Antonella Giannantoni1 , Silvia Proietti2, Massimo Porena1, Stephen Mock3 , Melissa Kaufman3, W. Stuart Reynolds3, Roger Dmochowski3 , Scott MacDiarmid4 , Hendrikje M. K. van Breda5 and John Heesakkers5
(1)
Department of Urology, University of Perugia, Perugia, Italy
(2)
Humanitas Clinical and Research Centre, Rozzano, MI, Italy
(3)
Department of Urology, Vanderbilt University Medical Center, Nashville, TN, USA
(4)
Department of Urology, Alliance Urology Specialists, University of North Carolina, Chapel Hill, NC, USA
(5)
Department of Urology, Radboud University Medical Center, Nijmegen, The Netherlands
Antonella Giannantoni
Email: agianton@libero.it
Stephen Mock
Email: stephen.mock@vanderbilt.edu
Roger Dmochowski
Email: roger.dmochowski@vanderbilt.edu
Scott MacDiarmid (Corresponding author)
Email: smacdiarmid@allianceurology.com
Hendrikje M. K. van Breda
Email: jetske.vanbreda@radboudumc.nl
John Heesakkers
Email: John.heesakkers@radboudumc.nl
Introduction
Antonella Giannantoni6 , Silvia Proietti7 and Massimo Porena6
(6)
University of Perugia, Perugia, Italy
(7)
Humanitas Clinical and Research Centre, Rozzano, MI, Italy
Antonella Giannantoni
Email: agianton@libero.it
Overactive bladder is a complex of symptoms defined by International Continence Society as “urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence (UUI), in the absence of urinary tract infection (UTIs) or other obvious pathology.”
Overactive bladder (dry or wet). In studies on prevalence of OAB conducted in both Europe and United States, many OAB patients complained of urgency without reporting urinary incontinence. This is the case of OAB dry, a condition usually more affecting men than women, in whom, conversely, there is a higher prevalence of OAB wet. The higher proportion of OAB wet in females could be explained by the anatomic characteristics of bladder neck, urethral sphincter, and pelvic floor, which make women more subjected to lose urine. OAB dry and wet equally exert a negative impact on patients’ quality of life (QoL).
Patient Presentation and Initial Assessment
To date, there is still insufficient evidence-based literature to address diagnosis of OAB, which remains a mere clinical diagnosis. Thus, it is important for clinicians to provide whether a diagnosis of OAB is appropriate and make the necessary differentiations. When daytime and nighttime urinary frequency and urinary urgency, with or without UUI, in the absence of infection or other pathologic conditions, are reported as bothersome and inconvenient by the patient, he/she may be diagnosed to have OAB.
History Taking and Physical Examination
A careful history and physical examination can be just useful to make a diagnosis of OAB (Fig. 3.1). Type of symptoms and of incontinence (UUI, stress urinary incontinence (SUI) or mixed UI incontinence) should be clearly assessed in the initial clinical evaluation. Furthermore, all predisposing or precipitant factors should be identified in order to require additional investigations and treatments. Current medications should be adequately controlled to ascertain that OAB symptoms are not related to medications. Clinician’s questions should be addressed to investigate type, timing, duration, and severity of urgency and urgency incontinence, by investigating on the behavior of both bladder filling and emptying, and the level of patient’s bother should be assessed.
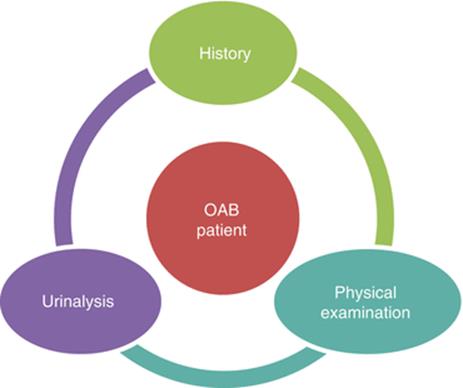
Fig. 3.1
Initial assessment of non-neurogenic patients with OAB: the minimum requirements for this diagnosis is history, physical examination, and urinalysis. American Urological Association (AUA) Guideline. Statement 1
In female patients, physical examination including abdominal, pelvic, and perineal evaluation with digital examination of the vagina should be performed to assess voluntary pelvic floor muscle contraction, estrogen status, and presence of urogenital prolapse (POP). The cough test should be performed in order to objectively assess presence and type of incontinence. In male patients, a rectal/genitourinary evaluation with digital rectal examination should be performed, in order to assess the urethral and prostate status.
Risk factors in men for the development of UI and OAB include increasing age, prostatectomy, UTIs, and cognitive and functional impairment. Presence of neurologic diseases or other genitourinary conditions (i.e., benign prostatic enlargement, bladder tumor, bladder lithiasis, congenital pathologies) should be adequately considered due to the impact of these conditions on bladder function. When a neurologic disease has been identified as responsible for the presence of bladder-sphincter dysfunction with OAB, a specialized assessment and management are required.
Urinalyses and culture are also useful to confirm or exclude the diagnosis of OAB (Fig. 3.2). In the presence of isolated hematuria, without signs of infection, patients should perform an in-depth urologic evaluation.
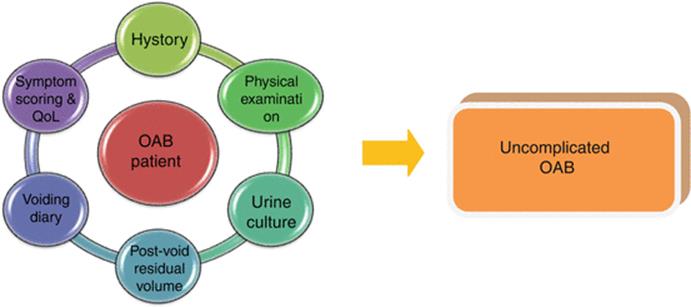
Fig. 3.2
Initial assessment of non-neurogenic patients with OAB: urine culture and/or postvoid residual assessment, bladder diaries, and/or symptom questionnaires are useful to make diagnosis. American Urological Association (AUA) Guideline. Statement 2
The assessment of residual urine by measuring the postvoid urinary volume with bladder scan or catheterization is useful particularly when patients present with symptoms suggestive of voiding dysfunction, POP, or recurrent UTIs (Fig. 3.2).
According to Nice and EAU Guidelines on urinary incontinence, in the initial assessment of patients with OAB, the use of micturition time charts, frequency volume charts, or voiding diaries should be strongly encouraged. Micturition time charts are used to record only the times of micturitions for a minimum of 24 h; frequency volume charts are used to record urinary volumes and times of each micturition for a minimum of 24 h. “Voiding” or “bladder” diary is used to report more detailed information on daily frequency of micturition, fluid intake, episodes of urgency and/or urgency incontinence, and pad usage. A minimum of 3 days voiding diary is usually required, and it should be completed during patients’ usual activities, such as both working and leisure days (Fig. 3.2).
The use of PAD test in the routine clinical assessment of patients with UI is not recommended by the Nice, EAU, and AUA Guidelines. Nevertheless, there is at least a good evidence to show that repeat pad testing can detect change following treatment for UI.
Symptom Scoring and Quality of Life Assessment
Although there is no information about whether using questionnaires to assess urinary symptoms and QoL improves outcomes in patients with UI or OAB, the use of validated symptom questionnaires is useful to quantify and characterize urinary symptoms and their impact on patient’s QoL (Fig. 3.2). Health-Related Quality Of Life questionnaires (HRQoLq) may be generic, as SF-36 or specific for a particular condition, as Incontinence Impact Questionnaire, the King’s Health Questionnaire, OAB-q, to study OAB. Additionally, questionnaires can help in evaluating the response to treatment along follow-up and to better monitoring patients’ condition along time. Questionnaires should have been validated for the language in which they are being used. Some of validated questionnaires mainly used for the assessment of OAB and urgency are showed in Table 3.1.
Table 3.1
Some of validated questionnaires used for the assessment of OAB and urgency
|
Assessment of severity and impact of OAB |
Abbreviations |
|
Urinary Incontinence-Specific Quality of Life Instrument |
I-QOL (ICIQ-Uqol) |
|
Overactive bladder questionnaire |
ICIQ-OABqol |
|
King’s Health Questionnaire |
KHQ |
|
OAB Satisfaction questionnaire |
OAB-SAT |
|
Overactive Bladder Symptom Score |
OAB-SS |
|
OAB-S = Overactive Bladder Satisfaction measure |
OAB-S |
|
Assessment of the Impact of Urgency |
|
|
Patient’s Perception of Intensity of Urgency |
PPIUS |
|
Scale Urge Incontinence Impact Questionnaire |
U-IIQ |
|
10-Item Scale to Measure Urinary Urgency |
UU Scale |
|
Urge Urogenital Distress Inventory |
U-UDI |
|
Urgency Severity and Intensity Questionnaire: Quality of Life |
USIQ-S |
Urodynamic Testing
Multichannel cystometry, ambulatory urodynamics (UDS), or videourodynamics are diagnostic instruments including multiple tests to study lower urinary tract function during both bladder storage and emptying. While it is now recognized that UDS is not required to diagnose and manage OAB in non-neurogenic patients, information about the presence or absence of detrusor overactivity (DO) and the volume at which patients experiment OAB symptoms can be helpful in their management. In patients refractory to conservative treatment, UDS can help to identify DO, high pressure of uninhibited detrusor contractions, and a reduced compliance, which can all induce UUI and OAB. Importantly, it should be reminded that the diagnoses of UUI are not related to urodynamic presence of DO, and the presence of DO is neither sensitive nor specific for UUI. This is in agreement with the AUA Urodynamics Guidelines, which state that multichannel filling cystometry should be performed when it is important to determine whether “altered compliance, DO, or other urodynamic abnormalities are present or not in patients in whom invasive, potentially morbid, or irreversible treatments are considered.” This is the case when OAB patients who are refractory to conventional anticholinergics need to be treated with surgical intervention, such as sacral neurostimulation or intradetrusor onabotulinumtoxinA injections. The AUA, OAB Guidelines and the EAU Guidelines on UI support these statements and underline that UDS should not be used in the initial assessment of patients with uncomplicated OAB (Fig. 3.3). Also the Nice Guidelines do not recommend the use of UDS before starting conservative management. Only after a detailed history and physical examination, UDS should be performed before surgery in women who present with OAB symptoms suspected for DO and voiding dysfunction, or who had previous surgery for stress incontinence or POP.
