Amanda Corbett, Rosa Yeh, Julie Dumond, and Angela D.M. Kashuba
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Explain the routes of transmission for HIV, and its natural disease progression.
2. Identify typical and atypical signs and symptoms of acute and chronic HIV infection.
3. Identify the desired therapeutic outcomes for patients with HIV infection.
4. Recommend appropriate first-line pharmacotherapy interventions for patients with HIV infection.
5. Recommend appropriate second-line pharmacotherapy interventions for patients with HIV infection.
6. Describe the components of a monitoring plan to assess effectiveness and adverse effects of pharmacotherapy for HIV infection.
7. Educate patients about the disease state, appropriate lifestyle modifications, and drug therapy required for effective treatment.
KEY CONCEPTS
![]() The treatment goals for HIV infection are to maximally and durably suppress HIV replication, avoid the development of drug resistance, restore and preserve immune function, prevent opportunistic infections, and minimize adverse effects.
The treatment goals for HIV infection are to maximally and durably suppress HIV replication, avoid the development of drug resistance, restore and preserve immune function, prevent opportunistic infections, and minimize adverse effects.
![]() HIV RNA plasma concentrations and CD4+ T-cell counts are used to assess risk of progression to AIDS (or risk for opportunistic infection) and to monitor efficacy and durability of treatment.
HIV RNA plasma concentrations and CD4+ T-cell counts are used to assess risk of progression to AIDS (or risk for opportunistic infection) and to monitor efficacy and durability of treatment.
![]() Effective and complete treatment of HIV infection involves a multidisciplinary approach, which includes pharmacists, clinicians, social workers, and others.
Effective and complete treatment of HIV infection involves a multidisciplinary approach, which includes pharmacists, clinicians, social workers, and others.
![]() Treatment with two nucleoside reverse transcriptase inhibitors (NRTIs) and either a nonnucleoside reverse transcriptase inhibitor (NNRTI) or a ritonavir-boosted protease inhibitor (PI) is the mainstay of initial treatment for HIV infection.
Treatment with two nucleoside reverse transcriptase inhibitors (NRTIs) and either a nonnucleoside reverse transcriptase inhibitor (NNRTI) or a ritonavir-boosted protease inhibitor (PI) is the mainstay of initial treatment for HIV infection.
![]() All patients with HIV infection relapse if medication is withdrawn. Therefore, long-term maintenance treatment is required.
All patients with HIV infection relapse if medication is withdrawn. Therefore, long-term maintenance treatment is required.
![]() Eventually, HIV becomes resistant to current medication therapy. To prolong this time to resistance, both strict adherence to the drug regimen and avoidance of deleterious drug interactions is critical.
Eventually, HIV becomes resistant to current medication therapy. To prolong this time to resistance, both strict adherence to the drug regimen and avoidance of deleterious drug interactions is critical.
![]() The majority of antiretroviral medications are metabolized by the cytochrome P-450 enzyme system (CYP). Therefore, it is important to review patient medication profiles for drugs that may interact with antiretroviral drugs.
The majority of antiretroviral medications are metabolized by the cytochrome P-450 enzyme system (CYP). Therefore, it is important to review patient medication profiles for drugs that may interact with antiretroviral drugs.
![]() Most antiretroviral medications cause acute and chronic adverse effects. Patients should be closely monitored for these toxicities so that interventions can occur quickly.
Most antiretroviral medications cause acute and chronic adverse effects. Patients should be closely monitored for these toxicities so that interventions can occur quickly.
The acquired immune deficiency syndrome (AIDS) was first recognized in 1981, and described in a cohort of young homosexual men with significant immune deficiency. Since then, human immunodeficiency virus type 1 (HIV-1) has been clearly identified as the major cause of AIDS.1 HIV-2 is much less prevalent than HIV-1, but also causes AIDS. HIV primarily targets CD4+ lymphocytes, which are critical to proper immune system function. If left untreated, patients experience a prolonged asymptomatic period followed by rapid, progressive immunodeficiency. Therefore, most complications experienced by patients with AIDS involve opportunistic infections and cancers.
HIV is primarily transmitted by sexual contact, by contact with blood or blood products, and from mother to child during gestation, delivery, or breast-feeding. The prevalence and incidence of HIV is rising globally, and to date there are no treatments which eradicate HIV from the body. Combinations of potent antiretroviral agents (called highly active antiretroviral therapy, or HAART) can suppress HIV replication to undetectable levels, delay the onset of AIDS, and prolong survival. However, there are a number of drug-induced, long-term toxicities that challenge effective patient management. This chapter will address HIV treatment options and challenges, and give practical suggestions for patient management.
EPIDEMIOLOGY
Since the first cases of AIDS were identified in 1981, over 25 million people have died as a result of HIV infection.1 This makes AIDS one of the most destructive epidemics in recorded history. The epidemic remains extremely dynamic, and no country in the world is unaffected. It is estimated that HIV currently infects approximately 35 million people worldwide. Approximately 68% of these cases are in subsaharan Africa, with a prevalence of approximately 5%. East Asia, Central Asia, and Eastern Europe are also seeing rapidly rising infection rates.
In 2007 alone, approximately 2.1 million people died from AIDS and 2.5 million people were newly infected with HIV. Most of these infections were acquired through heterosexual transmission. As of December 2007, women accounted for 47% of all people living with HIV worldwide; in subsaharan Africa, women account for 61%. Persons aged 15 to 24 years accounted for nearly one-half of new HIV infections worldwide.
In the United States, at the end of 2006, an estimated 1,039,000 to 1,185,000 persons were living with HIV/AIDS. Approximately 30% of these are undiagnosed and unaware of their HIV infection, and could be unknowingly transmitting the virus to others. In 2006, of the total number of HIV-infected patients, approximately 509,681 were living with AIDS, while 14,627 died with AIDS. The cumulative estimated number of diagnoses of AIDS through 2006 was 1,014,797, one-half of whom (565,927) had died. Approximately 56,300 people were newly infected with HIV in 2006.
Compared with their distribution in the U.S. population, African American and Hispanic populations are disproportionately affected by HIV/AIDS, representing 48% and 19% of cases, respectively. HIV/AIDS is among the top four causes of death for African American men aged 25 to 44 years and is among the top three causes of death for African American women of the same age. In 2006, HIV/AIDS rates for African American males were seven times those for white males and two times those for Hispanic males. HIV/AIDS rates for African American females were 19 times the rates for white females and four times the rates for Hispanic females.
ETIOLOGY AND PATHOGENESIS
HIV-1 is a retrovirus and member of the genus Lentivirus. These viruses have a characteristically prolonged latency period. There are two molecularly and serologically distinct but related types of HIV: HIV-1 and HIV-2. HIV-2 is a less common cause of the epidemic and is found primarily in West Africa. HIV-1 is categorized by phylogenetic lineages into three groups (M [main], N [new], and O [outlier]). HIV-1 group M can be further categorized into nine subtypes: A through D, F through H, and J and K. HIV-1 subtype B is primarily responsible for the North American and Western European epidemic.
HIV in humans is believed to result from cross-species transmission from primates infected with simian immunodeficiency virus (SIV). HIV-2 is closely related to the SIV found in sooty mangabeys in West Africa, and HIV-1 is similar to the SIV found in chimpanzees. The earliest known human HIV infection was in central Africa in 1959. Cultural practices such as the preparation and eating of bush-meat, or keeping primates as pets, may have allowed the virus to transmit from animal to human. The rapid spread of the virus throughout the world can be primarily attributed to sexual promiscuity, drug abuse, and high mobility due to modern transportation.
HIV infection occurs through three primary modes of transmission: sexual, parenteral, and perinatal. The most common method for transmission is receptive anal and vaginal intercourse, with the probability of transmission up to 30% per sexual contact. The probability of transmission increases when the index partner has a high level of viral replication (which occurs at the beginning of infection or late in disease), or when the uninfected partner has ulcerative disease, compromised mucosal surfaces, or (in the case of men) has not been circumcised.
Parenteral transmission of HIV primarily occurs through injection drug use by sharing contaminated needles or injection-related supplies. As a result of a comprehensive North American screening program, less than 1% of all cases of HIV infection occurs as a result of transfusions of contaminated blood or blood products, or infected transplant organs. Health care workers have a 0.3% estimated risk of acquiring HIV infection through percutaneous needlestick injury.
Perinatal infection (also known as vertical transmission or mother-to-child transmission [MTCT]) can occur during gestation, at or near delivery, and during breast-feeding. The risk of MTCT up to and including delivery is approximately 25%, while the risk of transmission during breast-feeding is approximately 15% to 20% within the first 6 months of life. Because a high rate of HIV replication in the blood is a significant risk factor for transmission of HIV, it is important to treat women for their HIV infection during pregnancy. After delivery, mothers are strongly recommended not to breast-feed if safe alternatives are available.
Understanding the life cycle of the virus is important to know how antiretroviral drugs are combined for optimal therapy (Fig. 87–1). Once HIV enters the body, an outer glycoprotein called gp120 binds to CD4 receptors found on the surface of dendritic cells, T lymphocytes, monocytes, and macrophages. This allows further binding to other chemokine receptors on the cell surface called CCR5 and CXCR4. Greater than 95% of newly infected patients have viruses that preferentially use CCR5 to enter the cell, and most patients with advanced disease have viruses that preferentially use CXCR4 to enter the cell. This becomes important in understanding the place in therapy for some of the new drugs in development.
