Q. You are referred a man who is 32 years old and presents with infertility. He has never fathered a child, but has been married for 5 years. His wife is 37 years old. They have been trying to start a family for 15 months. How do you define infertility?
A. The inability of a sexually active couple to achieve a pregnancy within 12 months following regular unprotected sexual intercourse.
Q. What is the baseline fertility rate?
A. The chance of a normal couple conceiving is estimated to be 20%-25% per month, 75% by 6 months, and 90% by 1 year. The baseline pregnancy rate is 1%-3% per month (in non- azoospermic couples).
Q. What proportion of infertility cases are due to female or male causes?
A. Approximately 20% of cases of infertility are caused entirely by a male factor, with an additional 30% of cases due to both male and female factors. There fore, a male factor is present in one-half of infertile couples. The remaining 50% are due to female factors only.
Q. When would you begin investigations of this couple if at all?
A. Of infertile couples without treatment, 25%-35% will conceive at some point by intercourse alone. In the past it was recommended not to investigate patients until 12 months of attempted conception. However, with the advancing age of infertile couples at presentation, a basic, simple, cost-effective evaluation of both the male and the female may be initiated at the time of presentation.
Q. How would you assess this man with infertility?
A. I would ensure that his wife is also investigated and this is often best done in a joint infertility clinic, with a female fertility specialist. First, I would establish whether the period of unprotected intercourse has been regular and long enough to meet the criteria for infertility (see definition above). Once confirmed, I would try to establish the underlying cause and identify risk factors by taking a focussed urological and sexual history, performing a clinical examination and undertaking relevant investigations.
Q. What would you ask in the history?
A. In the history, I would ask about the duration of infertility, details of any previous pregnancies, contraceptive use in the past, the couple’s frequency of sexual intercourse, as well as the timing of coitus. Both erectile and ejaculatory function should be assessed, and the use of any vaginal lubricants during intercourse should be noted as they may affect sperm quality.
The developmental history of the patient should be noted such as any history of cryptorchidism, age at puberty, and development of secondary sexual characteristics. The patient’s past surgical history may be of particular importance such as previous history of orchidopexy, testicular torsion or inguino-scrotal surgery particularly hernia repairs and epididymal cyst excision.
The patient should be questioned for a history of urinary tract infections or sexually transmitted diseases as well as a history of mumps orchitis.
A history of previous chemotherapy, radiation therapy or gonadotoxic treatment should also be ascertained.
A history of chronic upper respiratory tract infections should be actively sought (may indicate cystic fibrosis).
Anabolic steroid abuse and recreational drugs such as marijuana are also risk factors.
Q. What would you be looking for in the physical examination of this man?
A. On examination, the patient’s body habitus (obesity) as well as the pattern of virilization should be recorded. After this a focused urogenital examination is performed. The scrotal contents should be examined with the patient standing and lying down in a warm room to allow for relaxation of the cremasteric muscle. The testes should be carefully palpated to determine consistency and to exclude the presence of an intratesticular mass. The dimensions of the testes should be measured and documented. Palpation of the epididymis should determine the presence of the head, body and tail. The possibility of epididymal obstruction is suggested by the presence of a dilated epididymal head and body. Palpation of the vas deferens is performed to ensure its presence bilaterally. The scrotum should be examined with the patient standing up to check for the presence of a varicocele. (Use Valsalva if not obviously palpable.)
Q. What initial tests would you perform on this patient?
A. A semen analysis and hormone profile comprising serum FSH, LH and testosterone will diagnose the cause of infertility in the majority of men. All patients should have at least two semen analyses performed to confirm an abnormal result.
Q. How would you counsel this patient to perform a semen analysis?
A. An accurate semen analysis is an important investigation for the evaluation of the infertile male. To compare different semen samples from the same patient with accuracy, it is important to maintain consistency in the duration of sexual abstinence before collection of the specimen. It is recommended that the patient abstains from ejaculation for 2-5 days before providing the sample. The sample should be provided into a clean, wide-mouthed container at either the laboratory (ideal scenario) or at the patient’s home but must be delivered to the laboratory within 1 hour. It is recommended that specimens produced at home should be brought to the lab by placing the container in a shirt pocket next to the body to keep it warm during transit.
