António Avelino1 , Francisco Cruz2 , Jalesh N. Panicker3 and Martina D. Liechti4
(1)
Department of Experimental Biology, Faculty of Medicine of the University of Porto, I3S and IBMC of University of Porto, Porto, Portugal
(2)
Department of Renal, Urologic and Infectious Disease, Faculty of Medicine of the University of Porto, I3S and IBMC of University of Porto, Porto, Portugal
(3)
Department of Uro-neurology, The National Hospital for Neurology and Neurosurgery and UCL Institute of Neurology, London, UK
(4)
UCL Institute of Neurology, London, UK
António Avelino
Email: aavelino@med.up.pt
Francisco Cruz (Corresponding author)
Email: cruzfjmr@med.up.pt
Jalesh N. Panicker (Corresponding author)
Email: j.panicker@ucl.ac.uk
The present review highlights the morphology and neurophysiology, as well as the pharmacology, of the lower urinary tract (LUT).
The central element of the LUT is a reservoir, the urinary bladder, which stores and periodically eliminates urine. To perform this function, an efficient coordination is needed between storage and voiding mechanisms. During the storage phase, the bladder wall must accommodate a relatively high volume of urine at low pressure and the bladder outlet must remain closed by the urethral sphincter. Naturally, this coordination is carried out by a neural control system present in the brain, the spinal cord, and the peripheral ganglia.
LUT: Anatomy
The bladder wall is formed by a mucosal and a muscular layer, surrounded by a serosa. The mucosa is lined by a stratified epithelium, the urothelium, comprising three layers: a basal cell layer, an intermediate layer, and an apical layer of umbrella cells. The lamina propria contains several cell types including fibroblasts, mast cells, sensory nerve endings, and myofibroblasts (also referred to as interstitial cells; see below). It is also rich in afferent and efferent nerve endings, vascular and lymphatic vessels, collagen and elastic fibers, and more or less well-defined smooth muscle fascicles [1].
The detrusor muscle is composed of three layers of smooth muscle. They are typical smooth muscle cells, without particular features that distinguish them from those found in other viscera. The cells of the outer and inner layers tend to be oriented longitudinally and those of the middle layer circularly. In the human detrusor, bundles of muscle cells of varying size are surrounded by connective tissue rich in collagen. Those muscular bundles vary extensively in size. In the human detrusor they are large, often a few millimeters in diameter, and composed of several smaller sub-bundles.
The Urothelium
The major (and classical) function of the urothelium is to act as a barrier to block the passage of substances excreted in the urine into the bladder wall. The urothelium is a transitional-type epithelium, containing three layers of cells: a basal cell layer, an intermediate layer, and a superficial layer containing hexagonal cells (umbrella cells). The latter have particular characteristics that concur to the barrier function of the urothelium. Among them, they have specialized membrane lipids, uroplakin-rich asymmetric unit membranes, and a plasmalemma with stiff plaques. Uroplakins are transmembrane proteins that further reduce the permeability of the urothelium to small molecules like water, urea, and protons and can be used as a differentiation marker of umbrella cells. To reduce intercellular movement of molecules, occludin- and claudin-rich tight junctions connect the lateral membrane of superficial cells. The luminal membrane of umbrella cells is covered by a glycosaminoglycan (GAG) layer that further reinforces the barrier function and seems to have antibacterial and anti-adherence properties. The origin of the GAG layer is still not clear, with at least part of it being synthetized by the kidneys to increase surface area. Fusiform or discoidal-shaped cytoplasmic vesicles docked in the apical cytoplasm fuse with the apical membrane, thereby increasing the luminal area. The presence of SNARE proteins essential for exocytosis was recently reported in urothelial cells and one of its isoforms (SNAP23) is extremely abundant in umbrella cells (Fig. 1.1).

Fig. 1.1
Guinea pig urothelium stained for SNAP23 (green) and nuclei (Blue). Umbrella cells are strongly labeled. Lamina propria can be seen in faint green (background) (Avelino et al. 2015)
While the urothelium has been historically viewed primarily as a barrier, it is becoming increasingly appreciated as a responsive structure capable of detecting physiological and chemical stimuli and able to release a significant number of signaling molecules Thus, urothelial cells display a number of properties similar to sensory neurons and can use several signal-transduction mechanisms that perform an intricate cross talk with primary afferents located in the sub-urothelium. Urothelial cells express receptors for bradykinin, neurotrophins, as well as purinergic, cholinergic, adrenergic, and protease-activated receptors. Amiloride/mechanosensitive Na + channels and several transient receptor potential channels are also found in the urothelium (Table 1.1). In addition, urothelial cells can release neurotransmitters and signaling molecules such as nitric oxide, ATP, acetylcholine, prostaglandins, substance P, and neurotrophins (Table 1.1) that influence the excitability of suburothelial afferent nerves. Mediators released by urothelial cells may eventually act indirectly on suburothelial myofibroblasts (also referred to as “interstitial cells”) that lie in close proximity to afferent nerves. Thus, it is believed that urothelial cells and myofibroblasts can participate in sensory mechanisms in the urinary tract in an autocrine/paracrine way.