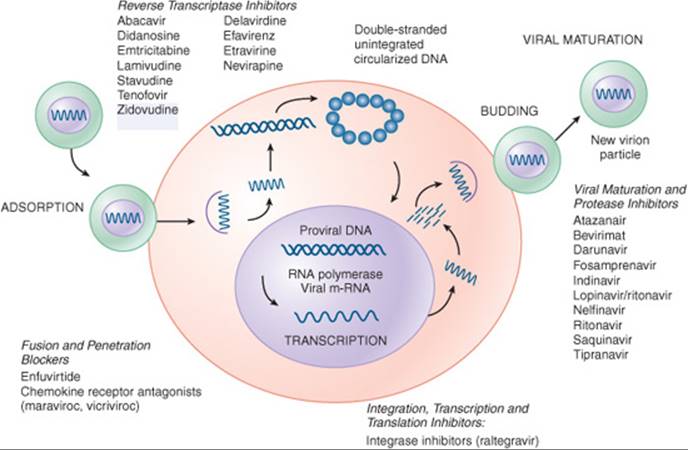
FIGURE 87–1. Life cycle of HIV and targets for antiretroviral drugs. (From Fletcher CV, Kakuda TN. Human immunodeficiency virus infection. In: DiPiro JT, Talbert RL, Yee GC, et al., eds. Pharmacotherapy: A Pathophysiologic Approach. 6th ed. New York: McGraw-Hill; 2005: 2258, with permission.)
After the virus has attached to CD4 and chemokine receptors, another viral glycoprotein (gp41) assists with viral fusion to the cell and internalization of the viral contents. The viral contents include single-stranded RNA, an RNA-dependent DNA polymerase (also known as reverse transcriptase), and other enzymes. Using the single-stranded viral RNA as a template, reverse transcriptase synthesizes a complementary strand of DNA. The single-stranded viral RNA is removed from the newly formed DNA strand by ribonuclease H, and reverse transcriptase completes the synthesis of double-stranded DNA. The viral reverse transcriptase enzyme is highly error-prone, and many mutations occur in the conversion of RNA to DNA. This inefficient reverse transcription activity is responsible for HIV’s ability to rapidly mutate and develop drug resistance.
A chronic infection is established when the double-stranded DNA migrates to the host cell nucleus and is integrated into the host cell chromosome by an HIV enzyme called integrase. Once the cell becomes activated by antigens or cytokines, HIV replication starts: host DNA polymerase transcribes viral DNA into messenger RNA, and messenger RNA is translated into viral proteins. These proteins assemble beneath the bilayer of the host cell, a nucleocapsid forms containing these proteins, and the virus buds from the cell. After budding, the virus matures when an HIV protease enzyme cleaves large polypeptides into smaller functional proteins. Without this process, the virus is unable to infect other cells.
During the early stages of infection, approximately 10 billion virions can be produced each day. Most of the cells containing these viruses will be lysed as a result of budding virions, killed by cytotoxic T-lymphocytes, or undergo apoptosis. However, virus will be protected within some cells (macrophages, T cells in lymph nodes), which can stay dormant for years. The initial immune response against HIV is relatively effective, but it is unable to completely clear the infection, and the patient enters a latent, asymptomatic or mildly symptomatic stage lasting 5 to 15 years. During this time, a high rate of viral replication can be seen in the lymph nodes. Eventually immune deficiency occurs when the body is no longer able to replenish helper T cells at a rate equal to that at which HIV is destroying them.
![]() The goal of therapy is to maximally and durably suppress HIV replication in order to restore and preserve immune system function and minimize morbidity and mortality. Because HIV replication has been found in all areas of the body, it is important to use potent drug therapy that can achieve adequate concentrations in all tissues, including protected sites such as the brain and genital tract.
The goal of therapy is to maximally and durably suppress HIV replication in order to restore and preserve immune system function and minimize morbidity and mortality. Because HIV replication has been found in all areas of the body, it is important to use potent drug therapy that can achieve adequate concentrations in all tissues, including protected sites such as the brain and genital tract.
CLINICAL PRESENTATION AND DIAGNOSIS
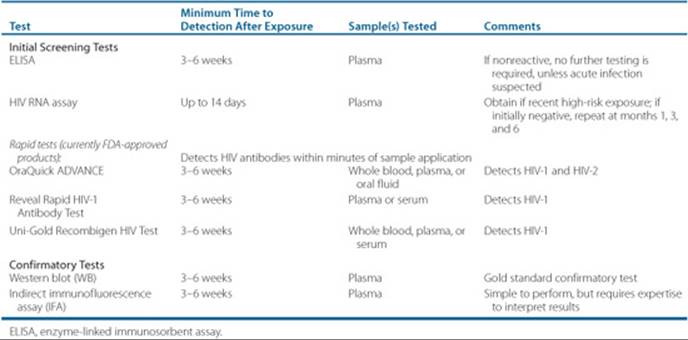
HIV diagnosis is made either by a positive HIV enzyme-linked immunosorbent assay (ELISA) or rapid test (these tests may be positive as soon as 3–6 weeks after infection) and then confirmed by a positive confirmatory test, usually the HIV Western blot (WB) (Table 87–1).
Patients who are acutely infected with HIV may be asymptomatic or present with signs and symptoms associated with any viral infection, such as fever, myalgias, lymphadenopathy, pharyngitis, or rash. Taken together, these are the “acute retroviral syndrome.” Providers should consider the possibility of HIV infection in any patients with these findings and inquire about recent high-risk sexual encounters or other modes of exposure. Risk factors for HIV/AIDS infection include: men who have sex with men (MSM); history of or current IV drug use (needle or equipment sharing); unprotected sexual intercourse with high-risk individuals; the presence of other sexually transmitted infections (e.g., Chlamydia trachomatis or Neisseria gonorrhoeae); persons with coagulation/hemophilia disorders; and previous blood product recipients. Up to 50% of HIV-infected individuals are not aware of their status, thus identifying acutely infected patients and providing referral into HIV care is critical for preventing HIV transmission.2 In acute infection, HIV RNA concentrations in blood and the genital tract are very high, increasing the risk of transmission to others.3 Increased infectiousness coupled with undiagnosed HIV infection in these patients may account for a substantial proportion of sexual HIV transmission.
If patients are not identified during acute infection, they may later present with various nonspecific symptoms such as myalgias, fatigue, weight loss, thrush, or symptoms associated with opportunistic infections. The U.S. Centers for Disease Control (CDC) currently recommends that patients aged 13 to 64 years in all health care settings undergo opt-out HIV testing, meaning that a separate consent form for testing is not needed after the patient has been informed that testing will be performed. For those patients in the high-risk groups mentioned above, HIV testing should be performed on an annual basis.4
The diagnosis of HIV infection is made either by a baseline serologic screening test such as the ELISA or a rapid test. If reactive, then a confirmatory test is performed. The WB is the gold standard confirmatory test and is commonly used. The WB is considered reactive if two of the three major bands (p24, gp41, and/or gp120/160) change color. The test is nonreactive if no viral bands are visible. If the test is indeterminate (one band visible), patients are retested in 2 to 3 months. This is most likely if a recent (i.e., less than 3–6 weeks) infection has occurred, and HIV antibodies have not yet been fully formed. In this case, a plasma HIV RNA concentration (reverse transcriptase polymerase chain reaction [RT-PCR]) should be evaluated. Patients with acute infection will generally have HIV RNA concentrations greater than 106 copies/mL. ![]() HIV severity is determined by following: (a) the CD4+ lymphocyte count (CD4 count) and percentage and (b) HIV RNA (viral load). The CD4 percentage is followed because the absolute count may fluctuate and does not necessarily indicate a change in the patient’s condition.
HIV severity is determined by following: (a) the CD4+ lymphocyte count (CD4 count) and percentage and (b) HIV RNA (viral load). The CD4 percentage is followed because the absolute count may fluctuate and does not necessarily indicate a change in the patient’s condition.
Table 87–1 HIV Diagnostic Tests
Clinical Presentation and Diagnosis of HIV
Patients with acute HIV infection may display symptoms described as “acute retroviral syndrome.” Patients with chronic HIV infection may present with these same nonspecific symptoms and/or opportunistic infections.