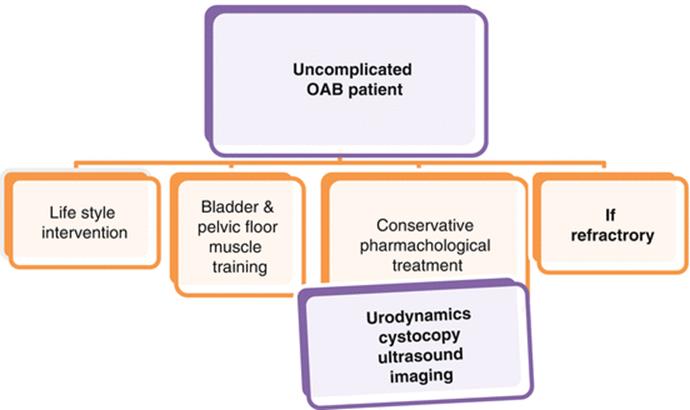
Fig. 3.3
Diagnostic investigations in patients with uncomplicated OAB
The use of videourodynamics or ambulatory urodynamics should be considered for OAB patients when the diagnosis remains unclear after standard UDS or for patients with OAB symptoms affected by neurogenic diseases.
Other Investigations
For the routine assessment of patients with isolated OAB syndrome, the use of cystoscopy and imaging (MRI, CT, X-ray) is not required.
With regard to the use of ultrasound in the evaluation of OAB patients, it has been suggested that lower urinary tract ultrasound should be useful to study detrusor and bladder wall thickness, as bladder hypertrophy can be related to the severity of detrusor contractions in OAB patients. Indeed, ultrasound measurement of detrusor and bladder wall thickness may be influenced by several factors, as the ability of the operator, type of ultrasound probes and image resolution, and degree of bladder filling. Actually, there is no demonstration that studying these measures could improve the management of patients affected by OAB. Thus, the use of ultrasound should be limited to the evaluation of postvoid residual urine volume.
Differential Diagnosis
Differential diagnoses should be made between OAB syndrome and some conditions that can mimic the same symptoms as polydipsia, nocturnal polyuria, interstitial cystitis, and/or bladder pain syndrome. In the latter case, the presence of bladder and/or urethral pain can help to perform a correct diagnosis. Additionally, OAB symptoms could be referred in the presence of the following conditions: neurogenic diseases, depression, erectile dysfunction, dementia, clinically benign pelvic masses, associated fecal incontinence, symptoms of voiding difficulty due to BPE or POP, previous continence surgery, previous pelvic cancer surgery, and previous pelvic radiation therapy. These conditions require a further, specialized assessment and management.
Conservative Treatment
Stephen Mock8 , Melissa Kaufman8, W. Stuart Reynolds8 and Roger Dmochowski8
(8)
Vanderbilt University Medical Center, Nashville, TN, USA
Stephen Mock
Email: stephen.mock@vanderbilt.edu
Roger Dmochowski (Corresponding author)
Email: roger.dmochowski@vanderbilt.edu
All patients with OAB should be provided with education and an explanation of normal lower urinary tract structure and function and what is known about OAB. According to the AUA panel, “explaining what is normal can help the patient understand their condition and give a comparator for establishing mutually identified and realistic goals for treatment. Education empowers the patient to participate in their treatment, an essential factor when interventions rely on behavioral change.” Patients with OAB will likely require long-term or lifelong therapy to control their symptoms, and as a result, a treatment plan should weigh the burden of symptoms on a patients’ QoL, as well as factor that potential benefit of a particular treatment against the treatment’s risk. Treatment initiation presumes a perceived improvement can be generated. The patient has to be a willing and engaged participant who understands that OAB has a variable and chronic course, and acceptable symptom control may require multiple therapeutic trials before it is achieved. It may not be possible to eliminate all symptoms. It must be stressed that no treatment is an acceptable choice made by some patients and caregivers.
First-Line Therapy
Lifestyle and behavioral modification should be offered to all patients with OAB. Lifestyle changes include cessation of smoking, weight reduction, elimination of known dietary bladder irritants such as alcohol or caffeine, adequate fluid intake, and regular bowel habits. Nuotio et al. showed a correlation between smoking and urinary urgency in a large population-based survey. In further support, the EUA gave a grade A to smoking cessation advice in line with good medical practice in patients with UI, but the level of evidence that it improves UI symptoms was only based of expert opinion and not randomized data (Lucas 2012). Obesity has been shown to be an independent risk factor for the development of SUI and mixed UI in women, and evidence exists that nonsurgical weight loss intervention has the potential to improve UI in overweight women. Indeed, a weight loss of 8 % in obese women reduced overall incontinence episodes per week and UUI episodes by 47 and 42 % vs. 28 and 26 % in controls. Reduced fluid intake is a widely used, inexpensive and noninvasive intervention but can predispose to UTI, dehydration, stone formation, and constipation. The data available is conflicting on whether fluid modification changes symptoms and QoL, but a recent trial showed a 25 % reduction in fluid intake improved symptoms with OAB but not UI. Caffeine has an excitatory effect on detrusor muscles, and randomized controlled data demonstrates significant improvements in urgency-frequency and UUI in those who reduced caffeine to <100 mg/day. Alcohol has a diuretic effect by reducing the release of antidiuretic hormone. Newman et al. reported a nearly uniform positive association between straining at stool and constipation and lower urinary tract symptoms which the authors surmised to be due to progressive neuropathy in the pelvic floor secondary to repeated straining efforts.
Behavioral treatments are a group of risk-free therapies which improve individual symptoms by changing patient behavior or the patient’s environment. They can be tailored individually and include bladder training, bladder control strategies, pelvic floor muscle training, and fluid management. This form of therapy relies on a patient with an intact nervous system to “relearn” to inhibit a detrusor contraction or a sensation of urgency. It generally consists of patient education, scheduling voiding with an aim to achieve an interval of 3–4 h between voids, and positive reinforcement that provides psychological support and encouragement. There is level 1 evidence in support of supervised bladder training but whose benefits are of short duration unless the program is practiced repeatedly. In the MOTIVE trial (Male overactive Bladder Treatment in Veterans), where Burgio et al. demonstrated equivalence between behavioral and antimuscarinic therapy, behavioral treatment involved verbal instruction followed by guided practice using verbal feedback based on anal palpation to teach participants how to contract and relax pelvic floor muscles while keeping abdominal muscles relaxed, as well as delayed voiding, monitoring with bladder diaries, and urge suppression techniques to inhibit or suppress detrusor contractions and thus reduce urgency-frequency and incontinence (Burgio 2011). Similar findings from randomized trials have been published that have demonstrated equivalence or superiority to medications in reducing symptoms, while more limited literature exists that indicates behavioral and drug therapy simultaneously may improve outcomes.
Pelvic floor muscle training (PFMT) is used frequently in conjunction with bladder training. The purpose is to increase the strength and durability of contraction of the pelvic floor muscles, which may help inhibit bladder contractions in patients with OAB. Biofeedback therapy can be used to augment PFMT and involves the use of monitoring equipment to detect, measure, and amplify internal physiological responses to provide patients with feedback concerning those responses. It increases patient awareness of the pelvic floor muscles, using visual, tactile, or auditory stimuli and is used to help teach patients to exercise their musculature more effectively. The most common modalities include electromyography (EMG) and manometry that measures the activity of the anal or urinary sphincter, the pelvic floor musculature, as well as the detrusor muscle with the aim to restore bladder control by teaching patients to modulate the mechanism of continence. There is level 1 evidence supporting both PFMT and the addition of biofeedback for added benefit. Berghmans et al. conducted a meta-analysis study assessing the efficacy of biofeedback in UUI and concluded that patients were able to achieve improvement after biofeedback. They stated that the actions were easy to perform in cognitively intact patients but took personal effort and time to perform the exercises.
Second-Line Treatment
Antimuscarinic agents act via competitive inhibition at the postganglionic receptor sites and, as a result, reduce bladder contractility and particularly exert their effects on the filling phase of the bladder, reduce urgency, increase bladder capacity, and minimize the effects of the parasympathetic system. While effective in reducing symptoms, they are commonly associated with side effects (e.g., dry mouth, constipation, dry eyes, dyspepsia, urinary retention, impaired cognitive function) as a result of the affinity of the drugs to muscarinic receptors widely distributed in the body. Additionally, for those patients with narrow angle glaucoma, impaired gastric emptying, or a history of urinary retention, consultation with each respective specialist should be sought prior to initiation of therapy. Clinicians must use caution in prescribing these agents in patients who are using other medications with anticholinergic properties. Agents available include darifenacin, fesoterodine, oxybutynin, solifenacin, tolterodine, or trospium. Both the EUA and AUA performed an extensive review of the randomized trials evaluating these agents for OAB and found no compelling evidence for differential efficacy across medications. Thus, the choice of medication is largely determined by patient factors that include age, side-effect profile, and out-of-pocket costs. However, if extended-release formulations are available, they should be preferentially prescribed because of the lower rates of side effects (Grade A recommendation, EUA). For likewise reasons, transdermal preparations may be offered, if available (Grade B recommendation, EUA). In those individuals who experience inadequate symptom relief and/or unacceptable adverse events, with one antimuscarinic agent, then a dose modification or a different antimuscarinic medication may be tried.
Special attention should be given to the frail elderly, a group where data on the use of antimuscarinic is not as robust and side effects may be more severe. Elderly people have been underrepresented in randomized controlled trials (RCTs) of antimuscarinic agents, despite having a higher prevalence. There is level 2 evidence that oxybutynin IR may worsen cognitive function and as a result received a grade A recommendation against its usage in those at risk of cognitive impairment (Lucas 2012). However, the effectiveness and risk of adverse events, including cognitive dysfunction, of solifenacin, tolterodine, and darifenacin, do not differ with patient age (level 1b evidence (Lucas 2012)). Nocturia, a symptom with a multifactorial etiology, is an another especially concerning issue in the elderly as it has been linked to a risk of falls, fractures, and even death. The role of antimuscarinics for the management of nocturia in the context of OAB is limited. Pooled analysis and/or post hoc analyses have demonstrated significant reduction of nocturnal voids per night, but the effect size was low and the clinical significance doubtful. To our knowledge, only one RCT that compared tolterodine with placebo focused on nocturia as a primary endpoint. In this study, micturitions were classified as either OAB related or not depending on the degree of urgency prior to that micturition. Eight hundred and fifty patients were randomized, and while tolterodine did significantly reduce OAB specific symptoms, there was no difference between the two groups for nocturia for the non-OAB micturitions and when both non-OAB and OAB micturitions were considered together. This clearly demonstrates the complex nature of nocturia resulting from its multiple contributing etiologies.