Table 1.1
Receptors and signaling molecules present in the urinary bladder
|
Receptor/channel/signaling molecule |
Location |
Neurotransmitter/agonist/stimulus |
|
Receptors |
||
|
Cholinergic muscarinic (M2, M3) |
D, U, S |
Acetylcholine, muscarine |
|
Cholinergic nicotinic |
U, S |
Acetylcholine, nicotine |
|
Adrenergic alpha |
U, D, S |
Norepinephrine; epinephrine |
|
Adrenergic beta |
U, D, S |
Norepinephrine; epinephrine |
|
Degenerin/ENaC |
U, S |
Amiloride, mechanical |
|
Neurotrophins (trkA; p75; trkB) |
U, D, S |
NGF, BDNF |
|
Bradykinin receptor [1, 3] |
U, D, S |
Bradykinin |
|
Tachykinin receptor [1–3] |
U, D, S |
Substance P, neurokinin A |
|
Estrogen receptor |
U, D, S |
Estradiol |
|
Proteinase-activated receptor |
U, D, S |
Thrombin; trypsin |
|
TRPV1 |
U, S |
Heat (43 °C), low pH, anandamide, vanilloids |
|
TRPV2 |
U, S |
Noxious heat (53 °C) |
|
TRPV4 |
U, S |
Cell stretch, moderate heat (24 °C), 4 alpha-PDD |
|
TRPM8 |
U, S |
Cold (8–28 °C), menthol, icilin |
|
TRPA1 |
U, S |
Cinnamaldehyde, mechanical |
|
P2X |
U, D, S |
ATP |
|
P2Y |
U, D |
ATP; UTP; ADP |
|
P1 |
U, D |
Adenosin |
|
Cannabinnoid [1, 3] |
U, D, S |
Anandamide; cannabinoids |
|
VEGF receptor |
U, D, S |
Vascular endothelial growth factor |
Note: the order of the initials in the table indicates relative (decreasing) expression
U urothelium, D detrusor muscle, S sensory afferents
Examples of this phenomenon are well documented: distension of the bladder causes ATP release from urothelial cells that consequently stimulates P2 × 2/P2 × 3 receptors in suburothelial sensory; stimulation of urothelial cells with the vanilloid receptor agonists capsaicin and RTX increases their intracellular calcium and evokes transmitter (nitric oxide and ATP) release. It is noteworthy to mention that in aged mice, increased levels of ATP are released in the bladder lumen, resulting in increased primary afferent activity, suggesting that aging results in aberrant urothelial function.
Interstitial Cells
Interstitial cells of Cajal, ICC-like cells, or myofibroblasts found in the urinary bladder resemble the interstitial cells of Cajal (ICC) of the gastrointestinal tract. After some debate, there is some consensus that these cells should be referred as interstitial cells of Cajal (ICC) or just as interstitial cells (IC).
Based on their location within the bladder wall, several subgroups of IC have been identified. ICC of the lamina propria are stellate-shaped cells that have been found to be extensively linked by gap junctions. Detrusor IC are elongated non-networked cells arranged in circular, longitudinal, and oblique orientation, on the boundary of smooth muscle bundles; see Fig. 1.2. They make close structural connections with nerves, but the origin of the nerve fibers (afferent or efferent?) was never determined.