Acute Retroviral Syndrome
The majority of patients may present with fever, lymphadenopathy, pharyngitis, and/or rash. Other symptoms include:
• Myalgia or arthralgia
• Diarrhea
• Headache
• Nausea and vomiting
• Hepatosplenomegaly
• Weight loss
• Thrush
• Neurologic symptoms (meningoencephalitis, aseptic meningitis, peripheral neuropathy, facial palsy, or cognitive impairment or psychosis)
Opportunistic Infections
Depending on the severity of immunosuppression (the CD4+ T lymphocyte count), patients may present with the following opportunistic infections (grouped by CD4+ count):
Any CD4+ count
• Mycobacterium tuberculosis disease
• Bacterial pneumonia (commonly Streptococcus pneumoniae, Haemophilus influenzae, Pseudomonas aeruginosa, and Staphylococcus aureus)
• Herpes simplex virus disease
• Varicella zoster virus disease
• Bacterial enteric disease (most commonly Salmonella, Campylobacter, and Shigella)
• Syphilis
• Bartonellosis
Less than 250 cells/mm3
• Coccidioidomycosis
• Pneumocystis jiroveci (formerly carinii) pneumonia (PCP)
• Oropharyngeal and esophageal candidiasis
• Kaposi’s sarcoma or human herpesvirus-8 disease
Less than 150 cells/mm3
• Disseminated histoplasmosis
Less than 100 cells/mm3
• Cryptosporidiosis
• Microsporidiosis
Less than 50 cells/mm3
• Disseminated Mycobacterium avium complex disease
• Cytomegalovirus disease
• Cryptococcosis, aspergillosis, and Toxoplasma gondii encephalitis
Patient Encounter, Part 1
A 46-year-old Caucasian man with a history of hypertension and gastroesophageal reflux disease (GERD) comes to your clinic complaining of increased fatigue, shortness of breath, and cough. He has noticed feeling tired more easily for the past 3 months, but the difficulty breathing and cough appeared 2 weeks ago. After questioning him further, he says he has sex with men, but he has had the same sexual partner for the past 8 years. They do not use condoms. He also says that he smokes about one pack of cigarettes per day.
What information is suggestive of HIV/AIDS?
What risk factors are present for having HIV/AIDS?
What additional information do you need to know before creating a treatment plan for this patient?
TREATMENT
![]() The goals of treatment are to maximally and durably suppress viral replication, avoid the development of drug resistance, restore and preserve immune function, prevent opportunistic infections, and minimize drug adverse effects. Elimination of HIV is not possible with currently available therapies. Instead, maximal suppression of viral replication (defined as HIV RNA concentrations undetectable by the most sensitive assay available) is desired. After the initiation of antiretroviral therapy, a rapid decline to undetectable HIV RNA in 16 to 24 weeks is a predictor of improved clinical outcomes.5
The goals of treatment are to maximally and durably suppress viral replication, avoid the development of drug resistance, restore and preserve immune function, prevent opportunistic infections, and minimize drug adverse effects. Elimination of HIV is not possible with currently available therapies. Instead, maximal suppression of viral replication (defined as HIV RNA concentrations undetectable by the most sensitive assay available) is desired. After the initiation of antiretroviral therapy, a rapid decline to undetectable HIV RNA in 16 to 24 weeks is a predictor of improved clinical outcomes.5
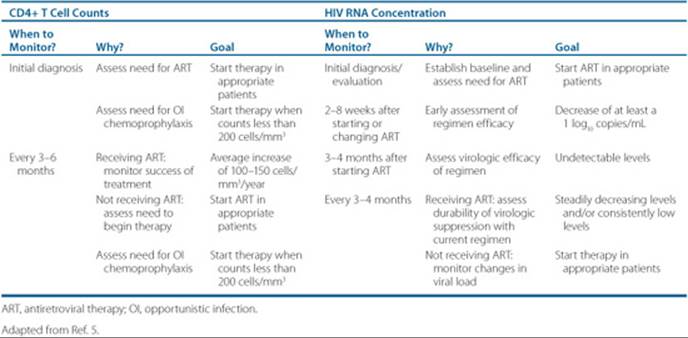
![]() Degree of immune function preservation also correlates with decreased viral replication, and is measured by CD4+ T-cell counts. CD4 measures are the best predictor of progression to AIDS, and help decide when to initiate treatment. At CD4+ T-cell counts of 200 cells/mm3 and lower, patients require drug prophylaxis for opportunistic infections. Table 87–2 details the monitoring end points of HIV treatment for HIV RNA and CD4+ T-cell counts.
Degree of immune function preservation also correlates with decreased viral replication, and is measured by CD4+ T-cell counts. CD4 measures are the best predictor of progression to AIDS, and help decide when to initiate treatment. At CD4+ T-cell counts of 200 cells/mm3 and lower, patients require drug prophylaxis for opportunistic infections. Table 87–2 details the monitoring end points of HIV treatment for HIV RNA and CD4+ T-cell counts.
Six classes of drugs are available to treat HIV infection: nucleoside (NRTI)/nucleotide (NtRTI) reverse tran-scriptase inhibitor, protease inhibitor (PI), nonnucleoside reverse transcriptase inhibitor (NNRTI), fusion inhibitors, CCR5 inhibitors, and integrase inhibitors. ![]() Currently, combination antiretroviral drug therapy with three or more active drugs is the standard of care, which increases the durability of viral suppression and decreases the potential for the development of resistance.
Currently, combination antiretroviral drug therapy with three or more active drugs is the standard of care, which increases the durability of viral suppression and decreases the potential for the development of resistance. ![]() Two nucleoside (nucleotide) reverse transcriptase inhibitors and either a NNRTI or a ritonavir-boosted PI are the mainstay regimens of combination therapy in initial treatment. In the late 1980s, when only zidovudine was available, achieving and maintaining viral suppression for more than 4 months was rarely possible. As more agents became available in the mid-1990s (most notably the PIs), HIV RNA was suppressed to undetectable concentrations and maintained there for long periods of time. Currently recommended combination regimens decrease HIV RNA to less than 50 copies/mL in 80% to 90% of patients in clinical trials. Therefore, monotherapy with any agent or the use of NRTIs without a PI or NNRTI are not routine treatment options. Fusion inhibitors, CCR5 inhibitors, and integrase inhibitors are only FDA-approved for use in treatment experienced patients with drug resistance to NRTIs, NNRTIs, and/or PIs, although clinical trials are ongoing to determine their role in the initial treatment of HIV infection. Figure 87–1 details the mechanisms of action of the drug classes within the life cycle of HIV.
Two nucleoside (nucleotide) reverse transcriptase inhibitors and either a NNRTI or a ritonavir-boosted PI are the mainstay regimens of combination therapy in initial treatment. In the late 1980s, when only zidovudine was available, achieving and maintaining viral suppression for more than 4 months was rarely possible. As more agents became available in the mid-1990s (most notably the PIs), HIV RNA was suppressed to undetectable concentrations and maintained there for long periods of time. Currently recommended combination regimens decrease HIV RNA to less than 50 copies/mL in 80% to 90% of patients in clinical trials. Therefore, monotherapy with any agent or the use of NRTIs without a PI or NNRTI are not routine treatment options. Fusion inhibitors, CCR5 inhibitors, and integrase inhibitors are only FDA-approved for use in treatment experienced patients with drug resistance to NRTIs, NNRTIs, and/or PIs, although clinical trials are ongoing to determine their role in the initial treatment of HIV infection. Figure 87–1 details the mechanisms of action of the drug classes within the life cycle of HIV.
Table 87–2 Monitoring End Points for CD4+ T-Cell Counts and HIV RNA
Nonpharmacologic Interventions
![]() Patient adherence is a key component in treatment success. Drug therapy is required for a lifetime, as the virus begins to replicate at high levels when medications are stopped. Early HIV combination therapy was exceedingly complicated for patients, with multiple daily doses, varying food restrictions, and large pill burdens. Advances in delivery and formulation s now make possible once - or twice-daily dosing with fewer than six pills per day. Currently, a combination tablet of tenofovir + emtricitabine + efavirenz (Atripla) supplies a one pill, once-daily regimen. The use of low-dose ritonavir to enhance the concentrations of other PIs (known as pharmacokinetic enhancement or “boosting”) allows for significantly fewer doses and lower pill burdens. Atazanavir with ritonavir boosting is a potent, once-daily PI option. These advances, however, do not replace the need for patient counseling by a trained pharmacist and a multidisciplinary approach to promoting adherence.
Patient adherence is a key component in treatment success. Drug therapy is required for a lifetime, as the virus begins to replicate at high levels when medications are stopped. Early HIV combination therapy was exceedingly complicated for patients, with multiple daily doses, varying food restrictions, and large pill burdens. Advances in delivery and formulation s now make possible once - or twice-daily dosing with fewer than six pills per day. Currently, a combination tablet of tenofovir + emtricitabine + efavirenz (Atripla) supplies a one pill, once-daily regimen. The use of low-dose ritonavir to enhance the concentrations of other PIs (known as pharmacokinetic enhancement or “boosting”) allows for significantly fewer doses and lower pill burdens. Atazanavir with ritonavir boosting is a potent, once-daily PI option. These advances, however, do not replace the need for patient counseling by a trained pharmacist and a multidisciplinary approach to promoting adherence.
Counsel all patients initially and repeatedly on ways to prevent viral transmission. Preventing the spread of resistant virus is particularly important. Patients receiving antiretroviral therapy can still transmit virus to sexual partners, and to those with whom they share needles or other drug equipment. Where both partners are HIV-positive, safe sex and needle practices reduce the risk of superinfection with differing strains of HIV and the transmission of other sexually transmitted diseases. General guidelines for preventing viral transmission include using condoms with a water-based lubricant for vaginal or anal intercourse, using condoms without lubricant or dental dams for oral sex, and not sharing equipment used to prepare, inject, or inhale drugs. Treating other sexually transmitted infections (STIs), particularly genital herpes, in HIV-infected patients may help to prevent HIV transmission. The presence of STIs increases genital tract HIV viral load, and correspondingly the risk of HIV transmission to sexual partners.
