Most common neurologic conditions are imaged using CT, MRI, or both. In the emergent setting, CT is still the most commonly utilized modality. Patients with trauma or a suggestion of acute intracranial hemorrhage are routinely imaged with CT scans, whereas those with findings suggestive of ischemic stroke may be triaged to either CT or MRI. Frequently, the decision rests on the availability of the modality and the time needed to obtain the study. Advances in neuroimaging allow greater specificity in identifying metabolic and physiologic differences between normal tissue and pathology. Some of the most exciting advances include the use of diffusion-weighted imaging (DWI) with MRI that can demonstrate physiologic change within minutes of ischemic change, MR spectroscopy that can identify abnormal metabolites and help in differentiating pathology such as recurrent tumor versus radiation necrosis, and functional MRI (fMRI) that can be used to localize specific functional areas in the brain. The choice of most appropriate imaging modality is presented according to clinical presentation. The information available with medical imaging carry with it specific risks that must be balanced against diagnostic gain. Ionizing radiation from CT may result in significant radiation dose, especially when multisequence scans are performed. Although the actual risk of cancer induction is still an open debate, it is clear that scans should only be ordered that are absolutely necessary and that will affect patient management. Other risks to consider from imaging have been described from contrast use in both CT and MRI. Iodinated contrast can lead to a contrast-induced nephropathy in patients with preexisting renal insufficiency. Recently, gadolinium contrast has been linked to nephrogenic systemic fibrosis (NSF), a rare scleroderma-like systemic disease in patients with renal insufficiency. Current recommendations are to either forgo gadolinium contrast in patients in moderate renal insufficiency, or if medically indicated, reduce the dose and use one of the agents believed to be associated with less risk of NSF. Patients with severe renal failure should in most cases not receive gadolinium contrast.
I. TRAUMA
CT is the primary imaging modality in trauma because of ready availability, speed, ability to detect bony abnormalities, and superior accuracy in detecting acute intracranial blood. High-resolution, fast CT scanners are routinely installed either within emergency departments or in close proximity. CT is performed routinely in trauma patients, often including those without focal neurologic signs to screen for occult injury. Widely accepted standards for patient selection in this setting are still controversial.
MRI in the setting of acute trauma is still a secondary modality. The most important indication is in evaluating for diffuse axonal injury in which diffuse brain injury may be seen in the presence of a normal head CT. MRI may also demonstrate small extra-axial hemorrhages not seen by CT, particularly in the subtemporal and subfrontal regions.
A. Closed head injury. CT imaging parameters in trauma should include a narrow window (for acute blood), an intermediate window (for subacute blood), and a wide (bone) window. Acute blood may be present in the form of intracerebral (contusion), epidural, or subdural hematomas and subarachnoid hemorrhage (SAH). Fractures are well-evaluated on bone windows and from coronal and sagittal reconstruction images. Patients with acute trauma and favorable initial coma scores usually fare well and do not require extensive follow-up imaging. In patients with moderate or severe trauma, or low initial Glasgow Coma Score, sequential CTs allow evaluating the course of the initial trauma and the effects of therapy, including extension of initial hemorrhage, rehemorrhage, cerebral edema, herniation, response to external ventricular drainage, and intracranial pressure monitors. Diffuse cerebral edema, more common in younger patients, causes effacement of CSF spaces and may result in herniation (tonsillar, transtentorial, and subfalcine). In patients who come to medical attention several days or weeks after head trauma, with worsening headaches or seizures, MRI may be preferred because subacute hematomas may be isodense on CT, and therefore difficult to see. MRI is superior in evaluating for the presence of blood products in the brain parenchyma (Fig. 32.1A and B).
B. Penetrating head injury. CT is the modality of choice in patients with penetrating head injuries. Metallic objects (shrapnel) and glass appear hyperdense on CT, whereas wood objects appear hypodense (air-containing). CT permits an excellent evaluation of the extent of bone damage, and that of the underlying parenchyma, including hematomas, edema, infarction, and herniation.
