Matthew C. Bunte and Richard A. Grimm
The evaluation of cardiac patients undergoing noncardiac surgery is an important part of the day-to-day practice of the consulting cardiologist. Unfortunately, poor outcomes can and do occur in high-risk patients. Therefore, predicting risk of potential nonfatal myocardial infarction (MI), heart failure, pulmonary embolism, or death is imperative when managing especially high-risk patients. Several risk prediction indexes have been proposed by various authors with this aim in mind. Coronary revascularization before noncardiac surgery is rarely necessary, however, even among high-risk populations. In this chapter, the revised American College of Cardiology Foundation/American Heart Association (ACCF/AHA) Task Force on Practice Guidelines are used extensively as the primary reference for review of the literature and practice recommendations. These guidelines emphasize the importance of utilizing clinical predictors, evidence-based practice, along with a rational and common sense approach.
Approximately 30 million patients underwent noncardiac surgery in the United States in 2003. Of these, an estimated 4% had diagnosed coronary artery disease (CAD), 8% to 12% had multiple risk factors for CAD, and approximately 16% were over 65 years of age. Those at greatest risk for cardiac complications are 65 years or older with previously diagnosed CAD. This group accounts for nearly 80% of the estimated 1 million patients who suffer major cardiovascular complications annually. These cardiovascular complications cost an estimated $12 billion annually
Despite the increasing age of those undergoing noncardiac surgery, age alone represents a minor risk factor for perioperative complications. However, mortality associated with perioperative acute MI increases dramatically with advanced age. While the overall risk of suffering a postoperative MI is probably <1% with many elective operations, up to 50% of these events can be fatal. The highest risk of perioperative reinfarction occurs within the first 6 months of the index infarction and subsequently falls with time. Guidelines published by the ACC/AHA recommend that elective surgery may be performed before the 6-month time period so long as a postinfarction risk stratification has been performed. A negative stress test for ischemia or complete revascularization does reduce the risk of reinfarction with elective surgery. Nonetheless, waiting at least 4 to 6 weeks before proceeding with elective surgery represents a prudent approach suggested by these guidelines.
For patients with cardiac disease undergoing noncardiac surgery, surveillance for cardiac complications should be performed for at least 48 hours following surgery. This assessment begins with a baseline preoperative resting 12-lead electrocardiogram (ECG) for future comparison, particularly among patients with at least one clinical risk factor and/or those undergoing procedures of intermediate risk or greater. The peak risk of MI occurs within the first 3 postsurgical days but may persist for as long as 5 to 6 days. Most postoperative MIs are non–Q-wave MIs, usually detected within the first 24 hours as a result of surveillance with electrocardiographic and cardiac enzyme testing. Routine acquisition of an ECG in the immediate postoperative period has been shown to be useful in reevaluation of risk in both low- and high-risk populations after major noncardiac surgical procedures. Patients with evidence of ischemia on the immediate postsurgical ECG were found to have a higher risk of subsequent major cardiac complications. Postoperative infarctions are frequently silent, and their presence may only be revealed by the detection of new onset heart failure, hypertension, nausea, altered mental status, or arrhythmias.
PREOPERATIVE CARDIAC RISK ASSESSMENT
Surgical risk assessment encompasses patient-specific, procedure-specific, and institution-specific factors that must be identified in order to estimate individual risk and in turn outline management plans. Variables associated with the perioperative state that require careful scrutiny include the type and urgency of operation, presence and severity of CAD, status of left ventricular (LV) function, advanced age, presence of severe valvular heart disease, significant cardiac arrhythmias, comorbid medical conditions (e.g., cerebrovascular disease, diabetes mellitus, chronic kidney disease), and overall functional status.
Clinical Markers of Increased Risk
Perioperative cardiovascular risk can be stratified further into major, intermediate, and low-risk categories. Acute conditions that often require hospitalization carry more risk than stable chronic conditions. For example, decompensated heart failure would be considered a major risk predictor, likely to require further evaluation and therapy, whereas compensated heart failure would be considered an intermediate-risk condition. Major clinic predictors of high risk for perioperative morbidity and mortality include unstable coronary syndromes (unstable or severe angina or recent MI), decompensated heart failure, significant arrhythmias, and/or severe valvular heart disease. Intermediate predictors include mild angina pectoris, prior MI (by history or pathologic Q waves), compensated or prior heart failure, diabetes mellitus (particularly insulin-dependent), and renal insufficiency. Notably, a history of MI is defined as an intermediaterisk factor. However, an acute (a documented MI <7 days before the exam) or recent MI (>7 days but <1 month before the exam) is considered a major predictor. Minor clinical predictors include advanced age, abnormal ECG, rhythm other than sinus, low functional capacity, history of stroke, and uncontrolled systemic hypertension.
