Pulmonary valve is quite often overlooked while performing echocardiography in adults because of poor acoustic window and rare organic involvement. However, study of pulmonary valve and its hemodynamics provide useful information. Pulmonary valve can get affected by rheumatic process, carcinoid heart disease, trauma, infective endocarditis, myxomatous degeneration and congenital dysplasia. Functional pulmonary regurgitation is frequent and provides information about pulmonary pressures. Severe pulmonary regurgitation may result in the right ventricular (RV) volume overload and heart failure. Echocardiographic examination of the pulmonary valve is critical before the Ross procedure.
ANATOMY OF THE PULMONARY VALVE
The pulmonary valve divides the right ventricular outflow tract (RVOT) from the pulmonary artery. In normal conditions, the pulmonary valve prevents regurgitation of blood from the pulmonary artery back to the right ventricle. It is a semilunar valve with three cusps, and it is located anterior, superior and slightly to the left of the aortic valve (Fig. 8.1).
The pulmonary valve is formed by three cusps, each with a fibrous node at the midpoint of the free edges as well as lunulae, which are the thin, crescent-shaped portions of the cusps that serve as the coaptive surfaces of the valve (Figs 8.2 and 8.3).

Fig. 8.1 : Schematic short-axis view showing relationship of pulmonary valve leaflets. Left lower panel shows transesophageal echocardiographic image.
• The cusps of the pulmonary valve are supported by freestanding musculature with no direct relationship with the muscular septum.1 The normal pulmonary valve is enclosed in a proximal sleeve of free-standing right ventricular infundibulum supporting the fibroelastic walls of the pulmonary sinuses at the anatomical ventriculoarterial junction. The valvular leaflets are attached in semilunar fashion across this junction, delimiting the extent of the valvular sinuses.
• The cusps are much thinner and lack a fibrous continuity with the anterior leaflet of the right atrioventricular valve.
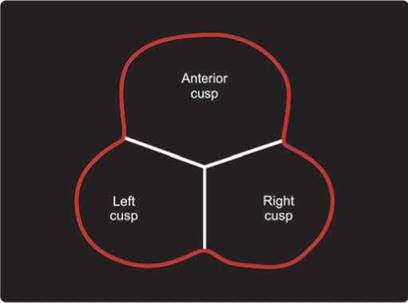
Fig. 8.2 : Schematic diagram showing three cusps or leaflets of the pulmonary valve without any definite fibrous annulus.