C. Cervical spine injury. Plain X-rays remain the first-line imaging method in patients with cervical spine trauma and should include at least anteroposterior, lateral, and open-mouth (odontoid) views. Cervical spine CT offers submillimeter resolution allowing the detection of subtle fractures not seen on plain films. MRI is used to evaluate for spinal cord injury, which appears bright on T2 pulse sequences and dark on gradient echo sequences because of the magnetic susceptibility effect of acute blood.
D. Vascular injury. Blunt injury to the neck may result in traumatic arterial dissections, pseudo-aneurysms, or occlusions. In stable patients, magnetic resonance angiography (MRA) or CT angiography (CTA) are excellent imaging methods. Unstable patients may go directly to angiography, particularly if endovascular management is contemplated. Penetrating injuries may result in similar lesions, in addition to significant bleeding from both arterial and venous (jugular vein) injuries. CTA is the imaging method of choice in these patients because current fast scanners allow the evaluation of large anatomical areas with high-resolution and limited-contrast administration. Unstable patients may also require emergency angiography and possibly lifesaving endovascular occlusion of a bleeding artery.

FIGURE 32.1 CT of a 10-year-old boy who fell from a tree. Noncontrast head CT demonstrates a right frontal epidural hematoma (A) and parenchymal contusions (B).
II. HEADACHES
Headache is one of the most common indications for neurologic consultation, with 50% of adults being seen for a severe headache at least once in their life. Fortunately, the vast majority have a benign origin. Pain sensitive structures in the cranial area include the scalp, the scalp blood vessels, head and neck muscles, dural sinuses, the dura and large cerebral arteries at the skull base, meningeal arteries, and pain sensitive fibers of the fifth, ninth, and tenth cranial nerves. Serious conditions that cause headaches include hemorrhage (subarachnoid, subdural, and intracerebral), infections (meningitis and brain abscess), tumors (primary or metastatic), hydrocephalus, and hypertensive crises. Patients at higher risk for significant pathology include those with (1) severe headache of sudden onset; (2) mental status changes, fever, focal neurologic deficits, or seizures; and (3) onset after the age of 50.
A. Acute headaches. Acute headache associated with nausea, vomiting, nuchal rigidity, and transient alteration in mental status is suggestive of SAH and should prompt immediate evaluation. CT is currently the preferred neuroimaging method for SAH, with reported accuracy rates in the 98% to 99% range.
1. SAH and aneurysms. Nontraumatic SAH is caused by a ruptured intracranial aneurysm in 80% to 85% of adult patients (Fig. 32.2A and B). About 10% of patients with SAH, usually in the younger age group, have a nonaneurymal perimesencephalic hemorrhage, a benign and self-limiting venous hemorrhage. Catheter angiography remains the gold standard to evaluate SAH, followed by endovascular or surgical aneurysm obliteration if an aneurysm is identified. CTA is increasingly used as a reliable replacement for cerebral angiography, including for the surgical planning of ruptured and unruptured aneurysms. Uncommon causes of SAH include cerebral and dural arteriovenous malformations (AVMs), arterial dissections, cerebral tumors, vasculitides, and moyamoya disease.
2. Intracerebral hemorrhage and other causes of acute headache. Intracerebral hemorrhage is most commonly caused by arterial hypertension and may be putaminal, thalamic, lobar, cerebellar, or pontine. Cerebral amyloid angiopathy may cause lobar hemorrhage in the nonhypertensive elderly. CT is the primary method of evaluation. In younger, nonhypertensive patients, a cerebral or dural AVM may be the cause of hemorrhage, requiring further workup with MRI and cerebral angiography, which may need to be repeated or delayed if there is a large and compressive hematoma. Contrast-enhanced MRI and possibly MR spectroscopy may be useful if a tumor is suspected.

FIGURE 32.2 CT of a 38-year-old woman with acute onset severe headache. Noncontrast head CT demonstrates subtle subarachnoid blood in right sylvian fissure (A). CTA demonstrates an anterior communicating artery aneurysm (B).