Nearly all studies for OAB either require UUI for study inclusion or include a mix of patients with both OAB dry and wet. For this latter case, rarely is subgroup analysis performed and thus depriving the ability to compare efficacy of treatment between OAB dry and wet. While it is believed, based on expert interviews, that OAB is represented by a spectrum of severity, from mild OAB with no leakage (OAB dry) to severe OAB with frequent leakage (OAB wet), there is no evidence to substantiate this. Lacking this, it is unknown whether each represents a unique clinical entity with differential treatment outcomes. This may largely be an academic exercise as urgency rather than incontinence has been cited as the most bothersome symptom and the one with the greatest impact on quality of life. Urgency data is available as an endpoint in most studies and is favorable (Table 3.2). Since OAB treatment is aimed at symptom relief, this may be more important than the dichotomy of OAB into wet or dry.
Table 3.2
Selected published series of randomized controlled trials of various anticholinergic agents
|
Author |
Drug |
Inclusion |
Length of study |
n |
Results |
Adverse events |
Notes |
|
Rudy et al. (2006) |
Trospium 20 mg BID |
Females and males, >18 years old, OAB >6 months; urinary frequency ≥10/day, +urgency, ≥7 UUI/week |
12 weeks |
658 |
Daily voids: −2.67 vs. −1.76, p < 0.0001 Daily UUI episodes: −1.86 vs. −1.29, p < 0.0001 Urgency score: −0.21 vs. −0.02, p < 0.001 Volume voided per void/24 h: +35.59 vs. +9.44, p < 0.0001 |
Dry mouth: 19.8 % vs. 5.2 % Constipation: 10.9 % vs. 5.8 % Discontinuation rate: 7.3 % vs. 4.6 % |
Quaternary amine and ionized at physiological pH; low lipophilicity and reduced CNS penetrance |
|
Dmochowski et al. (2008) |
Trospium 60 mg extended release, daily |
Females and males, >18 years old, OAB >6 months; urinary frequency ≥10/day, >1 severe urgency with void, mean >1 UUI/day |
12 weeks |
564 |
Daily voids: −2.5 vs. −1.8, p < 0.001 Daily UUI episodes: −2.4 vs. −1.6, p < 0.001 Urgency score: −0.28 vs. −0.13, p < 0.001 Volume voided per void/24 h: +31.5 vs. +17.8, p < 0.01 |
Dry mouth: 12.9 % vs. 4.6 % Constipation: 7.5 % vs. 1.8 % Discontinuation rate: 6.4 % vs. 2.8 % |
|
|
Van Kerrebroeck et al. (2001) |
Tolterodine ER 4 mg daily, tolterodine IR 2 mg BID |
Females and males, >18 years old, OAB >6 months; urinary frequency ≥8/day, ≥5 UUI/week |
12 weeks |
1529 |
Daily voids : ER −3.5 vs. −2.2, p < 0.001; IR −3.3 vs. −2.2, p = 0.0002 Weekly UUI episodes: ER −11.8 vs. −6.9, p < 0.0001; IR −10.6 vs. −6.9, p = 0.001 Volume voided per void/24 h: ER +34 vs. +14, p < 0.0001; IR +29 vs. +14, p < 0.0001 |
Dry mouth: ER 23 %, IR 30 %, placebo 8 % Constipation: ER 6 % IR 7 %, placebo 4 % Discontinuation rate: ER 5 % IR 5 %, placebo 6 % |
Prodrug and requires cytochrome P450 activation; ER formulation 18 % more effective than IR in UUI episodes; ER 23 % less dry mouth than IR |
|
Chapple et al. (2005) |
Darifenacin 7.5 mg and 15 mg daily |
Females and males, >18 years old, OAB >6 months; urinary frequency ≥8/day, 5–50 UI/week, mean of ≥1 urgency episode/24 h |
12 weeks |
1059 |
Mean voids/day: 7.5 mg −1.6 vs. −0.9, p < 0.001; 15 mg: −1.9 vs. −1.0, p < 0.001 Mean UI episodes/week: 7.5 mg −4.0 vs. −2.0, p < 0.001; 15 mg −4.8 vs. −2.7, p < 0.001 Mean urgency episodes/day: 7.5 mg −2.0 vs. −1.0, p < 0.001; 15 mg −2.3 vs. −1.2, p < 0.001 Average void volume: 7.5 mg +15 vs. +8, p = 0.007; 15 mg +27 vs +6, p < 0.001 |
Dry mouth: 7.5 mg 20.2 %, 15 mg 35.3 %, placebo 8.2 % Constipation: 7.5 mg 14.8 %, 15 mg 21.3 %, placebo 6.2 % Discontinuation rate: 7.5 mg 1.5 %, 15 mg 5.1 %, placebo 2.6 % |
Selective muscarinic M3 receptor antagonist; randomized data available demonstrating no change in cognition or QTc intervals |
|
Cardozo et al. (2005) |
Solifenacin 5 mg and 10 mg daily |
Females and males, >18 years old, OAB >3 months; urinary frequency ≥8/day, >3 urgency episodes and/or >3 UI during 3 day diary |
12 weeks |
907 |
Daily void: 5 mg −2.37 vs. −1.59, p = 0.002, 10 mg −2.81 vs. −1.59, p = 0.0001 Daily UI: 5 mg −1.63 vs −1.25, p = 0.002; 10 mg −1.57 vs. 1.25, p = 0.016 Urgency episodes: 5 mg −2.84 vs −1.98, p = .003; 10 mg −2.9 vs. −1.98, p = 0.002 Mean voided volume: 5 mg +30.75 vs. +10.75, p = 0.0001; 10 mg +36 vs. +10.67, p = 0.0001 |
Dry mouth: 5 mg 7.7 %, 10 mg 23.1 %, placebo 2.3 % Constipation: 5 mg 3.7 %, 10 mg 9.1 %, placebo 2.0 % |
|
|
Abrams et al. (2005) |
Solifenacin 5 and 10 mg daily |
Females and males, >18 years old, OAB >3 months; urinary frequency ≥8/day, ≥1 urgency episodes /day |
12 weeks |
975 |
Daily voids: 5 mg −3.6 vs. −1.6, p < 0.001; 10 mg −2.8 vs. −1.6, p < 0.001 Urgency episodes/day: 5 mg −3.2 vs. −2.1, p < 0.001; 10 mg −3.2 vs. −2.1, p < 0.001 Mean voided volume 5 mg +25 vs +7, p < 0.001; 10 mg +33.9 vs. +7, p < 0.001 |
Dry mouth: 5 mg 10.8 %, 10 mg 24.4 %, placebo 3.6 % Constipation: 5 mg 4 %, 10 mg 12.2 %, placebo 1.3 % Discontinuation rate: 5 mg 2.8 %, 10 mg 7.8 %, placebo 6.2 % |
Pooled analysis of 4 RCTs with subgroup analysis solely for OAB dry |
|
Nitti et al. (2007) |
Fesoterodine 4 mg and 8 mg daily |
Females and males, >18 years old, OAB >6 months; urinary frequency ≥8/day and mean of >6 urgency episode or >3 UUI over 3 day diary |
12 weeks |
836 |
Daily voids: 4 mg −1.61 vs −1.08, p < 0.001; 8 mg −2.09 vs. −1.08, p < 0.001 Daily UUI: 4 mg −1.65 vs. −0.96, p < 0.001; 8 mg −2.28 vs. 0.96, p < 0.001 Daily urgency episodes: 4 mg −1.91 vs. −.079, p < 0.001; 8 mg −2.3 vs. −0.79, p < 0.001 Voided volume: 8 mg +33.6 vs. +8.38, p < 0.001 |
Dry mouth: 4 mg 16 %, 8 mg 36 %, placebo 7 % Constipation: 4 mg 5 %, 8 mg 8 %, placebo 3 % Discontinuation rate: 4 mg 6 %, 8 mg 9 %, placebo 4 % |
Prodrug with same active metabolite as tolterodine; dual excretion pathways |
|
Versi et al. (2000) |
Oxybutynin 5 mg/day IR vs ER, 5 mg dose increases q7 days to max of 20 mg/day |
Females and males; >7 UUI/week; +prior response to oxybuytnin |
226 |
IR group 76 % reduction in UUI ER 83 % reduction-no significant difference |
Dose dependent significant difference in dry mouth between both groups |
Has direct muscle relaxant effect as well |
|
|
Dmochowski et al. (2003) |
Oxybutynin transdermal patch 3.9 mg/day; tolterodine LA 4 mg/day |
Females and males; +prior prestudy response; >4 UUI, >24 voids, <350 cc/void over 3 day diary |
12 weeks |
361 |
Daily UUI: patch −3 vs. placebo −2, p = 0.01 Average void volume: patch +24 vs. placebo +5.5, p = 0.001 |
Site erythema: patch 8.3 % vs placebo 1.7 % Pruritus: patch 14 % vs placebo 4.3 % Dry mouth: patch 4.1 %, tolterodine 7.3 %, placebo 1.7 % Constipation: patch 3.3 %, tolterodine 5.7 % |
Avoidance of GI and hepatic first pass metabolism to DEO, which is responsible for anticholinergic side effects |
As seen, while anticholinergic agents are the mainstay of pharmacological treatment for OAB, results may be suboptimal or can have numerous side effects and contribute to poor patient compliance and high discontinuation rates. Mirabegron is a first-in-class medication whose mechanism of action is via β3-adrenoceptor agonism and resultant detrusor relaxation and facilitation of urine storage. It gained Food and Drug Administration (FDA) approval in June 2012 and European Union approval in January 2013. Nitti et al. published the 12-week results of the phase 3 RCT of mirabegron vs. placebo. The study population was required to have an average of eight or more micturitions per 24 h and three or more urgency episodes with or without incontinence. Other OAB medications had to be stopped at screening. Patients were randomized to receive placebo, mirabegron 50 or 100 mg. Nearly 60 % had previously received antimuscarinic OAB medication. As a result of inclusion of both wet and dry OAB patients, the authors performed analysis on the composite study group (1270 patients) as well as just the OAB wet patients (933 patients). Both mirabegron treatment groups demonstrated significant greater reductions from baseline vs. placebo in mean number of incontinence episodes (−1.13, −1.47, −1.63), micturitions per 24 h (−1.05, −1.66, −1.75), mean level of urgency, mean number of grade 3 or 4 urgency episodes per 24 h, as well as greater increases in mean volume voided per micturition (7.0, 18.2, 18). Adverse events that included hypertension and cardiac arrhythmias were low and comparably to placebo, but small dose-dependent differences in a.m. and p.m. pulse rates were noted.