Procedure-Specific Risks
Risks associated with the planned surgical procedure require consideration and can be categorized into high, intermediate, and low-risk categories. High-risk procedures include emergency major operations (particularly in the elderly), aortic and other major vascular surgery, peripheral vascular surgery, aortic surgeries, as well as surgical procedures that are expected to be prolonged and associated with large fluid shifts and/or blood loss. Such high-risk procedures are often associated with a perioperative event rate (i.e., heart failure or MI) of over 5%. Blood loss, large intra- and extravascular fluid shifts, aortic cross clamping (in the case of aortic surgery), duration, and postoperative hypoxemia are factors believed to contribute to this increased risk. The risk of peripheral vascular surgical procedures relates to the likelihood of associated CAD in this patient population. Intermediate-risk procedures (cardiac risk generally <5%) include carotid endarterectomy, head and neck surgeries, intraperitoneal and intrathoracic surgery, orthopedic surgeries, and prostate surgery. Examples of low-risk surgeries (cardiac risk generally <1%) include endoscopic surgery, superficial procedures, breast surgery, and cataract surgery (Table 54.1).
TABLE
54.1 Cardiac Risk Stratification with Examples of Noncardiac Surgical Procedures

aReported cardiac risk, including combined incidence of nonfatal MI and death.
Modified from Fleisher LA, Beckman JA, Brown KA, et al. 2009 ACCF/AHA Focused Update on Perioperative Beta Blockade Incorporated Into the ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. et al. J Am Coll Cardiol. 2009;54:e13–e118, with permission from Elsevier.
Functional Capacity and Stress Testing for Preoperative Assessment of Risk
A patient’s functional capacity is a good indicator of his or her ability to safely tolerate noncardiac surgery. Estimating functional capacity can be accomplished using readily available energy requirement correlates of daily activities (Fig. 54.1). Exercise stress testing represents a particularly useful tool if it will change management among intermediate- or high-risk patients. It is important to note that noninvasive testing is not useful for patients who are at low cardiovascular risk undergoing low- or intermediate-risk procedure. Noninvasive stress testing provides an objective determination of functional status with concurrent assessment for myocardial ischemia or cardiac arrhythmias during stress evaluation. A final aim of supplemental preoperative stress testing is the provision of an objective measure of perioperative and long-term prognosis. The onset of myocardial ischemia at low exercise workload is associated with a significantly elevated risk of both perioperative and long-term cardiac events.
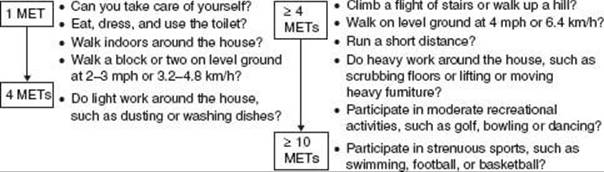
FIGURE 54.1 Estimated energy requirements for typical daily activities. (Adapted from Eagle KA, Berger PB, Calkins H, et al. Guideline update for perioperative cardiovascular evaluation for noncardiac surgery. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines [Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery]. J Am Coll Cardiol. 2002;39:542–553, with permission from Elsevier.)
A majority of patients in need of further preoperative risk stratification are either unable to exercise or have an ECG that is not interpretable. In these instances, a pharmacologic stress agent is utilized in substitute for physical activity, thereby inducing a hyperemic response, which enhances discrepancies in coronary flow, and enabling detection of myocardial ischemia. Dobutamine stress echocardiography and intravenous vasodilator myocardial perfusion scintigraphy utilizing either single positron emission computed tomography (SPECT) or positron emission tomography (PET) detectors represent the two most common techniques in preoperative stress testing for those who cannot exercise. The negative predictive value of these tests is high (99% for dipyridamole thallium, and 93% to 100% for dobutamine stress echo), though the positive predictive value of these tests for perioperative events is low (4% to 20% for intravenous dipyridamole thallium and 7% to 23% for dobutamine stress echocardiography). Therefore, incorporating clinical markers of risk is essential for improving the specificity and positive predictive value of these diagnostic studies. As a guideline, noninvasive testing in preoperative patients is indicated if two or more of the following are present: (a) intermediate clinical predictors (Canadian Class I or II angina, prior MI based on history or pathologic Q waves, compensated or prior heart failure, or diabetes), (b) a poor functional capacity (<4 METs), or (c) a high–surgical-risk procedure (aortic repair or peripheral vascular, prolonged surgical procedures with large fluid shifts or blood loss).
Abnormalities in thallium redistribution and coronary flow among patients with one or more clinical risk factors are associated with a higher incidence of perioperative cardiac events as compared to patients without clinical risk factors. Furthermore, postoperative events increased from 29% to 50% in a population of patients with thallium perfusion defects in the group of patients that were found to have three clinical risk factors as opposed to only one or two variables present. These data, again, highlight the importance of eliciting clinical markers of risk. Finally, the extent of ischemia (i.e., number of abnormal segments), as well as the severity of ischemia, correlates with perioperative cardiac events.