Severe unilateral headache with cervicalgia and/or a Horner syndrome may be caused by an acute carotid or vertebral arterial dissection. MRI/MRA, especially with precontrast fat-saturated axial T1 imaging, is diagnostic, showing the true lumen as a flow void and the mural thrombus as a bright crescent, so that there is generally no need for conventional angiography unless intracranial extension is suspected or endovascular treatment is contemplated. Migraines can cause severe acute headaches, usually periorbital, hemifacial, and frontal. The diagnosis is clinical, and CT may be sufficient to rule out hemorrhage in typical cases. Sinusitis is also diagnosed clinically; coronal CT shows soft tissue material obstructing sinus drainage pathways and filling the sinuses, and air–fluid levels in acute sinusitis. Glaucoma, retrobulbar optic neuritis, hydrocephalus, and infection may also cause acute headaches, evaluated with CT in clinically uncertain cases.
B. Subacute headaches. Subacute headaches, particularly in the elderly, may be caused by a subdural hematoma. CT is usually adequate for both the initial evaluation and the follow-up, showing subdural space crescentic collections. Acute blood is hyperdense, whereas chronic collections are hypodense. CT is also the modality of choice to diagnose hydrocephalus (the temporal horns and the third ventricle are early reliable indicators of hydrocephalus). Cerebral tumors and infections may be evaluated with postcontrast CT (although contrast-enhanced MRI is superior). Spontaneous intracranial hypotension, possibly associated with a chronic CSF leak, is a potential cause for headaches causing recurrent ER visits; postcontrast MRI (particularly in the coronal plane) may be diagnostic, showing thickened and densely enhancing meninges as well as “sagging of the midbrain” and decreased volume in the suprasellar and basilar cisterns.
C. Chronic headaches. Unruptured AVMs, temporal arteritis, vasculitides, colloid cysts of the third ventricle, and cervical spondylosis are all potential causes of chronic headaches, in addition to migraine, cluster headaches, and chronic sinusitis. MRI has the highest yield in screening patients with a suspected structural intracranial anomaly.
III. CEREBRAL ISCHEMIA
Cerebral ischemia may be the result of acute arterial occlusion, hypoxic or anoxic injury, or may result from venous occlusion with increased venous pressure. Arterial occlusion may be an acute event, a chronically progressive process or an acute process superimposed on chronic.
A. Acute stroke. It is a true emergency (time is brain). There is a 4.5-hour window after the ischemic stroke onset for the delivery of intravenous tissue plasminogen activator. Past that, and up to 6 hours in the carotid circulation (possibly longer in the vertebrobasilar circulation), intra-arterial thrombolysis may be an option. One proposed algorithm for the emergency evaluation of acute stroke is to obtain a plain CT, followed (if no hemorrhage) by a contrast CTA and CT perfusion study to evaluate the perfusion deficit. In some centers, MRI, MRA, and MR perfusion are utilized in acute stroke patients. DWI is positive for acute stokes as early as 30 minutes and up to 10 days after the onset and is therefore particularly well-suited to differentiate acute and subacute from chronic events (Fig. 32.3A and B).
B. Dural sinus and cortical vein thrombosis. The venous intracranial circulation should always be evaluated, particularly if the patient’s neurologic deficit is accompanied by headache or does not fit a recognizable vascular distribution. Recent thrombus within a dural sinus may be difficult to identify on plain CT (although possibly seen as a spontaneously hyperdense filling structure) or on plain MRI (hyperacute blood may appear gray on both T1 and T2 sequences). Therefore, postcontrast imaging (CT and MRI) has higher accuracy, showing clots as filling defects within a dural sinus or cortical vein. An MR venography (MRV) provides 3-D visualization of the venous system, which may be selectively imaged owing to its lower velocity profile compared with arterial structures (Fig. 32.4A and B).
C. Intermittent and chronic deficits. Transient ischemic attacks (TIAs) and chronic ischemic deficits are best evaluated with MRI/MRA. DWI MRI is most often normal in TIAs and ischemic lesions 10 days or older. MRI is superior to CT in evaluating stroke mimics including demyelinating disease and tumor. MRA is effective in screening the intracranial and cervical arterial vasculature although the resolution of CTA (0.625 mm with current 64-detector scanners) provides excellent definition, nearing that of conventional angiography in certain locations. Severe arterial stenoses are a known pitfall of MRA, which exaggerates the degree of the lesion owing to signal loss. Therefore, conventional angiography remains useful in equivocal cases whenever corrective therapy is contemplated.