Third-Line Treatment
Please see chapter on chemical and electrical neuromodulation for OAB for more details.
Additional Treatments
Indwelling catheters are not recommended as a management strategy for OAB and, in our experience, can exacerbate symptoms. Additionally, indwelling catheterization has a high risk of catheter-associated UTIs, urethral erosion/destruction, and urolithiasis. Adjunctive measures such as pads, special undergarments, and condom catheters for men are valuable if symptoms prove refractory, especially in those where the risk of therapy outweighs the benefit. However, an indwelling catheter may be considered as a last resort in cases where UI contributes to progressive skin breakdown or decubiti. Intermittent catheterization may be an option when OAB symptoms are associated with incomplete emptying, a commonly seen phenomena in the elderly, but this approach requires patient willingness and/or caregiver support.
Very rarely is major abdominal surgery such as augmentation cystoplasty, detrusor myectomy to disrupt coordinated detrusor contraction, or urinary diversion needed for idiopathic OAB. It may be considered in cases of severe and refractory OAB, but the patient must know the substantial risk to these procedures, and the data to support it are poor (level 3 evidence, EUA (Lucas 2012)). Use of these options has decreased substantially due to its short- and long-term risks as well as the effectiveness of Botox and neuromodulation.
Figure 3.4 provides an algorithm for the diagnosis and treatment for idiopathic OAB.
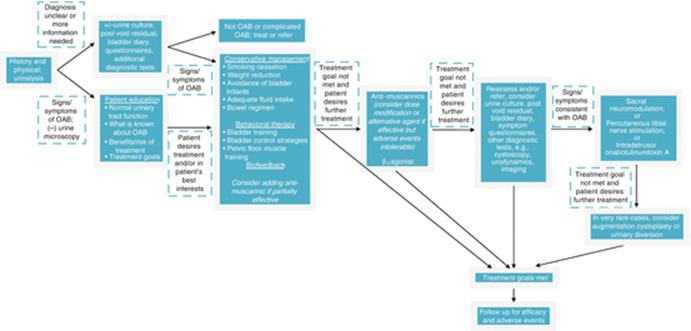
Fig. 3.4
Algorithm for diagnosis and treatment of idiopathic overactive bladder
Conclusion
OAB is a common symptom-driven clinical diagnosis that can have an adverse impact on a patient’s QoL. After a full evaluation to exclude other possible pathologies, counseling and education on OAB should be discussed with the patient. The risk and benefits of the available treatment options should be given. Every OAB patient should be offered conservative and behavioral therapy as a first-line treatment as these are largely risk-free and tailorable to patients and their symptoms. If the response is inadequate, treatment should proceed in stepwise fashion from available pharmacotherapy to surgical intervention. While a lifelong problem, patients now have the benefit of multiple lines of therapy to pursue and which continue to expand.
Tips and Tricks
1.
2.
3.
4.
Botulinum Toxins
Antonella Giannantoni9 , Silvia Proietti10 and Massimo Porena9
(9)
University of Perugia, Perugia, Italy
(10)
Humanitas Clinical and Research Centre, Rozzano, MI, Italy
Antonella Giannantoni
Email: agianton@libero.it
Botulinum neurotoxins (BoNT) are protein complexes produced by Clostridium botulinum, an anaerobic gram-positive Bacillus. BoNT interferes with the release of acetylcholine (Ach) from presynaptic terminals, what clinically prevents muscle contractions and/or gland secretion. Since BoNT is used as a local injectable treatment, with fewer side effects as compared to systemic administered medications, and due to its ability to determine a robust muscle relaxation, it is used as treatment of choice in many neurologic diseases such as dystonia and limb spasticity and in other conditions, as glabellar lines, hyperhidrosis, and sialorrhea. Since the end of 1990, there has been an increasing use of onabotulinumtoxinA in the treatment of urinary incontinence due to neurogenic or idiopathic DO.
Botulinum Neurotoxin: Structure, Serotypes, and Subtypes
The neurotoxin is produced as a 150 kDa polypeptide and contains a heavy chain of 100 kDa (Hc) which is attached to a light chain molecule of 50 kDa (Lc) by a disulfide non-covalent bond. The heavy chain is divided into an amino domain (Hcn) and a carboxyl terminal domain (Hcc) (Fig. 3.5).
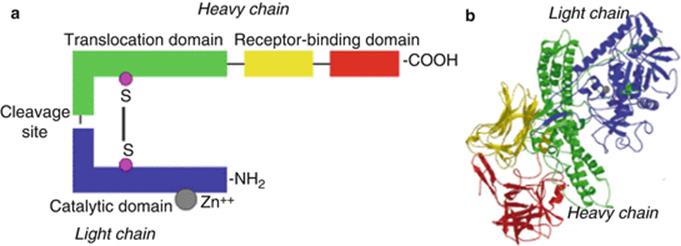
Fig. 3.5
OnabotulinumtoxinA molecular structure. OnabotulinumtoxinA molecular structure depicted in a schematic way (a) and threedimensional way (b)
Seven serologically different neurotoxins (designated as neurotoxin types A-G) are produced by four strains/species of Clostridium botulinum. Each of the A-G serotypes of BoNTs are divided in subtypes on the basis of differences in amino acid sequences and, consequently, of different immunological and biological properties. Five subtypes of BoNT/A have been described (A1–A5), all of which have a similar affinity for binding SNAP25 somewhat with less efficiency (i.e., BoNT/A3 and BoNT/A4).
Mechanisms of Action
Neuromuscular transmission starts with an action potential reaching the presynaptic endplate, which then transiently depolarizes the axon terminal and opens the calcium channels. The increase in intracellular calcium leads to the fusion of vesicle membranes containing Ach with the presynaptic endplate. The propagation of the action potential along the terminal axon of the motoneuron produces the release of ACh from the cytosol into synaptic clefts by the soluble N-ethylmaleimide-sensitive factor activating protein receptor complex (SNARE). Once in the synaptic cleft, ACh binds to postsynaptic membrane inducing a muscle fiber contraction. In the presence of BoNT, the carboxyl terminal of the heavy chain of the toxin binds to specific receptors from the presynaptic terminal. BoNT is endocytosed and then the N-domain of the heavy chain translocates the light chain into the cytoplasm. Once in the cytoplasm, the light chain cleaves SNAP25 of the SNARE complex protein, thus preventing Ach release (Fig. 3.6). This process can be subdivided into four steps: binding, internalization, membrane translocation, and proteolysis of specific SNARE proteins. More recently, it has been demonstrated that BoNTs are also able to act at the level of sensitive nerve fibers, by blocking the vesicular exocytosis of different substances, such as CGRP, SP, NGF, and ATP, which are involved in the afferent neurotransmission.
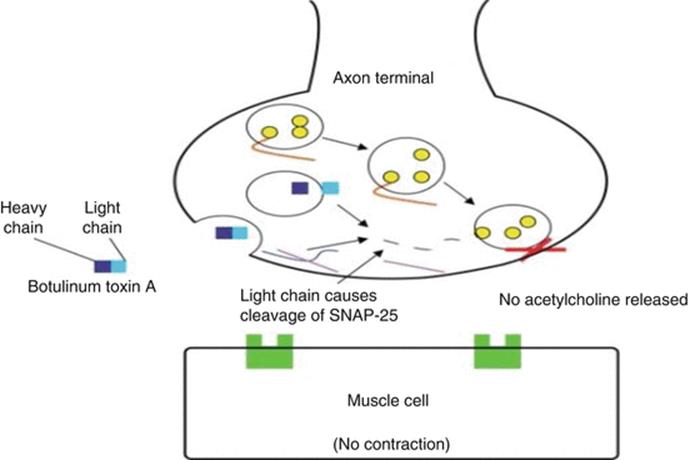
Fig. 3.6
OnabotulinumtoxinA mechanisms of action
Commercially Available Neurotoxins
Actually, there are only two serotypes available: BoNT/A (onabotulinumtoxinA, abobotulinumtoxinA, and incobotulinumtoxinA) and a BoNT/B, patented by rimabotulinumtoxinB. Each brand has been approved by the US Food and Drug administration (FDA). Commercial preparations of BoNTs include the neurotoxin molecule, nontoxic hemagglutinin proteins, and non-hemagglutinin proteins, plus excipients. The associated proteins are thought to stabilize the neurotoxin. Protein complexes are not present in incobotulinumtoxinA, which is a formulation of botulinum toxin serotype A. Units of biological activity of different botulinum toxin A cannot be converted into units of any other botulinum toxin.
BoNTs and OAB
Although the use of botulinum toxin A (American and English brands) to treat neurogenic DO and idiopathic OAB was introduced about 15 years ago, only more recently, the use of onabot/A has been licensed for the treatment of neurogenic and idiopathic DO in US and most European countries, in patients who have been inadequately managed by ≥1 anticholinergic therapy. Onabot/A is not interchangeable with other botulinum toxin preparations.