Fig. 8.3 : Three-dimensional echocardiographic en face view of the pulmonary valve in late diastole showing all the three leaflets and lack of any definite annulus. (AV: Aortic valve; A: Anterior pulmonary leaflet; L: Left; R: Right leaflets).
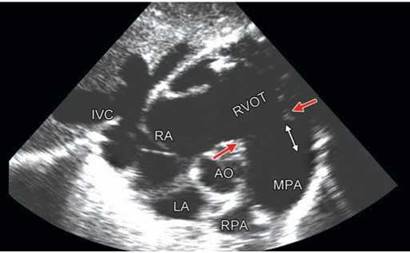
Fig. 8.4 : Transthoracic echocardiographic subcostal coronal view. Red arrows point to ventriculoarterial junction and the white arrow shows the extent of pulmonary sinuses of Valsalva.
• the cusps are defined by their relationship to the aortic valve and are thus termed anterior or nonseptal, right and left cusps.2,3 ttey can also be defined by their relationship to a commissure found in the pulmonary and aortic valves and hence termed right adjacent (right facing), left adjacent (left facing) and opposite (non-facing).
• Anatomical studies have failed to demonstrate any well-defined fibrous “annulus” that could be of clinical relevance.
Pulmonary root is that part of the RVOT that supports the leaflets of the pulmonary valve.4 It is limited distally by the sinotubular junction (Fig. 8.4). Its components are:
• Sinuses of Valsalva
• Valvular leaflets
• Interleaflet triangles
• Right ventricular muscular infundibulum
the sinutubular junction separates the pulmonary sinuses from the tubular component of the pulmonary trunk. It also is the level of insertion of the peripheral ends of the zones of apposition between the leaflets.
the sinuses of Valsalva are the three parts of the pulmonary root confined proximally by attachments of the valvular leaflets and distally by the sinotubular junction. According to their relation to the aorta, these are labeled as left-facing, right-facing and non-facing sinuses.
Commissures are the areas on the coapting surface of the arterial valvular leaflets.
the interleaflet triangles are the areas of arterial wall proximal to the attachments of the leaflets that are incorporated within the ventricular cavity.
The ventriculoarterial junction is the zone of union between the muscular right ventricular infundibulum and the fibroelastic pulmonary arterial wall. It has no fibrous continuity with the tricuspid annulus (Fig. 8.5).
Pulmonary valve disorder can either present as right ventricular outflow obstruction [pulmonary stenosis (PS)] or valvular regurgitation (PR) or combination of the two.
Pulmonary stenosis is described as lesions that are collectively associated with obstruction to the RVOT. Stenosis may be valvular, subvalvular or supravalvular
(Figs 8.6 to 8.10). It is the most common cause of congenital outflow tract obstruction, resulting in decreased flow from the right ventricle to the pulmonary arteries.5 Isolated RVOT obstruction is pulmonary valvular stenosis in 80% of the cases.
Stenosis proximal to the pulmonary valve may result from a number of causes, both congenital and acquired. Congenital ventricular septal defect may also be associated with RVOT obstruction secondary to development of obstructive midcavitary or infundibular muscle (Figs 8.6 and 8.7).