FIGURE 32.3 Noncontrast head CT demonstrates an area of hypodensity in the right temporal region (A). Diffusion weighted MRI scan (B) demonstrates larger area of restricted diffusion, consistent with acute ischemic change.

FIGURE 32.4 Severe headache and mental status changes. Noncontrast head CT demonstrates parenchymal hemorrhage in the left hemisphere with high density noted in the superior sagittal sinus (A). MRV demonstrates absent signal in the superior sagittal sinus consistent with sinus thrombosis (B).
IV. ALTERED LEVEL OF CONSCIOUSNESS
Evaluation of a patient with an altered level of consciousness (LOC) a common indication for neurologic consultation requiring immediate neuroimaging. Causes of altered LOC may include intracranial hemorrhage (including traumatic), stroke, metabolic disorders, toxic substance ingestion (suicide attempts or accidental), and cerebral tumors (primary or metastatic).
The immediate concern in these patients is to rule out a major indication for emergent intervention, including intracranial hemorrhage, acute infarctions, impending tonsillar or transtentorial herniation, or other life-threatening conditions. CT scanning is the preferred modality in these patients because of ready access and accuracy in identifying intracranial hemorrhage (intracerebral, SAH, SDH, and epidural). It is also possible to assess the risk for herniation if a lumbar is indicated to rule out SAH or infection. In patients without CT evidence of acute abnormal findings, MRI should be obtained as soon as to detect CT occult process including dural sinus thrombosis, acute basilar artery thrombosis, and posterior reversible encephalopathy syndrome, which show characteristic T2 and FLAIR cortical and subcortical lesions.
V. DEMENTIA
Cognitive decline may be related to a number of clinical conditions including (1) depression, (2) structural lesions (cerebral tumor, subdural hematoma, and hydrocephalus), (3) chronic cerebral ischemia, or (4) primary neurodegenerative conditions, the most common of which is dementia of the Alzheimer’s type (DAT). A thorough clinical evaluation plays a major role in these patients who are often in the older age group. Neuroimaging detects correctable causes of dementia, found in about 5% of patients with progressive cognitive decline. CT is adequate to identify severe hydrocephalus and chronic subdural hematomas. However, MRI is superior to CT in the vast majority of patients, although unfortunately effective therapy is available in only a few conditions. Assessment of generalized cerebral and focal hippocampal atrophy in DAT is more easily done with MRI, including computerized volumetric measurements of the hippocampus. PET and SPECT may allow detection of DAT earlier by showing decreased hippocampal glucose metabolism. Also MRS may show increased myo-inositol and decreased N-acetyl-aspartate peaks in the gray matter. Vascular dementia is also well-evaluated by MRI, particularly FLAIR imaging, which demonstrates lacunar infarcts and white matter abnormalities. Cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) may have a typical distribution (periventricular, anterior temporal, and subinsular) that may suggest the diagnosis (Fig. 32.5A and B). In Pick’s disease and other frontotemporal dementias, predominance of frontal lobe atrophy is well-evaluated on multiplanar MRI. In suspected normal pressure hydrocephalus, transependymal CSF flow may be present, seen as periventricular bright T2 signal. The pattern and velocity of CSF flow may be evaluated at the foramen magnum using phase contrast flow techniques.
VI. SEIZURES
A. New onset adult seizures. Although patients presenting with seizures are often scanned initially with CT to rule out hemorrhage or tumor, MRI is the preferred initial imaging method to investigate new onset adult seizures, owing to its superior contrast resolution and multiplanar capability. Magneto-encephalography (MEG) is a new technique that measures magnetic fields caused by neuronal activity, with a spatial resolution of a few millimeters and a temporal resolution of milliseconds. Magnetic source imaging uses MEG in combination with MRI in the same machine and has currently the highest yield detecting epileptogenic foci. New onset adult seizures may be caused by tumors (primary or metastatic), AVMs, inflammatory conditions, vasculitides, ischemic lesions, and gliosis from prior injury (Fig. 32.6A and B).