Which Stress Test Is Best?
For most outpatients able to exercise with a normal resting ECG requiring further preoperative risk assessment, treadmill exercise ECG testing represents a cost-effective assessment of functional capacity, if not a slightly less sensitive and specific assessment for myocardial ischemia compared with stress testing utilizing advanced nuclear or echocardiographic imaging. Low-risk patients include those able to exercise without cardiac ischemic symptoms beyond stage II of the Bruce protocol (>7 METs), or achieve a heart rate (HR) over 130 bpm, or over 85% age-predicted maximum HR.
Among patients unable to perform sufficient exercise (at least 4 to 6 METs) for reliable test interpretation or those with abnormal resting ECGs (e.g., LV hypertrophy, left bundle branch block, or digitalis effect), stress testing utilizing an imaging modality such as myocardial perfusion imaging or dobutamine echocardiography is most effective. In many locations, specific expertise and familiarity with a particular stress imaging technique determines which test is used. Either stress imaging technique may be appropriate when used selectively, provided the expertise in a specific institution is satisfactory and commensurate with published investigations. Certain characteristics of these imaging techniques should be kept in mind when deciding on the ideal choice of a test. Both imaging techniques provide high sensitivity for detecting patients at risk for perioperative events, with a commensurate high negative predictive value but suffer from low specificity with corresponding low positive predictive values. Risk for perioperative events is proportional to the amount of myocardium at risk as detected by either modality. Myocardial perfusion imaging utilizing a vasodilator such as dipyridamole, adenosine, or regadenoson should be avoided in patients with significant bronchospasm, critical carotid disease, or in patients with a condition that prevents them from being withdrawn from theophylline preparations. Dobutamine should not be used as a stressor in patients with serious arrhythmias, severe hypertension, or hypotension.
Indications for Angiography
For patients with suggested or proven CAD, coronary angiography should be considered among those with high-risk results during noninvasive testing, unstable angina, and nondiagnostic or equivocal noninvasive tests in a high-risk patient undergoing a high-risk procedure. Coronary angiography may be considered in the setting of intermediate results during noninvasive testing, a nondiagnostic or equivocal noninvasive test in a patient at lower risk undergoing a high-risk procedure, urgent noncardiac surgery in a patient recovering from an acute MI, and in the setting of perioperative MI.
GENERAL APPROACH TO SUCCESSFUL PERIOPERATIVE EVALUATION OF CARDIAC PATIENTS UNDERGOING NONCARDIAC SURGERY
The joint ACC/AHA Task Force approach to the patient undergoing elective noncardiac surgery is based on a Bayesian strategy reliant on clinical markers, including prior CAD evaluation and treatment, functional capacity, and magnitude of the proposed surgical procedure. These general guidelines may be useful in a majority of preoperative assessment. Since publication of the original guidelines in 1996, several studies and subsequent guideline revisions have demonstrated that this stepwise approach is both efficacious and cost effective (Fig. 54.2).
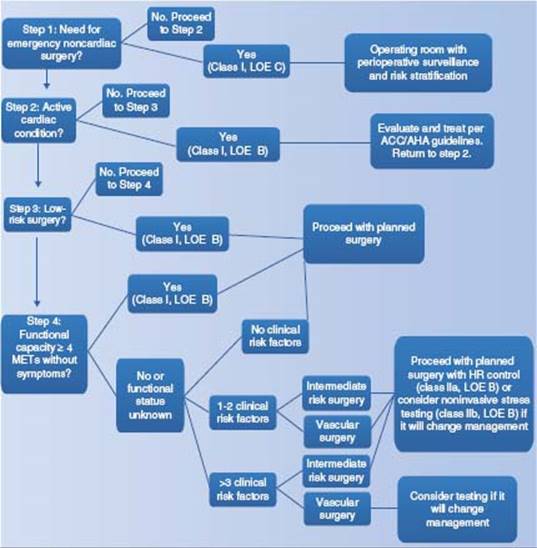
FIGURE 54.2 Algorithm of preoperative cardiac evaluation for patients over age 50 years undergoing noncardiac surgery. LOE: level of evidence; METs: metabolic equivalent of task. (Modified from Fleisher LA, Beckman JA, Brown KA, et al. 2009 ACCF/AHA Focused Update on Perioperative Beta Blockade Incorporated Into the ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. J Am Coll Cardiol. 2009;54:e13–e118, with permission from Elsevier.)
The first step in preoperative assessment for noncardiac surgery is determining the urgency of the procedure. When encountering a patient in need of emergency surgery, time often does not allow for a preoperative evaluation. In such cases, proceeding directly to the surgical suite with careful perioperative surveillance and postoperative risk stratification is most appropriate. When the proposed surgery is determined to be elective, clinical risk stratification based on type of surgery is important (see Table 54.1).