I would inform the patient that the specimen should be produced by masturbation without the use of latex condoms as these may interfere with the viability of sperm due to the presence of spermicides. The specimen should be examined in the laboratory within 1 to 2 hours of collection. A label on the container should state the patient’s name, the date, the time of collection and the abstinence period.
Q. Can the specimen be produced using coitus interruptus?
A. Although interrupted coitus may be used as an alternative method for obtaining specimens, this is not recommended because the initial portion of the ejaculate may be lost and bacteria and acidic vaginal secretions may contaminate the specimen which can result in inaccurate results.
Q. What are the normal characteristics of the semen analysis?
A. The World Health Organization (2010) defines the following reference values:
|
Volume: |
>1.5 mL |
|
pH: |
>7.2 |
|
Sperm concentration: |
>15 x l06/mL |
|
Total sperm number: |
>39 x 106 or more spermatozoa per ejaculate |
|
Motility: |
>32% with progressive motility |
|
Morphology: |
>4% of normal forms |
|
Vitality: |
>58% |
|
White blood cells: |
< 1 million/mL |
Q. This particular patient in question has a semen analysis performed. His results are as follows on two occasions:
|
Volume: |
2.4 mL |
|
pH: |
7.5 |
|
Sperm concentration: |
6 X 106 sperm/mL |
|
Total sperm number: |
14.4 X 106 spermatozoa |
|
Motility: |
20% with > grade 2 and 11% with grade 4 |
|
Morphology: |
2% of normal forms |
|
White blood cells: |
< 1 million/mL |
His hormone profile is normal. What is this man’s diagnosis?
A. Combined defects in sperm density, motility and morphology are known as oligoasthenoteratospermia or OAT syndrome.
Q. What are the potential causes of the OAT syndrome?
A. The OAT syndrome can be associated with a varicocele. Other causes include cryptorchidism, temporary insults to spermatogenesis such as heat, drugs or environmental toxins, or idiopathic causes. A heat effect may be either environmental or endogenous, such as a systemic illness resulting in fever.
Q. Physical examination in this patient’s scrotum revealed a large left varicocele. What is a varicocele and how are these graded?
A. A varicocele is defined as an abnormal dilatation of the pampiniform plexus of spermatic veins, and is graded (Hudson classification) according to physical characteristics:
|
Grade III |
Palpable and visible |
|
Grade II |
Palpable on standing only, but not visible |
|
Grade I |
Palpable on Valsalva manoeuvre, not visible |
|
Grade 0 |
Subclinical (ultrasound scan diagnosis only) |
Q. How common are varicoceles?
A. The prevalence is about 15% in the healthy general population. However in men presenting with infertility they are present in 20%-40%.
Q. Why do patients develop varicoceles?
A. Approximately 90% of varicoceles are on the left side. Differences in the venous drainage patterns of the right and left testicular veins may account for this left-sided predominance. The left testicular vein normally drains directly into the left renal vein, whereas the right testicular vein drains into the inferior vena cava. In addition, an absence of the venous valves is more commonly found on the left side than on the right. Finally, the left renal vein may be compressed between the superior mesenteric artery and the aorta. This ‘nutcracker phenomenon’ may result in impaired venous drainage in the left testicular venous system.
Q. Are varicoceles associated with infertility?
A. ttere does appear to be an association with impaired semen parameters. Semen samples from infertile men with varicoceles demonstrate decreased motility in 90% of patients and reduced sperm concentrations in 65% of patients.
Q. Why are varicoceles associated with infertility?
A. It is possible that the countercurrent exchange mechanism which normally maintains a lower intrascrotal temperature is impaired in the presence of a varicocele. Oligospermic patients with varicoceles have a higher intrascrotal temperature (0.6°C higher). Other causes may include reflux of renal and adrenal metabolites from the renal vein, decreased blood flow and hypoxia and impaired sperm DNA quality.
Q. When should varicoceles be treated in the context of fertility?
A. Adolescent patients with grade II/III varicoceles with a reduced ipsilateral testicular volume should undergo treatment. Sub-fertile males with impaired semen parameters can also be offered treatment as there is likely to be an improvement in the semen parameters. This may be enough to allow a natural pregnancy or improve the parameters prior to assisted reproduction which may either be intrauterine insemination (IUI), in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI).
Q. How would you manage the varicocele in this patient?
A. Improvement in semen parameters is demonstrated in approximately 70% of patients after a varicocele repair. Improvements in motility are most common, occurring in 70% of patients, with improved sperm densities in 51% and improved morphology in 44% of patients. Semen characteristics usually improve between 3 months to 1 year following surgery or embolisation.
