Tommaso Cai1 and Truls E. Bjerklund Johansen2, 3
(1)
Department of Urology, Santa Chiara Regional Hospital, Trento, Italy
(2)
Oslo University Hospital, Oslo, Norway
(3)
Institute of Clinical Medicine, University of Aarhus, Aarhus, Denmark
Truls E. Bjerklund Johansen
Email: tebj@medisin.uio.no
Introduction
Humans have a huge number of microorganisms in the intestine and on the body surface. Most of the time, we live in friendly commensalism with them, but sometimes infections break out. This can be due to own microorganisms crossing physiological barriers as in honeymoon cystitis or the intrusion of foreign microorganisms as in venereal diseases.
A common feature in UTI is that the host reaction is the cause of the symptoms, as when urothelial cells are being shed from the bladder mucosa in acute cystitis or when immunological reactions counteract each other in the physiological chaos of urosepsis. The development of systemic inflammatory response syndrome into sepsis, severe sepsis, and septic shock is accompanied by a death risk of 20–40 % (Fig. 8.1, Table 8.1). While antibiotic treatment remains the cornerstone of treatment, modification of the host reaction has become an important aspect in acute cystitis as well as in urosepsis.

Fig. 8.1
Abdominal skin of a woman in uroseptic shock showing areas of bleeding and ceased microcirculation as a sign of physiological chaos with counteracting host reactions (Courtesy of F. Wagenlehner)
Table 8.1
Clinical presentation of cystitis (CY), pyelonephritis (PN), and urosepsis (US) and grading of severity
|
Clinical diagnosis |
Acronym |
Clinical symptoms |
Grade of severity |
|
Cystitis |
CY-1 |
Dysuria, frequency, urgency, suprapubic pain; sometimes unspecific symptoms (see Table 8.1) |
1 |
|
Mild and moderate pyelonephritis |
PN-2 |
Fever, flank pain, CV tenderness; sometimes unspecific symptoms (see Table 8.1) with or without symptoms of CY |
2 |
|
Severe pyelonephritis |
PN-3 |
As PN-2, but in addition nausea and vomiting with or without symptoms of CY |
3 |
|
Urosepsis (simple) |
US-4 |
Temperature >38 °C or <36 °C Heart rate >90 beats min Respiratory rate >20 breaths/min or PaCO2 <32 mmHg (<4.3 kPa) WBC > 12,000 cells/mm3 or <4000 cells/mm3 or ≥10 % immature (band) forms With or without symptoms of CY or PN |
4 |
|
Severe urosepsis |
US-5 |
As US-4, but in addition associated with organ dysfunction, hypoperfusion, or hypotension Hypoperfusion and perfusion abnormalities may include but are not limited to lactic acidosis, oliguria, or an acute alteration of mental status |
5 |
|
Uroseptic shock |
US-6 |
AS US-4 or US-5, but in addition with hypotension despite adequate fluid resuscitation along with the presence of perfusion abnormalities that may include, but are not limited to lactic acidosis, oliguria, or an acute alteration in mental status. Patients who are on inotropic or vasopressor agents may not be hypotensive at the time that perfusion abnormalities are measured |
6 |
Diagnostics
Patient evaluation is based on a careful history, physical examination, and urine analysis.
The diagnosis of a urinary tract infection is based on clinical signs and the demonstration of microorganisms considered to be causing the infection. In most cases, the diagnosis is easy like in acute cystitis in a young woman, but in spinal cord injured patients with urinary catheters, the symptoms are totally different and urine findings are difficult to interpret.
Patient assessment always starts with an evaluation of severity. The clinical presentation forms correspond to grades of severity ranging from cystitis to urosepsis (Table 8.1). Severity is modified by risk factors, which can be described by means of phenotyping in a system called ORENUC (Table 8.2). Each letter in the word ORENUC refers to special patient features such as R – recurrent UTI without other known risk factor, E – extraurogenital risk factors like pregnancy, and C – presence of a catheter. According to a new classification presented by European Section for Infection in Urology (ESIU), a full section of the European Association of Urology (EAU), severity grades, and phenotyping is intended to replace the old classification into uncomplicated and complicated UTI.
Table 8.2
Host risk factors in urinary tract infections categorized according to the ORENUC system
|
Category of risk factor |
Examples of risk factors |
Phenotype |
|
NO known risk factor |
Otherwise healthy premenopausal women |
O |
|
Risk factors for Recurrent UTI, but no risk of more severe outcome |
Sexual behavior (frequency, spermicide) Hormonal deficiency in postmenopause Secretor type of certain blood groups Well-controlled diabetes mellitus |
R |
|
Extra-urogenital risk factors with risk of more severe outcome |
Prematurity, newborn Pregnancy Male gender Badly controlled diabetes mellitus Relevant immunosuppression (not well defined) |
E |
|
Nephropathic diseases with risk of more severe outcome |
Relevant renal insufficiency (not well defined) Polycystic nephropathy Interstitial nephritis, e.g., due to analgesics |
N |
|
Urological risk factors with risk of more severe outcome, which can be resolved during therapy |
Ureteral obstruction due to a ureteral stone Well-controlled neurogenic bladder disturbances Transient short-term external urinary catheter Asymptomatic bacteriuria |
U |
|
Permanent urinary Catheter and nonresolvable urological risk factors with risk of more severe outcome |
Long-term external urinary catheter Nonresolvable urinary obstruction Badly controlled neurogenic bladder disturbances |
C |
Symptoms
Urinary tract infections (UTIs) often affect young people and are an important cause of reduced quality of life due to disabling symptoms. In most cases, UTIs are accompanied by typical signs and symptoms, but asymptomatic infections also occur. For male genital infections, the clinical symptoms are not related to the severity of the infection. Asymptomatic chronic bacterial prostatitis due to Chlamydia trachomatis infection may cause severe complications such as decreased fertility. Generally, the symptoms of UTI depend on which part of the urinary tract that is affected. Doctors should learn the symptom language of each infection site.