Stenosis of the pulmonary artery above the valve (distal to the valve) may occur in the main pulmonary trunk at the bifurcation, or more distally in the branch vessels. In rare instances, a membrane just above the valve may cause stenosis (Figs 8.9 and 8.10).
Valvular PS is of four types:
1. Classic PS with fusion of commissures with no or mild leaflet thickening and doming of the leaflets (Figs 8.8 and 8.11)
Most cases of pulmonary valvular stenosis are congenital. Often times, the valvular abnormality is associated with syndromes such as Noonan syndrome and Leopard syndrome. The trileaflet pulmonary valve ranges from thickened or partially fused commissures to an imperforate valve.5 The pulmonary valve in valvular PS can be unileaflet, bileaflet or trileaflet (Figs 8.12 and 8.13).
2. Dysplastic PS with no commissural fusion6 but obstruction due to bulky and voluminous leaflets (Figs 8.14 and 8.15)
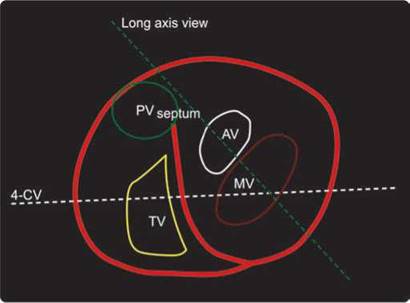
Fig. 8.5 : Anatomical position of the four cardiac valves.
3. Annular stenosis with rudimentary or absent leaflets7-9
4. Any combination of the above (Figs 8.16 to 8.20)
Supravalvular lesion may occur in the setting of
tetralogy of Fallot, Williams syndrome, Alagille syndrome, as well as Noonan syndrome.10
The dysplastic valves show cauliflower-like changes that severely thicken the leaflets and obstruct the right ventricular outflow merely by their size.9 The zones of apposition between the dysplastic leaflets are not fused, the sinuses are deep and the lines of attachment are semilunar, all as seen in normal hearts. Only the distal parts of the leaflets are affected by the myxomatous degeneration (Fig. 8.21); the most proximal part, originating from the right ventricular musculature, is smooth and delicate.
Acquired stenosis of the pulmonary valve is very uncommon. Rheumatic PS is rare even when the valve is affected by the rheumatic process.11 Carcinoid disease is the commonest cause of acquired pulmonary valve disease.12 Various tumors may compress the RVOT.
Pulmonary valve stenosis may also occur as part of more complex congenital lesions such as tetralogy of Fallot,13 complete atrioventricular canal, double outlet RV and univentricular heart (Figs 8.22 and 8.23).
Peripheral pulmonary artery stenosis may coexist with valvular PS such as in Noonan syndrome and Williams syndrome (Fig. 8.24).
Quantitative assessment of PS severity is based mainly on the transpulmonary pressure gradient.14 Calculation of pulmonary valve area by planimetry is not possible
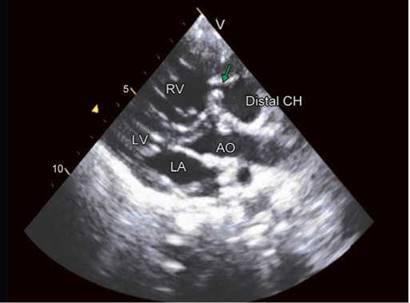
Fig. 8.6 : Parasternal long-axis view showing discrete stenosis in the right ventricular cavity (arrow) at the level of aortic valve.
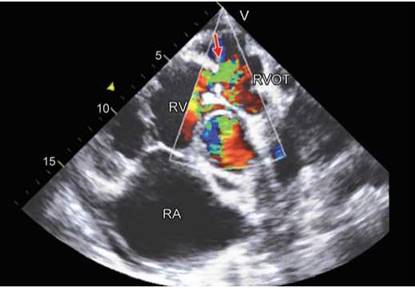
Fig. 8.7 : Short-axis view showing narrow turbulent color Doppler jet through the subvalvular stenosis just proximal to the pulmonary valve (arrow).