Clinical risk factor assessment is especially critical for patients with three or more clinical risk factors if further testing will affect management. Patients with known CAD should be assessed for symptoms as well as a history of coronary revascularization within the preceding 5 years. Screening for interval development of ischemic heart disease signs or symptoms within that time frame may allow for further invasive or noninvasive testing prior to the planned operation. If the patient has one or two clinical risk factors, often the patient can proceed with the planned surgery. If a history for interval ischemic heart disease signs or symptoms is determined to be negative, further testing is not necessary. If the patient has undergone a coronary evaluation within the previous 2 years (assuming risk was adequately assessed and findings were favorable) and is currently without symptoms, further testing is usually not necessary. The presence of one or more major clinic predictors of risk (i.e., unstable coronary disease, decompensated heart failure, symptomatic arrhythmias, and/or severe valvular heart disease) mandates more intensive preoperative evaluation and management. Cancellation or delay of surgery until these active cardiac conditions has been satisfactorily identified and treated is usually necessary to mitigate this risk.
A number of cardiovascular risk prediction models have been developed from broad screening for markers and multivariate analyses that predict preoperative cardiac mortality. The six independent risk correlates for perioperative MI of the Revised Cardiac Risk Index have been extensively studied and validated, and are among the most widely used. The presence or history of ischemic heart disease (by history or ECG), compensated or prior heart failure, cerebrovascular disease, insulin-dependent diabetes mellitus, and renal insufficiency (defined as preoperative creatinine >2 mg/dL) stratify clinical risk for perioperative coronary events. Assessment of functional capacity (see Fig. 54.1) and magnitude of surgeryspecific risk (see Table 54.1) along with clinical risk factor analysis together allows for a rational approach to identify patients most likely to benefit from noninvasive testing.
Patients without major predictors but with intermediate predictors of clinical risk, and moderate or excellent functional capacity, generally may undergo intermediaterisk surgery with little likelihood of perioperative death or MI. Conversely, further noninvasive testing is often considered for patients with poor or moderate functional capacity undergoing higher-risk surgery or patients with more than two intermediate predictors. Assessment of LV function is reasonable for patients with preoperative dyspnea of unknown origin. In addition, patients with heart failure with worsening dyspnea or change in clinical status who have not undergone noninvasive assessment of ventricular function within 12 months may also benefit from reassessment. Conversely, routine perioperative assessment of LV function is not recommended in patients without such histories.
Noncardiac surgery is generally safe for patients without major or intermediate clinical predictors and a functional capacity of more than 4 METs. Additional testing may be considered for patients without clinical markers but considered to have a poor functional capacity faced with high-risk operations, particularly those with several minor predictors of risk who are scheduled for vascular surgery. Finally, the results of noninvasive testing should be used to determine further preoperative management, including initiation of beta-blockers and pursuit of coronary revascularization if indicated.
Class I ACCF/AHA Recommendations for Perioperative Cardiac Assessment:
![]() Patients who have a need for emergency noncardiac surgery should proceed to the operating room and continue perioperative surveillance and postoperative risk stratification and risk factor management. (Level of Evidence: C)
Patients who have a need for emergency noncardiac surgery should proceed to the operating room and continue perioperative surveillance and postoperative risk stratification and risk factor management. (Level of Evidence: C)
![]() Patients with active cardiac conditions should be evaluated and treated per ACC/AHA guidelines and, if appropriate, consider proceeding to the operating room. (Level of Evidence: B)
Patients with active cardiac conditions should be evaluated and treated per ACC/AHA guidelines and, if appropriate, consider proceeding to the operating room. (Level of Evidence: B)
![]() Patients undergoing low-risk surgery are recommended to proceed to planned surgery. (Level of Evidence: B)
Patients undergoing low-risk surgery are recommended to proceed to planned surgery. (Level of Evidence: B)
![]() Patients with poor (< 4 METs) or unknown functional capacity and no clinical risk factors should proceed with planned surgery. (Level of Evidence: B)
Patients with poor (< 4 METs) or unknown functional capacity and no clinical risk factors should proceed with planned surgery. (Level of Evidence: B)
MANAGEMENT OF OTHER CARDIOVASCULAR CONDITIONS
Hypertension
Severe hypertension (systolic blood pressure >180 mm Hg, diastolic blood pressure >110 mm Hg) should be controlled preoperatively when possible. While hypertension is not an independent risk factor for perioperative complications, the condition does serve as a marker for potential CAD. The decision to delay surgery because of severe hypertension needs to take into account the urgency of the noncardiac surgery. Continuation of preoperative antihypertensive treatment through the perioperative period is important.