· Kidneys (i.e., acute pyelonephritis): upper back and flank pain; fever (not always); shaking and chills, nausea and/or vomiting (not always).
· Bladder and prostate (i.e., cystitis and prostatitis): lower abdominal discomfort; a strong, persistent urge to urinate; a burning sensation when urinating; pelvic pressure; blood in urine (cystitis); rectal or perineal pain (prostatitis); sexual dysfunction (erectile dysfunction/premature ejaculation in case of prostatitis); in women symptoms of UTI occur without vaginal discharge or irritation.
· Urethra (urethritis): Burning with urination and/or urethral discharge.
The most common clinical presentations of UTIs are outlined below.
Acute Uncomplicated Cystitis
The typical patient with acute (uncomplicated) cystitis is a female of reproductive age who presents with irritative urinary symptoms. The diagnosis can be made with a high probability based on a focused history of dysuria, frequency, and urgency in the absence of vaginal discharge or irritation. No risk factors for complicated urinary tract infections should be present.
The most common symptoms are dysuria, frequent voiding of small volumes, and urgency, sometimes hematuria and less often suprapubic discomfort or pain. In case of recurrent cystitis without other risk factors, the young patients may show psychological symptoms such as depression with a subsequent reduction in quality of life. Recently, a simple standardized self-reporting questionnaire for acute uncomplicated cystitis was presented. This is an 18-item self-reporting questionnaire, named Acute Cystitis Symptom Score (ACSS), including (a) six questions about “typical” symptoms of acute uncomplicated cystitis, (b) four questions regarding differential diagnoses, (c) three questions on quality of life, and (d) five questions on additional conditions which may affect therapy. The questionnaire has been validated and can be recommended for clinical studies and for initial diagnosis and monitoring of treatment of acute uncomplicated cystitis. Also, Clayson et al. developed a 14-item UTI Symptoms Assessment questionnaire (UTISA), to measure the severity and bothersomeness of the most frequently reported symptoms and signs of uncomplicated urinary tract infections [5]. This instrument comprises three four-item domains (urination regularity, problems with urination, and pain associated with UTI) with two additional items measuring hematuria. This questionnaire has demonstrated excellent psychometric properties and good accuracy in evaluation of severity and bothersomeness of UTI symptoms. The authors highlighted the importance of specific symptoms of UTIs that can decrease the patient’s quality of life: frequency and urgency, pain or burning on urination, feeling of incomplete emptying, pain/pressure in lower abdomen, and low back pain.
Key Message
The combination of newly onset frequency and dysuria, in the absence of vaginal discharge, is diagnostic for an acute uncomplicated cystitis.
Acute Uncomplicated Pyelonephritis
The presence of flank or back pain in an otherwise healthy patient with lower urinary tract symptoms (dysuria, frequency, urgency, hematuria, suprapubic pain) is highly indicative of acute uncomplicated pyelonephritis. Moreover, these patients can sometimes present with systemic symptoms, such as nausea, vomiting, fever, chills, and abdominal pain. The history of recurrent lower urinary tract infections or a recent episode of acute uncomplicated cystitis is a risk factor of acute pyelonephritis. Other risk factors like diabetes, ureteral reflux, and incontinence may support the diagnosis of acute pyelonephritis in the presence of typical symptoms.
Key Message
The combination of newly onset frequency and dysuria, with flank or back pain, with or without systemic symptoms, in an otherwise healthy patient, is highly indicative of an acute uncomplicated pyelonephritis.

Fig. 8.2
Pain at costovertebral angle
Risk Factors, Phenotyping, and Complicated UTIs
A urinary tract infection in a patient with a structural or functional abnormality of the genitourinary tract was used to be called a complicated urinary tract infection, and the clinical presentations varied across a wide spectrum, ranging from mild lower tract irritative symptoms, such as frequency and urgency, to severe systemic manifestations, such as bacteremia and sepsis. In particular, complete urinary obstruction or trauma to the genitourinary tract with hematuria is associated with more severe clinical presentation forms. This complex picture was a main argument for the introduction of severity grades and phenotyping of risk factors thus enabling a more differentiated description of the patient and the symptoms than just a “complicated” urinary tract infection.
An important feature of the new ESIU/EAU classification is the status of asymptomatic bacteriuria which is regarded as a risk factor, not a specific type of UTI. The prevalence of asymptomatic bacteriuria is very high (almost 100 %) in patients with chronic indwelling catheters and 30–40 % in patients with a neurogenic bladder managed by intermittent catheterization.
Key Messages
· Patients with indwelling urological devices and systemic symptoms, such as fever should be suspected of having UTI even in the absence of local genitourinary signs and symptoms.
· Bacteriuria in patients with indwelling urological devices should only be treated if clinical symptoms of UTI are present.
Acute and Chronic Bacterial Prostatitis
Even if bacterial prostatitis represents a small portion of UTIs (about 10–15 %) of all urological visits in outpatient clinical setting, the impact on patient’s quality of life is considerable. Patients with acute bacterial prostatitis present with typical signs and symptoms of an acute urinary tract infection including irritative and/or obstructive voiding complaints and often have additional symptoms of systemic infections like malaise, nausea, vomiting, chills, and fever and sometimes present with signs of urosepsis. They also complain of perineal and suprapubic pain, associated with pain or discomfort of the external genitalia. Chronic bacterial prostatitis represents the most frequent cause of recurrent urinary tract infections in young and middle-aged men. The initial clinical presentation of chronic prostatitis is similar to acute prostatitis, but chronic bacterial prostatitis can be a devastating disease, characterized by relapsing febrile episodes, if not treated adequately from the beginning.
Key Message
The new-onset irritative and/or obstructive voiding symptoms associated with perineal or testicular discomfort/pain are highly indicative of acute bacterial prostatitis. Moreover, all perineal discomfort/pain with or without typical signs and symptoms of urinary tract infection should be investigated in order to exclude a bacterial prostatitis due to possible future complications.
