelines for management of urinary and male genital tract infections. Urinary Tract Infection Working Group of the Health Care Office of the European Associated of Urology. Eur Urol 2001;40:576–588.