Clinical Effects
In the treatment of idiopathic OAB, two large, double-blind placebo-controlled phase III trials demonstrated that onabot/A100U intravesically injected significantly reduced all symptoms of OAB, including episodes of incontinence, urgency, micturition, and nocturia compared with placebo. In these studies, improvements in OAB symptoms were also accompanied by clinically relevant improvements in patients’ HRQoL. In patients with NDO, onabot/A has been observed to induce a reversible chemical denervation lasting for about 9 months. The efficacy of onabot/A has been demonstrated in patients with neurogenic diseases and urological disorders in phase III RCT randomized placebo-controlled trials. What is evident is that repeated injections seem to be possible without loss of efficacy. Histological studies have not found ultrastructural changes after injection. In the EAU Recommendations of minimally invasive treatment for NDO, botulinum toxin intradetrusorial injection is considered the most effective treatment to reduce NDO.
The efficacy of onabotulinumtoxinA can be attributed to its inhibitory effects at the level of both efferent and afferent nervous pathways controlling bladder function. In recent times, great importance has been attributed to the action of onabotulinumtoxinA into the bladder wall. It decreases the mucosal levels of the afferent receptors TRPV1 and P2X3, which are involved in sensory transmission and can reduce the levels of neurotransmitters involved in the mechanisms of urgency. Thus, the current notion is that onabotulinumtoxinA modulates the intrinsic reflexes thought to cause the OAB condition.
Side Effects
The most important adverse events are UTI and an increase in postvoid residual volume, which may require the use of intermittent catheterization. The emergence of high PVR should be obviously avoided in patients who void spontaneously. Conversely, in patients with neurogenic disease unable to spontaneously empty the bladder and using CIC, the goal is properly to obtain detrusor areflexia with higher capacity. In this patient population, generalized muscular weakness can be an occasional adverse effect.
Doses
In patients with idiopathic OAB with urinary incontinence, the established licensed dose of onabotulinumtoxinA is 100U diluted in 10 ml saline solution. In NDO, the established licensed dose of onabotulinumtoxinA is 200U diluted in 30 ml saline solution.
Recommendations of OnabotulinumtoxinA in the Treatment of NDO and IDO
In synthesis, EAU Guidelines state that onabot/A (100 U) intravesically injected can be offered to patients with urgency urinary incontinence refractory to antimuscarinic therapy. Patients have to be advised about the “risk of UTI and the possible prolonged need to self-catheterize (ensure that they are willing and able to do so).” In addition, the EAU Guidelines outline some evidences related to the repeat use of onabot/A, the eventual need to perform CIC, the superiority of onabot/A in comparison with solifenacin per os, and other important information.
How to Perform OnabotulinumtoxinA Intradetrusorial Injection
· In adult males and females with IDO and urgency urinary incontinence : after a local anesthesia, or a mild general sedation, by using flexible or rigid cystoscope, 100U onabot/A diluted in 10 ml saline solution, intradetrusorial injections can be performed (20 injections into the bladder wall, with the exclusion of the trigone).
· In adult males and females with NDO : after a local anesthesia, or a mild general sedation, with flexible or rigid cystoscope, 200 U onabot/A diluted in 30 ml saline solution, intradetrusorial injections can be performed (30 injections into the bladder wall, with the exclusion of the trigone).
In the procedure, the needle for the injection should be typically 22–27 gauge and 4 mm in length.
While some information exist on repeat intradetrusorial injections of onabot/A in patients with NDO, very few data exist on long-term follow-up of intravesical onabot/A treatment in patients with IDO and OAB. Indeed in these few studies, onabot/A has been observed to induce a stable and significant amelioration of OAB symptoms, without serious side effects, and a long-lasting improvement of patients’ QoL.
Points of Interest
· Overactive bladder (OAB) is a complex of symptoms defined by International Continence Society as “urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence (UUI), in the absence of urinary tract infection (UTIs) or other obvious pathology”.
· The initial assessment of a patient with uncomplicated OAB symptoms should include a careful history and physical examination, urinalyses and culture, and the assessment of residual urine by measuring the postvoid urinary volume. The use of micturition time charts, frequency volume charts, or voiding diaries allow to confirm the clinical diagnosis. Validated symptom questionnaires are useful to quantify and characterize urinary symptoms and their impact on patient’s QoL.
· The use of urodynamics is not recommended before starting conservative management. It should be performed before surgery in women who present with OAB symptoms suspected for detrusor overactivity and voiding dysfunction, or who had previous surgery for stress incontinence or urogenital prolapse.
· First-line treatment consists of conservative measures (bladder training and anticholinergics). When OAB is refractory to these interventions, therapeutic options such as onabotulinumtoxinA intradetrusor injections, electrical stimulation, or surgical interventions can be applied.
· OnabotulinumtoxinA intravesically injected can be offered to patients with idiopathic urgency urinary incontinence and to patients with neurogenic detrusor overactivity refractory to antimuscarinic therapy. Patients have to be advised about the risk of urinary tract infections and the possible need to self-catheterize.
Percutaneous Tibial Nerve Stimulation: Neuromodulation for the Masses
Scott MacDiarmid11
(11)
Alliance Urology Specialists, Greensboro, North Carolina, USA
Scott MacDiarmid
Email: smacdiarmid@allianceurology.com
Overactive bladder (OAB) has a deleterious effect on nearly every aspect of daily life, greatly affecting the overall health and well-being of those who suffer from it. The mainstay of therapy for OAB is behavioral therapy and antimuscarinic agents and now a β-3 agonist. Percutaneous tibial nerve stimulation (PTNS) is a neuromodulation therapy that offers patients an attractive alternative when conservative measures fail.
In this section, I will discuss the significant clinical need supporting the liberal use of neuromodulation therapies in the treatment of OAB. I will review the literature regarding the efficacy and safety of PTNS as well as provide clinical tips to help make PTNS successful in your practice. Importantly, an algorithm for the treatment of OAB will also be presented. My personal mission is to inspire clinicians to strive for excellence in the management of OAB and to liberally offer neuromodulation therapies to their patients.
A New Disease in Town
Health providers worldwide are well aware that the primary treatment of OAB is behavioral modification and medication with the goal of relieving symptoms and balancing drug treatment efficacy with side effects and costs. Unfortunately, many patients do not reach their treatment goal with conservative therapy and suffer from a new disease that in day-to-day practice is seldom talked about by patients and healthcare providers. This disease is called “refractory OAB” and to which I have coined the term “ROAB.”
Who Has ROAB?
Simply stated – millions do! In fact, I believe that ROAB is more prevalent than OAB that is currently effectively managed by medical therapy. Let’s face it; more than 50 % of OAB patients treated do not respond favorably to or do not reach their treatment goal with medication.
The American Urological Association (AUA) Guideline panel defines the refractory OAB patient as one who has failed appropriate behavioral therapy of sufficient length and a trial of at least one antimuscarinic medication administered for 6–12 weeks. Failure may include lack of efficacy and/or inability to tolerate adverse drug effects. The panel notes the importance of combining behavioral and medication and trying alternative antimuscarinics.
A more real-world definition of patients with ROAB include those who:
· Do not respond to medication in spite of trying multiple agents
· Cannot tolerate medication
· Cannot afford mediation
· Have a contraindication to taking medication
· Do not want to take medication
Patients with ROAB are those who keep coming back cycling through various agents and are still symptomatic. They are those who do not respond to their 2 weeks of samples and who simply give up thinking they cannot be helped and unfortunately surrender to their condition. In addition, think about the millions of men and women with OAB symptoms who are only partially satisfied with their antimuscarinic agent, alpha blocker, or β-3 agonist and who would jump at the chance of achieving better symptom relief by adding a noninvasive treatment to their current medical therapy.
We Have a Problem
Unfortunately, we have a significant clinical problem when it comes to the treatment of OAB, and that problem is that we are dramatically under treating ROAB.
The mainstay of OAB therapy consists of non-pharmacologic and pharmacologic approaches, either as monotherapy or in combination. Non-pharmacologic treatment includes dietary and fluid modifications, behavioral therapy, and pelvic floor muscle rehabilitation. The antimuscarinics darifenacin, fesoterodine, oxybutynin, solifenacin, tolterodine, and trospium chloride are recommended as second-line therapy. Mirabegron, a newly approved β-3 agent, is an alternative for both the treatment naive and for those who have failed antimuscarinics.
Although published improvements in urgency incontinence episodes with behavioral therapy range from 50 to 80 %, these were achieved in highly motivated patients treated by experts using strict behavioral protocols. Such improvement in daily practice may be unrealistic and many offices do not offer non-pharmacological treatments. Many patients benefit from antimuscarinics but the data and clinical experience support a low adherence rate with them. Brostrom reported continuation rates with OAB agents of less than 50 % at 6 months, less than 25 % at 1 year, and less than 10 % at 2 years or longer.
The good news is that sacral nerve stimulation (InterStim®), percutaneous tibial nerve stimulation (Urgent®PC), and onabotulinumtoxinA (Botox®) are all FDA approved for treatment of refractory OAB. The bad news is that we are not liberally using them in our OAB population. For instance, there have been approximately 150,000 sacral nerve stimulators placed worldwide since 1997, with 25,000 implanted in the USA in 2012. In the same year, approximately 15,000 and 35,000 patients were treated with PTNS and onabotulinumtoxinA, respectively. Estimating 10,000 US Urologists and Urogynecologists, we are each treating on average 7 or 8 ROAB patients annually. Clearly we need to do better.
What Is Neuromodulation?
The International Neuromodulation Society defines therapeutic neuromodulation as “the alteration of nerve activity through the delivery of electrical stimulation or chemical agents to targeted sites of the body.” By altering neurotransmission processes and restoring neural balance, neuromodulation can have profound effects on pain relief, restoration of normal bowel and bladder control, tremor control, and many others.
Neuromodulation for the Masses
In spite of the effectiveness of sacral nerve stimulation, most urologists do not perform InterStim®, don’t refer to those who do, and it is perceived by some as too invasive or expensive, especially in elderly patients and in those who are frail or have a number of medical comorbidities. OnabotulinumtoxinA injected directly into the detrusor muscle endoscopically is an excellent neuromodulation treatment, but its use in clinical practice is somewhat limited because of the risk of retention associated with the treatment.
Clearly we need new and less invasive therapies. Percutaneous tibial nerve stimulation (Urgent®PC) offers patients an in-office noninvasive treatment alternative and I believe it is neuromodulation for the masses: the masses of physicians who are not currently liberally using neuromodulation therapies and the masses of untreated ROAB patients including the frail and elderly.
What Is PTNS and How Does It Work?