Physical Examination
The patient history and urinalysis are the most important tools for diagnosing UTIs. Even if there are no key diagnostic signs, a focused physical examination is still an important part of the diagnostic work-up. However, sometimes the physical examination is diagnostic as in cases of acute bacterial prostatitis and acute pyelonephritis.
|
Clinical condition |
Local signs |
Possible signs |
Systemic signs |
|
Acute uncomplicated cystitis |
None |
Suprapubic tenderness |
Rarely |
|
Acute uncomplicated pyelonephritis |
Costovertebral angle tenderness |
Discomfort during kidney palpation |
Commonly (fever, tachycardia) |
|
Acute bacterial prostatitis |
Painful, swollen prostate |
Perineal pain/discomfort Perineal pain and anal sphincter spasm |
Usually (fever, nausea) |
|
UTIs in patients with risk factors (complicated UTI) |
Depending on risk factor |
Not specific |
Commonly (fever, tachycardia) |
|
Prostatic abscess |
Fluctuation during prostate palpation |
Perineal pain/discomfort |
Rarely |
|
Chronic bacterial prostatitis |
Painful prostate |
Perineal pain/discomfort |
Rarely |
The aim of the physical examination is to assess the severity of the infection and to look for risk factors. In septic patients, one should always look for focal manifestations of circulatory failure (Fig. 8.3). In adults, the physical examination should be done in order to specifically evaluate:

Fig. 8.3
Necrotic fingers in a young woman in septic shock due to delayed diagnosis of pyonephrosis resulting from an obstructing ureteral stone
· Abdomen
· Previous surgical scars
· Abdominal meteorism and signs of ileus
· Costovertebral tenderness (pain elicited by blunt striking of the back, flanks, and the angle formed by the 12th rib and lumbar spine – with a fist) (Fig. 8.4)

Fig. 8.4
Localization of the costovertebral angle
· Palpable renal mass
· Dullness to percussion in the lower abdomen (bladder distension)
· In women, genitals should be evaluated for:
· Vaginal discharges
· Vaginal mucosa
· Urethral secretion
· Signs of infection of subcutaneous tissue (Fournier gangrene)
· In men, the physical examination of genitals should evaluate:
· Urethral secretion (the penis should be examined by retracting the foreskin)
· Testicular tenderness
· Painful/swollen prostate
· Perineal pain and anal sphincter spasm
· Signs of infection of subcutaneous tissue (Fournier gangrene)
· In children, one should also evaluate the external urethral orifice.
In case of risk factors, the physical examination should be focused on genitourinary signs and symptoms related to each risk factor. In case of patients with indwelling catheter, the physical examination should always include systemic signs such as fever, nausea, and tachycardia (systemic inflammatory response syndrome).
Lab and Culture
Even if the diagnosis of UTIs can be made with a high probability based on history and physical examination, the verification of causative microorganism is the definitive diagnostic measure in UTIs. The microbiological evaluation is mandatory in cases of recurrent UTIs, in the presence of risk factors, and in patients with unusual signs and symptoms. As a rule, a urine sample should always be taken for culture before initiation of empiric antibiotic treatment. A good microbiological evaluation of a patient with UTIs requires that the urine specimen is appropriately collected, that a uropathogen is isolated, and the number of organisms is counted.
Sampling Technique
There is general consensus about the importance of using a midstream, clean-caught urine specimen to diagnose UTIs, although some authors found no significant difference in numbers of contaminated or unreliable results between specimens collected with and without preparatory cleansing.
Dipstick
In most cases of UTIs without risk factors, the urine dipstick testing has the same sensitivity and specificity, convenience, and cost-effectiveness, as conventional urinalysis and urine microscopy. This is due to the fact that nitrites and leukocyte esterase are the most accurate indicators of UTIs in symptomatic patients. A diagnosis of UTI can be safely made in patients with typical symptoms who are found to have a positive urine dipstick test or urinalysis, without obtaining a urine culture.
It is important to know that nitrite tests may be negative if the UTI is caused by a non-nitrate-producing pathogen such as Enterococci, S. saprophyticus, and Acinetobacter and if the urine is too dilute. Pyuria is frequently present in patients with lower urinary tract infection and always in those with acute pyelonephritis. However, the absence of pyuria does not exclude a urinary tract infection in patients with typical symptoms. Nitrites and leukocyte esterase may be negative in urine of patients with bacterial prostatitis.
Urine Culture
Urine cultures are highly recommended for:
· Patients with suspected acute pyelonephritis
· Patients with symptoms that do not resolve or that recur within 2–4 weeks after therapy
· Patients with atypical symptoms
· Patients with suspected bacterial prostatitis
· Patients with suspected UTIs and systemic signs or symptoms
In the emergency room setting, it is recommended to take a urine sample for microbiological evaluation before starting empirical treatment in patients thought to be at high risk of pyelonephritis or bacteremia/urosepsis, as well as in those expected to have uncommon or resistant organisms. Routine posttreatment urinalysis or urine cultures in asymptomatic patients are not necessary.
