Definition
• A variant of malignant mesothelioma (MM) that is composed of cells morphologically similar to epithelial cells
Clinical features
Epidemiology
• The most common histological variant; accounts for 65% to 70% of MM
Presentation
• Same as other pleural mesotheliomas
Prognosis and treatment
• Relatively better prognosis than other histological variants
• Pleomorphic diffuse epithelioid variant and MM with high nuclear grade have worse prognosis
• Surgical (extrapleural pneumonectomy or pleurectomy and decortication) treatment and talc pleurodesis are palliative; multiple trials for chemoradiation ongoing
Pathology
Histology
• Tumor cells are polygonal, oval, or cuboidal in shape with round nuclei, prominent nucleoli, and moderate amount of eosinophilic cytoplasm
• Usually bland in appearance; mimic nonneoplastic mesothelial cells
• May have necrosis, atypical mitotic figures, psammoma bodies; may have sharply defined vacuoles with metachromatic hyaluronic acid, representing intracytoplasmic lumina
• Stroma may be desmoplastic or myxoid containing hyaluronic acid
• Common growth patterns: tubulopapillary, acinar (glandular), adenomatoid (microglandular), sheets (solid), or mixed
• Less common growth patterns: clear cell, deciduoid, adenoid cystic, signet ring, small cell, and rhabdoid
• Lymphohistiocytoid pattern: composed of discohesive, atypical histiocytoid-appearing malignant mesothelial cells with an intense lymphoplasmacytic infiltrate
Immunopathology/special stains
• Calretinin is the most useful marker for epithelioid mesothelioma, showing strong and diffuse cytoplasmic and nuclear staining; note that the nuclear staining is usually more intense than the cytoplasmic
• CK5/6 is expressed in the cytoplasm of mesothelial and squamous cells; rarely, expressed in adenocarcinoma
• WT-1 is expressed in mesothelioma as well as in serous carcinoma of the ovary with a nuclear staining pattern. Cytoplasmic staining is seen in endothelial cells and some carcinomas
• If a carcinoma is the differential diagnosis, an initial workup should include at least two mesothelial markers and two carcinoma markers in addition to a pancytokeratin; additional stains can be used if the initial panel is nondiagnostic
• Pancytokeratin is virtually positive for all epithelioid mesotheliomas; if pancytokeratin staining is negative, other possible differential diagnoses including malignant melanoma, epithelioid hemangioendothelioma or angiosarcoma, and lymphoma should be considered
Main differential diagnoses
• Reactive mesothelial hyperplasia (see previous section)
• Adenocarcinoma
• Adenocarcinoma markers: MOC-31, BG8, CEA, Leu-M1 (CD15), Ber-Ep4, TTF-1
• ER, PR for breast and gynecological carcinomas
• Squamous cell carcinoma
• CK5/6 is not useful; positive in 100% of squamous cell carcinoma
• D2-40 is not useful; positive in 50% of squamous cell carcinoma
• Markers useful for squamous cell carcinoma: p63, MOC31, BG8, and Ber-Ep4
• Malignant melanoma
• Negative for pancytokeratin
• Positive for melanoma markers: S100, HMB45
• Angiosarcoma
• D2-40 is not a good marker for this differential diagnosis
• Positive for endothelial markers CD31, CD34
• Some can be keratin positive
• Lymphoma
• Initial panel includes CD45 (LCA), CD3, and CD20
• Negative for pancytokeratin

Fig 1 Epithelioid malignant mesothelioma. Common tubulopapillary growth pattern of epithelioid MM (A and B).

Fig 2 Epithelioid malignant mesothelioma. Epithelioid MM with small tubular/acinar growth pattern (A) with invasion of vessel wall (B).

Fig 3 Epithelioid malignant mesothelioma. Adenomatoid growth pattern.

Fig 4 Epithelioid malignant mesothelioma. Sheetlike (solid) growth pattern invading fat.