Figs 8.8A and B: Two-dimensional echocardiographic image of valvular pulmonary stenosis. Doming of the pulmonary valve with narrowed orifice (arrows); (B) Three-dimensional echocardiographic image of valvular pulmonary stenosis. Doming of the pulmonary valve with narrowed orifice (arrows).

Fig. 8.9 : Transthoracic echocardiographic short-axis view of the right ventricular outflow tract. Red arrows point to the pulmonary valve, while the yellow arrows show discrete supravalvular membranous stenosis.

Fig. 8.10: Color Doppler image of the right ventricular outflow tract. Note turbulent flow across the pulmonary valve due to valvular pulmonary stenosis (red arrows) and very narrow jet across supravalvular obstruction (yellow arrows).

Fig. 8.11: Valvular pulmonary stenosis with continuous wave Doppler interrogation in the right panel.
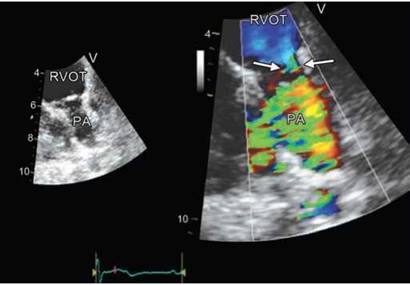
Fig. 8.12: Two-dimensional echocardiographic appearance of a unicuspid pulmonary valve (left panel) with no doming in systole. The right panel shows eccentric and narrow orifice due to stenosis.
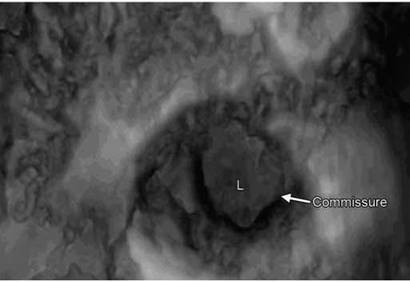
Fig. 8.13: Unicuspid pulmonary valve with single leaflet and single commissure.

Fig. 8.14: Dysplastic pulmonary valve filling the right ventricular outflow tract and causing obstruction. Right panel shows continuous wave interrogation with peak velocity of 500 cm/s.
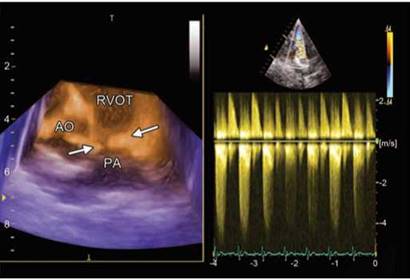
Fig. 8.15: Three-dimensional echocardiographic short-axis image showing dysplastic pulmonary valve (arrows) with evidence of stenosis as well as regurgitation (right panel).
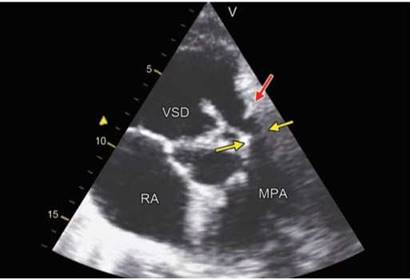
Fig. 8.16: Transthoracic echocardiographic modified apical short- axis image showing ventricular septal defect (VSD) and rudimentary pulmonary valve (red arrow), supravalvular pulmonary stenosis (yellow arrows) and main pulmonary artery hypoplasia.