Each clinical presentation requires specific sampling and microbiological assessment in order to isolate the bacterial strains and localize the infection site.
|
Clinical condition |
Sample to obtain |
Additional samples |
|
Acute uncomplicated cystitis |
Midstream urine |
None |
|
Acute uncomplicated pyelonephritis |
Midstream urine |
Blood |
|
UTIs with risk factors (complicated) |
Midstream urine |
Blood |
|
Acute bacterial prostatitis |
Midstream urine |
Blood In mild cases, only samples may be obtained by using Meares-Stamey test |
|
Prostatic abscess |
Samples obtained by using Meares-Stamey test |
Blood/secretion from surgical drainage |
|
Chronic bacterial prostatitis |
Samples obtained by using Meares-Stamey test |
None |
Acute Uncomplicated Cystitis
A colony count of greater than or equal to 103 CFU/ml of a uropathogen in midstream urine is now the commonly accepted microbiologic diagnostic criterium for acute uncomplicated cystitis. Some authors argue that more than 102colony-forming units per mL in women with typical symptoms of UTI represent a positive culture. Moreover, it is well known that about 10–20 % of symptomatic women have negative urine cultures, and yet the clinical response to treatment of these women is similar to women with positive urine cultures. An important aspect to consider is the potential role of contamination of voided specimens by periurethral microorganisms in premenopausal women. In fact, when cultures do not yield Escherichia coli in women with symptoms of cystitis, the usefulness of midstream urine cultures is debatable. In such cases, the presence of Escherichia coli in midstream urine was highly predictive of bladder bacteriuria even at very low counts, but Enterococci and group B streptococci were not predictive of bladder bacteriuria at any colony count. This aspect is important to take into account in order to avoid unnecessary antibiotic therapy.
Key Messages
· In women with typical symptoms of a UTIs, a count >102 colony-forming units per mL represent a positive culture.
· In women with symptoms suggestive of cystitis, a midstream portion of voided urine is recommended, but if Enterococci and group B streptococci are detected, more attention is due.
Acute Uncomplicated Pyelonephritis
Up to 95 % of episodes of acute uncomplicated pyelonephritis are associated with greater than or equal to 105 CFU/mL of a uropathogen. However, due to the fact that some patients with acute uncomplicated pyelonephritis have lower colony counts, it is proposed that a quantitative count of greater than or equal to 104 CFU/mL may be considered as significant bacteriuria. Although blood cultures are commonly performed for patients with febrile UTIs, the role of blood culture in diagnosis of acute uncomplicated pyelonephritis is controversial. Many authors argue that blood cultures do not provide useful information for the clinical management of acute uncomplicated pyelonephritis, and it may therefore not be routinely performed.
Key Messages
In acute uncomplicated pyelonephritis, blood culture may not be routinely performed.
UTI with Risk Factors (Complicated UTI)
Significant bacteriuria in a UTI with risk factors is defined by counts of ≥105 cfu/mL and ≥104 cfu/mL, in the midstream urine of women and men, respectively. If a straight catheter urine sample is taken, ≥104cfu/mL can be considered relevant. The requirement for pyuria is ≥10 white blood cells per high-power field (×400) in the resuspended sediment of a centrifuged aliquot of urine or per mm3 in unspun urine. A dipstick method can also be used for routine assessment, including a leukocyte esterase test, hemoglobin, and a nitrite reaction.
Pyuria only as identified by urinalysis or a positive leukocyte esterase dipstick test is consistent with, but not diagnostic of, urinary tract infection. On the contrary, a urinalysis without pyuria may reliably exclude symptomatic urinary infection due to the high negative predictive value of pyuria. In all cases, however, a urine specimen for culture should be obtained before the initiation of antimicrobial therapy in order to identify the infective organism and its susceptibilities. Moreover, a count of at least 105 cfu/mL is required for microbiological verification of symptomatic UTI in patients on intermittent catheterization or with indwelling catheters.
Key Messages
A urine specimen should be obtained for culture and susceptibility testing before starting antimicrobial therapy in every episode of urinary tract infection in patients with risk factors.
Bacterial Prostatitis
Acute Bacterial Prostatitis
The Meares-Stamey test is not recommended during the early phase of acute bacterial prostatitis. Hence, midstream urine culture is the only laboratory evaluation of the lower urinary tract and usually shows typical uropathogens. Blood cultures should also be taken in case of SIRS. Even if elevated levels of PSA have been described in more than 70 % of men with acute bacterial prostatitis as a consequence of increased vascular permeability and disrupted epithelium of the gland, the role of PSA in the differential diagnosis and evaluation of acute bacterial prostatitis is not clear.
Chronic Bacterial Prostatitis
The Meares-Stamey four-glass test is, however, the standard method of assessing inflammation and the presence of bacteria in the lower urinary tract in men presenting with prostatitis symptoms. This test not only gives information about bacterial strains and susceptibility but also gives useful data to plan treatment in abacterial prostatitis. Moreover, the Meares-Stamey test is the best way to localize the infection site. Unfortunately, very few urologists use the four-glass test in routine diagnostic work-up. Therefore, the two-glass pre- and post-prostatic massage test is a valuable alternative to the preferred Meares-Stamey test for diagnosing prostatitis. Other laboratory testing is not routinely needed.
The four-glass test comprises the following samples: first-void early morning urine (VB1), midstream urine (VB2), expressed prostatic secretion (EPS), and post-prostatic massage urine (VB3) (Fig. 8.5). The four-glass test can be considered positive for bacterial prostatitis if the bacterial load in EPS or in VB3 was at least 1000 CFU/mL and at least ten times higher than in VB1 and VB2.

Fig. 8.5
The Meares-Stamey test
Key Messages
· Midstream urine culture is considered the only laboratory evaluation needed in patients with suspected acute bacterial prostatitis.
· However, in the case of SIRS, blood cultures should also be taken.
· The two-glass pre- and post-prostatic massage test is a reasonable alternative to the preferred Meares-Stamey four-glass test for diagnosing chronic bacterial prostatitis.
Imaging
The role of imaging in UTI is to detect conditions that need to be resolved immediately (i.e., pyonephrosis), to detect conditions that can be corrected to prevent recurrences (i.e., obstruction and reflux), and to diagnose pyelonephritis in children.
Urinary Tract Infections
Imaging is rarely indicated in diagnosis and management of UTIs in adult women. Imaging should be considered in acute cystitis and acute pyelonephritis when urological and nephrological risk factors are suspected, in patients who present with atypical symptoms, and in those who do not respond to appropriate antimicrobial therapy. Imaging may reveal a renal abscess or an obstructing stone.