![]() Nutrition and dietary counseling should also be included in the care of the HIV patient, as poor nutrition leads to poorer outcomes and complicates treatment. Antiretroviral therapy itself introduces a host of nutritional issues, including drug-food interactions, GI adverse effects that may affect appetite and limit dietary intake, lipid abnormalities, and fat redistribution. The American Dietetics Association currently recommends assessing HIV-infected patients for their level of nutritional risk and involving a registered dietician as part of the clinical team for optimal nutrition care.6
Nutrition and dietary counseling should also be included in the care of the HIV patient, as poor nutrition leads to poorer outcomes and complicates treatment. Antiretroviral therapy itself introduces a host of nutritional issues, including drug-food interactions, GI adverse effects that may affect appetite and limit dietary intake, lipid abnormalities, and fat redistribution. The American Dietetics Association currently recommends assessing HIV-infected patients for their level of nutritional risk and involving a registered dietician as part of the clinical team for optimal nutrition care.6
Pharmacologic Therapy for Antiretroviral-Naïve Patients
Two major panels of experts publish guidelines for the treatment of HIV-infected individuals. Although the recommendations are quite similar, slight differences do exist between the Department of Health and Human Services (DHHS) Guidelines5 and the International AIDS Society-USA (IAS-USA) Panel Recommendations.7 The DHHS Guidelines are updated every 6 months and current and archived versions are available online at www.aidsinfo.nih.gov. The IAS-USA Guidelines were last updated in 2008, and in 2006 prior to that revision. Due to the intense research and constant modifications to therapeutic approaches in the treatment of HIV, the majority of the treatment algorithms and recommendations presented herein follow the most up-to-date information found in the DHHS recommendations.
![]() The decision of when to begin antiretroviral therapy is complex. Recommendations are based on the CD4+ T-cell count, which predicts disease-free survival (Table 87–3). Other factors to consider include the patient’s viral load, willingness to begin therapy and maintain medication adherence, and the risk versus benefit of treating an asymptomatic patient. The clinical evidence is strongest for beginning treatment at CD4+ counts less than 200 cells/mm3, but more recent evidence from long-term studies and the availability of potent drugs with improved tolerability support earlier treatment at higher CD4+ counts. Once the decision is made to initiate treatment, the regimen is selected based on patient-specific factors.
The decision of when to begin antiretroviral therapy is complex. Recommendations are based on the CD4+ T-cell count, which predicts disease-free survival (Table 87–3). Other factors to consider include the patient’s viral load, willingness to begin therapy and maintain medication adherence, and the risk versus benefit of treating an asymptomatic patient. The clinical evidence is strongest for beginning treatment at CD4+ counts less than 200 cells/mm3, but more recent evidence from long-term studies and the availability of potent drugs with improved tolerability support earlier treatment at higher CD4+ counts. Once the decision is made to initiate treatment, the regimen is selected based on patient-specific factors. ![]() All recommended regimens for initial treatment contain either an NNRTI or a ritonavir-boosted PI in combination with two NRTIs (or NtRTI). The preferred agents are:
All recommended regimens for initial treatment contain either an NNRTI or a ritonavir-boosted PI in combination with two NRTIs (or NtRTI). The preferred agents are:
Table 87–3 Summary of Recommendations for Initiating Antiretroviral Therapy

1. NRTI/NtRTI combinations:
a. Tenofovir and emtricitabine
2. PIs:
b. Lopinavir/ritonavir (dosed once or twice daily)
c. Atazanavir/ritonavir (dosed once daily)
d. Fosamprenavir/ritonavir (dosed twice daily)
e. Darunavir/ritonavir (dosed once daily)
3. NNRTI:
a. Efavirenz
The decision to choose a NNRTI- or PI-based regimen as initial therapy is based on many patient- and clinician-specific factors. Drug resistance testing should be performed at diagnosis, and again prior to initiating treatment, if time has elapsed between diagnosis and treatment (see Pharmacologic Treatment for Antiretroviral-Experienced Patients for further discussion of drug resistance testing). The results of resistance testing may dictate which drug class is preferred; 6% to 16% of newly diagnosed patients will have drug-resistant virus. This initial resistance pattern often involves the NNRTIs, but may involve other drug classes. NNRTI-based regimens have low pill burdens and may have decreased incidences of long-term adverse effects (e.g., dyslipidemia) in comparison to some PI-based regimens. However, this class also has a low threshold for drug resistance (the K103N mutation causes high level cross-class resistance), and patient adherence is a critical consideration. In pregnant women, or women with the potential to become pregnant, a PI-based regimen is preferred due to the potential teratogenicity of efavirenz (pregnancy category D).
In patients who cannot tolerate the above preferred firstline therapies, or have a compelling reason to choose a different agent, the following alternatives are recommended.
1. NRTI:
a. Zidovudine and lamivudine
b. Didanosine and (emtricitabine or lamivudine)
c. Abacavir and lamivudine
2. PIs:
a. Atazanavir (if tenofovir is included in the regimen, ritonavir must be used)
b. Fosamprenavir (dosed twice daily)
c. Fosamprenavir/ritonavir (dosed once daily)
d. Saquinavir/ritonavir
3. NNRTI:
a. Nevirapine in selected populations (due to a more frequent incidence of hepatotoxicity, nevirapine should only be used in patients with low to moderate CD4+ T-cell counts: less than or equal to 250 cells/mm3 for females, less than or equal to 400 cells/mm3 for males)
If abacavir is included in a regimen, patients should undergo HLA-B*5701 testing prior to initiation to reduce the risk of abacavir hypersensitivity. Patients who test positive for the allele are at high risk (approximately 70%) of developing this reaction, and should not be given abacavir. An abacavir allergy should also be documented in the patient’s medical record to prevent future administration. Those patients with a negative test may receive abacavir, but should still be monitored for the development of hypersensitivity.
Therapies not recommended for initial treatment due to poor potency or significant toxicity include triple-NRTI regimens, delavirdine, nevirapine in patients with moderate to high CD4+ T-cell counts, indinavir ± ritonavir, saquinavir used without ritonavir (“unboosted”), ritonavir used without another PI, nelfinavir, tipranvir/ritonavir, and tenofovir with didanosine. Due to lack of data in antiretroviral naïve patients, maraviroc, etravirine, enfuvirtide, and raltegravir are not recommended in the DHHS guidelines.
Drugs that should not be combined due to overlapping toxicities include: atazanavir plus indinavir (due to enhanced hyperbilirubinemia), two NNRTIs, and didanosine plus stavudine. Emtricitabine and lamivudine should not be combined because of their similar chemical structures, and antagonism can result when stavudine is combined with zidovudine.
Pharmacologic Therapy for Antiretroviral-Experienced Patients
![]() Ongoing viral replication, whether at low levels in the face of adequate drug concentrations or at higher levels due to inconsistent systemic concentrations (or low concentrations in sanctuary sites; e.g., male and female genital fluids, cerebrospinal fluid, or lymph nodes), will eventually lead to resistance to the prescribed medications. There is no consensus on the optimal time to change therapy based on virologic or immunologic failure (Table 87–4).
Ongoing viral replication, whether at low levels in the face of adequate drug concentrations or at higher levels due to inconsistent systemic concentrations (or low concentrations in sanctuary sites; e.g., male and female genital fluids, cerebrospinal fluid, or lymph nodes), will eventually lead to resistance to the prescribed medications. There is no consensus on the optimal time to change therapy based on virologic or immunologic failure (Table 87–4). ![]() Virologic failure is defined as HIV RNA greater than 400 copies/mL after 24 weeks, greater than 50 copies/mL after 48 weeks, or a repeated HIV RNA greater than 400 copies/mL after prior suppression to less than 400 copies/mL.5Some clinicians may change therapy with any repeated, detectable viremia (HIV RNA greater than 50–400 copies/mL), while others will set arbitrary thresholds of 1,000 to 1,500 copies/mL. Immunologic failure is defined as having an increase of less than 25 to 50 cells/mm3 in CD4+ T lymphocyte count above baseline after 1 year of therapy, or a decline in CD4+ cell count below baseline while taking antiretroviral therapy.
Virologic failure is defined as HIV RNA greater than 400 copies/mL after 24 weeks, greater than 50 copies/mL after 48 weeks, or a repeated HIV RNA greater than 400 copies/mL after prior suppression to less than 400 copies/mL.5Some clinicians may change therapy with any repeated, detectable viremia (HIV RNA greater than 50–400 copies/mL), while others will set arbitrary thresholds of 1,000 to 1,500 copies/mL. Immunologic failure is defined as having an increase of less than 25 to 50 cells/mm3 in CD4+ T lymphocyte count above baseline after 1 year of therapy, or a decline in CD4+ cell count below baseline while taking antiretroviral therapy.
Treatment considerations for antiretroviral-experienced patients are much more complex than for patients who are naïve to therapy. Prior to changing therapy, the reasons for treatment failure should be identified. A comprehensive review of the patient’s severity of disease, antiretroviral treatment history, adherence to therapy, intolerance or toxicity, concomitant drug therapies, comorbidities, and results of current and past HIV resistance testing should be performed. If patients fail therapy due to poor adherence, the underlying reasons must be determined and addressed prior to initiation of new therapy. Reasons for poor adherence include: problems with medication access, active substance abuse, depression and/or denial of the disease, and a lack of education on the importance of 100% adherence to therapy. Medication intolerance or toxicity can be remedied with therapy for the adverse event, exchanging the drug causing the toxicity with another in the same class, or changing the entire regimen. Pharmacokinetics or systemic drug exposure can be optimized by ensuring maximal drug absorption (taking the drug with or without food can alter exposure by up to 30%), and avoiding interactions with concomitant prescription or nonprescription medications and dietary supplements or natural products (e.g., antacids, St. John’s wort, and garlic). When causes for treatment failure are identified, appropriate strategies for therapy can be determined.