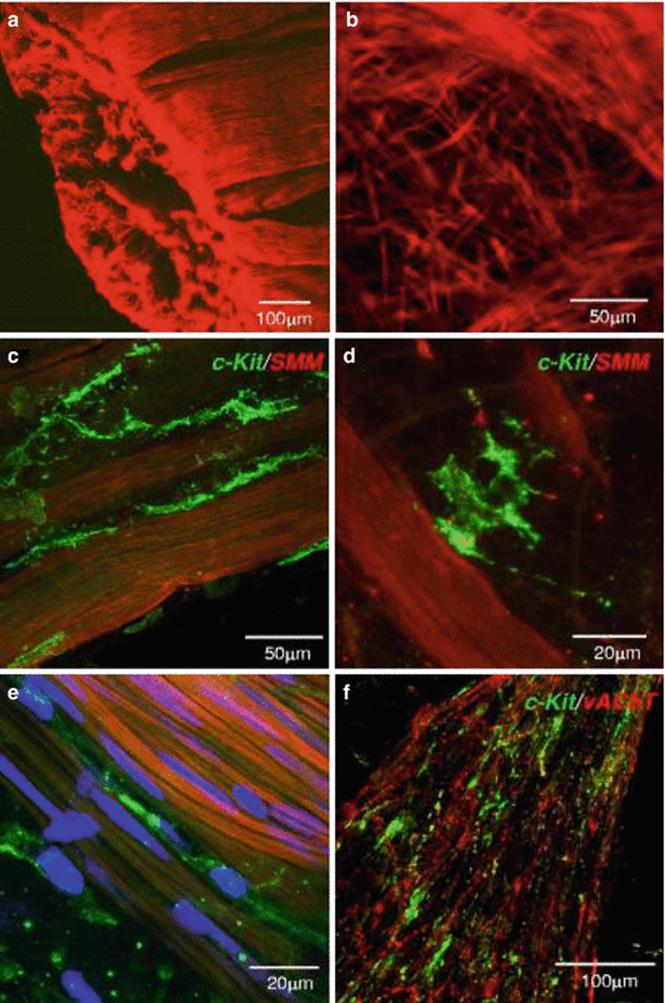
Fig. 1.2
Human detrusor ICC. (a) human detrusor labeled with anti-SM myosin (SMM). (b) At mucosa-detrusor interface, SMC were arranged in loose network, which was organized into distinct bundles in detrusor muscularis. (c–e) Representative human detrusor colabeled with anti-SM myosin and anti-KIT (green areas). Elongated, branched KIT-positive ICC track SM bundles and occupy spaces between bundles with obvious spiky morphology. (e) Nuclei counterstained with 4,6-diamidino-2-phenylindole (blue areas). (f) Detrusor ICC (green areas) associated with vAChT nerves (red areas) (From Johnston et al. J Urol. 2010;184:370–7)
Ultrastructurally, IC of the lamina propria present the characteristics of the classical gut myofibroblasts. These include bundles of fine cytoplasmic filaments, dense bodies, linear arrays of subsurface vacuoles, and the presence of an interrupted basal lamina. In the detrusor, electron microscopy studies showed that interstitial cells have flattened, dendritic-like processes, forming a branching network around individual muscle fascicles. In the cell soma and cytoplasmic processes, mitochondria, rough endoplasmic reticulum, and Golgi apparatus were observed, as well as intermediate filaments. Gap junctions and close appositions were described, but not specialized contacts with smooth muscle.
IC can be detected by immunostaining for the intermediate filament, vimentin. A subpopulation of these cells, in addition, also expresses the tyrosine kinase receptor, KIT. Some data is available regarding the functional role of IC in overactive bladders, but the emerging picture is still not clear. Some reports showed that overactive human bladder tissues had upregulated numbers of IC and enhanced contractility, which was more sensitive to a tyrosine kinase inhibitor. On the other hand, C-kit immunoreactivity was similar in patients with neurogenic bladders and controls and remained unchanged after treatment.
IC form an heterogeneous population, with some cells responding to cholinergic stimulation (detrusor IC) whereas others (lamina propria IC) do not. In contrast, they respond to application of ATP with calcium inflow. In this way, they appear to be ideally placed to respond to ATP released from urothelial cells, providing a putative transduction signaling system between the suburothelial sensory fibers and the detrusor muscle. In addition, they may eventually contribute to myogenic activity (nerve-dependent or spontaneous) due to their syncytial-like network.
Detrusor Muscle
The smooth muscle cells of the detrusor muscle are not clearly arranged in distinct layers, but run in all directions, forming groups of small functional units, or fascicles. Here, individual cells are evenly separated by an approximately 200 nm thick space containing collagen fibrils attached to a basal lamina. Specialized junctions, like zonulae adherens, and areas of sarcolemma apposition can be found in contiguous cells. Connexin-rich gap junctions were described in normal detrusor human bladder biopsies. Experimental studies suggest that gap junctions may have an important role in detrusor function. In animals with bladder outlet obstruction (BOO) leading to hypocontractility of the detrusor, connexin expression and the number of gap junctions were significantly reduced.
Innervation of the detrusor can be seen coursing the interstitium as Schwann cell-ensheathed axon bundles. Many axons form varicosities closely apposed to the muscle cells, forming neuroeffector junctions. Ultrastructural studies have shown that many of the varicosities are probably autonomic motor efferents, while others are undoubtedly primary afferents (Fig. 1.3).