The preferred modalities in adults are ultrasonography of the urinary tract and/or computed tomography (CT) scan with contrast. CT may also be used to diagnose subcutaneous gas-producing infections as Fournier gangrene. Bladder ultrasonography is helpful in diagnosing post-void residual volumes. Plain kidney ureter and bladder films still have a role in patients with a history of stones in the urinary tract and in the case of emphysematous pyelonephritis (Fig. 8.6). Renography should be considered if kidney function needs to be assessed. In patients with a history of considerable radiation exposure, MRI of kidneys may replace CT.

Fig. 8.6
CT scan of a severe case of emphysematous ureteropyelonephritis due to a left-out ureteral stent
Bacterial Prostatitis
Imaging is indicated in patients with suspected prostatic abscess and in those who fail to respond to appropriate antimicrobial therapy. The preferred modalities are transrectal ultrasound (TRUS) or MRI of the prostate. The main aim is to rule out and sometimes drain a prostatic abscess. In patients who do not respond to appropriate antibiotic therapy and in particular in immunocompromised patients, TRUS should not be postponed for >48 h. CT scans and magnetic resonance imaging offer no advantage over TRUS, unless the abscess has penetrated, the confines of the prostate gland or further abscess foci are suspected. TRUS can be performed in patients with acute bacterial prostatitis without increasing the risk for urosepsis. In patients with chronic bacterial prostatitis, TRUS should be performed also in order to evaluate concomitant prostatitic disease such as BPH or seminal vesciculitis. The role of prostatic calcifications in diagnosis and management of bacterial prostatitis is controversial. There is currently no indication to evaluate the presence, location, size, and number of prostate calcifications in patients with chronic bacterial prostatitis unless in extreme cases.
Treatment
Daily Life Changes as Prophylaxis
UTIs have a high prevalence in the population, especially among otherwise healthy young sexually active women and may cause considerable morbidity and expenses. Preventive measures of these events are therefore desirable. Having a mother with a history of UTI and having had UTI during childhood put patients at greater risk of developing UTI themselves in adult age.
Among all UTIs, acute cystitis is that more suitable to respond to daily life changes. Recommendations on preventive measures have to be based on the most common behavioral risk factors associated with UTIs. In young women, most behavioral risk factors for cystitis and pyelonephritis are related to sexual activity. These are:
· Frequency of sexual intercourse
· Spermicide use
· Getting a new sexual partner
· Having more than one sexual partner
While sexual intercourse is a strong predictor of UTI in young women, studies in postmenopausal women have yielded inconsistent and weak relations. Moore et al., recently, showed absent or only moderately increased risks of UTI related to sexual intercourse in postmenopausal women.
Other behaviors that are thought to increase the risk of UTI are reduced fluid intake, habitually delaying urination, delaying postcoital urination, wiping from back to front after defecation, douching, and wearing occlusive underwear. Some factors are associated with higher risk of getting a second UTI: hormonal status, bowel function, number of previous UTI episodes, and type of pathogens isolated.
Even if there are no good studies that have evaluated the impact of behavioral changes in the prevention UTIs, some strategies are highly recommended:
· Reducing use of spermicides
· Reducing sexual activity
· Early postcoital micturition
· Increasing daily fluid intake
· Regularize the bowel function
Due to the low cost of daily life changes, all women with a second episode of UTI should be counseled about the pathogenesis of UTIs and how behavior modification might be useful in reducing one’s risk.
Key Messages
· All safe strategies to prevent recurrent UTIs that avoid the use of antimicrobials are highly desirable.
· Education and counseling with the aim of behavioral modification should be offered to all women with more than one episode of UTI.
Antimicrobial Treatment of Urinary Tract Infections
In everyday clinical practice, a diagnosis is frequently made and treatment is started without the results of urine culture. However, even if empirical therapy is still recommended, it is very important to take into account that in geographic areas or in age groups where antimicrobial resistance is high, preemptive culturing (a culture secured prior to instituting empirical therapy) is recommended.
Empirically based treatment means that we know beforehand which pathogen is the most likely in the current situation and what is the most probable susceptibility.
The aim of antimicrobial treatment is to relieve the patients’ symptoms, eradicate the pathogen, and preserve kidney function. However, systemic antibiotic treatment affects all microorganisms in our body. Unfortunately, some bacteria are able to develop resistance to antibiotics and to reattack the host.
Recommendations on which antibiotic to use are based on general pathogen susceptibility, pharmacokinetic and pharmacodynamics properties of the antibiotic, and evidence form clinical studies. Pharmacokinetics refers to concentrations of antibiotics being obtained in the serum, and pharmacodynamics refers to the concentrations being obtained at the site of infection. The aim is to achieve a certain concentration for a certain length of time. Too low dosing will facilitate development of resistance. Recommendations on empirical antimicrobial treatment are shown in Table 8.3.