Valvular Heart Disease
The indications for evaluation and treatment of valvular heart disease are the same as those in the nonoperative setting. Symptomatic stenotic lesions are associated with an increased risk of perioperative congestive heart failure or shock, and may require balloon valvuloplasty or valve replacement before the cardiac surgery. Symptomatic regurgitant lesions, on the other hand, are usually well tolerated perioperatively and can often be optimized with medical therapy and monitoring. This strategy assumes normal LV function, and therefore, relatively adequate cardiac reserve. These recommendations are appropriate if a delay in the noncardiac surgery is thought to have potentially dire consequences. In the case of severe valvular regurgitation and LV dysfunction in which cardiac reserve is limited, instability during noncardiac surgery is likely, and therefore, valve surgery prior to noncardiac surgery may be warranted.
Hypertrophic Obstructive Cardiomyopathy
Patients with hypertrophic obstructive cardiomyopathy generally tolerate surgery well, provided that attention is given to a few critical factors. Anesthetic-associated decrease in peripheral vascular resistance, hypovolemia, and adrenergic stimulation may result in tachycardia and depletion of LV preload. These factors may precipitate hemodynamic deterioration and should be avoided if possible. HR control to allow for adequate ventricular filling is useful. For these reasons, perioperative beta-blockade may provide a reduced HR and negative inotropic effects that can help to avoid hemodynamic instability.
Dilated Cardiomyopathy
Patients with dilated cardiomyopathy also are at increased risk for perioperative heart failure. Management is directed at optimizing preoperative hemodynamics and providing intensive postoperative medical therapy and surveillance.
PERIOPERATIVE MEDICAL THERAPY
Although few randomized controlled trials have been performed to determine the optimal perioperative medical regimen for patients with cardiac disease undergoing noncardiac surgery, study data strongly support perioperative betablocker therapy among individuals at elevated risk for cardiovascular events. Among patients with more than one major clinical risk factor undergoing at least intermediate-risk surgery, beta-blockade reduces the frequency of postoperative ischemic events, MI, and perioperative death. A randomized trial of beta-blocker therapy versus standard treatment in a series of patients with positive dobutamine stress echocardiograms demonstrated a positive benefit to bisoprolol in reducing perioperative death and nonfatal MI. These data, as well as prior studies, support the use of beta-blocker therapy unless it is contraindicated. Beta-blocker therapy should be initiated days to weeks before the procedure and the dose titrated to a resting HR of 50 to 60 beats/min (bpm). Fixed-dose regimens of perioperative beta-blockade are to be generally avoided, as they have not been shown to be as efficacious in intermediate-risk groups. Perioperative withdrawal of beta-blockers should be avoided, because of associated risks of angina pectoris and MI. Hypotension (systolic blood pressure <100 mm Hg) and bradycardia (HR <50 bpm) associated with beta-blocker use is to be avoided, as hypotension is associated with increased perioperative morbidity and mortality.
Statin therapy and reduction of lipid levels has proven to be highly efficacious in secondary prevention of cardiac events in a number of large randomized trials. The effectiveness of statins to reduce cardiovascular events in the perioperative setting is less conclusive, although data suggest an overall protective effect against cardiac complications during noncardiac surgery. Statin therapy should be continued if already initiated. It may be reasonable to start statin therapy preoperatively for patients undergoing high-risk procedures and in those with one clinical risk factor or more undergoing intermediate-risk surgical procedures.
PERIOPERATIVE CORONARY REVASCULARIZATION
Preoperative risk stratification tools provide the greatest advantage to patients who are correctly stratified as high risk, allowing for preparation and preoperative management that may mitigate cardiac risk associated with noncardiac surgery. Those stratified to the highest-risk group often have known CAD with symptoms of ischemic heart disease, reduced LV function, or other clinical risk factors. Nevertheless, prophylactic preoperative revascularization of severe coronary stenosis may not reduce perioperative complications related to ischemia. Attempts to invasively characterize CAD and potentially perform revascularization should be reserved for a select group of patients and circumstances. In essence, only those patients who would otherwise gain a mortality benefit from coronary revascularization, regardless of whether a noncardiac surgery is planned, should undergo such attempts at preoperative coronary intervention.
Coronary revascularization before noncardiac surgery is useful for patients with stable angina and significant left main trunk stenosis or 3-vessel disease. Patients with stable angina and 2-vessel disease that includes proximal stenosis of the left anterior descending artery and either LV ejection fraction of 0.50 or demonstrable ischemia on noninvasive testing also enjoy a benefit from preoperative revascularization. Coronary revascularization should also be considered before noncardiac surgery for those with high-risk unstable angina, non–ST-segment elevation MI, or acute ST-segment elevation MI as is recommended in the general percutaneous intervention (PCI) guidelines. Routine prophylactic coronary revascularization among patients with stable CAD is not recommended.