Fig. 8.17: Subpulmonic stenosis (small arrow) with vestigial pulmonary valve and annular stenosis.

Fig. 8.18: Absent pulmonary valve with dilated pulmonary artery and hour-glass right ventricular outflow tract.

Fig. 8.19: Three-dimensional echocardiographic image. Absent pulmonary valve with so-called annular stenosis. Nodular constriction at the ventriculoarterial junction.
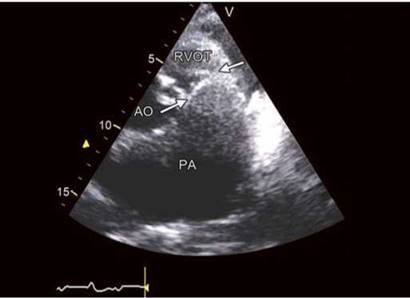
Fig. 8.20: Massive pulmonary artery dilatation with vestigial pulmonary valve tissue.
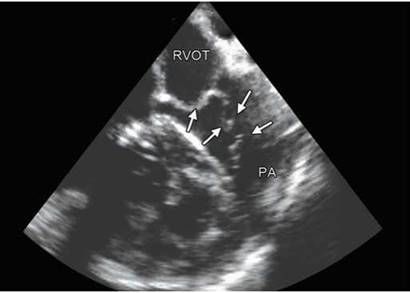
Fig. 8.21: Dysplastic pulmonary valve with redundant distal parts.
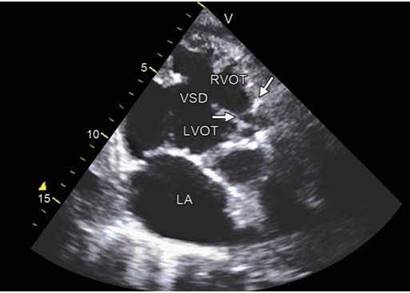
Fig. 8.22: Tetralogy of Fallot with narrow right ventricular outflow tract and pulmonary valve stenosis (arrow).
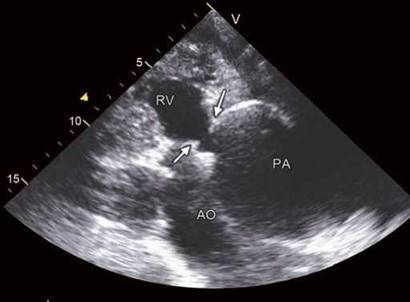
Fig. 8.23: Double-outlet right ventricle with pulmonary stenosis and aneurysmal dilatation of pulmonary artery.
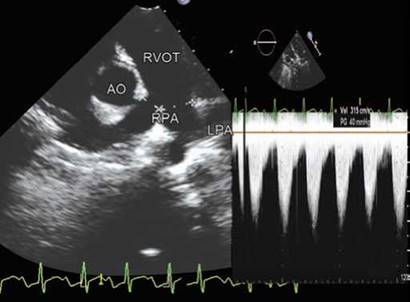
Fig. 8.24: Right pulmonary artery stenosis with a peak gradient of 40 mm Hg in Williams syndrome. Diastolic spilling maintains the flow in the affected lung (right panel).

Fig. 8.25: Hemispheric proximal isovelocity surface area in a patient with severe pulmonary stenosis. Narrow vena contracta is seen.

Fig. 8.26: Color-flow map-adjusted continuous wave Doppler to obtain transpulmonary gradients. A peak velocity of 4.88 m/s indicates severe pulmonary stenosis.
since the required image plane is in general not available. Continuity equation or proximal isovelocity surface area method, although feasible in principle, has not been validated in PS and is rarely performed (Fig. 8.25).
• The estimation of the systolic pressure gradient is derived from the transpulmonary velocity flow curve using the simplified Bernoulli's equation ДР = 4v2 (Fig. 8.26).
• This estimation is reliable, as shown by the good correlation with invasive measurement using cardiac catheterization.15,16
• Continuous wave (CW) Doppler is used to assess the severity when even mild stenosis is present. It is important to line up the Doppler sample volume parallel to the flow with the aid of color flow mapping where appropriate.
• In adults, this is usually most readily performed from a parasternal short-axis view but in children and in some adults, the highest gradients may be found from the subcostal window.
• A modified apical five-chamber view may also be used where the transducer is angled clockwise to bring in the RVOT. Ideally, the highest velocity in multiple views should be used for the determination.15,16

Fig. 8.27: Severe pulmonary regurgitation causing over-estimation of the forward flow velocities.