FIGURE 32.5 Forty-one-year-old man with migraines with aura, TIAs, and cognitive decline diagnosed with CADASIL. Axial FLAIR images at the level of the lateral ventricles (A) and through the temporal lobes (B) demonstrates scattered areas of signal abnormality. The symmetric anterior temporal lobe abnormalities are typical in this disease.

FIGURE 32.6 T2 axial image demonstrates abnormal signal in the inferior right frontal lobe (A). Postcontrast T1 axial image (B) demonstrates a minimally enhancing cystic ganglioglioma in this 19 year old with a history of seizures.
B. Known seizure disorder. In patients with temporal lobe epilepsy, coronal MRI (FLAIR and T2) has a high overall detection rate (up to 80%) for mesio-temporal sclerosis, showing an atrophic hippocampus with high T2 signal and indirect signs including dilated choroidal fissure and forniceal atrophy. Presurgical evaluation includes the evaluation of hemispheric language dominance, usually obtained by Wada’s testing (selective internal carotid artery injection of sodium amobarbital). The fMRI has been used to detect language dominance, although its accuracy is as of yet unproven compared with Wada’s test.
C. Pediatric seizures. Infants with germinal matrix and traumatic hemorrhages, intracranial neonatal infections, and perinatal ischemia may be evaluated and followed with CT. In stable, nonfebrile infants, most neonatal seizures are related to congenital disorders (migrational anomalies and structural defects), which are best evaluated with MRI, although traumatic lesions are detectable on CT. In childhood-onset seizures, clinical and EEG evaluations are usually adequate and imaging may not be necessary for certain forms of seizure activity, including febrile, absence (petit mal) seizures, infantile spasms (Lennox–Gastaut’s syndrome), and benign focal epilepsy, unless the child has abnormal physical findings or delayed development. Certain forms of childhood epilepsy, like juvenile myoclonic epilepsy, are associated with a higher frequency of structural anomalies requiring MRI evaluation.
VII. HEARING LOSS AND TINNITUS
A. Hearing loss. Clinical and audiometric data should guide neuroimaging. The deficit may be (1) sensorineural (SNHL), conductive (CHL) or mixed; (2) unilateral or bilateral; and (3) congenital or acquired.
1. Sensorineural hearing loss. Unilateral or asymmetrical SNHL in adults is best evaluated with MRI. The most common cause is a vestibular schwannoma. Even small lesions of the internal auditory canal and cerebellopontine angle are diagnosed with thin T1, T2, and postcontrast axial and coronal MRI, and the addition of a high-resolution volume (Fast Imaging Employing Steady sTate Acquisition). MRI should also evaluate the remainder of the acoustic pathway for possible ischemic or demyelinating lesions, particularly the medullary cochlear nuclear complex (which lesions mimic those caused by vestibular schwannomas), thalamus, and temporal lobe (see Fig. 56.2).
SNHL in children, unilateral or bilateral, is usually related to congenital inner ear diseases, requiring high-resolution noncontrast CT as the initial evaluation to assess the cochlea, vestibule, semicircular canals, vestibular aqueduct, and endolymphatic duct and sac. Enlarged vestibular aqueduct syndrome is a common cause of SNHL.
2. Conductive hearing loss. CHL is caused by disruption of the mechanical components of the auditory apparatus. CHL is therefore best evaluated by noncontrast high resolution CT. It is most commonly caused by temporal bone inflammatory disease, particularly otomastoiditis and otitis media. Otosclerosis (also called otospongiosis) causes both CHL and SNHL (bilateral in 80%) and tinnitus. It is because of the replacement of endochondral bone by spongious bone at the oval window (fenestral) or the cochlea (retrofenestral). Other causes for CHL include middle ear cholesteatomas, tumors (glomus tympanicum), and traumatic ossicular dislocations, all well-evaluated with CT.
B. Tinnitus. It (ringing in the ear) may be very disturbing to patients. It may be pulsatile or nonpulsatile. Objective tinnitus, heard by both the patient and the examiner, commonly leads to findings. Subjective tinnitus, only heard by the patient, has a low diagnostic yield.