Fig. 1.3
(a) Normal detrusor. Normal detrusor myocytes are spindle-shaped mononuclear cells evenly aligned with minimal interstitial collagen and elastin in fascicles. (b) Ultrastructural features that correlated with each other and with poor postoperative voiding outcome were loss of fascicle arrangement, variable cell size, collagen in fascicles, and abnormal cell shape (From Blatt et al. J Urol. 2012;188:2294–9)
Several bladder dysfunctions show ultrastructural changes in the detrusor. These are particularly evident in muscle cells and in the interstitium (Fig. 1.4). In overactive human bladders, a well-described disjunction pattern was recognized, consisting of widened spaces between cells, reduction of intermediate cell junctions, and increase of protrusion junctions and ultraclose cell abutments. In a recent study, the detrusor muscle of patients suffering from bladder underactivity subsequent to bladder outlet obstruction also showed morphological abnormalities. These consisted of variations in muscle cell size and shape, abnormal fascicle arrangement, and collagenosis, loosely corresponding to the myohypertrophy pattern originally described by.

Fig. 1.4
Ultrastructural visualization of VR1 (presently know as TRPV1) immunoreactivity in nerve profiles found in the muscular layer. Immunoreactive varicosities lie in shallow grooves in the surface of smooth muscle cells. Small clear and large dense-core vesicles (arrows) can be seen (Adapted from Avelino et al. Neuroscience. 2002;109:787–98)
The detrusor muscle contains receptors for numerous transmitters/modulators, released from nerves or generated locally. In humans the smooth muscle contraction is almost exclusively cholinergic. The predominant receptors for contraction are the muscarinic M2 and M3 receptors. In the normal human detrusor, the M2 muscarinic receptor is more abundant, although the M3 receptor is more relevant for detrusor contraction.
Muscarinic receptors are coupled to G proteins but with varying signal-transduction pathways. M3 receptors couple to Gq/11, activating phosphoinositide hydrolysis, leading to mobilization of intracellular calcium stores and causing a direct smooth muscle contraction. M2 receptors couple to pertussis toxin-sensitive Gi/o, leading to inhibition of adenylyl cyclase activity. The most important receptor for relaxation is the β3-adrenoreceptor. It is also coupled to a Gs protein, and its prototypical signaling pathway is activation of adenylyl cyclase to elevate intracellular cAMP concentrations. Sequentially, cAMP activates protein kinase A (PKA) decreasing intracellular calcium by stimulation of the sarcoplasmic reticulum calcium pump ATPase. Thus, in theory M2 receptor activation oppose to sympathetically mediated smooth muscle relaxation, mediated by β3-adrenoreceptor agonists. This forms the rationale for the therapeutical use of agents that block M3 receptors or drugs that activate the β3-adrenoreceptor.
The other mechanisms that influence detrusor contraction are collectively known as non-adrenergic, non-cholinergic mechanisms. They involve mainly purinergic neurotransmission, but also nitric oxide release, neuropeptides, and prostaglandins.
ATP acts on two families of purinergic receptors: an ion channel family (P2X) and a G protein-coupled receptor family (P2Y). Seven P2X subtypes and eight P2Y subtypes have been identified. The detrusor response to ATP seems to be mediated by activation of a ligand-gated cation channel (P2X receptor) that promotes the influx of extracellular Ca, whereas uridine triphosphate and adenosine act through G protein-coupled receptors (P2Y2 or P2Y4) to induce smooth muscle contractions via a phospholipase C/IP3 signaling pathway.
Although purinergic stimulation is considered to be the only minor contributor to normal bladder function in healthy humans, some studies show that this may not be true in abnormal conditions. The aging bladder seems to have a higher contribution of purinergic transmission. A significant positive correlation between age and purinergic neurotransmission, accompanied by a reduction of cholinergic neurotransmission, was detected in bladder strips of aging human bladders. Additionally, upregulation of purinergic activity through P2X receptors has been demonstrated in the bladder following chronic bladder outlet obstruction.
Nitric oxide (NO) has a strong role on the inhibitory non-adrenergic, non-cholinergic mechanisms. NO is generated by a reaction catalyzed by the enzyme NO synthase (NOS), of which three isoforms are currently known: neuronal NOS (nNOS), epithelial NOS (eNOS), and inducible/inflammatory NOS (iNOS). In a very convenient way, the three isoforms can be detected using NADPH histo- or immunohistochemistry. Using this approach, NO synthesis was identified throughout the bladder, in the detrusor muscle blood vessels, urothelium, nerves, and pelvic ganglia.
The most prominent effect of NO is its action on soluble guanylate cyclase, producing guanosine 3′, 5′- cyclic monophosphate (cGMP) and mediating relaxation of muscle. Accordingly, there is strong evidence that NO contributes to bladder reflex activity, since, e.g., the intravesical administration of the NO scavenger oxyhemoglobin induces bladder overactivity in experimental animals.
The effect of NO in bladder contraction opens a window of opportunity for the use of phosphodiesterase inhibitors, targeting a nitric oxide-cyclic guanosine monophosphate pathway, in the treatment of lower urinary tract symptoms.
Neuropeptides are abundant in the urinary bladder, but their functional role is far from being fully understood.