Fig. 8.28: Continuous wave Doppler pattern of dynamic infundibular obstruction.
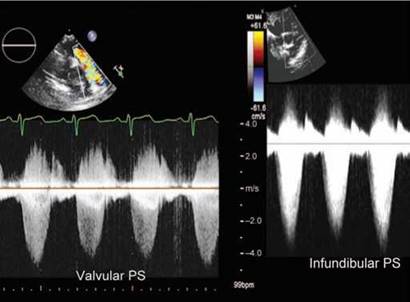
Fig. 8.29: Continuous wave Doppler envelope of valvular PS (left panel) versus fixed infundibular PS (right panel). The latter begins late and is triangular in shape. (PS: Pulmonary stenosis).
• In most instances of valvular PS, the modified Bernoulli's equation is applicable and there is no need to account for the proximal velocity as this is usually < 1 m/s.
• In the setting of subvalvular or infundibular stenosis and PS as part of a congenital syndrome or as a result of RV hypertrophy, the presence of two stenoses in series may make it impossible to ascertain precisely the individual contribution of each.
• In addition, such stenoses in series may cause significant PR resulting in a higher Doppler gradient compared with the net pressure drop across both stenoses (Fig. 8.27).
• Pulsed wave Doppler may be useful to detect the sites of varying levels of obstruction in the outflow tract and in lesser degrees of obstruction.
• Muscular infundibular obstruction is frequently characterized by a late peaking systolic jet that appears “dagger-shaped" reflecting the dynamic nature of the obstruction; this pattern can be useful is separating dynamic muscular obstruction from fixed valvular obstruction, where the peak velocity is generated early in systole (Figs 8.28 and 8.29).
TRANSESOPHAGEAL ECHOCARDIOGRAPHY IN PULMONARY STENOSIS
Transesophageal echocardiography (TEE) may allow a more accurate assessment of the pulmonary valve and RVOT in patients with poor transthoracic echocardiography (TTE) window. The pulmonary valve may be identified from a midesophageal window at varying transducer positions from 50° to 90°, anterior to the aortic valve. The RVOT is often well seen in this view.
It is difficult to correctly align CW Doppler cursor to accurately ascertain maximal flow velocity. Other windows in which the pulmonary outflow tract may be interrogated include the deep transgastric view in which, by appropriate torquing of the transducer, the RV inflow and outflow may be appreciated in a single image. This view can allow accurate alignment of the Doppler beam with the area of subvalvular/valvular stenosis through the RVOT.
The following definitions of severity have been proposed in the 2006 American College of Cardiology/ American Heart Association (ACC/AHA) guidelines on the management of valvular heart disease:14
Severe PS: Peak jet velocity > 4 m/s (peak gradient
> 64 mm Hg)
Moderate PS: Peak jet velocity of 3-4 m/s (peak gradient 36-64 mm Hg)
Mild stenosis: Peak jet velocity is < 3 m/s (peak gradient < 36 mm Hg).
A useful index of severity is to determine the RV systolic pressure in patients with PS from the tricuspid regurgitant velocity and the addition of an estimate of right atrial pressure. The pulmonary artery systolic pressure should be RV systolic pressure—pulmonary valve pressure gradient. Severe PS is characterized by near-systemic RV systolic pressures in subjects with no hypertension.
consequences and associations of pulmonary stenosis
Severe PS may be associated with right ventricular hypertrophy, eventually right ventricular enlargement and right atrial enlargement. Given the unusual shape of the RV and its proximity to the chest wall, accurate estimation of RV hypertrophy and enlargement may be difficult. The parasternal long-axis and subcostal long- axis views are often best in assessing RV hypertrophy. The normal thickness of the RV is approximately 2-3 mm, but given the difficulties in estimating thickness, a thickness of
> 5 mm is usually considered abnormal. RV enlargement is typically assessed in the apical or subcostal four-chamber view.
PS may be a part of other syndromes or may be associated with other congenital lesions. Dilatation of the pulmonary artery beyond the valve is common and is due to weakness in the arterial wall in a manner analogous to bicuspid aortic valve and is not necessarily commensurate with the degree of obstruction.
Detection of other lesions such as infundibular stenosis, ventricular septal defect or tetralogy of Fallot is all important in the assessment of these patients.
assessment of pulmonary regurgitation
Pulmonary regurgitation is quite frequent although mostly functional. Causes of organic tricuspid regurgitation (TR) are:
• Congenital: Bicuspid or quadricuspid valve, absent pulmonary valve
• Infective endocarditis
• Carcinoid heart disease
• Rheumatic heart disease
• Myxomatous degeneration
• Pulmonary valve prolapse
• Postoperative17
• Hypoplastic right heart syndrome
Assessment of PR requires detailed understanding of morphology by TTE, TEE and real-time three-dimensional (3D) echocardiography.
PR is diagnosed by documenting a diastolic jet in the RVOT originating from the pulmonary valve. It is normally present in > 50% of the echocardiographic examinations.
• Pathological PR is distinguished from physiological PR by a longer duration of flow (pan-diastolic) and a wider jet as the regurgitant jet crosses the pulmonary valve (Fig. 8.30).
• Functional PR jets are usually very small, central and spindle-shaped (Fig. 8.31).
• In severe PR, where equalization of diastolic pulmonary artery and RV pressures occurs early in diastole, the color jet area can be brief and inaccurate (Fig. 8.32).
• The assessment of PR severity is usually estimated by the diameter of the jet at its origin. The maximum color jet diameter (width) is measured in diastole immediately below the pulmonary valve.1819
• A jet width that occupies > 65% of the RVOT width measured in the same frame is in favor of severe PR (Fig. 8.33).
Pulmonary Regurgitation Vena Contracta
Vena contracta (VC) width is probably a more accurate method than the jet width to evaluate PR severity by color Doppler. A VC width of > 6 mm is indicative of severe PR.20 Advantages and limitations of the VC are same as applicable to other regurgitant lesions (Fig. 8.34).
• The shape of the VC is complex in organic PR. Hence, simple jet width in short-axis plane may not be valid.
• The 3D VC is correlated with the two-dimensional (2D) VC but provides more quantitative assessment of PR.
• The effective regurgitant orifice area values of < 20, 21 to 115 and > 115 mm2 have been proposed to serve as cut-offs for PR grade mild, moderate and severe.21
• By multiplying the 3D VC with the PR velocity-time integral of the PR jet, the regurgitant volume can be obtained.