PTNS is a minimally invasive neuromodulation system designed to deliver retrograde electrical stimulation to the sacral nerve plexus through percutaneous electrical stimulation of the posterior tibial nerve. The posterior tibial nerve contains mixed sensory-motor nerve fibers that originate from L4 through S3 which modulate the innervation to the bladder, urinary sphincter, and pelvic floor. Although the specific mechanism of action of PTNS is unclear, it appears that stimulation of these nerves inhibits bladder activity by depolarizing somatic afferent fibers, which provides central inhibition of the preganglionic bladder motor neurons through a direct route in the sacral cord. It has been postulated that neuromodulation may also have a direct effect on the detrusor or a central effect on the micturition centers of the brain [7].
Using a battery-powered, handheld stimulator and a 34 gauge needle electrode, the tibial nerve is accessed and stimulated. Patients receive a 30-min weekly treatment in office for 12 weeks. Patients treated with PTNS may begin to see changes in their voiding parameters after 4–6 treatments. Following 12 weeks, treatment responders require additional therapy at individually defined treatment intervals for sustained relief of OAB symptoms.
Effectiveness of PTNS
The literature clearly supports the efficacy of Urgent®PC in the treatment of overactive bladder. The robustness of the PTNS data is supported by:
· Its consistency
· Its objective efficacy as measured by urodynamics
· Its comparison to medical therapy
· Its effectiveness compared to sham
· Its long-term effectiveness
What is noteworthy of the efficacy data is its consistency as measured by its effect on bladder diary variables. Govier et al. performed a well-designed, prospective, multicenter trial which assessed the efficacy and safety of PTNS for the treatment of refractory OAB. The primary endpoint was the change in mean daytime frequency from baseline to 12 weeks, typical for an OAB trial.
The treatment resulted in at least a 25 % reduction in daytime frequency in 55.2 % of patients, an overall 21 % reduction in nighttime voids, and a mean reduction of urge incontinence episodes of 35 %. Overall, 71 % of patients were classified as a treatment success based on acceptable criteria and were continued on long-term therapy.
In a prospective, 3-month, multicenter study, van Balken et al. evaluated the efficacy of PTNS for the treatment of OAB. Voiding diaries and general and disease-specific quality-of-life measures were utilized, and responders were defined as those patients who requested for long-term maintenance therapy after 12 weeks of treatment.
In all 33 OAB patients, the mean reduction in urinary frequency and nocturia was statistically significant and numerically similar to the results achieved with antimuscarinic agents in independent studies. In the patients defined as “responders,” the mean reduction in daytime and nocturnal voids was impressive: −4.8 and −1.4 voids, respectively. Similarly, the statistically significant reduction in the number of leaking episodes and pad usage experienced in the 30 patients with urge incontinence at baseline was impactful (−4.8 and −2.5, respectively, for all patients; −6.2 and −3.3, respectively, for responders).
Klinger et al. evaluated the benefit of PTNS as measured by its effect on objective urodynamic parameters. The mean maximum cystometric capacity increased from 197 to 252 mL and the mean volume at first unstable bladder contraction from 95 to 133 mL. In responders, the mean increase in total bladder capacity increased from 175 to 266 mL. Similarly, Vandoninck et al. evaluated 46 PTNS patients with urodynamics and demonstrated an increase in cystometric capacity. Notably, the increases in bladder capacity achieved with PTNS are greater than the approximate 20–30 mL increase customarily observed with antimuscarinics, further supporting the efficacy of neuromodulation.
The Overactive Bladder Innovative Therapy (OrBIT) Trial was a randomized, multicenter, controlled study that compared the effectiveness of PTNS to extended-release tolterodine in 100 patients with OAB [8]. The global response assessment demonstrated that subject assessment of OAB symptoms compared to baseline was statistically significant in the PTNS arm with 79.5 % reporting cure or improvement compared to 54.8 % of subjects on tolterodine (p < 0.01). After 12 weeks of therapy, objective measures improved similarly in both groups for reductions in urinary frequency, urge urinary incontinence episodes, urge severity, and nighttime voids, as well as for improvement in voided volume.
The Study of Urgent® PC vs. Sham Effectiveness in the Treatment of Overactive Bladder Symptoms (SUmiT) was a multicenter, double-blind, randomized, controlled trial comparing the efficacy of PTNS to sham through 12 weeks of therapy [9]. Two hundred twenty adults were randomized 1–12 weeks of treatment with weekly PTNS or sham therapy. The 13-week subject global response assessment for overall bladder symptoms demonstrated that PTNS subjects achieved statistically significant improvement in bladder symptoms with 54.5 % reporting moderately or markedly improved responses compared to 20.9 % of sham subjects from baseline (p < 0.001). Voiding diary parameters after 12 weeks of therapy showed PTNS subjects had statistically significant improvements in frequency, nighttime voids, voids with moderate to severe urgency, and urinary urge incontinence episodes compared to sham. The level I evidence provided by this pivotal study demonstrated that PTNS is a safe and effective therapy in treating OAB symptoms. In a second sham study, Finazzi Agro demonstrated a > 50 % reduction in urgency incontinence episodes in 71 % of the PTNS group vs. 0 % with placebo (p < 0.0001).
In the second phase of the OrBIT Trial, the investigators assessed the sustained therapeutic efficacy of PTNS in OAB subjects over 1 year [10]. After 12 weeks, subjects randomized to weekly PTNS therapy with Urgent® PC were offered an additional 9 months of treatment with assessments at 6 and 12 months from baseline. Thirty-three PTNS responders continued therapy with 32 and 25 subjects completing 6 and 12 months of treatment, respectively. Subjects received a mean of 12.1 treatments over an average of 263 days and a mean of 21 days between treatments. At 12 months, mean improvements from baseline included frequency 2.8 voids/day (p < 0.001), urge incontinence 1.6 episodes/day (p < 0.001), and nocturia 0.8 voids (p < 0.05). The durability of response demonstrated supported that PTNS is effective as a viable, long-term OAB therapy.
Peters et al. followed 50 participants from the SUmiT Trial who met the primary effectiveness endpoint after 12 weekly PTNS. The patients were prescribed a fixed-schedule 14-week tapering protocol followed by a personal treatment plan aimed at sustaining OAB symptom improvement. Of this group, 29 patients completed the 36-month protocol and received a median of 1.1 treatments per month. At 3 years, 77 % maintained moderate or marked improvement in OAB symptoms. Compared to baseline, median voids per day decreased from 12.0 to 8.7, and urge incontinence episodes per day decreased from 3.3 to 0.3. All quality-of-life parameters remained markedly improved from baseline through 3 years.
Special Populations
The efficacy of PTNS has been demonstrated in patients with neurogenic bladder dysfunction. Nine of 14 patients (multiple sclerosis, Parkinson’s, stroke, or spinal cord injury) showed a significant improvement of symptoms and of urodynamic parameters after PTNS. In Parkinson’s patients, Kabay demonstrated statistically significant improvements in the volume at the first involuntary detrusor contraction and maximum cystometric capacity. Similar benefits in urodynamic parameters were observed in multiple sclerosis patients treated with PTNS.
Kim found that 90 % of patients with painful bladder syndrome showed an improvement >25 % in the VAS score for pain, with 60 % reaching improvement >50 %. Van Balken demonstrated efficacy in 42 % of pelvic pain patients. Kabay evaluated the efficacy of PTNS vs. sham in a large cohort of patients with category IIIB chronic nonbacterial prostatitis. A complete response on pain and symptoms was observed after PTNS in 40 and 66.6 % of patients, respectively, vs. no response from sham.
The benefit of PTNS in the treatment of fecal incontinence has also been demonstrated and is FDA approved for bowel complaints in Europe. Fecal incontinence episodes decreased from 19.6 ± 21.0 at baseline to 9.9 ± 15.5 (p = 0.082) at 6 weeks and to 3.6 ± 4.8 (p = 0.029) at 1 year. Findlay reported a 100 % response in 13 female patients with fecal incontinence, and Mentes concluded that PTNS could be effective in treating the same in those with partial spinal cord injury.
In a single-center unpublished retrospective analysis, we evaluated the effectiveness of PTNS in 286 primarily elderly patients with OAB (median age 78.5 years, range 19–96 years). In addition to demonstrating improvements in voiding parameters, 76.2 % desired to continue maintenance therapy after 12 weeks of therapy.
Hoebeke evaluated the effectiveness of PTNS in children with non-neurogenic voiding dysfunction. Seventeen of 28 patients (60 %) with urgency and 16 of 23 (69 %) with urgency incontinence significantly benefitted from therapy. Seventeen percent of children became completely dry. De Gennaro found that PTNS is generally well accepted by children, with low scores measured by a visual analog pain score that further decreased during the therapy. The same author found that OAB children had symptom improvement in 80 % of cases and in 71 % with urinary retention. Durability of response with PTNS at 2 years has also been demonstrated.
Little data are available about prognostic factors for success or failure with PTNS. Studies on urodynamic changes by PTNS suggest that in cases of overactive bladder, without detrusor overactivity, patients are more prone to a successful treatment outcome. Clinical parameters for predicting SNS outcome were also tested in 132 patients treated with PTNS, but showed no significance.
A number of manuscripts have been published about the cost-effectiveness of neuromodulation therapies. In a comparative analysis, the initial and ongoing 2-year costs associated with PTNS and SNS were evaluated. During initial therapy, costs per patient were similar at $1773 for 12 weeks of PTNS treatment and $1857 for the SNS test. After 1 year, the costs were more than five times higher for SNS than PTNS ($13,746 vs. $2772) and more than three times higher at 2 years ($14,160 vs. $3850).
Clinical Relevance of PTNS Data
In addition to its robustness, the data supporting the efficacy of PTNS is clinically relevant for a number of reasons. In the majority of cases, the patients studied had failed prior medical and behavioral therapy. Not only does this potentially select for a more difficult-to-treat population, the patients evaluated are similar to the refractory patients we treat with PTNS in clinical practice. More importantly, I believe that most of the positive responders in the literature and in clinical practice may not have been offered onabotulinumtoxinA and sacral nerve stimulation or perhaps declined them due to their perceived invasiveness. If so, these PTNS responders would otherwise have been left untreated and living with their bothersome symptoms.