Class I ACCF/AHA Recommendations for Preoperative Coronary Revascularization with coronary artery bypass graft (CABG) or PCI:
![]() Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have significant left main coronary artery stenosis. (Level of Evidence: A)
Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have significant left main coronary artery stenosis. (Level of Evidence: A)
![]() Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have 3-vessel disease. (Level of Evidence: A)
Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have 3-vessel disease. (Level of Evidence: A)
![]() Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have 2-vessel disease with significant proximal left anterior descending coronary artery stenosis and either ejection fraction <0.50 or demonstrable ischemia on noninvasive testing. (Level of Evidence: A)
Coronary revascularization before noncardiac surgery is useful in patients with stable angina who have 2-vessel disease with significant proximal left anterior descending coronary artery stenosis and either ejection fraction <0.50 or demonstrable ischemia on noninvasive testing. (Level of Evidence: A)
![]() Coronary revascularization before noncardiac surgery is recommended for patients with high-risk unstable angina or non–ST-segment elevation MI. (Level of Evidence: A)
Coronary revascularization before noncardiac surgery is recommended for patients with high-risk unstable angina or non–ST-segment elevation MI. (Level of Evidence: A)
![]() Coronary revascularization before noncardiac surgery is recommended for patients with acute ST-segment elevation MI. (Level of Evidence: A)
Coronary revascularization before noncardiac surgery is recommended for patients with acute ST-segment elevation MI. (Level of Evidence: A)
When coronary revascularization before noncardiac surgery is indicated, the mode of revascularization depends on guideline consensus statements. Indications for CABG before noncardiac surgery are identical to those reviewed in the ACC/AHA 2004 Guideline Update for CABG Surgery Preoperative PCI in patients undergoing elective noncardiac surgery follows the ACC/AHA 2005 Guideline Update for PCI.
In the uncommon event that preoperative PCI with stent placement is indicated, important considerations may affect the timing of coronary intervention and the proposed operation (see Fig. 54.3). Premature discontinuation of antiplatelet therapy, including aspirin or thienopyridine, may unnecessarily increase the risk of in-stent thrombosis and subsequent acute MI. Elective noncardiac surgery is not recommended within 4 to 6 weeks of bare metal stent or within 12 months of drug-eluting stent implantation in patients in whom thienopyridine or aspirin therapy requires discontinuation perioperatively. Balloon angioplasty represents an alternative modality for coronary revascularization, although noncardiac surgery is not recommended within 4 weeks of such a procedure.
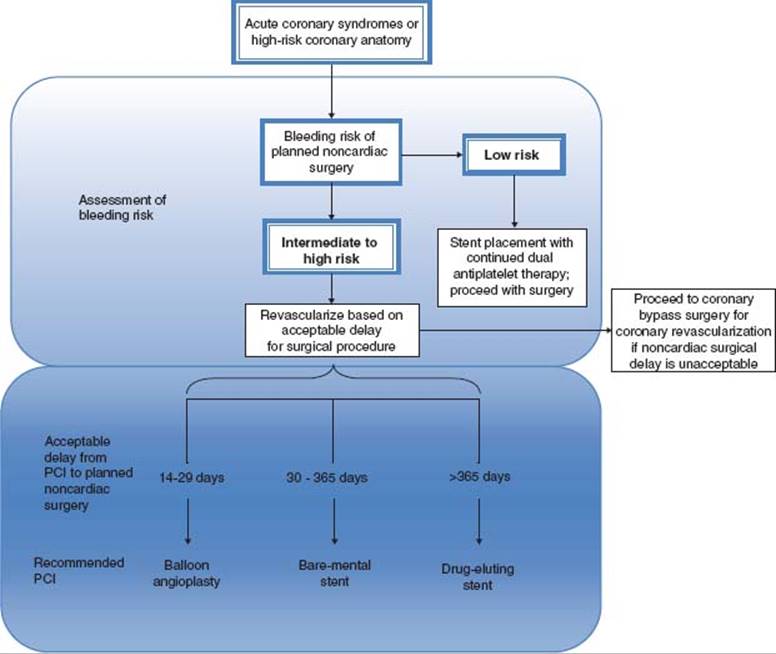
figure 54.3 Pathway for coronary arterial revascularization prior to non-elective noncardiac surgery in setting of acute coronary syndromes or high-risk coronary anatomy. PCI: percutaneous intervention. (Modified from Fleisher LA, Beckman JA, Brown KA, et al. 2009 ACCF/AHA Focused Update on Perioperative Beta Blockade Incorporated Into the ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. J Am Coll Cardiol. 2009;54:e13–e118, with permission from Elsevier.)
POSTOPERATIVE MYOCARDIAL INFARCTION MANAGEMENT
Perioperative MI is commonly associated with minimal symptoms, nonspecific ST- or T-wave changes, and exhibit small increases in cardiac enzyme leaks. Surveillance for perioperative MI with troponin measurement is recommended in patients with ECG changes or chest pain typical for acute coronary syndrome. Patients undergoing intermediate-risk or vascular surgical procedures who are otherwise asymptomatic may also benefit from such monitoring.