Fig. 8.30: Wide bifurcated PR jet (arrows) across the pulmonary valve in an operated tetralogy of Fallot.

Fig. 8.31: Functional PR with structurally normal pulmonary valve. Note small, central jet with a length of 10 mm.
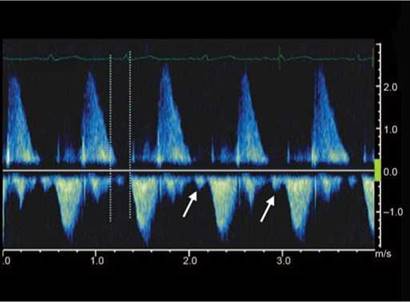
Fig. 8.32: Non-holodiastolic severe PR as judged by rapid deceleration; regurgitation confined to first half of diastole. In late diastole, because of the RV diastolic pressure exceeding PA diastolic pressure, there is forward flow in diastole (arrows).

Fig. 8.33: Measuring jet width of PR and comparison with right ventricular outflow tract width in the same frame. A ratio > 65% is suggestive of severe PR.
• the regurgitant volume values of < 15, 15 to 115 and > 115 mL have also been proposed to serve as cut-offs for PR grade mild, moderate and severe, respectively.21 However, these data require validation.
Proximal Isovelocity Surface Area Method
In some patients, the flow convergence zone can be assessed. However, no studies have examined the clinical accuracy of this method in quantifying the severity of
PR. There are practical difficulties in assessing proximal isovelocity surface area (PISA) in many cases as shown in Figure 8.35.
Pulsed wave Doppler assessment of the forward and the reverse flows at the pulmonary annulus and in the pulmonary artery can been used to calculate regurgitant volume and regurgitant fraction.
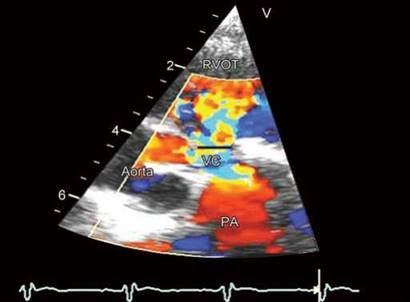
Fig. 8.34: Measuring vena contracta of PR in transthoracic echocar- diographic short-axis view.
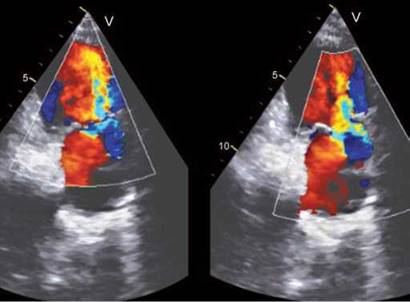
Fig. 8.35: Two examples of severe PR. Proximal isovelocity surface areas is not well-visualized in the left panel, while in the right panel, it is bifid.