Tachykinins like substance P (SP), neurokinin A (NKA), and neurokinin B (NKB) are abundant in primary afferent nerves of the human bladder. Despite their afferent functions, peripheral release of these peptides seems to have an important role on detrusor function by inducing “neurogenic inflammation.” Tachykinins bind to G-coupled receptors, and in the human bladder, NKA is the most potent tachykinin, through binding to the NK2 type.
Although detrusor smooth muscle contraction is normally triggered by the release of neurotransmitters, a certain degree of spontaneous activity is also present. Spontaneous contractile and electrical activities can be detected in isolated cells, bladder strips, or whole bladders. Spontaneous activity can affect the muscle from simple changes of tone to contractions involving several muscle bundles. Interestingly, spontaneous activity is increased in overactive bladders of experimental animals and patients. Recent reports showed that treatment of rat bladders with botulinum toxin, a neurotoxin that disrupts nerve vesicle exocytosis, abolished afferent and efferent nerve discharge without affecting spontaneous muscular activity.
Detrusor smooth muscle cells are electrically connected via gap junctions composed of the connexin subtype C × 45. This fact may contribute to increased spontaneous activity, since gap junction blockers inhibit spontaneous contractions in animal models of overactive bladders. Furthermore, an increase of C × 45 was reported in the detrusor muscle of patients with neurogenic detrusor overactivity and of patients with lower urinary tract symptoms (LUTS).
Innervation
Three sets of nerves (sacral parasympathetic, thoracolumbar sympathetic, and somatic nerves) innervate the bladder [2]. The nerves consist of efferent and afferent axons originating at thoracolumbar and sacral spinal levels. Parasympathetic efferent nerves contract the bladder while sympathetic efferent nerves relax the bladder and contract the urethra. Somatic efferent nerves contract the EUS. Sympathetic postganglionic efferent pathways in the pelvic and hypogastric nerves elicit similar effects in the bladder, for example, firstly, inhibition of the detrusor smooth muscle via release of noradrenaline from nerve terminals causing activation of ß-adrenoceptors and, secondly, excitation of the urethra via noradrenaline release and activation of urethral α1-adrenoceptors
Parasympathetic preganglionic neurons are located in the intermediolateral gray matter (laminae V–VII) in the sacral segments of the spinal cord, whereas sympathetic preganglionic neurons are located in medial (lamina X) and lateral (laminae V–VII) areas in the rostral lumbar spinal cord. Intramural parasympathetic ganglion cells are present in the bladder wall of humans and some other species (curiously, are not present in the rat). The majority of the ganglia are small sized, containing one to six neurons, and typically stain for choline acetyltransferase (ChAT).
Afferent fibers project into Lissauer’s tract in the lumbosacral spinal cord and extend through lamina I laterally and medially around the dorsal horn into deeper laminae (V–VII and X). Unmyelinated peptidergic bladder afferents project to spinal cord lamina I and non-peptidergic afferents project to spinal cord inner lamina II.
Myelinated Aδ-fibers are located primarily in the detrusor muscle layer, respond primarily to detrusor stretching during bladder filling, and convey sensations of fullness. These nerves enter the bladder through the neck and branch extensively, forming a rich plexus in the trigone. Unmyelinated sensory C-fibers are more widespread and reside in the muscle, close to the urothelium in the mucosa and directly adjacent to the urothelial cells. In some species, including humans, peptidergic afferent axons are also located in close proximity to intramural parasympathetic ganglion cells, where they participate in local reflex networks within the bladder wall.
Bladder afferent neurons display immunoreactivity for several neuropeptides, such as SP, CGRP, PACAP, leucine-enkephalin, galanin, and corticotrophin-releasing factor as well as nitric oxide synthase, glutamic acid, and aspartic acid. Co-expression of neuropeptides is highly prevalent, namely, of CGRP, SP, and PACAP. Several transient receptor potential channels, namely, the vanilloid receptor TRPV, are also highly expressed in primary afferent fibers [3].
The micturition reflex starts when stretch receptors in the bladder wall project sensory information regarding the degree of bladder distension via pelvic afferent nerve fibers to the spinal cord and convey this information to the pontine micturition center (PMC) and periaqueductal gray (PAG). During the initial part of the filling phase, afferent nerve firing is low, inhibiting the sacral parasympathetic nerves and exciting lumbar sympathetic preganglionic neurons. Stimulation of sympathetic neurons initiates the release of noradrenaline which acts on ß-adrenoceptors in the bladder detrusor muscle to cause relaxation of the muscle and acts on α-adrenoceptors on the urethral sphincter causing contraction of the sphincter muscle, promoting continence.