Table 87–4 Treatment Options Following Virologic Failure With the Initial Regimen Initial Regimen

An additional consideration when stopping or changing therapy is a staggered discontinuation of antiretrovirals with different half-lives. For example, in patients taking Atripla (tenofovir, emtricitabine, and efavirenz) tenofovir and emtricitabine should be continued for at least 4 days after discontinuation of efavirenz due to the much prolonged half-life of efavirenz as compared to tenofovir and emtricitabine. Otherwise, the potential for monotherapy with efavirenz exists. If new antiretroviral therapy is to be initiated immediately, no overlap is necessary; however, it should be noted that efavirenz concentrations will persist for some period of time.
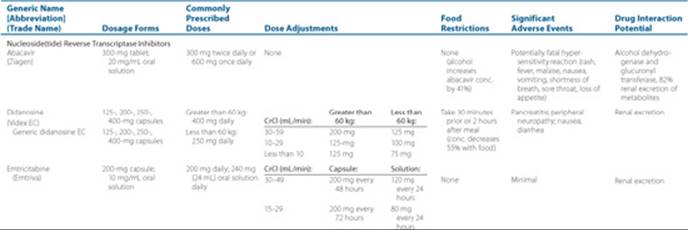
Drug interactions between antiretrovirals and between antiretrovirals and concomitant medications should be evaluated for each patient to avoid under- and/or overexposure of either therapy. ![]() NNRTIs and PIs are metabolized by CYP450 enzymes and are inducers and/or inhibitors of this enzyme system. In addition, some of the antiretrovirals are substrates, inhibitors, and/or inducers of transporters such as P-glycoprotein, and therefore may lead to drug interactions. Information provided in Table 87–5 describes the drug interaction potential of each antiretroviral. Due to the ever-changing drug interactions with this class of medications, the regularly updated DHHS Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents are a recommended source of specific drug interactions.5
NNRTIs and PIs are metabolized by CYP450 enzymes and are inducers and/or inhibitors of this enzyme system. In addition, some of the antiretrovirals are substrates, inhibitors, and/or inducers of transporters such as P-glycoprotein, and therefore may lead to drug interactions. Information provided in Table 87–5 describes the drug interaction potential of each antiretroviral. Due to the ever-changing drug interactions with this class of medications, the regularly updated DHHS Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents are a recommended source of specific drug interactions.5
Table 87–5 Summary of Currently Available Antiretroviral Agents









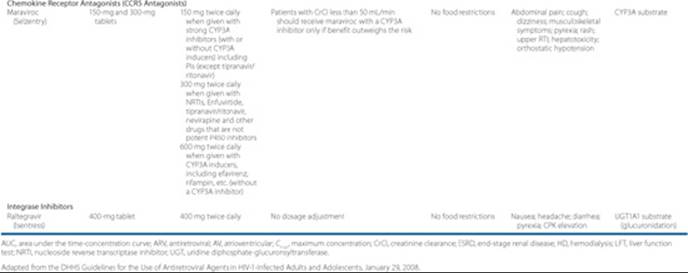
The goals of therapy differ for antiretroviral-experienced patients that have limited drug exposure (i.e., developing resistance to their first antiretroviral regimen) versus those with extensive exposure (i.e., developing resistance to their third or fourth antiretroviral regimen). It is reasonable to expect maximal viral suppression in those with limited drug exposure. However, this may not be feasible for patients with prior exposure to multiple medications. ![]() In antiretroviral-experienced patients, a reasonable goal is to simply preserveimmune function and prevent clinical progression.
In antiretroviral-experienced patients, a reasonable goal is to simply preserveimmune function and prevent clinical progression.
Several issues need to be considered in choosing a salvage regimen for HIV infection. Knowing prior medication exposure can assist in identifying which drugs to avoid. However, direct HIV resistance testing can better identify the resistance and susceptibility patterns of the major viral strains. Because HIV may be susceptible to certain components of the failing antiretroviral regimen, these drugs can be recycled into future regimens. Resistance testing should be used when all patients enter into care, in patients with virologic failure on a current ARV regimen, or with suboptimal suppression after initiation of ARV therapy. Testing is generally preferred for antiretroviral naïve patients. For resistance testing to be useful, the patient should have a plasma HIV RNA of at least 1,000 copies/mL, and should be currently taking their antiretroviral medications (or be within 4 weeks of discontinuing antiretroviral therapy). This viral concentration is necessary to yield reliable amplification of the virus, and the antiretroviral medications are needed because the dominant viral species reverts to wild-type within 4 to 6 weeks after medications are stopped.
Two types of HIV resistance testing are available, genotyping and phenotyping. Genotyping involves detecting mutations by genetically sequencing the virus, while phenotyping determines the ability of the virus to replicate in the presence of varying ARV concentrations. Genotyping is more rapid and less costly than phenotyping, but results in a list of mutations that may be more difficult to interpret than phenotyping. A virtual phenotype report may also be obtained when genotypes are ordered.8–15 This compares the patient’s viral sequence to a database of matched genotypes and drug susceptibilities. Because the predictability of virtual phenotypes are dependent on the robustness of the database from which they are derived, some clinicians believe their utility is limited. Web-based tools are available to assist with interpretation of resistance mutations (e.g., Stanford University’s HIV Drug Resistance Database; http://hivdb.stanford.edu/index.html). However, expert interpretation of genotype and phenotype reports is recommended.
Certain guiding principles should be considered when treating ARV-experienced patients, and expert opinion is advised before selecting therapy. ![]() As with ARV-naïve patients, three or more active drugs should be prescribed.11,16–18 Because considerable cross-resistance can occur between medications within an antiretroviral class, simply using drugs to which the patient has not been exposed may be insufficient. Complete cross-resistance occurs within the NNRTI class, whereas the NRTIs and PIs have variable overlapping resistance patterns. For this reason, HIV resistance assays are important tools for choosing subsequent effective therapies. The following factors are associated with superior virologic response: lower viral load at the time therapy is changed, using a new class of antiretroviral agent, and using ritonavir enhanced PIs in patients previously exposed to PIs.19,20
As with ARV-naïve patients, three or more active drugs should be prescribed.11,16–18 Because considerable cross-resistance can occur between medications within an antiretroviral class, simply using drugs to which the patient has not been exposed may be insufficient. Complete cross-resistance occurs within the NNRTI class, whereas the NRTIs and PIs have variable overlapping resistance patterns. For this reason, HIV resistance assays are important tools for choosing subsequent effective therapies. The following factors are associated with superior virologic response: lower viral load at the time therapy is changed, using a new class of antiretroviral agent, and using ritonavir enhanced PIs in patients previously exposed to PIs.19,20
Table 87–4 provides general treatment options based on previous drug use. If patients fail therapy with resistance to only one drug, one or two active agents may be substituted for this drug while retaining the remaining drugs in the regimen. If patients fail therapy with resistance to more than one drug, changing classes of antiretrovirals and/or adding new active drugs is warranted. New NRTIs should be selected from resistance testing. If this is not available, the assumption should be made that resistance has developed to all NRTIs used in the failing regimen. In general, HIV that is resistant solely to lamivudine and/or emtricitabine will be susceptible to other NRTIs. If HIV develops resistance solely to tenofovir, then it may have reduced susceptibility to didanosine, but should remain susceptible to zidovudine, stavudine, lamivudine, emtricitabine, and abacavir. Cross-resistance occurs between zidovudine and stavudine.
Three new antiretrovirals have been FDA-approved for use in antiretroviral experienced patients. Two of these agents, Isentress (raltegravir) and Selzentry (maraviroc) have unique mechanisms of action compared to previous agents lending to their advantage in treating antiretroviral experienced patients with limited treatment options and resistance. The final agent, Intelence (etravirine), a member of the NNRTI class, is known as a second-generation NNRTI which is effective at reducing viral load in patients with first generation NNRTI resistance virus. These agents add to the antiretroviral armamentarium in HIV infected treatment experienced patients.
If a patient appears to fail an antiretroviral regimen without detectable HIV resistance, adherence should be investigated, and the adequacy of the plasma HIV RNA concentration in the resistance sample confirmed. Options include continuing the current regimen or starting a new regimen and repeating the resistance test 2 to 4 weeks after adherence is verified. An increasing number of patients have extensive HIV resistance, such that antiretroviral regimens cannot be designed to which the virus is fully susceptible. For these patients, continuing the current regimen may be beneficial because drug-resistant virus may have a compromised replication capacity. Other strategies may be considered for this type of patient, including pharmacokinetic enhancement with ritonavir, retreatment with prior antiretroviral agents, treatment with multidrug regimens (four or more antiretroviral drugs), and the use of new agents through expanded access programs or clinical trials.