Patients with CAD, especially those with prior MI, are at risk for postoperative MI. Most commonly, perioperative MI is secondary to increased myocardial oxygen demand outstripping coronary arterial oxygen supply Whether as a case of “supply–demand mismatch” or a more traditional scenario of coronary thrombosis and acute vascular occlusion, perioperative MI engenders a high rate of cardiovascular morbidity and mortality. Coronary stent thrombosis represents a unique problem after discontinuation of aspirin or thienopyridine antiplatelet therapy that carries a fatality rate as high as 45%. Stent thrombosis may also represent an untoward consequence of PCI, which lends credence to a strategy of careful screening before performing prophylactic preoperative coronary revascularization.
AREAS OF CONTROVERSY AND FURTHER RESEARCH
The latest release of the ACC/AHA Perioperative Guidelines for Cardiovascular Evaluation and Care for Noncardiac Surgery includes a number of important studies that have clarified the importance of perioperative beta-blockade and the limited role of coronary revascularization prior to noncardiac surgery. Nevertheless, a number of important questions and topics would benefit from further research seeking to clarify remaining concerns. The following lists important topics to be addressed with further research pertaining to cardiac risk of noncardiac surgery:
1. The efficacy and cost-effectiveness of various noninvasive stress testing methods in determining risk and reducing cardiac complications
2. Establishment of optimal guidelines for selected subgroups of patients undergoing noncardiac surgery, especially among the elderly and women
3. The optimal time delay between an acute (≤7 days) or recent (8 to 30 days) for myocardial infarction among patients undergoing elective noncardiac surgery is uncertain and requires further evaluation
4. Establishing improved guidelines for patients with valvular heart disease undergoing noncardiac surgery
5. Establishment of the efficacy of surveillance and monitoring of patients for myocardial ischemia and infarction perioperatively
CONCLUSIONS
The effective preoperative cardiovascular evaluation requires careful regard to individual clinical risk factors, surgery-specific considerations, and communication to determine the appropriate timing of further cardiac testing and treatments before proceeding to noncardiac surgery. Preoperative testing should be limited to those instances in which the results of testing will impact subsequent care. The patient is best served by recommendations that lower their immediate perioperative risk while assessing the need for subsequent postoperative surveillance and risk stratification. The recently updated 2009 ACC/AHA Guidelines on Perioperative Cardiac Evaluation and Care for Noncardiac Surgery provide a rational, conservative, and systematic approach. These guidelines promote clinical risk-factor stratification while highlighting the importance of perioperative beta-blockade in selected groups and acknowledgment that neither routine noninvasive stress testing nor prophylactic coronary artery revascularization is routinely required to lower the risk for most patients undergoing a noncardiac operation.
SUGGESTED READINGS
Devereaux PJ, Yang H, Yusuf S, et al. Effects of extendedrelease metoprolol succinate in patients undergoing non-cardiac surgery (POISE trial): a randomised controlled trial. Lancet. 2008;371:1839–1847.
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). J Am Coll Cardiol. 2002;39:542–553.
Eagle KA, Coley CM, Newell JB, et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med. 1989;110:859–866.
Eagle KA, Guyton RA, Davidoff R, et al. ACC/AHA 2004 guideline update for coronary artery bypass graft surgery: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1999 Guidelines for Coronary Artery Bypass Graft Surgery). J Am Coll Cardiol. 2004;44:e213–310.
Fleisher LA, Beckman JA, Brown KA, et al. 2009 ACCF/AHA focused update on perioperative beta blockade incorporated into the ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery. J Am Coll Cardiol. 2009;54:e13–e118.
Goldman L, Caldera DL, Nussbaum SR, et al. Multifactorial index of cardiac risk in noncardiac surgical procedures. N Engl J Med. 1977;297:845–850.
Landesberg G. The pathophysiology of perioperative myocardial infarction: facts and perspectives. J Cardiothorac Vasc Anesth. 2003;17:90–100.
Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100: 1043–1049.
McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med. 2004;351:2795–2804.
Poldermans D, Boersma E, Bax JJ, et al. The effect of bi- soprolol on perioperative mortality and myocardial infarction in high-risk patients undergoing vascular surgery. Dutch Echocardiography Cardiac Risk Evaluation Applying Stress Echocardiography Study Group. N Engl J Med. 1999;341:1789–1794.
Poldermans D, Schouten O, Vidakovic R, et al. A clinical randomized trial to evaluate the safety of a noninvasive approach in high-risk patients undergoing major vascular surgery: the DECREASE-V Pilot Study. J Am Coll Cardiol. 2007;49:1763–1769.
Smith SC Jr, Feldman TE, Hirshfeld JW Jr, et al. ACC/AHA/SCAI 2005 Guideline Update for Percutaneous Coronary Intervention-Summary Article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/SCAI Writing Committee to Update the 2001 Guidelines for Percutaneous Coronary Intervention). J Am Coll Cardiol. 2006;47:216–235.