1. Pulsatile tinnitus. Pulsatile (pulse synchronous) tinnitus is best evaluated by MRI/MRA, whether or not direct otoscopic examination shows a retrotympanic mass. A vascular-appearing tympanic membrane may be associated with arterial (aberrant carotid artery, carotid stenosis or dissection, and petrous carotid artery aneurysms), venous (dehiscent or high-riding jugular bulb), inflammatory (cholesterol granuloma, middle ear mastoiditis) causes or tumors (glomus tympanicum or jugulo tympanicum and meningioma). Tinnitus with a normal otoscopic examination should raise suspicion for a dural arteriovenous fistula (of the sigmoid sinus or the tentorium); MRI may show suspicious flow voids, MRA source images may demonstrate transosseous arterial structures, and postcontrast MRI and MRV may demonstrate an occluded dural sinus. Confirmation (and therapy) is provided via catheter angiography. Other conditions include venous sinus stenosis, idiopathic intracranial hypertension (pseudotumor cerebri), chronic anemia, and thyrotoxicosis.
2. Nonpulsatile tinnitus. It is most commonly caused by Ménière’s disease, which also manifests as episodes of vertigo and SNHL. Increased volumes of endolymph causing enlarged endolymphatic spaces have been incriminated. The diagnosis is clinical, but when indicated, neuroimaging should be done with CT. Other causes include otosclerosis and middle ear inflammatory disease, also best studied with CT.
VIII. VERTIGO AND ATAXIA
Vertigo and ataxia may indicate posterior fossa lesions where MRI is the modality of choice because of superior contrast resolution and beam hardening artifact with CT.
Causes of vertigo that may be diagnosed with imaging include vestibular schwannomas, viral labyrinthitis, or perilymphatic fistulae. Central vertigo may be caused by posterior fossa lesions including demyelinating disease, tumors, strokes, Arnold–Chiari malformation, and trauma. The preferred neuroimaging method to investigate vertigo is MRI. Even small vestibular schwannomas may be diagnosed with a good quality MRI, appearing as small enhancing masses. Rarely, viral labyrinthitis will show as bright T1 signal within the vestibular apparatus indicative of hemorrhagic products. Small multiple sclerosis plaques and ischemic lesions appear as bright lesions on T2 and FLAIR imaging.

FIGURE 32.7 Sagittal T1 image demonstrates prominent volume loss of the cerebellum, pons, and brainstem, in this patient with multiple system atrophy of the cerebellar type (olivopontocerebellar atrophy).
Ataxia usually reflects cerebellar dysfunction, although it may also be sensory or vestibular in origin. Again, MRI is the preferred imaging modality to study patients with ataxia because of its superior contrast resolution in demyelinating diseases, ischemia, and tumors. Other causes of ataxia include chronic ethanol and phenytoin intoxication, a number of degenerative conditions, paraneoplastic syndromes, all accompanied with cerebellar atrophy, well-demonstrated on sagittal and coronal MRI (Fig. 32.7).
IX. DISTURBANCES OF VISION
The optic pathways and the globe are both well-evaluated with MRI, which is the preferred neuroimaging study in patients with visual disturbance.
A. Visual loss (including amaurosis fugax). Gradual monocular vision loss is usually related to ocular pathology like cataract. Sudden unilateral vision loss most commonly results from diabetic retinopathy, followed by ocular ischemic syndrome, which may be caused by retinal vein occlusion, retinal artery occlusion, anterior ischemic optic neuropathy, an ischemic syndrome of the anterior ciliary vasculature, and rarely to demyelinating disease of the optic nerve, well-evaluated with pre- and postcontrast MRI. Amaurosis fugax designates vision loss caused by reduced blood flow to the eye, heralding a stroke and prompting therapy. The most common cause of amaurosis fugax is stenotic carotid disease, which is well-evaluated by MRA. CTA is an excellent alternative to evaluate carotid disease, although heavily calcified plaques remain a limitation.
Visual field deficits related to lesions affecting the optic chiasm, tracts, and radiations should be studied with contrast-enhanced MRI. Lesions affecting the chiasm, like pituitary adenomas, and suprasellar lesions are particularly well-suited for coronal and sagittal MRI. Most demyelinating and inflammatory conditions and tumors involving the postchiasmatic optic pathways appear bright on T2 and FLAIR imaging and enhance after contrast administration. Cerebral AVMs of the temporal and occipital lobes appear as dark, tortuous flow voids on T1 and T2 imaging.