Table 8.3
Recommended antimicrobial therapy in acute urinary tract infections without risk factors (uncomplicated)
|
Antibiotics |
Daily dose |
Duration |
Comments |
|
Severity grade 1 (acute cystitis) |
|||
|
First choice |
|||
|
Fosfomycin trometamol |
3 g single dose |
1 day |
|
|
Nitrofurantoin macrocrystal |
100 mg bid |
5 (−7) days |
Avoid in G6PD deficiency |
|
Pivmecillinam |
400 mg tid |
3 days |
|
|
Alternatives |
|||
|
Ciprofloxacin |
250 mg bid |
3 days |
Not during pregnancy |
|
Levofloxacin |
250 mg qd |
3 days |
Not during pregnancy |
|
Norfloxacin |
400 mg bid |
3 days |
Not during pregnancy |
|
Ofloxacin |
200 mg bid |
3 days |
Not during pregnancy |
|
If local resistance pattern is known (E. coli resistance <20 %) |
|||
|
TMP |
200 mg bid |
5 days |
TMP not in the first trimester of pregnancy |
|
TMP-SMX |
160/800 mg bid |
3 days |
SMX not in the last trimester of pregnancy |
|
Severity grade 2 (mild and moderate pyelonephritis) |
|||
|
Ciprofloxacin |
500–750 mg bid |
7–10 days |
|
|
Levofloxacin |
500 mg qd |
7–10 days |
|
|
Levofloxacin |
750 mg qd |
5 days |
|
|
Alternatives (clinical but not microbiological equivalent efficacy compared with fluoroquinolones) |
|||
|
Cefpodoxime proxetil |
200 mg bid |
10 days |
|
|
Ceftibuten |
400 mg qd |
10 days |
|
|
Only if the pathogen is known to be susceptible (not for initial empirical therapy): |
|||
|
Trimethoprim/sulphamethoxazole |
160/800 mg bid |
14 days |
|
|
Co-amoxiclava,b |
0.5/0.125 g tid |
14 days |
|
|
Severity grade 3 (severe uncomplicated pyelonephritis) |
|||
|
Initial empirical parenteral antimicrobial therapy |
|||
|
Ciprofloxacin |
400 mg bid |
Fluoroquinolones are contraindicated during pregnancy Re: duration, kindly see below under alternatives |
|
|
Levofloxacina |
250–500 mg qd |
||
|
Levofloxacin |
750 mg qd |
||
|
Alternatives |
|||
|
Cefotaximeb |
2 g tid |
After improvement, the patient can be switched to an oral regimen using one of the above mentioned antibacterials (if active against the infecting organism) to complete a 1–2-week course of therapy. Therefore, only daily dose and no duration of therapy are indicated |
|
|
Ceftriaxonea,d |
1–2 g qd |
||
|
Ceftazidimeb |
1–2 g tid |
||
|
Cefepimea,d |
1–2 g bid |
||
|
Co-amoxiclavb,c |
1.5 g tid |
||
|
Piperacillin/tazobactama,d |
2.5–4.5 g tid |
||
|
Gentamicinb |
5 mg/kg qd |
||
|
Amikacinb |
15 mg/kg qd |
||
|
Ertapenemd |
1 g qd |
||
|
Imipenem/cilastatind |
0.5/0.5 g tid |
||
|
Meropenemd |
1 g tid |
||
|
Doripenemd |
0.5 g tid |
||
|
Severity grade 4, 5, and 6 (the sepsis syndrome) |
|||
|
Any of the drugs recommended for severity grade 3, usually in combination with gentamicin |
|||
aLower dose studied, but higher dose recommended by experts
bNot studied as monotherapy in acute uncomplicated pyelonephritis
cMainly for gram-positive pathogens
dSame protocol for acute uncomplicated pyelonephritis and complicated UTI (stratification not always possible)
If risk factors are present, as described in the ORENUC phenotyping, the severity of the clinical situation might be upgraded and the empiric antibiotic treatment adjusted accordingly. Some practical advices can be found in the textbox below:
State-of-the-Art Expert Advice on Antibiotic Treatment of UTI
· Determine the clinical presentation of the infection, check for risk factors, and determine the severity grade.
· Check the resistance rates to the most commonly used antibiotics in your country.
· Clarify if the infection is community acquired or health care acquired.
· Clarify which is the most likely pathogen and if inherent resistance is likely.
· Chose the most relevant antibiotic from Table 8.3.
· Adjust treatment according to culture results as soon as they are available.
· In febrile infections, continue treatment until 3–5 days after defeverescence.
Prevention of Recurrent UTI (rUTI)
Recurrent UTI is said to occur when a patient has more than three episodes of microbiologically verified UTIs in 1 year or two episodes within 6 months. A reinfection should be differentiated from a relapse which means that an infection with the same pathogen occurs within 2 weeks after completion of antibiotic treatment of a UTI. In the ORENUC system, phenotype R means that all other risk factors are excluded. Recurrent UTI is common among young, healthy women. Several preventive measures are available.
Behavioral Modifications
Sexual intercourse is highly associated with rUTI in young and old women alike. Spermicide use and a new sex partner add to the risk. Several other behaviors are also thought to increase the risk of rUTI. These include reduced fluid intake, habitually delaying urination, delaying postcoital urination, wiping from back to front after defecation, douching, and wearing occlusive underwear. Their association with UTI is, however, not proven in clinical trials. Micturition training and biofeedback accompanied by long-term antibiotic prophylaxis have significant effect on rUTI in children, but no corresponding studies are available in adults.
Hormonal Replacement
In postmenopausal women, local, vaginal estrogen replacement is thought to strengthen the mucosa and influence the local bacterial flora by stimulating the growth of Lactobacillus sp. which prevents the growth of Enterobacteria that are urinary tract pathogens. Studies have showed a trend toward preventing UTI recurrences, but vaginal irritation is common.
Immunoactive Prophylaxis
Immunoactive prophylaxis is thought to stimulate the innate immune system. Three routes of administration have been tested: oral, vaginal, and parenteral.
OM-89 (Uro-Vaxom®) are tablets for oral administration which contain a dried lysate of several bacteria and have a good safety profile. The drug has been shown to be more effective than placebo in several randomized trials but has not yet been compared with antimicrobial prophylaxis.
Urovac® is a vaginal vaccine that has been shown to reduce UTI recurrence and to increase time to reinfection. Two parenteral immunotherapeutic products have been studied in phase II studies, StroVac® and Solco-Urovac®. Both have been shown to be effective when administered with a booster cycle of the same agents.
Probiotics
Vaginal application of Lactobacillus crispatus has been shown to reduce the rate of recurrent UTI in premenopausal women, while oral lactobacilli prophylaxis did not decrease UTI recurrence. Clinically proven probiotics for UTI prophylaxis are currently not universally available.