Fig. 8.36: Comparing continuous wave Doppler spectrum of mild versus severe PR. Note rapid deceleration of severe PR in the right panel with increased forward flow (forward flow velocity of 1.6 m/s versus 0.8 m/s).
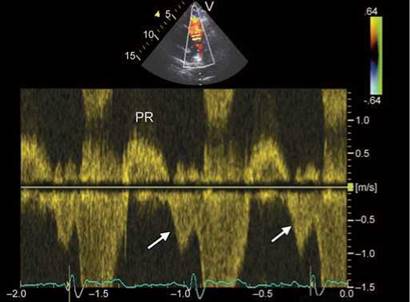
Fig. 8.37: Early diastolic PR with late diastolic antegrade flow across the right ventricular outflow tract. Such PR is usually mild unless associated with rapid deceleration.
The pulmonary annulus should be measured carefully during early systole just below the valve. This technique is subject to errors in geometric assumptions.
Continuous Wave Doppler and Pulmonary Regurgitation Severity22
• The density of the CW signal provides a qualitative measure of regurgitation.
• In mild PR, there is a slow deceleration of the jet velocity.
• A rapid deceleration rate with termination of flow in mid to late diastole is not specific but compatible with severe regurgitation (Fig. 8.36).
consequences of pulmonary regurgitation
Evaluation of the size and function of the RV in the absence of pulmonary hypertension provides indirect clues to the severity of PR. Evidence of RV dilatation is, however, not specific for severe PR. Its absence suggests milder degree of PR. Antegrade diastolic flow across the pulmonary valve without prolonged PR interval is suggestive of markedly elevated RV diastolic pressure and indirectly suggests milder PR if deceleration time is not short (Fig. 8.37).
In summary, echocardiographic assessment of PR includes integration of morphological data from 2D/3D
imaging of the pulmonary valve and RV as well as Doppler measures of regurgitant severity. Validation studies of PR severity are still few.
references
1. Hokken RB, Bartelings MM, Bogers AJ, Gittenberger-de Groot AC. Morphology of the pulmonary and aortic roots with regard to the pulmonary autograft procedure. J ttorac Cardiovasc Surg. 1997;113(з):453-61.
2. Wilcox BR, Cook AC, Anderson RH. Surgical anatomy of the valves of the heart. In: Wilcox BR, Cook AC, Anderson RH (Eds). Surgical Anatomy of the Heart. Cambridge: Cambridge University Press; 2005:45-82.
3. Bateman MG, Quill JL, Hill AJ, et al. the clinical anatomy and pathology of the human atrioventricular valves: implications for repair or replacement. J Cardiovasc Transl Res. 2013;6(2):155-65.
4. Stamm C, Anderson RH, Ho SY. Clinical anatomy of the normal pulmonary root compared with that in isolated pulmonary valvular stenosis. J Am Coll Cardiol. 1998;31(6): 1420-5.
5. Gikonyo BM, Lucas RV, Edwards JE. Anatomic features of congenital pulmonary valvular stenosis. Pediatr Cardiol. 1987;8(2):109-16.
6. Koretzky ED, Moller JH, Korns ME, et al. Congenital pulmonary stenosis resulting from dysplasia of valve. Circulation. 1969;40(1):43-53.
7. Vancini M, Roberts KD, Silove ED, et al. Surgical treatment of congenital pulmonary stenosis due to dysplastic leaflets and small valve anulus. J ttorac Cardiovasc Surg. 1980;79(3):464-8.
8. Milo S, Fiegel A, Shem-Tov A, et al. Hour-glass deformity of the pulmonary valve: a third type of pulmonary valve stenosis. Br Heart J. 1988;60(2):128-33.
9. Edmunds LH. Pulmonary valvular dysplasia [editorial]. Ann ttorac Surg. 1985;19:497.
10. Ergul Y, Nisli K, Kayserili H, et al. Cardiovascular abnormalities in Williams syndrome: 20 years' experience in Istanbul. Acta Cardiol. 2012;67(6):649-55.
11. Bandin MA, Vargas-Barron J, Keirns C, et al. Echocardiographic diagnosis of rheumatic cardiopathy affecting all four cardiac valves. Am Heart J. 1990;120(4):1004-7.
12. Carrilho-Ferreira P, Silva D, Almeida AG, et al. Carcinoid heart disease: outcome after balloon pulmonary valvuloplasty. Can J Cardiol. 2013;29(6):751.e7-e9.
13. Kobayashi J, Nakano S, Matsuda H, et al. Quantitative evaluation of pulmonary regurgitation after repair of tetralogy of Fallot using real-time flow imaging system. Jpn Circ J. 1989;53(7):721-7.
14. Bonow RO, Carabello BA, Chatterjee K, et al. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardi- ology/American Heart Association Task Force on Practice Guidelines (writing Committee to Revise the 1998 guidelines for the management of patients with valvular heart disease) developed in collaboration with the Society of Cardiovascular Anesthesiologists endorsed by the Society for Cardiovascular Angiography and Interventions and the Society of 'boracic Surgeons. J Am Coll Cardiol. 2006;48:e1- 148.
15. Lima CO, Sahn DJ, Valdes-Cruz LM, et al. Noninvasive prediction of transvalvular pressure gradient in patients with pulmonary stenosis by quantitative two-dimensional echocardiographic Doppler studies. Circulation. 1983;67(4):866-71.
16. Lancellotti P, Tribouilloy C, Hagendorff A, et al.; Scientific Document Committee of the European Association of Cardiovascular Imaging. Recommendations for the echocar- diographic assessment of native valvular regurgitation: an executive summary from the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2013;14(7):611-44.
17. Yoo BW, Park HK. Pulmonary stenosis and pulmonary regurgitation: both ends of the spectrum in residual hemodynamic impairment after tetralogy of Fallot repair. Korean J Pediatr. 2013;56(6):235-41.
18. Williams RV Minich LL, Shaddy RE, et al. Comparison of Doppler echocardiography with angiography for determining the severity of pulmonary regurgitation. Am J Cardiol. 2002;89(12):1438-41.
19. Goldberg SJ, Allen HD. Quantitative assessment by Doppler echocardiography of pulmonary or aortic regurgitation. Am J Cardiol. 1985;56(1):131-5.
20. Williams RV, Minich LL, Shaddy RE, et al. Comparison of Doppler echocardiography with angiography for determining the severity of pulmonary regurgitation. Am J Cardiol. 2002;89(12):1438-41.
21. Pothineni KR, Wells BJ, Hsiung MC, et al. Live/real time three-dimensional transthoracic echocardiographic assessment of pulmonary regurgitation. Echocardiography. 2008;25(8):911-7.
22. Lei MH, Chen JJ, Ko YL, et al. Reappraisal of quantitative evaluation of pulmonary regurgitation and estimation of pulmonary artery pressure by continuous wave Doppler echocardiography. Cardiology. 1995;86(3):249-56.