Treatment Considerations in Special Populations
Acute HIV Infection
Diagnosis of acute HIV infection is difficult, since many patients are asymptomatic, or have nonspecific clinical symptoms similar to other common respiratory infections. If acute HIV infection is suspected, HIV antibody tests and a plasma HIV RNA concentration should be obtained. A clear diagnosis is made when an HIV antibody test is negative and the plasma HIV RNA concentration is high. There are limited outcomes data for treating acutely infected patients. Treatment of acute infection can decrease the severity of acute disease and decrease the viral set point; this may decrease progression rates and reduce the rate of viral transmission.21–25 Limitations include an increased risk of chronic drug-induced toxicities and the development of viral resistance. Resistance testing should be performed prior to initiation of therapy due to an increase in resistance of antiretroviral naïve patients.5
Adolescent Patients
As a result of similar modes of HIV transmission, adolescents infected after puberty are treated with similar considerations as adults. In this population, dosing of antiretroviral drugs should not be based on age, but on the Tanner stage (which considers external primary and secondary sexual characteristics). Adolescents in early puberty should be dosed according to pediatric guidelines, while those in late puberty should be dosed as adults. During growth spurts, adolescents should be monitored closely for drug efficacy and toxicity, since rapid changes in weight can lead to altered drug concentrations. Adherence is of concern in this population due to denial of the disease, misinformation, distrust of health care professionals, low self-esteem, and lack of family and/or social support. Additionally, asymptomatic patients this age find it more difficult to adhere to therapy while feeling well.
Pediatric Patients
There are unique considerations in the treatment of HIV-infected children. Specific treatment guidelines exist,26 but a thorough review is outside the scope of this chapter. Most children acquire HIV infection through perinatal transmission either in utero, intrapartum, or postpartum through breast-feeding, although antiretroviral interventions have dramatically reduced transmission rates.5 Antiretroviral therapy is limited in pediatric patients, as some drugs have no dosing recommendations for this population, or are not available in a formulation that can be easily administered to children. Additionally, drug exposures can change dramatically during ontogeny due to altered drug-metabolizing enzyme and drug transporter activities.
Drugs of Abuse
Treatment challenges in illicit drug users include comorbidi-ties (such as hepatitis infection), limited access to care, inadequate adherence to therapy, side effects and toxicities, and the need for treatment of substance abuse which can lead to drug interactions. Many drugs of abuse have the potential to interact with antiretroviral medications, and a number of case reports have documented drug overdose when combined with PI therapy.27 In these populations, without addiction control, adherence is suboptimal and treatment failure is common.2 Most PIs and NNRTIs decrease methadone concentrations up to 50%. As this can result in the development of withdrawal symptoms, patients should be closely monitored for 4 to 8 weeks after initiation of antiretroviral therapy. Withdrawal symptoms can be alleviated with a methadone dose increase of 5 to 10 mg. Although there are fewer data, buprenorphine concentrations may be similarly affected, and therefore this drug requires close monitoring.
Pregnancy and Women ofReproductive Potential
The goals of antiretroviral therapy for women of reproductive age and pregnant women are the same as for other adult patients. Specific guidelines for HIV-infected pregnant women are available.28Recommended therapies in pregnancy include zidovudine, lamivudine, lopinavir/ritonavir, and if the CD4+ count is less than 250 cells/mm3, nevirapine. Drugs to be avoided include efavirenz (due to potential teratogenicity), the combination of didanosine and stavudine (due to a high incidence of lactic acidosis), nevirapine in patients with a CD4 count greater than 250 cells/mm3 (due to an increased risk of hepatotoxicity), and the liquid formulation of amprenavir (due to high concentrations of propylene glycol). The goal of therapy is to reduce plasma HIV RNA below 1,000 copies/mL and prevent MTCT of HIV. Limited data are available on antiretroviral pharmacokinetics in pregnancy, and standard doses of antiretroviral drugs are currently recommended with close HIV RNA and CD4 monitoring in the third trimester of pregnancy. Some experts will consider increasing lopinavir/ritonavir dosing in the third trimester from 2 to 3 tablets twice daily; however, studies are underway to better assess this approach.28
Women of reproductive potential prescribed efavirenz should be counseled on its potentially teratogenic effects and the importance of birth control. ![]() Additionally, nevirapine, nelfinavir, ritonavir, lopinavir/ritonavir, and tipranavir/ritonavir decrease the concentrations of estrogens and/or progestins in oral contraceptives, which could lead to failure.5 For patients prescribed these drugs, barrier forms of contraception are preferred to prevent pregnancy. Atazanavir may be taken with oral contraceptives with caution, as it can increase or decrease the exposure to estrogen and progesterone, depending on whether it is used in combination with ritonavir. DepoProvera may be a safe alternative, as it does not affect nelfinavir, efavirenz, or nevirapine concentrations; although the effect of antiretrovirals on medroxyprogesterone concentrations has not been examined, no evidence of ovulation has been seen in women on these combinations.29
Additionally, nevirapine, nelfinavir, ritonavir, lopinavir/ritonavir, and tipranavir/ritonavir decrease the concentrations of estrogens and/or progestins in oral contraceptives, which could lead to failure.5 For patients prescribed these drugs, barrier forms of contraception are preferred to prevent pregnancy. Atazanavir may be taken with oral contraceptives with caution, as it can increase or decrease the exposure to estrogen and progesterone, depending on whether it is used in combination with ritonavir. DepoProvera may be a safe alternative, as it does not affect nelfinavir, efavirenz, or nevirapine concentrations; although the effect of antiretrovirals on medroxyprogesterone concentrations has not been examined, no evidence of ovulation has been seen in women on these combinations.29
Hepatitis B Coinfection
HIV-infected patients coinfected with hepatitis B virus (HBV) have higher concentrations of DNA and hepatitis B early antigen (HBeAg), and higher rates of HBV-associated liver disease. Therapy for HBV should be offered to patients who are HBeAg-positive, or have HBV DNA greater than 105 copies/mL and have either liver serologies (alanine aminotransferase) greater than two times the upper limit of normal or histologic evidence of moderate disease or fibrosis. Options include interferon alfa 2a or 2b and nucleoside/tide analogs. Nucleoside/tide analogs that treat HBV but not HIV are adefovir and entecavir. Nucleoside/tide analogs that treat HBV and HIV are lamivudine, emtricitabine, and tenofovir. These latter agents should be considered when treating HIV infection in HBV coinfected patients.
Hepatitis C Coinfection
Patients coinfected with hepatitis C virus (HCV) and HIV have a threefold increase in rate of progression to cirrhosis compared to those with HCV alone. Therapy for HCV is considered in patients with detectable plasma HCV RNA and a liver biopsy showing bridging or portal fibrosis. Patients with HCV genotype 2 and 3 (and a CD4 count greater than 200 cells/mm3) treated with pegylated interferon plus ribavirin have a better sustained viral response at 48 weeks (60%–70%) compared to those with HCV genotype 1 (15%–28%). Comprehensive treatment guidelines for HIV/HCV coinfected patients are available.30–32 Important considerations addressed in these guidelines include avoiding the combination of ribavirin with didanosine, and stavudine (due to increased risk of pancreatitis and/or lactic acidosis) and ribavirin with zidovudine (due to increased risk of anemia). Growth factors and erythropoietin may be needed to treat neutropenia from interferon and anemia from ribavirin. Antiretrovirals such as nevirapine, efavirenz, and tipranavir are hepatotoxic and in most cases should be avoided in HCV/HIV-infected patients.
Patient Encounter, Part 2: Medical History, Physical Exam, and Diagnostic Tests
PMH: Hypertension for 5 years; it is often not well controlled because of poor patient adherence; GERD, currently controlled on histamine antagonists; history of hepatitis B
FH: Father died of myocardial infarction at the age of 68 years; mother is still alive with history of diabetes
SH: Works as a truck driver; reports distant history of IV drug use in his 20s; drinks alcohol occasionally
Meds: Hydrochlorothiazide 25 mg by mouth once daily; famotidine 20 mg by mouth twice daily
ROS: (+) weight loss, decreased appetite, shortness of breath, and cough; (−) chest pain, nausea, vomiting, diarrhea
PE:
VS: BP 144/84 mm Hg, P 100 bpm, RR 22 per minute,
T 38.3°C (100.9°F)
HEENT: Mild thrush on tongue
CV: RRR, normal S1, S2; no murmurs, rubs, gallops
Abd: Soft, nontender, nondistended; (+) bowel sounds, no hepatosplenomegaly
Rectal: Deferred
Labs: Sodium 135 mEq/L (135 mmol/L), potassium 3.6 mEq/L (3.6 mmol/L), chloride 100 mEq/L (100 mmol/L), bicarbonate 24 mEq/L (24 mmol/L), blood urea nitrogen 14 mg/dL (5 mmol/L), creatinine 1.0 mg/dL (88 μmol/L), WBC 5.2 × 103/mm3 (5.2 × 109/L), hemoglobin 11.5 g/dL (115 g/L or 7.1 mmol/L), hematocrit 34.1%, platelets 151 × 103/mm3 (151 × 109/L), neutrophils 58%, bands 9%, lymphocytes 32%, monocytes 1%, eosinophils 0%, basophils 0%, CD4 150 cells/mm3
HIV ELISA: Pending
CXR: Diffuse interstitial infiltrates bilaterally
Given this additional information, what is your assessment of the patient’s condition?
What other laboratory tests would you recommend?
Identify your treatment goals for the patient.
What nonpharmacologic and pharmacologic alternatives are available for the patient?