B. Impairment of ocular motility. Ocular motility dysfunction most commonly results from diabetic cranial neuropathy and traumatic lesions of the orbit or the superior orbital fissure. Traumatic lesions involving bony structures are best evaluated with thin-cut CT with coronal reconstructions. Nontraumatic pathology may result from a variety of lesions affecting the oculomotor, trochlear, and abducens nerves anywhere between their brainstem nuclei and the orbit including brainstem strokes, demyelinating and inflammatory lesions, tumors, petrous apex lesions, cavernous sinus and orbital apex tumors, aneurysms, and inflammatory lesions. MR is the modality of choice in evaluating all of these possibilities. A sudden third cranial nerve palsy suggestive of an internal carotid artery—posterior communicating artery junction aneurysm may be initially evaluated with either MRI/MRA or CT/CTA. The extraocular muscles are also well-evaluated with coronal MRI of the orbits.
C. Chemosis and proptosis. Carotid-cavernous fistulae (CCFs) are the most common causes of ophthalmic venous system flow reversal and engorgement. Direct CCFs are caused by arterial wall rupture of the intracavernous carotid segment, most commonly from a traumatic arterial laceration, less commonly from spontaneous rupture of a small aneurysm, Ehlers–Danlos’ syndrome type IV (vascular type), fibromuscular dysplasia, or a spontaneous arterial dissection. Indirect CCFs are because of spontaneous arteriovenous shunting to the ophthalmic vein from dural arterial branches of the external carotid. The most common associations being pregnancy, dehydration, and sinus infections. Contrast-enhanced CT and MRI show dilated ophthalmic vein and cavernous sinus in direct CCFs. Indirect CCFs may be much more subtle, sometimes only suspected on the basis of small flow voids around a dural sinus. Catheter angiography is diagnostic and provides the route for transvascular therapy.
X. NECK PAIN AND CERVICAL RADICULOPATHY
Cervical spondylosis is the most common cause of neck pain and cervical radiculopathy, and its incidence increases with age. It is characterized by hypertrophic arthropathy of the facet joints, osteophyte formation at the disk margins, and progressive intervertebral disk degeneration and herniation. All these changes result in central canal and neural foraminal stenoses, with resulting restriction of the spinal cord and the nerve roots. In younger patients, sudden disc herniation may cause acute symptoms. Other causes of cervical pain and radiculopathy include syringomyelia with or without Chiari’s malformation, benign tumors of the spinal canal or the neural foramina like schwannomas and meningiomas, demyelinating disease, and post-traumatic myelomalacia. MRI is the preferred method to image these patients because it allows superior evaluation of the cervicomedullary junction, the cord, the spinal canal, and the neural foramina. CT myelography may be useful in patients with cervical spondylosis and contraindications or intolerance to MRI (Fig. 32.8A and B).
XI. BACK PAIN
Low back pain is one of the most common presenting complaints to physicians with up to 85% of the population having experienced symptoms at least once.
A. General causes and evaluation. Most causes of transient low back pain are benign and related to degenerative disease, muscle strain, mild trauma, excess weight, and poor posture. There is a widely held perception in both the medical community and the public is that imaging for back pain is overutilized. Imaging is recommended for back pain associated with certain findings, including radiculopathy or lower motor neuron deficit, sudden onset, older age, signs of systemic infection, known or suspected malignancy, and trauma. MRI is the preferred study in these cases. The relationship between persistent back pains including radicular pain may be striking. Instability may be defined as loss of support of a segment of the spine, subjecting it to abnormal displacement during physiologic movements. Plain X-rays of the spine, in neutral position, flexion, and extension are the most widely used screening method and allow identification of patients who are likely to benefit from corrective surgery, that is, those with spondylolisthesis and lumbar intervertebral instability. CT may be useful to provide further detail.

FIGURE 32.8 T2 sagittal image (A) and axial image (B) demonstrate a large left paracentral disc protrusion at the C5-6 level with marked deformity of the underlying cord with increased signal in the cord that may represent edema or myelomalacia.