When the volume of the bladder nears the micturition threshold, afferent nerve signals produce nerve firing in the sacral parasympathetic pathways and inhibit the sympathetic and somatic pathways. Voiding of urine consists of two phases, firstly parasympathetically mediated relaxation of the urethral sphincter by nitric oxide as well as cessation of excitatory adrenergic and somatic inputs to the urethra and secondly the release of ACh which acts on muscarinic receptors on detrusor muscle by the parasympathetic nerves of the bladder, eliciting bladder contraction and urine flow.
Summary and Conclusions
This review pretends to stress the complexity of the mechanisms involved in an apparently simple voiding-filling cycle.
From its inner lining, the urothelium, to the outermost layer, the detrusor, a delicate balance of various inhibitory and excitatory pathways must exist. The molecular cross talk between several structures is being further dissected every day, expanding knowledge and improving therapeutics.
Pelvic Neurophysiology
Jalesh N. Panicker5 and Martina D. Liechti6
(5)
The National Hospital for Neurology and Neurosurgery and UCL Institute of Neurology, London, UK
(6)
UCL Institute of Neurology, London, UK
Jalesh N. Panicker
Email: j.panicker@ucl.ac.uk
Introduction
Neurophysiology tests involve the recording of bioelectrical activity from muscles and the nervous system to investigate nerve functions. It is practical to distinguish recordings from muscle (electromyography, EMG) and tests of nerve and nervous pathway function (conduction studies). Although less widely available in functional urology, some neurophysiology tests may be useful in the assessment of patients with pelvic organ complaints.
Electromyography
Electromyography (EMG) is a diagnostic procedure to assess the integrity of muscle functions and their innervation. In this way, muscles that are difficult to test clinically can be investigated, for example, those of the pelvic floor. Kinesiological EMG recordings during specific tasks may be useful in specific situations, such as during biofeedback applications to address disturbances of pelvic floor muscles activity which enable patients to be actively involved in the treatment of their symptoms.
Pelvic floor EMG was first introduced as part of urodynamic studies to assess the extent of relaxation of the urethral sphincter during voiding, with the aim of recognizing detrusor-sphincter dyssynergia. For this purpose, either surface or needle electrodes may be used. The best signal is obtained using a concentric needle electrode (CNE), but the discomfort from the needle itself is likely to influence relaxation of the pelvic floor. Surface recording electrodes often provide sufficient information; however they also record EMG signals from other pelvic floor muscles. EMG allows an ongoing assessment of muscle activity during the entire urodynamics test; however the test is performed less often nowadays because of increasing availability of video-urodynamic investigations for diagnosing detrusor-sphincter dyssynergia.
However, CNE EMG studies of the external urethral and anal sphincter muscles, performed separately from urodynamics, have been proven useful to assess the integrity of the innervations to the pelvic floor. It provides information on insertion activity, spontaneous activity, interference pattern, and motor unit potentials. The normal sphincter EMG shows continuous “tonic” activity at rest, which may be increased voluntarily or reflexly. The number of recorded motor units recorded depends upon the uptake area of the electrode. Using a CNE, activity from one to five motor units is usually recorded per site in the anal sphincter at rest. Electromyography has been used to demonstrate changes of reinnervation in the urethral or anal sphincter in a few neurological disorders. Well-established values exist for the normal duration and amplitude of motor units recorded from the sphincter muscles.
Sphincter EMG in the Evaluation of Suspected Cauda Equina Lesions
Lesions of the cauda equina are an important cause for pelvic floor dysfunction and patients present with lower urinary tract (LUT) dysfunction and often sexual and bowel dysfunction as well. Most often, EMG of the external anal sphincter demonstrating changes of chronic reinnervation, with a reduced interference pattern with fewer motor units and enlarged polyphasic motor units (>1 mV amplitude), can be found in patients with long-standing cauda equina syndrome. Though EMG may demonstrate pathological spontaneous activity 3 weeks or more after injury, these changes of moderate to severe partial denervation (i.e., fibrillations and positive sharp waves) often become lost in the tonically firing motor units of the sphincter.
Sphincter EMG in the Diagnosis of Multiple System Atrophy
Multiple system atrophy (MSA), a variant of Parkinson’s disease, is a progressive neurodegenerative disease which often, particularly in its early stages, is mistaken for Parkinson’s disease but is poorly responsive to antiparkinsonian treatment. Autonomic failure causing postural hypotension and cerebellar ataxia causing unsteadiness and clumsiness may be additional features. Urinary incontinence in both women and men occurs early in this condition, often appearing some years before the onset of obvious neurological features, and not uncommonly, patients may present to the urologist. Degenerative changes occur both in the brain and spinal cord resulting in LUT dysfunction. Neuropathological studies have shown that the anterior horn cells in Onuf’s nucleus are selectively lost in MSA and this results in changes in the sphincter muscles that can be identified by EMG. The anal sphincter is once again most often studied and changes of chronic reinnervation in MSA tend to result in prolonged duration motor units and these changes can be detected easily (Fig. 1.5).