OUTCOME EVALUATION
![]() The success of antiretroviral therapy is measured by the degree to which the therapy: (a) restores and preserves immunologic function, (b) maximally and durably suppresses HIV RNA, (c) improves quality of life, (d) reduces HIV-related morbidity and mortality, and (e) prevents opportunistic infections.
The success of antiretroviral therapy is measured by the degree to which the therapy: (a) restores and preserves immunologic function, (b) maximally and durably suppresses HIV RNA, (c) improves quality of life, (d) reduces HIV-related morbidity and mortality, and (e) prevents opportunistic infections. ![]() The major outcome parameters are CD4+ lymphocyte absolute count and percentage, and plasma HIV RNA. Adequate immunologic response in antiretroviral-naïve patients consists of an increase in CD4+ cell count that averages 50 to 150 cells/mm3 (with a faster response in the first 3 months), and a 1 log decrease in HIV RNA by 2 to 8 weeks after starting medications, followed by concentrations less than 50 copies/mL by 12 to 16 weeks (if HIV RNA less than 100,000/mL or by 16–24 weeks if HIV RNA greater than 100,000/mL). Upon initiating or changing antiretroviral therapy, HIV RNA should be measured after 2 to 8 weeks and every 4 to 8 weeks until undetectable. Once stable, HIV RNA and CD4 count are monitored generally every 3 to 6 months. In highly treatment-experienced patients, adequate immunologic response may be only a stable, or slightly increased, CD4 T-cell count, and a stable HIV RNA. This may be enough to prevent clinical progression. However, with the recent new agents and new therapeutic classes of antiretrovirals available for treatment-experienced patients, the goal of treatment should be to reestablish maximal viral suppression to less than50 HIV RNA copies/mL.
The major outcome parameters are CD4+ lymphocyte absolute count and percentage, and plasma HIV RNA. Adequate immunologic response in antiretroviral-naïve patients consists of an increase in CD4+ cell count that averages 50 to 150 cells/mm3 (with a faster response in the first 3 months), and a 1 log decrease in HIV RNA by 2 to 8 weeks after starting medications, followed by concentrations less than 50 copies/mL by 12 to 16 weeks (if HIV RNA less than 100,000/mL or by 16–24 weeks if HIV RNA greater than 100,000/mL). Upon initiating or changing antiretroviral therapy, HIV RNA should be measured after 2 to 8 weeks and every 4 to 8 weeks until undetectable. Once stable, HIV RNA and CD4 count are monitored generally every 3 to 6 months. In highly treatment-experienced patients, adequate immunologic response may be only a stable, or slightly increased, CD4 T-cell count, and a stable HIV RNA. This may be enough to prevent clinical progression. However, with the recent new agents and new therapeutic classes of antiretrovirals available for treatment-experienced patients, the goal of treatment should be to reestablish maximal viral suppression to less than50 HIV RNA copies/mL.
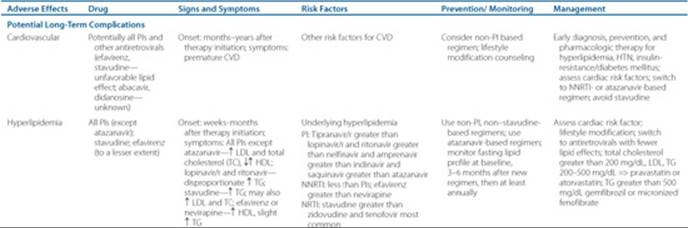
Each patient should have a plan to assess the effectiveness of antiretroviral therapy after initiation. At each clinic visit, patients should be evaluated for the presence of adverse drug reactions, drug allergies, medication adherence, and potential drug interactions. ![]() Antiretrovirals have both class-associated and drug-specific adverse effects (see Table 87–5). If the patient experiences any of the serious, life-threatening effects (Table 87–6), the offending agent should be discontinued promptly, and in most cases the patient cannot be rechallenged. Potential long-term complications that may reduce the quality of life are listed in Table 87–7. For drugs with a high likelihood of intolerability (such as nelfinavir-associated diarrhea), patients should be counseled to anticipate these effects and have concomitant prescriptions available for preemptive management (such as an antidiarrheal agent). Patients should have follow-up within the 1st week after initiating a new drug regimen. If the patient does not tolerate a medication despite all efforts to the contrary, consider changing the drug.
Antiretrovirals have both class-associated and drug-specific adverse effects (see Table 87–5). If the patient experiences any of the serious, life-threatening effects (Table 87–6), the offending agent should be discontinued promptly, and in most cases the patient cannot be rechallenged. Potential long-term complications that may reduce the quality of life are listed in Table 87–7. For drugs with a high likelihood of intolerability (such as nelfinavir-associated diarrhea), patients should be counseled to anticipate these effects and have concomitant prescriptions available for preemptive management (such as an antidiarrheal agent). Patients should have follow-up within the 1st week after initiating a new drug regimen. If the patient does not tolerate a medication despite all efforts to the contrary, consider changing the drug.
![]() Currently, treatment of HIV infection is lifelong. Unplanned short-term treatment interruptions may be necessary due to drug toxicity or illness that precludes administration of oral therapy. If a patient must interrupt therapy due to toxicity, all drugs of the regimen should be stopped at the same time, regardless of half-life. The strategy of scheduling elective treatment interruptions (where patients stop and start antiretroviral therapy based on CD4 T-cell count criteria) has been evaluated in several clinical trials. Viral rebound occurs quickly after stopping therapy, and worsens immune function, clinical progression, and may even result in death. If either short-term (less than 7 days) or long-term treatment interruption is needed, drug half-life must be taken into consideration. For regimens where all components have similar half-lives, all drugs can be stopped simultaneously. If the regimen contains components with significantly different half-lives (e.g., Atripla), stopping all drugs at the same time could result in the drug with the longest half-life (usually NNRTIs) lingering in the body and functioning as monotherapy. The ideal time to stop the NNRTIs (efavirenz, etravirine, or nevirapine) is unknown as these drugs can continue to be detectable 1 to 3+ weeks in patients. Options include either: (a) stopping the NNRTI first and continuing the other drugs in the regimen for up to 4 weeks or (b) substituting the NNRTI with a PI and continuing the PI with dual NRTIs for up to 4 weeks. In this situation, therapeutic drug monitoring of the long half-life drug can be useful in determining when to stop the NRTI ± PI coverage.
Currently, treatment of HIV infection is lifelong. Unplanned short-term treatment interruptions may be necessary due to drug toxicity or illness that precludes administration of oral therapy. If a patient must interrupt therapy due to toxicity, all drugs of the regimen should be stopped at the same time, regardless of half-life. The strategy of scheduling elective treatment interruptions (where patients stop and start antiretroviral therapy based on CD4 T-cell count criteria) has been evaluated in several clinical trials. Viral rebound occurs quickly after stopping therapy, and worsens immune function, clinical progression, and may even result in death. If either short-term (less than 7 days) or long-term treatment interruption is needed, drug half-life must be taken into consideration. For regimens where all components have similar half-lives, all drugs can be stopped simultaneously. If the regimen contains components with significantly different half-lives (e.g., Atripla), stopping all drugs at the same time could result in the drug with the longest half-life (usually NNRTIs) lingering in the body and functioning as monotherapy. The ideal time to stop the NNRTIs (efavirenz, etravirine, or nevirapine) is unknown as these drugs can continue to be detectable 1 to 3+ weeks in patients. Options include either: (a) stopping the NNRTI first and continuing the other drugs in the regimen for up to 4 weeks or (b) substituting the NNRTI with a PI and continuing the PI with dual NRTIs for up to 4 weeks. In this situation, therapeutic drug monitoring of the long half-life drug can be useful in determining when to stop the NRTI ± PI coverage.
Patient Encounter, Part 3
Based on the information presented, create a care plan for this patient’s HIV/AIDS. Your plan should include:
(a) A statement of the best drug combinations and reasons supporting each drug recommended, as well as any adverse effects or potential drug-related problems.
(b) Goals of therapy.
(c) A patient-specific, detailed therapeutic plan.
(d) A plan for follow-up to determine whether the goals have been achieved and adverse effects avoided.
Patient Encounter, Part 4
Your patient begins treatment with atazanavir 300 mg by mouth once daily, ritonavir 100 mg by mouth once daily, tenofovir 300 mg by mouth once daily, and lamivudine 300 mg by mouth once daily. He initially does well on this regimen, but after about 3 months, he has difficulty taking his medications at the same time every day due to his busy schedule. Because of his job, he relocated to Alabama, and has not been seen in your clinic for 2 years. He returns today to see you in clinic and complains of feeling tired, but otherwise no specific complaints.
What laboratory tests do you recommend?
What additional information do you need to know before creating a treatment plan for this patient?