B. Lumbar radiculopathy (sciatica). It is pain that originates along the course of the sciatic nerve, which runs from the lumbar spine to the posterior thigh. Lumbar disc herniation, the most common cause of sciatica, is a break in the annulus fibrosus with subsequent displacement of nucleus pulposus, cartilage, or bone beyond the disc space. Other causes of sciatica include degenerative disease (including synovial cysts) and spinal stenosis, tumors (primary and metastatic), infections (osteomyelitis and abscess), and hematomas (epidural, subdural, and psoas). MRI is the study of choice for the initial evaluation of lumbar radiculopathy, as it demonstrates the conus, cauda equina, nerve roots, bony elements, and discs with exquisite contrast in multiple planes. Contrast enhancement is utilized in evaluating infections and tumors. Disc herniations are well-evaluated with MRI, although CT myelography may allow greater detail of bone abnormalities. MRI is inferior to CT in spondylolysis and spondylolisthesis.
XII. MYELOPATHIES
MRI is the preferred imaging method in myelopathies, both acute and chronic, providing multiplanar imaging, superior contrast between normal and abnormal spinal cord, CSF, fat and bony structures. Gating techniques help reduce artifacts from breathing and CSF, cardiac and vascular pulsations. Patients with acute myelopathy and major contraindications to MRI (pacemakers) may be evaluated with plain CT or CT myelography.
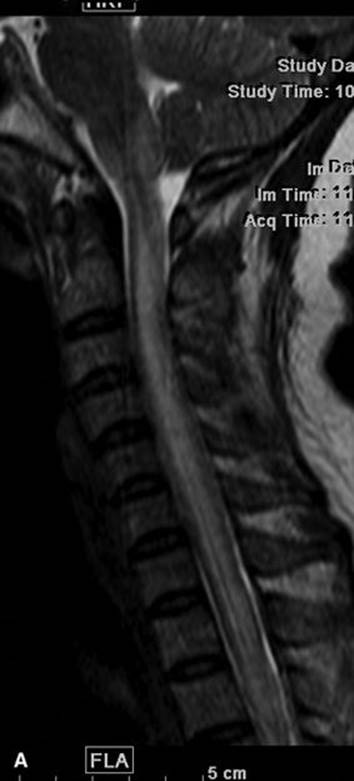



FIGURE 32.9 T2 sagittal (A) T1 sagittal (B), T1 sagittal with contrast (C) and T2 axial (D) of the cervical spine demonstrate increased signal on T2 with slight cord enlargement. Post contrast T1 sagittal images demonstrate enhancement in the areas of signal abnormality in this 42-year-old woman with Devic’s disease.
Nontraumatic acute myelopathy most often results from spinal cord compression by a retropulsed neoplastic vertebral compression fracture, usually in an elderly patient with an unknown (or known) cancer, best evaluated by noncontrast whole spine MRI. Acute inflammatory myelopathy caused by transverse myelitis or demyelinating conditions are also best evaluated with MRI, with the addition of contrast (Fig. 32.9A–D). Less commonly, acute myelopathy is caused by spontaneous epidural. Traumatic cord contusions are best evaluated by MRI using T2, STIR, and gradient echo sequences.
Causes of chronic myelopathy include spinal cord tumors, degenerative disease and disc herniations, syringomyelia, congenital anomalies, inflammatory and demyelinating disease, and spinal dural arteriovenous fistulae. Initial evaluation of these patients is also best undertaken with pre- and postcontrast MRI. CT myelography should be reserved for the rare patient with a major contraindication to MRI.
![]()
Recommended Readings
Brazis PW, Masdeu JC, Biller J. Localization in Clinical Neurology. 6th ed. Philadelphia, PA: Wolters Kluwer Lippincott Williams & Wilkins; 2011.
Grossman RI, Yousem DM. Neuroradiology: The Requisites. 3rd ed. St Louis, MO: Mosby; 2010.
Osborn AG, Salzman KL, Barkovich AJ. Diagnostic Imaging: Brain. 2nd ed. Philadelphia, PA: Wolters Kluwer Lippincott Williams & Wilkins; 2009.