There have been a number of studies reviewing the effects of a varicocele on fertility with conflicting results as the majority of the studies are uncontrolled with heterogenous patient and partner subgroups.
the widely quoted Evers meta-analysis suggested that varicocele treatment does not improve pregnancy rates [2]. However, this meta-analysis included patients with sub-clinical varicoceles and also patients with normal semen parameters. When these are excluded a further meta-analysis reported pregnancy rates of 36.4% versus 20% in the treated versus the untreated group. The recommendations of the Joint Committee of the American Urological Association and the American Society for Reproductive Medicine did not acknowledge the conclusions of a Cochrane meta-analysis and consider varicocele treatment to be suitable in patients with a palpable varicocele and abnormal semen parameters.
National Institute for Health and Care Excellence (NICE) guidelines recommend that men should not be offered surgical treatment for a varicocele because it does not improve pregnancy rates.
Q. What would you recommend to this couple?
A. I would recommend that they be seen by a urologist and also a fertility specialist. They should be appropriately counselled on the relative success rates of all the available options which would include either a varicocele ligation or IUI/ICSI/IVF depending upon their personal circumstances.
Q. Which assisted reproduction techniques (ARTs) are you aware of?
A. ARTs involve the manipulation of sperm or ova or both in an attempt to improve the chance of conception and resultant live birth rates. These are as follows:
Intrauterine insemination (IUI)
In vitro fertilization (IVF)
Intracytoplasmic sperm injection (ICSI)
Intracytoplasmic morphologically selected sperm injection (IMSI)
Q. Which sperm retrieval techniques are you aware of?
A. Surgical techniques to retrieve sperm include the following:
Percutaneous sperm aspiration (PESA)
Microsurgical epididymal sperm aspiration (MESA)
Testicular sperm aspiration (TESA)
Testicular sperm extraction (TESE)
Microdissection TESE (mTESE)
Q. What is a TESE?
A. This refers to the technique of testicular exploration and sperm extraction. The optimal technique of sperm extraction should be minimally invasive and avoid impairment of testicular function while maximising the chance of retrieving enough mature sperm to perform ICSI. Conventional TESE uses single or multiple open testicular biopsies to retrieve seminiferous tubules which are then dissected to retrieve mature sperm which can then be used for ART. MicroTESE utilises high magnification of the seminiferous tubules and more extensive dissection of the testicular tissue to retrieve sperm from the seminiferous tubules.
A smaller amount of tissue is removed with targeting of the best seminiferous tubules which are generally of a better calibre and colour. The tubules are then examined within the theatre setting under a light microscope to confirm the presence of mature sperm following which the tissue can either be frozen for future ICSI or sperm harvested for a same-day ICSI cycle.
The conventional TESE technique requires multiple, blind testicular biopsies with excision of a larger volume (>500 mg) of testicular tissue. The microTESE technique of sequential excision of microdissected seminiferous tubules (10-15 mg, or 2 mm in length, of seminiferous tubule) has been shown to be more successful, compared with the results achieved by conventional TESE, or random biopsies of testicular tissue.
Q. What is IVF and how is it performed?
A. IVF refers to in vitro fertilisation. Gonadotropins are used to stimulate multiple oocytes during each cycle of treatment. Follicular development is then monitored ultrasonically, and the ova are harvested before ovulation with the use of ultrasound-guided needle aspiration.
IVF is performed by mixing processed sperm with retrieved oocytes. In standard IVF, when fertilization occurs, the developing embryos are incubated for 2-3 days in culture and then placed trans-cervically into the uterus. Using this technique, 20%-30% of transferred embryos will implant and produce clinical pregnancies. More than 90% of inseminated oocytes are routinely fertilized when sperm function is normal. However, fertilization rates are reduced significantly when a male factor for infertility is present.
Q. What is the ICSI and when is it used?
A. ICSI refers to intracytoplasmic sperm injection whereby a single sperm is injected into the ovum. This allows for fertilization with extremely low numbers of sperm or sperm retrieved from testicular tissue. ICSI is indicated in cases of severe male factor infertility, in couples with prior failed or poor fertilization during regular IVF cycles, or in cases in which the sperm demonstrate significant fertilizing ability defects. In 2006, ICSI represented 47% of all IVF treatments in the United Kingdom.