Cranberry
Cranberry containing proanthocyanidin A is able to decrease the binding capacity of P-fimbriae of Escherichia coli to the mucosa in a dose-depending manner. Previous studies have suggested that cranberry (Vaccinium macrocarpon) was useful in reducing the rate of lower UTIs in women, but a recent meta-analysis comprising 4473 participants showed that cranberry products did not significantly reduce the occurrence of symptomatic UTI.
D-Mannose
D-mannose is a sugar thought to inactivate fimbria which is a virulence factor of E. coli. In a prospective randomized placebo-controlled clinical trial, it could be demonstrated that a daily dose of 2 g d-mannose was significantly superior to placebo and as effective, but better tolerated than 50 mg nitrofurantoin in preventing recurrent UTI.
Non-antimicrobial approaches to prevent recurrent UTIs
|
When |
How |
Mechanism |
Evidence |
|
|
Sexual behavioral changes |
Premenopausal women |
Education and counseling |
Reduce the bacterial passage through the urethra |
Some evidence from studies |
|
Use of topical estrogen |
Postmenopausal women |
Topical vaginal estrogens |
Reconstitution of vaginal flora |
Randomized placebo-controlled studies |
|
Cranberry products |
All women (especially in young women) |
Oral administration |
Block adherence of E. coli to uroepithelial cells via the proanthocyanidin type A |
Conflicting evidence – no evidence for cranberry powders |
|
Probiotics |
All women |
Oral administration |
Reconstituting vaginal lactobacilli |
Randomized studies |
|
Immunoprophylaxis |
All women |
Oral administration |
Stimulates urinary tract immunological defense |
Meta-analysis |
Antimicrobial Prophylaxis
Due to the risk of resistance, antimicrobial prophylaxis should only be given after other preventive measures have failed. Prophylaxis can be given continuously (daily, weekly) for longer periods of time (3–6 months), or as a single postcoital dose. Recurrences tend to occur in clusters and it is advisable to pay attention to the time intervals when the duration of prophylaxis is decided. Furthermore, many women with recurrent UTI can safely diagnose themselves and administer short-course regimens of an antimicrobial agent.
The choice of antibiotics should be based upon the identification and susceptibility pattern of the organism causing the rUTI, the patient’s history of drug allergies, and the ecological collateral effects including bacterial selection of resistance by the chosen antimicrobial. The spectrum of antibiotics is the same as for sporadic acute UTI (Table 8.3).
The most commonly used regimens for antimicrobial prophylaxis of rUTI include:
· Continuous regimens:
· Nitrofurantoin (macrocrystal) 50 or 100 mg once daily
· Fosfomycin trometamol 3 g every 10 days
· During pregnancy:
· Cephalexin 125 or 250 mg
· Cefaclor 250 mg once daily
· Postcoital prophylaxis should be considered in pregnant women with a history of frequent UTIs before onset of pregnancy.
Recently, several concerns have been raised against antimicrobial prophylaxis of rUTI. Oral fluoroquinolones and cephalosporins are no longer recommended due to the risk of ecological side effects. There are warnings against long-term prophylactic use of nitrofurantoin because of pulmonary and hepatic side effects. Increase of E. coli resistance against trimethoprim and resistance in ESBL-producing E. coli to fosfomycin underlines the need for non-antimicrobial preventive measures.
An important aspect is whether or not to treat persistent asymptomatic bacteriuria in young women after UTI. Recently, Cai et al. found that the antibiotic treatment of asymptomatic bacteriuria in young women with recurrent UTI is not only unnecessary, but harmful. They found that in women who had undergone antibiotic treatment, the rate of E. coli decreased over time, whereas the prevalence of E. faecalis increased gradually, suggesting that E. colimight be an important biological defense mechanism that effectively interferes with the establishment of other important enteric pathogens, such as E. faecalis.
The use of antimicrobial prophylaxis might appear highly effective in reducing the risk of recurrent UTI in a short time perspective, but a non-antimicrobial approach should always be tried first in order to avoid long-term antimicrobial side effects with selection of resistant strains.
Alternative Preventive Measures
Natural Antibiotics and Disinfectants
There are several components of our infection defense system acting as natural antibiotics and disinfectants. Antimicrobial peptides (AMPs) are produced by several species as a key component of the natural defense system. AMPs protect against bacteria, fungi, parasites, viruses, and even cancer cells. Keratinocytes in the perianal region produce psoriasin, a substance which destroys enteric bacteria. This is an intriguing new field of research, but so far there are no data from clinical studies.
Phytotherapy
Phytotherapeutic drugs have been shown to have inflammatory, antioxidative, antibacterial, diuretic, and spasmolytic effects. Well-designed studies have been performed showing the phytotherapeutic drug CanephronR to have similar protective effect as antimicrobial prophylaxis against rUTI. The initial results warrant follow-up with double-blind clinical studies.
Bladder Instillation
Intravesical instillation of hyaluronic acid (HA) or chondroitin sulfate (CS) has been attempted to strengthen the intravesical GAG (glycosaminoglycan) layer and thereby prevent rUTI. In a recent small double-blind, placebo-controlled trial, intravesical instillations of HA-CS were given during a five-month period. Compared with placebo, HA-CS instillations significantly reduced the recurrence rate over a 12-month period, and there were no severe side effects. However, further intravesical instrumentations are not recommended for the management of uncomplicated rUTI.
Surgery
Surgery has a double role related to urinary tract infections:
· Treatment of acute infections
· Prevention
· Of recurrences
· Of hospital acquired infections
Treatment of Acute Infections
The role of surgery in acute situations rests on the old principles: wherever there is pus, you should drain it (ubi pus ibi evacua), and that retention might cause infection. An important feature of the urinary tract is the reflux from the renal pelvis to the veins through the pyelovenous and pyelolymphatic drainage which occurs when the pressure in the renal pelvis exceeds a certain limit.