Maximizing Therapy with a Treatment Algorithm
In order to maximize efficacy and to help millions of OAB patients, we need to embrace and liberally offer all three neuromodulation therapies to them. In order to do so, I recommend the adoption of an OAB treatment algorithm in order to drive efficacy. Let me present to you mine (Fig. 3.7).
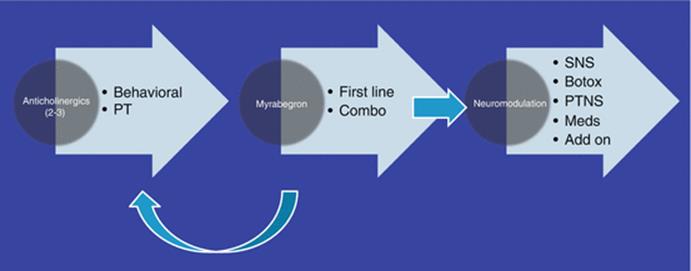
Fig. 3.7
Treatment algorithm for the treatment of overactive bladder
Most patients are initially treated with a combination of antimuscarinics and behavioral therapy. Nearly all patients are started on behavioral treatments including fluid modifications, bladder retraining, and pelvic floor rehabilitation. Many patients are offered to see our in-house physical therapy team who are experts in treating patients with voiding dysfunction.
When patients fail two (and perhaps three) antimuscarinics, they are offered mirabegron. We have had tremendous success treating ROAB with the β-3 agonist either as monotherapy or in combination with antimuscarinics. In milder patients, I tend to switch to mirabegron, and in the more severe partial responding population, I often add it to the antimuscarinic. The efficacy and tolerability of mirabegron has been such that I would not be surprised if it becomes first-line therapy for many urologists prior to starting antimuscarinics.
When medical and behavioral therapy does not reach the patient’s treatment goal, I offer nearly all ROAB patients neuromodulation. Each of the three therapies has its inherent advantages and disadvantages, and in my opinion, it should be the patient’s choice of which one best suits him, not mine. Utilizing a common sense approach, many elderly patients with multiple comorbidities are only offered PTNS.
Of course ROAB patients who have not exhausted all of the available antimuscarinics have the option of trying a third, fourth, or fifth medication, and in rare cases, this may be beneficial.
Patients declining neuromodulation and who have exhausted all pharmaceuticals unfortunately have reached the end of the treatment algorithm. Those who fail a neuromodulation therapy are usually reoffered the other two options, depending on patient factors. In addition, partial responders to neuromodulation may be restarted on an OAB agent that has helped minimally in the past to help augment their therapy.
The algorithm is simple and in my opinion helps maximize efficacy. The moment the pen or keypad orders an OAB agent, the patient is on a path to be offered neuromodulation unless they reach their treatment goal with conservative therapies. Patients are educated about the process with the help of an educational first-prescription treatment tool given to each and every one of them at the beginning of the treatment process. The booklet better educates them regarding their options and better equips them in making a more informed decision.
I find it equally rewarding to help patients with medication, behavioral therapy, neuromodulation, or with a combination of treatments. I strongly encourage my colleagues to adopt such an algorithm and no longer accept the mediocre care many of our patients are currently receiving.
Practical Tips for Success
Let me give you five practical tips that may help you be successful in establishing PTNS into your practice:
1.
2.
3.
4.
5.
Conclusions
PTNS offers patients with refractory OAB or “ROAB” a minimally invasive, office-based procedure that is safe, effective, and is an important addition to our therapeutic armamentarium. Utilizing a treatment algorithm and adopting a mindset of treating patients beyond medication is integral in driving excellence and maximizing therapeutic outcomes for our OAB patients.
Sacral Neuromodulation
Hendrikje M. K. van Breda12 and John P. F. A. Heesakkers12
(12)
Radboud University Medical Center, Nijmegen, The Netherlands
Hendrikje M. K. van Breda (Corresponding author)
Email: jetske.vanbreda@radboudumc.nl
John P. F. A. Heesakkers
Email: J.Heesakkers@uro.umcn.nl
Introduction
The International Continence Society (ICS) and the International Urogynecological Association (IUGA) have defined overactive bladder syndrome (OAB) as urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence in the absence of urinary tract infection or other obvious pathology. Symptoms of lower urinary tract dysfunction are often challenging to treat and have significant negative effect on the quality of life for many people; it lowers their self-esteem and health perception and are very expensive. An estimated 455 million people worldwide (10.7 %) experienced at least one OAB symptom in 2008, with the worldwide prevalence expected to increase to 500 million in 2013 and 546 million in 2018. In America, approximately 34 million inhabitants have OAB with associated costs exceeding $9 billion per year including direct care, health-related consequences, and lost productivity.
Conservative treatment, such as behavioral interventions and antimuscarinic drugs, does not always give sufficient improvement and/or is associated with considerable side effects such as dry mouth, blurred vision, constipation, erythema, fatigue, and urinary retention resulting in terminating the treatment in 70 % of the patients within 6 months. In the case of conservative treatment failure, sacral nerve stimulation (SNS) (InterStim®), percutaneous tibial nerve stimulation (PTNS) (Urgent®PC), and onabotulinumtoxinA (BTA) (Botox®) are all FDA approved for treatment of refractory OAB. The purpose of neuromodulation for the treatment of OAB is to target the innervation system that controls the pelvic floor organs and specifically the lower urinary tract. This chapter will discuss SNS treatment in clinical practice for OAB symptoms.
Historical Overview SNS Therapy
The history of electrical stimulation in the urology field started in 1864 when Budge observed detrusor contraction in animals when stimulating the sacral spinal cord. In 1954, Boyd suggested a technique for direct electrical stimulation of the bladder in humans. The first SNS implant in a urological patient was performed by Tanagho and Schmidt at the University of California in San Francisco in 1982. After that, in 1994, InterStim received the CE mark for treatment of chronic functional disorders of the pelvis, lower urinary tract, and intestinal tract in Europe. In 1997, InterStim received an FDA approval for treatment of urgency urinary incontinence in the USA which was expanded in 1999 for treatment of symptoms of urgency-frequency, and urinary retention. In 2002, the tined lead was introduced in SNS therapy to diminish the migration change of the lead.
Mode of Action
The precise mode of action of SNS is unknown. There are at least two presumed mechanisms of action: The first one claims that activation of efferent nerve fibers to the striated urethral sphincter reflexively causes detrusor relaxation. The second presumed mechanism states that activation of afferent nerve fibers causes inhibition of the voiding reflex at a spinal and/or supraspinal level; pudendal nerve afferents seem to be particularly important for the inhibitory effect on the voiding reflex. In OAB, the inhibition by SNS may, in part, modulate the sensory outflow from the bladder through the ascending pathways to the pontine micturition center, thereby preventing involuntary contractions by modulating the micturition reflex circuit but allowing voluntary voiding to occur.
The assumption that there is not only an effect at the spinal level but that there is also a central effect is strengthened by studies that are performed in complete spinal cord patients versus incomplete spinal cord patients. Schurch performed a PNE test in three complete SCI patients; after stimulation with a lead in the S3 foramen, a good anal reflex was seen, but after 5 days of stimulating, no improvement was observed in detrusor hyperreflexia. Hohenfellner concluded in his study of chronic sacral neuromodulation that only the patients with nearly complete or complete spinal lesions seem unlikely to benefit from this treatment.
Procedure
The procedure can be preceded by a preliminary test phase referred to as a percutaneous nerve evaluation (PNE). This test uses a non-anchored test lead placed in the S3 foramen and connected to an external stimulator. The test extends between 4 and 14 days, after which the lead is removed. Success of this test phase is generally defined as improvement of at least one major voiding symptom by more than 50 %. The overall response rate for PNE is around 55 %. Lead migration is considered the main factor of false-negative results. A true-negative response to PNE of about 20 % can be explained because the pudendal nerve afferents seem to be particularly important for the inhibitory effect on the voiding reflex, and those afferents are confined to the S2 level only in 18 % of all subjects.
SNS is nowadays often preformed in a two-stage technique with a quadripolar lead with self-anchoring tines, first described by Spinelli et al. in 2003. This technique enables test stimulation to be performed using the permanent anchored lead that reduces the risk of migration. This lead is usually placed through the back skin in the S3 foramen at sacral level (Fig. 3.8) and tunneled subcutaneously, where it is connected to a temporary extension lead that exits the body and is connected to an external test stimulator (Fig. 3.9). This test period mostly extends to 1–2 weeks. If the voiding symptoms improve by more than 50 %, the subject might undergo surgery to implant the internal pulse generator (IPG) (Fig. 3.10).
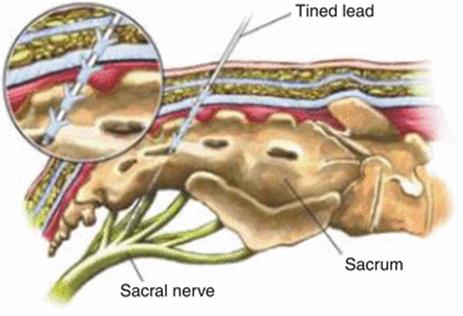
Fig. 3.8
Percutaneous insertion of the tined lead electrode through the sacral 3 (S3) foramen close the sacral nerve roots
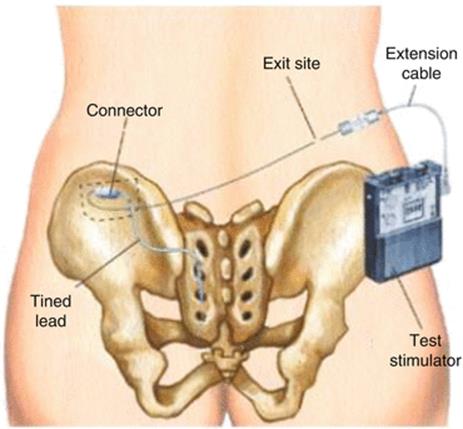
Fig. 3.9
Test situation of the first phase tined lead technique. The tined lead electrode has been inserted in the S3 foramen on the left side. It is connected to a test wire via an incision on the left lateral buttock side. The test wire is tunneled tot he right buttock side where it exits the skin and is connected to a external test stimulator
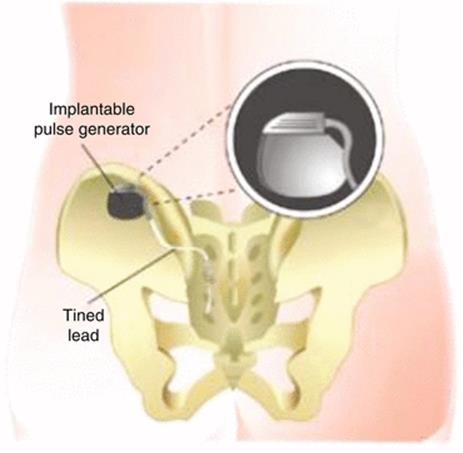
Fig. 3.10
The situation after the second phase tined lead technique. The tined lead electrode has been inserted in the S3 foramen on the left side. It is connected to the definite implanted pulse generator on the left lateral buttock side
Test Phase: PNE Versus First Stage Tined Lead
Leong et al. compared the patients’ response rate to PNE versus 1st stage tined lead procedure. Eighty-two patients, comprising 69 OAB patients, were screened with both PNE and with a 1st stage tined lead procedure. More than 50 % improvement in at least two relevant urinary symptoms was considered a positive response. The positive response rate on PNE was 47 %, while 1st stage tined lead showed a 69 % positive response rate. All patients with a positive 1st stage tined lead received SNS treatment. Failure rate after 2 years was 2.9 %. This study may suggest that 1st stage tined lead is a more sensitive screening rate method for SNS therapy.