Q. What are the success rates of ICSI?
A. The clinical pregnancy rates using ICSI average 20%-37% per initiated cycle. Pregnancy rates are affected by the age of the female when undergoing IVF or ICSI. The reported pregnancy rates are 36.9% in women <35 years and 10.7% in women >40 years. In the United Kingdom the HFEA reports a mean take-home baby rate of 23.1% with a rate of 31% for women under the age of 35.
Q. Is there a difference in congenital malformation rates between IVF and ICSI?
A. Children born as a result of ICSI (6.2%), compared to IVF (4.1%), may have an increased risk of malformations. Only the difference in the rates between ICSI children and the control group was statistically significant. Furthermore, a recent meta-analysis has suggested a small significant increase in malformations in children born following ARTs versus controls. More recently a malformation rate of 6.2% has been shown with ART versus 4.4% in normal pregnancies.
Q. Are there any specific concerns when treating patients with severe male factor infertility with ART?
A. Patients who are considered candidates for the ARTs with either severe oligospermia or azoospermia may have associated chromosomal defects which can be abnormal karyotypes (47XXY), Y microdeletions or carriers of the cystic fibrosis gene. As there is a risk for the offspring patients should undergo these tests and appropriate genetic counselling before treatment.
Q. Another patient is referred to see you. He is aged 26 and has recently married. This is his first sexual relationship. He has already been to a private laboratory and performed two semen analyses 3 months apart. The results of the latest one are below and are the same as the first one:
|
Volume: |
2.7 mL |
|
pH: |
7.4 |
|
Sperm concentration: |
None seen |
|
Total sperm number: |
Nil |
|
Motility: |
Not assessed |
|
Morphology: |
Not assessed |
|
White blood cells: |
<1 million/mL |
Which other tests would you perform?
A. After taking an appropriate history and a focused physical examination I would perform a baseline hormone profile. I would also consider performing genetic studies to check the karyotype and Y microdeletion.
Q. His testosterone is 7 and both his FSH and LH are significantly elevated, the FSH is more than three times normal. What is his differential diagnosis?
A. This man is likely to have non-obstructive azoospermia indicating abnormal spermatogenesis. This may be secondary to hypogonadotrophic hypogonadism (Kallmann’s syndrome, pituitary tumour) or abnormalities of spermatogenesis (chromosomal abnormalities, toxins, orchitis, previous torsion).
Q. What are you specifically looking for on examination?
A. I am looking specifically for secondary sexual characteristics, body habitus, the presence of gynaecomastia, the size and consistency of the testis and whether the vas are palpable.
Q. This patient has bilateral small soft testes. This is the biopsy from his testis (Figure 13.6). What does it show? What is a possible diagnosis if this was a chromosomal disorder?
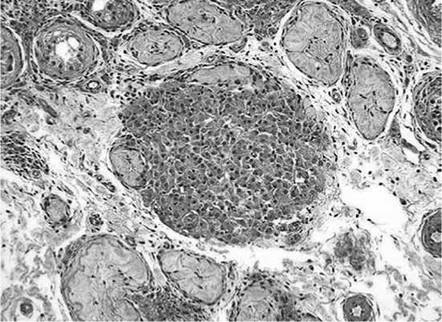
Figure 13.6
A. The testicular biopsy presented in Figure 13.6 shows small hyalinised seminiferous tubules and pseudo-adenomatous clusters of Leydig cells. The likely diagnosis here is Klinefelter’s syndrome where patients present with non-obstructive azoospermia, a high FSH with a low testosterone and small volume soft testicles. Although phenotypically they are normal looking males, genetic tests confirm that the karyotype is 47XXY.
Q. Assuming that the genetic testing shows a normal karyotype but the genetic tests report that a Y microdeletion has been detected, can you explain the significance of a Y microdeletion in the context of male infertility?
A. Patients with these microdeletions are usually phenotypically normal, with the only apparent abnormality being a defect in spermatogenesis. The defect occurs in one of three non-overlapping regions of the long arm of the Y chromosome referred to as AZFa (proximal), AZFb (middle) and AZFc (distal). Although there is no strict correlation betwen these microdeletions and the testis biopsy histology, the following are associated:
Microdeletion AZFa - Sertoli only
Microdeletion AZFb - Maturation arrest
Microdeletion AZFc - Severe oligzoospemia (no histological pattern)
Men with an AZFa or AZFb microdeletion will not have sperm within the testicle and a sperm retrieval is not indicated. However there is a small probability (10%-15%) of finding sperm in the presence of an AZFc microdeletion.