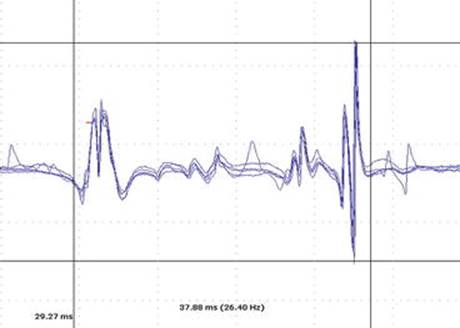
Fig. 1.5
Concentric needle EMG of the external anal sphincter from a 54-year-old gentleman presenting with voiding difficulties and incontinence on the background of a recent onset parkinsonism syndrome. Duration of the recorded motor unit is 37.9 ms, which is prolonged and suggests chronic reinnervation. The mean duration of MUPs during the study was 19 ms (normal <10 ms) and the EMG was compatible with a diagnosis of multiple system atrophy (gain 200 microvolts/division, sweep speed 10 ms/division)
The value of sphincter EMG in the differential diagnosis of parkinsonism has been widely debated over the years. Technically, the MUPs recorded in MSA are quite prolonged and automated MUP analysis results in a tendency to chop up these long polyphasic MUPs into individual components. Thereby the late components might be missed when measuring the motor unit duration and the duration of the MUP may be erroneously noted as being normal. It is advisable therefore to also identify MUPs manually when performing the EMG for a patient with suspected MSA to avoid this pitfall. Moreover, the EMG changes are not specific to MSA, and similar changes of chronic reinnervation may be found in long-standing Parkinson’s disease, other parkinsonian syndromes such as progressive supranuclear palsy, dementia with Lewy bodies, and other neurodegenerative conditions such as spinocerebellar ataxia type 3 and following obstetric injury with sphincter damage and cauda equina damage. Nevertheless, a body of opinion exists that maintains that a highly abnormal result in a patient with mild parkinsonism is of value in establishing a diagnosis of probable MSA. A highly abnormal EMG in the absence of other obvious causes in a patient with suspected MSA in first 5 years is significant [4]. This correlation is important not only for the neurologist but also for the urologist, because inappropriate surgery for a suspected prostate enlargement as the cause for bladder troubles can then be avoided.
Sphincter EMG in the Investigation of Urinary Retention in Young Women
Isolated urinary retention in young women may occur where the neurological examination is normal and investigations such as MRI exclude a neurological cause for voiding dysfunction. A characteristic abnormality, however, can be found on urethral sphincter EMG, consisting of decelerating bursts (Fig. 1.6), a signal somewhat like myotonia, and complex repetitive discharges (Fig. 1.7). It has been proposed that this abnormal spontaneous activity results in impairment of relaxation of the urethral sphincter, which may cause urinary retention in some women and obstructed voiding in others. This condition, nowadays known as Fowler’s syndrome [5], is also characterized by elevated urethral pressures with values regularly in excess of 100 cmH2O.

Fig. 1.6
Decelerating burst recorded during concentric needle EMG of the urethral sphincter in a young woman presenting with urinary retention (gain and sweep speeds provided in respective panels)

Fig. 1.7
Complex repetitive discharges recorded during concentric needle EMG of the urethral sphincter in a young woman presenting with urinary retention (gain and sweep speeds provided in respective panels)
Sacral Reflexes
Mechanical, electrical, or magnetic stimulation in the perineal region can be used to elicit reflex responses from the perineal and pelvic floor muscles. The penilo-cavernosus reflex and anal reflex both involve the sacral (S2,3,4) roots and pudendal nerve in their reflex arc and are often elicited during the clinical examination. However, reflex responses are more accurately assessed and can be quantified when tested using neurophysiology tests.
Similar to the anal reflex, which is elicited by perianal stimulation and recording from the anal sphincter muscle, the penilo-cavernosus reflex, formally known as the “bulbocavernosus” reflex, assesses the sacral root afferent and efferent pathways. For the penilo-cavernosus reflex, the dorsal nerve of penis (or clitoris) is electrically stimulated and recordings are made from the bulbocavernosus muscle or external anal sphincter, usually with a concentric needle. In those patients in whom the reflex is difficult to elicit, double electrical stimuli should be used. A sacral reflex arc lesion should not be inferred by absence of a response if only single pulse is used for stimulation. This test is complementary to the clinically assessed response and the EMG examination of the pelvic floor muscles in patients with suspected peripheral nervous lesions. It may be of value in patients with bladder dysfunction suspected to be secondary to cauda equina damage or damage to the lower motor neuron pathway.