QUESTIONS AND ANSWERS
Questions
1. You are asked to evaluate a 55-year-old man with a history of prior myocardial infarction (MI) in preparation for an abdominal aortic aneurysm repair. A dobutamine stress echocardiogram demonstrates a single segment of basal inferior akinesis on rest and stress imaging. Your recommendation to the referring physician is:
a. Clear the patient for surgery with beta-blocker prophylaxis.
b. Stress single positron emission computed tomography (SPECT) thallium nuclear imaging
c. Coronary angiography
d. Cancellation of surgery
e. Stent grafting, in hopes of avoiding major aortic surgery
2. A 70-year-old man with hypertension and a recently diagnosed solitary pulmonary nodule is scheduled for pulmonary wedge resection. He is otherwise healthy, active, and regularly plays 18 holes of golf. His Electrocardiogram (ECG) reveals left ventricular (LV) hypertrophy with secondary repolarization changes consistent with a strain pattern. Your recommendation is:
a. Stress echocardiography for risk stratification
b. Clear the patient for surgery.
c. Coronary angiography
d. Echocardiogram
e. Stress SPECT thallium imaging
3. An 80-year-old woman with hypertension, coronary artery disease (CAD), and chronic heart failure recently suffered a hip fracture requiring open reduction and internal fixation. She lives with family but is known to be inactive, primarily because of arthritis. Your recommendation is:
a. Clear the patient for the orthopedic procedure with beta-blocker initiated prior to her operation.
b. Coronary angiography
c. Dobutamine stress echocardiography for risk stratification
d. Echocardiogram, and if LV function is normal, clear the patient for surgery.
e. Exercise stress SPECT thallium
4. A 78-year-old woman with a history of chronic systolic heart failure, chronic obstructive pulmonary disease, insulin-dependent diabetes mellitus, and chronic kidney disease is scheduled for cataract surgery. She describes chronic angina after ascending over two flights of stairs that resolves with rest. Your recommendation is:
a. Dipyridamole stress SPECT thallium imaging
b. Coronary angiography
c. Clear the patient for cataract surgery.
d. Echocardiogram, and if LV function is normal, clear the patient for surgery.
e. Exercise stress echocardiography
5. A 55-year-old male with iliofemoral peripheral arterial disease and claudication presents for preoperative evaluation with plan for infrainguinal arterial bypass surgery. Which of the following provides greatest utility for further cardiac risk stratification in this patient?
a. Exercise ECG
b. Diagnostic coronary angiography
c. Dobutamine stress echocardiography
d. Dipyridamole thallium myocardial perfusion imaging
e. Clinical evaluation
Answers
1. Answer A: The dobutamine echocardiogram demonstrates a fixed regional wall motion abnormality involving the base of the inferior wall and base of the septum consistent with a right coronary artery territory scar. The remaining LV wall segments were contracting normally at rest with a resting ejection fraction of approximately 55%. Peak dobutamine stress images reveal an appropriate improvement in wall motion involving the left anterior descending and circumflex coronary territories with no change in the wall motion involving the inferior and basal septum. These results are consistent with right coronary artery territory scar and no evidence for ischemia. Based on this negative echocardiogram for ischemia, this patient with an intermediate clinical predictor, namely, prior MI may be considered at a relatively low perioperative risk for a cardiac event and therefore could be cleared for his procedure with beta-blocker prophylaxis.
2. Answer B: This 70-year-old man with hypertension is scheduled for an intermediate-risk surgery. He has a history of hypertension with ECG changes consistent with LV hypertrophy. Other than his age and an abnormal baseline ECG, this gentleman has no other significant clinical predictors of perioperative risk. Reportedly, he has a very good exercise capacity, which would place him in the moderate to excellent category for functional capacity based on his ability to regularly play 18 holes of golf without difficulty. Based on the absence of significant clinical predictors, as well as a good exercise capacity, this patient can be cleared for his wedge resection with a low perioperative risk of sustaining a cardiac event.
3. Answer C: Because of the one reported critical predictor, namely, the prior history of congestive heart failure, as well as a suspected poor functional capacity, this patient should undergo further risk stratification using a pharmacologic stress imaging study. Stress echocardiography can be considered a good stress imaging modality in a patient with hypertensive heart disease, as the microvascular disease associated with hypertensive heart disease may result in abnormalities in coronary flow reserve that may in turn lead to a false- positive result when using nuclear perfusion imaging.
4. Answer C: This 78-year-old woman does have > 3 clinical predictors of risk. However, she was noted to have good functional capacity and is undergoing a very lowrisk surgical procedure, so she can be cleared for the cataract surgery with a low anticipated risk for adverse events.
5. Answer E: In any patient undergoing preoperative evaluation, an assessment of clinical predictors of risk is central to initiating an appropriate workup prior to the noncardiac surgery. If this man with vascular disease were found, on clinical evaluation, to have a history of one or more intermediate or major clinical predictors of risk, or were found to be unable to exercise to a moderate level, further risk stratification with pharmacologic stress imaging would be indicated.