Q. Why is it important to test patients for microdeletions?
A. It is important because these gene deletions will be transmitted to male offspring. Couples in whom the husband has Y chromosome microdeletions should be offered genetic counselling before embarking on ART.
Q. Another 26-year-old man with azoospermia has a normal hormone profile (LH, FSH, testosterone) and a normal testicular volume when examined on his first visit. What do you suspect is the underlying diagnosis?
A. He is likely to have an obstructive cause for his azoospermia, however maturation arrest cannot be excluded without a testicular biopsy.
Q. What would have been an important finding in his clinical examination?
A. It is important to establish the presence of the vas deferens. If there are no palpable vasa bilaterally, the patient may have congenital bilateral absence of vas deferens (CBAVD). CBAVD is a clinical diagnosis and is due to an abnormality in the CFTR gene. This may occur in the absence of any respiratory symptoms.
A dilated vas or epididymis is usually indicative of obstruction which can be at the level of the ejaculatory ducts, vas or epididymis.
Q. What is the level of spermatogenesis in patients with obstructive azoospermia and how would you manage them?
A. Most of these patients have normal spermatogenesis within the testicles. A scrotal exploration is reserved for cases where a reconstruction is planned to bypass the obstruction at which time sperm retrieval can ensure that tissue is cryopreserved for future ART. Patients with ejaculatory duct obstruction (EDO) can be offered endoscopic resection, transurethral resection of ejaculatory ducts (TURED).
Q. What is the role of testicular biopsy in azoospermic patients?
A. Isolated diagnostic testicular biopsies are now seldom performed in azoospermic patients as most will be undergoing sperm retrieval for ICSI at which point a biopsy can be performed if no sperm is found. Testicular biopsy is not indicated in patients with oligospermia, because the results will not change the treatment options unless it is required to exclude the presence of ITGCN.
Q. The patient has a testicular biopsy performed at the time of sperm retrieval. How is it evaluated?
A. The most common system used to classify spermatogenesis on a testicular biopsy is the Johnsen score (Table 13.3).
Table 13.3 Johnsen score count
|
Score |
Description |
|
10 |
Complete spermatogenesis - organised epithelium |
|
9 |
Many spermatozoa - disorganised epithelium |
|
8 |
< 10 spermatozoa |
|
7 |
No spermatozoa, but many spermatids |
|
6 |
No spermatozoa, but < 10 spermatids |
|
5 |
No spermatozoa, spermatids, but many spermatocytes |
|
4 |
< 10 spermatocytes |
|
3 |
Spermatogonia |
|
2 |
Sertoli cells only |
|
1 |
No cells, tubular fibrosis |
Q. Figure 13.7 is a slide showing a testicular biopsy. What does it show?
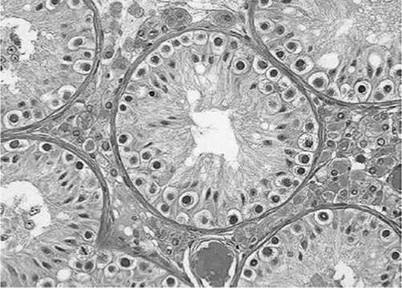
Figure 13.7
A. Figure 13.7 shows maturation arrest.
Q. If the biopsy showed normal spermatogenesis, what would you do next?
A. If reconstruction is contemplated I would perform a vasogram to determine the site of obstruction in azoospermic patients who have active spermatogenesis documented by testis biopsy. Vasography should be ideally performed in conjunction with reconstructive surgery because this procedure carries an inherent risk of vasal injury that could complicate future reconstructive surgery, if performed separately.
Vasography is performed at the level of the straight portion of the scrotal vas deferens by needle puncture with an orange needle on a 2.5 mL syringe containing injection of nonionic contrast agent.
Q. What is a normal vasogram?
A. A normal vasogram is documented when contrast agent is visualized throughout the length of the vas deferens, seminal vesicles, ejaculatory duct and bladder. Proximal patency of the epididymis is documented by microscopic (x400) visualization of sperm in the intravasal fluid.