Evoked Potential Studies
The acquisition of somatosensory evoked potentials (SSEP) is a well-established method to assess afferent/sensory nerve fiber function from various parts of the body. Stimulation of a certain nerve or dermatome elicits SSEPs, which can be measured along the spinal cord or over the cortex providing characteristic latency and amplitude measures.
Pudendal SSEP are typically recorded from the scalp following electrical stimulation of the dorsal nerve of penis or clitoral nerve (Fig. 1.8) and are compared to tibial evoked potentials. SSEP may be abnormal when a spinal cord lesion is the cause of sacral sensory loss or neurogenic detrusor over activity; however, such pathology is usually apparent from the clinical examination. Additional recordings over the cauda equina and lumbosacral cord help in localizing spinal lesions. Apart from its main application in intraoperative monitoring, normal pudendal SSEP can help to rule out a neurological cause in certain situations such as erectile dysfunction.

Fig. 1.8
Somatosensory evoked potential (SSEP) recorded at Cz-Fz during electrical stimulation (15 mA) of the dorsal penile nerve in a young man presenting with genital numbness. The P40 waveform was of normal latency
Similarly, though technically more demanding, LUT sensory evoked potentials can be obtained on stimulation of the bladder urothelium or urethra. Currently, these tests are limited to research applications, but considering the potential relevance for neurogenic LUT dysfunction, these tests need to be further developed for future clinical applications.
Motor evoked potentials (MEP) can be used to study efferent/motor pathways, which involve the stimulation of the motor cortex or spinal pathways and the recording from target muscles. While these tests can be useful to localize lesions in the central nervous system, they have a limited role in the investigation of pelvic floor problems.
Nerve Conduction Studies
Due to the limited accessibility in that area of the body, the only test of motor nerve conduction for the pelvic floor is the pudendal nerve terminal motor latency (PNMTL). The pudendal nerve is the most important motor nerve innervating the pelvic floor. The pudendal nerve is stimulated either per rectally or vaginally adjacent to the ischial spine using the St Mark’s electrode, a finger-mounted stimulating device with a surface EMG recording electrode 7 cm proximal located around the base of the finger which records from the external anal sphincter. Prolongation was initially considered evidence for pudendal nerve damage, although a prolonged latency is a poor marker of denervation. This test has not proved contributory in the investigation of patients with suspected pudendal neuralgia and can currently only be interpreted in combination with the results of other tests.
Autonomic Function Tests
The tests mentioned so far are restricted to evaluating the somatic nervous system. Considering the significant proportion of autonomic innervation relevant for the sympathetic and parasympathetic control of sacral organ functions, appropriate function tests would be important. The only test available is the sympathetic skin response (SSR) which can be recorded from palms, soles, and perineal skin. Although the responses are easily habituated and highly dependent on individual as well as environmental factors, the latencies can typically be interpreted which can be used for the evaluation of LUT dysfunction and sexual impotence.
Conclusion and Outlook
Neurophysiological tests are important to better understand and diagnose the neurological cause for pelvic organ complaints, which is also helpful for the choice of treatment. Apart from EMG and reflex tests, which have a clear role in patients with suspected peripheral nervous system lesions, pelvic neurophysiology remains technically demanding and often test only a subset of nerves. However, these tests are useful as a research tool to understand the basis of LUT dysfunction, and it is anticipated that they will gain clinical relevance for diagnosis of LUT dysfunction and intraoperative applications in the future.
Acknowledgments
This work was undertaken at UCLH/UCL Institute of Neurology which received a proportion of funding from the Department of Health’s NIHR Biomedical Research Centers funding scheme. The authors also acknowledge a postdoctoral fellowship to M.D.L. through the Swiss National Science Foundation.
References
1.
Andersson KE, McCloskey KD. Lamina propria: the functional center of the bladder? Neurourol Urodyn. 2014;33:9–16.CrossRefPubMed
2.
de Groat WC, Yoshimura N. Afferent nerve regulation of bladder function in health and disease. Handb Exp Pharmacol. 2009;2009:91–138.CrossRef
3.
Avelino A, Charrua A, Frias B, Cruz C, Boudes M, de Ridder D, Cruz F. Transient receptor potential channels in bladder function. Acta Physiol (Oxf). 2013;207:110–22.CrossRef
4.
Paviour DC, Williams D, Fowler CJ, Quinn NP, Lees AJ. Is sphincter electromyography a helpful investigation in the diagnosis of multiple system atrophy? A retrospective study with pathological diagnosis. Mov Disord. 2005;20(11):1425–30.CrossRefPubMed
5.
Fowler CJ, Christmas TJ, Chapple CR, Parkhouse HF, Kirby RS, Jacobs HS. Abnormal electromyographic activity of the urethral sphincter, voiding dysfunction, and polycystic ovaries: a new syndrome? BMJ. 1988;297(6661):1436–8.PubMedCentralCrossRefPubMed