Long-Term Effect
Long-term data about the success rates of SNS are scarce. Van Kerrebroeck et al. published a prospective multicenter trial evaluating the long-term safety and efficacy of SNS in patients with refractory urgency incontinence, urgency-frequency, and retention. A total of 17 centers worldwide enrolled 163 patients for test stimulation, of whom 152 patients received the implantation using InterStim. Voiding diaries were collected annually, and clinical success was defined as ≥50 % improvement from baseline in primary voiding diary variables. After 5 years, 105 patients completed the follow-up and of them 87 completed the voiding diary. Sixty-eight percent of patients with urgency incontinence, 56 % with urgency-frequency, and 71 % with retention had successful outcomes.
Groen et al. published the 5-year results of SNS in 60 women with refractory idiopathic urgency urinary incontinence. A non-tined lead procedure was used and success was defined as at least a 50 % decrease in the number of incontinence episodes or pads used daily. At 5 years, SNS was still used in 48 (80 %) patients of which 31 completed the voiding diaries. The success rate, patients with at least a 50 % decrease in the number of incontinence episodes or pads used daily, gradually decreased from 52 patients (87 %) at 1 month to 37 (62 %) at 5 years. Complete continence persisted in 15 % of patients.
As with all treatments, adverse events in SNS therapy are described, this mainly encompassed pain, infection, bowel dysfunction, and change in clinical effect often due to lead migration. Based on the summarized adverse event data, many wound complications and some changes in efficiency can be managed conservatively without repeated surgery. Published rates of infection range from 0 to 11 % in various studies. Rates of surgical revision and explanations vary in literature.
There is a decline in therapy effect of SNS over the years. If patients are successfully treated with SNS, the battery will eventually empty and a surgical intervention to change the battery will be necessary. The typical battery life of InterStim I is 8–10 years, depending on the parameters used for stimulation. The expected lifespan of InterStim II lies around 3–5 years when used 24 h/day. There is also a percentage of patients treated with SNS who, after a few years, don’t benefit from their therapy despite of adequate nerve stimulation. A suggested hypothesis for this phenomenon is habituation of the nerve system to prolonged stimulation of the sacral nerve.
If on-demand stimulation would be possible, it will lead to a longer battery life and it may decrease the risk of habituation of the nervous system. To answer this question, Oerlemans et al. completed a pilot intervention study for on-demand SNS in patients with urgency incontinence and urgency-frequency syndrome. Twenty-one patients participated of which 5 were randomized into a control group (continuous stimulation) and 16 into the on-demand group. The on-demand group was asked to switch the device off, and on again, when they felt recurrent symptoms. Of the study group, 10 patients reported no worsening of the symptoms by on-demand sacral nerve stimulation. The mean off-time was 12.4 h. The other six had worsening of their symptoms. Larger randomized studies with longer follow-up are needed, but this pilot study suggests that in the majority of patients, on-demand SNS treatment could be possible, implying less battery changes and therefore lower therapy costs. If on-demand stimulation reduces the decline in therapy effect over the years due to habituation of the nervous system, it could give better long-term results.
Costs
There are several publications about the cost-effectiveness of the treatment for OAB, in particular for PTNS and SNS. Various articles show different costs, since the medical care systems differ between countries. Leong et al. published that the cost of SNS as a treatment for OAB syndrome in the Netherlands is $15.743 over a 2-year time period. D’Ausilio et al. calculated the 10-year cost for SNS in Italy at €33.897. Staskin et al. compared the costs of PTNS to SNS for OAB syndrome in the USA. PTNS was the least costly with $7.565 for a three-year treatment, compared to SNS which costs $24.681 for the same treatment period. Martinson et al. also concluded that PTNS had substantially lower costs compared to SNS in the USA. Although it’s difficult to compare between countries, the treatment costs for SNS seem to be higher compared to PTNS treatment.
Practical Advice
· If you want to apply SNS therapy, you need a passionate and motivated team. Especially in the first year, you and your team will see the patients frequently for the implantation and to apply the best program for their therapy. It can be time consuming, but good and precise settings can lead to a very satisfying therapy.
· Before the implantation, inform your patient about the expectations, the procedure, the long-term results, and the possible complications.
· With strict selection criteria, you will have a high success rate. But in less clear cases, who have tried other therapies, you can give them the benefit of the doubt by performing a test phase with a PNE or a tined lead and still keep strict implantation criteria. Offering the patients a test phase will help to treat or, when it does not work, to accept their problem.
· Take your time with implanting the lead to confirm the right position. If you perform it under local anesthesia, ask for the sensory response. If you implant under general anesthesia, make sure no muscle relaxant is used, because the motor response is the only response you can rely on.
· Yes it’s expensive, but for patients the result can be priceless.
Further Reading
1.
Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Subcommittee of the International Continence Society. Neurourol Urodyn. 2002;21:167–78.CrossRefPubMed
2.
Nice. National Institute for Health and Care Excellence. The management of urinary incontinence in women. Issued September 2013. Nice Clinical Guideline 171.
3.
Lucas MG, Betredinova D, Bosch JRHL, Burkhard F, Cruz F, Nambiar H, Nilsson CG, de Ridder DJMK, Tubaro A, Pickard RS. Guidelines on urinary incontinence. 2014. Accessed from: www.uroweb.org/wp-content/uploads/EAU-Guidelines-Urinary-Incontinence-2015.pdf.
4.
Timbrook Brown E, Krlin RM, Winters C. Urodynamics: examining the current role of UDS testing. What is the role of urodynamic testing in light of recent AUA urodynamics and overactive bladder guidelines and the VALUE study? Curr Urol Rep. 2013;14:403–8.CrossRef
5.
Mangera A, Apostolidis A, Andersson KE, Dasgupta P, Giannantoni A, Roehrborn C, et al. An updated systematic review and statistical comparison of standardised mean outcomes for the use of botulinum toxin in the management of lower urinary tract disorders. Eur Urol. 2014;65:981–90.CrossRefPubMed
6.
M.G. Lucas (chair), D. Bedretdinova, et al. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188:2455–63.
7.
Finazzi-Agrò E, Rocchi C, Pachatz C, Petta F, Spera E, Mori F, Sciobica F, Marfia GA. Percutaneous tibial nerve stimulation produces effects on brain activity: study on the modifications of the long latency somatosensory evoked potentials. Neurourol Urodyn. 2009;28(4):320–4.CrossRefPubMed
8.
Peters KM, MacDiarmid SA, Wooldridge LS, et al. Randomized trial of percutaneous tibial nerve stimulation versus extended release tolterodine: results from the Overactive Bladder Innovative Therapy (OrBIT) Trial. J Urol. 2009;182:1044–61.CrossRef
9.
Peters KM, Carrico DJ, Perez-Marrero RA, et al. Randomized trial of percutaneous tibial nerve stimulation versus sham efficacy in the treatment of overactive bladder syndrome: results from the SUmiT Trial. J Urol. 2010;183:1438–43.CrossRefPubMed
10.
MacDiarmid SA, Peters KKM, Shobeiri A, et al. Long-term durability of percutaneous tibial nerve stimulation for the treatment of overactive bladder. J Urol. 2010;183:234–40.CrossRefPubMed
11.
Spinelli M, et al. New percutaneous technique of sacral nerve stimulation has high initial success rate: Preliminary results. Eur Urol. 2003;43(1):70–4.CrossRefPubMed
12.
van Kerrebroeck PEV, et al. Results of sacral neuromodulation therapy for urinary voiding dysfunction: outcomes of a prospective, worldwide clinical study. J Urol. 2007;178(5):2029–34.CrossRefPubMed
13.
Groen J, Blok BFM, Bosch JLHR. Sacral neuromodulation as treatment for refractory idiopathic urge urinary incontinence: 5-year results of a longitudinal study in 60 women. J Urol. 2011;186(3):954–9.CrossRefPubMed
Suggested Readings for Conservative Treatment
Andersson KE, Chapple CR, Cardozo L, et al. Pharmacological treatment of overactive bladder: Report from the International Consultation on Incontinence. Curr Opin Urol. 2009;19:380.CrossRefPubMed
Chapple CR, Khullar V, Gabriel Z, et al. The effects of antimuscarinic treatments in overactive bladder: an update of a systematic review and meta-analysis. Eur Urol. 2008;54:543.CrossRefPubMed
Gormley EA, Lightner DJ, Burgio KL, et al. Diagnosis and treatment of overactive bladder (Non neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188:2455.CrossRefPubMed
Lucas MG, Bosch RJL, Burkhard FC, et al. EAU guidelines on assessment and nonsurgical management of urinary incontinence. Eur Urol. 2012;62:1130.CrossRefPubMed
Lucas MG, Bosch RJL, Burkhard FC, et al. EAU guidelines on surgical management of urinary incontinence. Eur Urol. 2012;62:1118.CrossRefPubMed