In the most serious acute conditions like severe pyelonephritis and urosepsis, one should always look for dilatation of the upper tract, and drain it with a nephrostomy tube if pyonephrosis is suspected (Fig. 8.7). The reason why symptoms usually improve very fast is that the pyelovenous reflux of microorganisms is stopped.
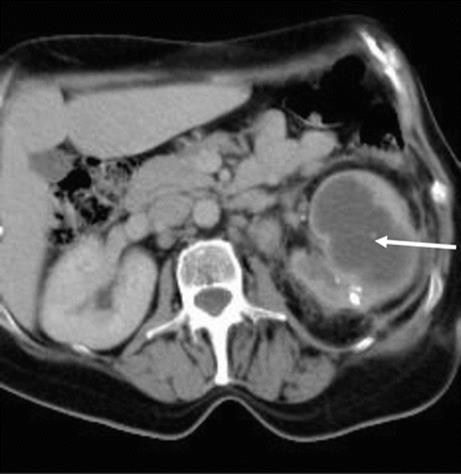
Fig. 8.7
CT scan showing left-sided pyonephrosis in a young woman (arrow)
Renal or perirenal abscesses should be drained outside the abdominal cavity. The peritoneum is a dialysis membrane and microorganisms may easily penetrate to the systemic circulation. Spread of infection may cause intra-abdominal adhesions and ileus at a later time. If open surgery is needed to drain an abscess, the surgical field will become infected. The surgeon should therefore ensure large drainage tubes and consider the old war surgical principle of delayed primary closure of the skin a week or so later.
Prevention of Recurrences
Prevention of recurrent UTI in children is a cornerstone of pediatric urology. We used to think that there was an etiological relationship between reflux causing ureteral dilation, causing infections, and causing renal failure, but recent studies indicate that there might be a common inborn defect behind all four conditions. Reflux may be prevented by periureteral instillation of blocking materials like collagen and ureteral reimplantation.
In adults, urology prevention is about removal of infection stones and relief of obstruction at all levels of the urinary tract. It is difficult to define a critical threshold for residual urine in patients with recurrent UTI and prostatic enlargement due to benign prostatic hyperplasia. The decision on when to do a transurethral resection (TURP) should be based on clinical experience and supported by urodynamic evaluation. One hundred milliliters might sometimes be enough to cause recurrent infections. One should be hesitant to do a TURP if recurrent UTI is thought to stem from chronic bacterial prostatitis.
Prevention of Hospital-Acquired Infections (HAUTI)
Some measures to prevent HAUTI lie on the level of the hospital and some lie on the personal level of the surgeon.
Hospital Level
The prevalence of HAUTI varies form 7 to 21 % in urology departments. This is a big problem, especially as more and more patients are coming in for elective surgery without having any symptoms. Prevention of HAUTI rests on:
· A recognition of the contamination status of the procedure
· The patient’s risk factors for developing infection
· Antibiotic prophylaxis (ABP)
Each contamination category has a certain peril of infection. Risk factors modify this peril. ABP should be decided according to the most likely pathogen, the resistance situation in the hospital, and the pharmacokinetic and pharmacodynamic properties of the antibiotic. Antibiotics used for prophylaxis should not cause collateral damage. This means development of resistance in causative pathogens and spread of resistance to other microorganisms is called cross-resistance and is a point of growing concern. As a rule, ABP should not extend beyond 24 h, and the most potent antibiotics should not be used for prophylaxis, but should be reserved for treatment in case infection occurs. This situation is already setting limits for urological practice in certain countries, especially in prostate biopsies and stone surgery.
The Surgeon Level
The single most important measure to prevent hospital-acquired urinary tract infections is to take out urinary catheters and stents (Fig. 8.6). As a rule, prophylaxis can never compensate for a poor surgical technique. Unfortunately, some surgeons have more complications than others.
The surgeon should strive for perfection in surgical technique and avoid damaging tissue and leaving dead tissue behind. Tissues must be handled gently with forceps, avoiding too extensive electrocoagulation and big ligatures. Consider carefully the blood supply to intestinal segments. One single improper suture may cause leakage and infection and ruin the result of an extensive procedure. Try and prevent hematomas, if necessary with drains, but never use a suction drain after opening the urinary tract. Leakage usually stops after 11 days. Learn the pathophysiology of sepsis and the clinical signs of imminent septic shock. Follow recommendations on basic hygienic principles.
Recommended Further Reading
1.
Bjerklund Johansen TE, Botto H, Cek M, Grabe M, Tenke P, Wagenlehner FME, Naber KG. Critical review of current definitions of urinary tract infections and proposal of an EAU/ESIU classification system. Int J Antimicrob Agents. 2011;38(Suppl):64–70.CrossRef
2.
Grabe M, Bjerklund Johansen TE, Botto H, Wullt B, Cek M, Naber KG, Pickard RS, Tenke P, Wagenlehner F. Guidelines on urological infections. Arnhem: European Association of Urology; 2013. Update. ISBN 978-90-79754-83-0.
3.
Cai T, Mazzoli S, Mondaini N, Meacci F, Nesi G, D’Elia C, Malossini G, Boddi V, Bartoletti R. The role of asymptomatic bacteriuria in young women with recurrent urinary tract infections: to treat or not to treat? Clin Infect Dis. 2012;55(6):771–7.CrossRefPubMed
4.
Naber KG, Scaeffer AJ, Heyns CF, Matsumoto T, Shoskes DA, Bjerklund Johansen TE. Urogenital infections (textbook 1182pp). Arnhem: International Consultation on Urological Diseases (ICUD) and European Association of Urology; 2010. ISBN 978-90-79754-41-0.
5.
Tandogdu Z, Cek M, Wagenlehner F, Naber K, Tenke P, Bjerklund Johansen TE. Resistance patterns of nosocomial urinary tract infections in urology departments: 8 year results of the global prevalence of infections in urology study. World J Urol. 2013. doi:10.1007/s00345-013-1154-8.PubMed