Table 87–6 Serious Adverse Effects and Management




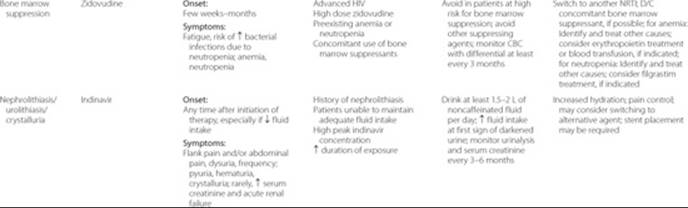
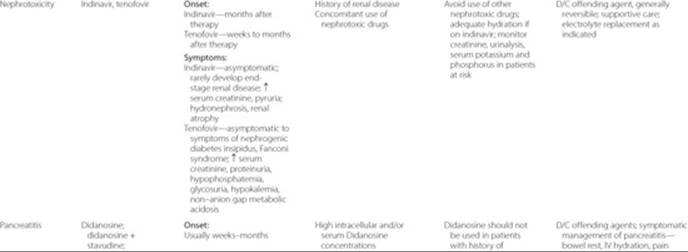

Table 87–7 Other Adverse Effects and Management



Patient Care and Monitoring
Patient Assessment
1. Medication history
• Get a thorough history of prescription, nonprescription, and natural drug product use. Determine what prior antiretroviral regimens, if any, were used in the past.
• Is the patient taking the appropriate dose of each medication? Are the doses adjusted for renal or hepatic failure? Are the doses adjusted for drug interactions with concomitant medications?
• Evaluate the patient for the presence of adverse drug reactions, drug allergies, and drug interactions.
• Assess improvement in quality-of-life measures such as physical, psychological, and social functioning and well-being. Is the patient experiencing any drug-induced adverse effects? What can you do to help manage these adverse effects?
2. Review any available diagnostic data to determine status of his HIV/AIDS.
3. Determine if initiation of antiretroviral therapy is indicated. Evaluate the patient’s ability to adhere to medications, daily routine, social support, and financial stability. Is the patient taking any medications that may interfere with the individual components of potential regimens? Does the patient have medical insurance and prescription coverage?
4. Develop a plan to assess the effectiveness and tolerability of antiretroviral therapy.
Patient Education
1. Educate the patient on HIV/AIDS disease and the importance of strict adherence to medication (the patient must ideally take his or her medications at the same time every day). Recommend a therapeutic regimen that is as easy as possible for the patient to take. Talk to the patient specifically about when and how the patient will be taking the medications. What time do they eat meals? When do they wake up? What other medications are they taking at the same time? Educate patients whether to take their medications with or without food.
2. Educate the patient on common adverse drug effects and a few of the key signs and symptoms of severe toxicity (i.e., jaundice and abacavir hypersensitivity reaction). Tell them to call their provider immediately if any of those symptoms occur. Make sure they have the correct telephone number for the clinic.
Abbreviations Introduced in This Chapter
 Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Gallo RC, Salahuddin SZ, Popovic M, et al. Frequent detection and isolation of cytopathic retroviruses (HTLV-III) from patients with AIDS and at risk for AIDS. Science 1984;224:500–503.
2. Centers for Disease Control and Prevention. HIV prevalence, unrecognized infection, and HIV testing among men who have sex with men—Five U.S. cities, June 2004-April 2005. MMWR 2005;54: 597–601.
3. Wawer MJ, Gray RH, Sewankambo NK, et al. Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J Infect Dis 2005;191:1403–1409.
4. Branson BM, Handsfield HH, Lampe MA, et al. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR Recomm Rep 2006;55:1–17.
5. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1 Infected Adults and Adolescents. Department of Health and Human Services. November 3, 2008:1–139. http://www.aidsinfo.nih.gov/ContentFiles/AdultandAdolescentGL.pdf.
6. Nerad J, Romeyn M, Silverman E, et al. General nutrition management in patients infected with human immunodeficiency virus. Clin Infect Dis 2003;36:S52–S62.
7. Hammer SM, Eron JJ, Reiss P, et al. Antiretroviral treatment of adult HIV infection: 2008 recommendations of the International AIDS Society-USA panel. JAMA 2008;300(5):555–570.
8. Baxter JD, Mayers DL, Wentworth DN, et al., for the CPCRA 046 Study Team for the Terry Beirn Community Programs for Clinical Research on AIDS. A randomized study of antiretroviral management based on plasma genotypic antiretroviral resistance testing in patients failing therapy. AIDS 2000;14:F83–F93.
9. Cohen CJ, Hunt S, Sension M, et al. A randomized trial assessing the impact of phenotypic resistance testing on a ntiretroviral therapy. AIDS 2002;16:579–588.
10. Durant J, Clevenbergh P, Halfon P, et al. Drug-resistance genotyping in HIV-1 therapy: The VIRADAPT randomised controlled trial. Lancet 1999;353:2195–2199.
11. Cingolani A, Antinori A, Rizzo MG, et al. Usefulness of monitoring HIV drug resistance and adherence in individuals failing highly active antiretroviral therapy: A randomized study (ARGENTA). AIDS 2002;16:369–379.
12. Meynard JL, Vray M, Morand-Joubert L, et al. Phenotypic or genotypic resistance testing for choosing antiretroviral therapy after treatment failure: A randomized trial. AIDS 2002;16:727–736.
13. Vray M, Meynard JL, Dalban C, et al. Predictors of the virological response to a change in the antiretroviral treatment regimen in HIV-1-infected patients enrolled in a randomized trial comparing genotyping, phenotyping and standard of care (Narval trial, ANRS 088). Antivir Ther 2003;8:427–434.
14. Wegner SA, Wallace MR, Aronson NE, et al. Long-term efficacy of routine access to antiretroviral-resistance testing in HIV type 1-infected patients: Results of the clinical efficacy of resistance testing trial. Clin Infect Dis 2004;38:723–730.
15. Tural C, Ruiz L, Holtzer C, et al. Clinical utility of HIV-1 genotyping and expert advice: The Havana trial. AIDS 2002;16:209–218.
16. Gallego O, Martin-Carbonero L, Aguero J, et al. Correlation between rules-based interpretation and virtual phenotype interpretation of HIV-1 genotypes for predicting drug resistance in HIV-infected individuals. J Virol Methods 2004;121:115–118.
17. Ravela J, Betts BJ, Brun-Vezinet F, et al. HIV-1 protease and reverse transcriptase mutation patterns responsible for discordances between genotypic drug resistance interpretation algorithms. J Acquir Immune Defic Syndr 2003;33:8–14.
18. Wensing A, Keulen W, Buimer M, et al. Analysis of the world-wide evaluation study on HIV-1 genotype interpretation: ENVA-3. Antivir Ther 2001;6:101.
19. Gulick RM, Hu XJ, Fiscus SA, et al. Randomized study of saquinavir with ritonavir or nelfinavir together with delavirdine, adefovir, or both in human immunodeficiency virus-infected adults with virologic failure on indinavir: AIDS Clinical Trials Group Study 359. J Infect Dis 2000;182:1375–1384.
20. Hammer SM, Vaida F, Bennett KK, et al. Dual vs single protease inhibitor therapy following antiretroviral treatment failure: A randomized trial. JAMA 2002;288:169–180.
21. Hoen B, Dumon B, Harzic M, et al. Highly active antiretroviral treatment initiated early in the course of symptomatic primary HIV-1 infection: Results of the ANRS 053 trial. J Infect Dis 1999;180:1342–1346.
22. Lafeuillade A, Poggi C, Tamalet C, et al. Effects of a combination of zidovudine, didanosine, and lamivudine on primary human immunodeficiency virus type 1 infection. J Infect Dis 1997;175:1051–1055.
23. Lillo FB, Ciuffreda D, Veglia F, et al. Viral load and burden modification following early antiretroviral therapy of primary HIV-1 infection. AIDS 1999;13:791–796.
24. Malhotra U, Berrey MM, Huang Y, et al. Effect of combination antiretroviral therapy on T-cell immunity in acute human immunodeficiency virus type 1 infection. J Infect Dis 2000;181:121–131.
25. Smith DE, Walker BD, Cooper DA, et al. Is antiretroviral treatment of primary HIV infection clinically justified on the basis of current evidence? AIDS 2004;18:709–718.
26. Working Group on Antiretroviral Therapy and Medical Management of HIV-Infected Children. Guidelines for the Use of Antiretroviral Agents in Pediatric HIV Infection. July 29, 2008. http://aidsinfo.nih.gov/contentfiles/PediatricGuidelines.pdf
27. Institute NYSDoHA. Drug-Drug Interactions Between HAART, Medications Used in Substance Use Treatment, and Recreational Drugs, 2008. http://www.hivguidelines.org.
28. Public Health Service Task Force. Recommendations for Use of Antiretroviral Drugs in Pregnant HIV-Infected Women for Maternal Health and Interventions to Reduce Perinatal HIV-1 Transmission in the United States. July 8, 2008. http://aidsinfo.nih.gov/contentfiles/PerinatalGL.pdf.
29. Cohn SE, Watts D, Lertora J, Park JG, Yu S. Depo-medroxyprogesterone in women on antiretroviral thearpy: Effective contraception and lack of clinically significant interactions. Clin Pharmacol Ther 2007;81:222–227.
30. Koziel MJ and Peters MG. Viral hepatitis in HIV infection. N Engl J Med 2007;356:1445–1454.
31. Tien PC. Management and treatment of hepatitis C virus infection in HIV-infected adults: Recommendations from the Veterans Affairs Hepatitis C Resource Center Program and National Hepatitis C Program Office. Am J Gastroenterol 2005;100:2338–2354.
32. Rockstroh JK, Bhagani S, Benhamou Y, et al. European AIDS Clinical Society (EACS) guidelines for the clinical management and treatment of chronic hepatitis B and C coinfection in HIV-infected adults. HIV Med 2008;9:82–88.