Fig 5 Epithelioid malignant mesothelioma. Clear cell change in epithelioid MM.

Fig 6 Epithelioid malignant mesothelioma. Microcystic growth pattern.

Fig 7 Epithelioid malignant mesothelioma. Deciduoid growth pattern.

Fig 8 Epithelioid malignant mesothelioma. Sharply defined vacuoles forming signet-ring cells.

Fig 9 Epithelioid malignant mesothelioma. Clusters of malignant mesothelial cells in a mucinous stroma.

Fig 10 Epithelioid malignant mesothelioma. Chondroid formation.

Fig 11 Epithelioid malignant mesothelioma. Calretinin immunostain of epithelioid MM with strong and diffuse cytoplasmic and nuclear staining patterns. Note that nuclear staining is darker than the cytoplasmic staining, a helpful feature in MM.

Fig 12 Epithelioid malignant mesothelioma. WT-1 immunostain of epithelioid MM with nuclear staining pattern. Note cytoplasmic staining of endothelial cells.

Fig 13 Epithelioid malignant mesothelioma. CK5/6 immunostain of epithelioid MM with cytoplasmic staining pattern showing membranous accentuation.
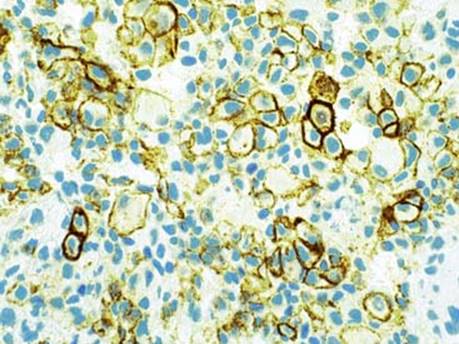
Fig 14 Epithelioid malignant mesothelioma. D2-40 immunostain of epithelioid MM with a membranous staining pattern.

Fig 15 Epithelioid malignant mesothelioma. Strongly positive for keratin CK7 (A); it may show focal/weak positivity for CK20 as shown here (B).
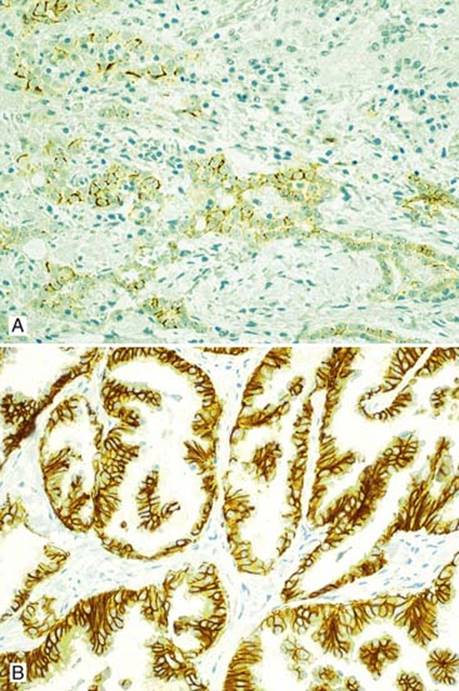
Fig 16 Epithelioid malignant mesothelioma. Usually negative for MOC31, can be focally positive (up to 10% of cells staining) as shown here (A); in contrast, adenocarcinoma is strongly positive with a membranous staining pattern (B).

Fig 17 Epithelioid malignant mesothelioma. Epithelioid MM is usually negative for BG8 but can be focally positive (A); in contrast, adenocarcinoma is strongly and diffusely positive with a membranous staining pattern (B).

Fig 18 Epithelioid malignant mesothelioma. Adenocarcinoma (A) and large-cell carcinoma (B) with strong and diffuse Ber-Ep4 positivity with membranous staining pattern.

Fig 19 Epithelioid malignant mesothelioma. CT scan of chest showing adenocarcinoma of lung involving the pleura mimicking MM.