More than 40,000 new cases of primary CNS tumors are diagnosed each year in the United States. Brain tumors are the second most frequent cause of cancer-related death among children. Brain tumors may affect adults at any age, and often have a devastating effect on patients and their families. This review will summarize the current clinical approaches to the most common primary brain tumors. For most if not all brain tumors the therapeutic strategies are in a state of evolution, and there are more than a few uncertainties and controversies. The past several years have seen an explosion of new knowledge of the molecular genetics and basic biology of brain tumors. Although the clinical progress seems painfully slow, these new discoveries are gradually being translated into more precise disease stratification and into targets for new therapies.
I. GLIOBLASTOMA AND ANAPLASTIC ASTROCYTOMA
A. Course of disease. The World Health Organization (WHO) stratifies malignant astrocytic neoplasms into two main groups, anaplastic astrocytoma (AA, WHO grade 3) and glioblastoma (GBM, WHO grade 4) based on the degree of hypercellularity, nuclear pleomorphism, mitoses, microvascular proliferation, and necrosis. GBM is the most common glioma in adults, accounting for 50% to 60% of all cases. Unfortunately, this tumor is also the most deadly. AA accounts for 15% to 20% of adult gliomas. There is a slight male predominance. Peak age incidence for GBM is approximately 55 years and for AA about 45 years. Recent epidemiologic evidence suggests an increasing incidence. Approximately 10% of patients with AA or GBM had a prior histologically proven diagnosis of astrocytoma or other lower-grade glioma.
Patients with GBM or AA generally present with a fairly short history of some combination of headache, seizures, and focal neurologic symptoms determined by the tumor location. Malignant gliomas appear on MR scans as an irregular mass lesion with heterogenous or ring enhancement (Fig. 52.1). There is a predilection to extend across the corpus callosum or to spread along other major white matter pathways. T2-weighted or fluid attenuated inversion recovery images typically show abnormal signal extending in an irregular shape for considerable distance beyond the margins of contrast enhancement. In most if not all patients there are infiltrating tumor cells within and beyond the area of abnormal T2/fluid-attenuated inversion recovery (FLAIR) signal. The variable topography and distance of tumor cell infiltration are serious obstacles to attempts at surgical resection or other “focal” therapies for these tumors.
B. Therapy. Standard treatment for patients with newly diagnosed GBM/AA is maximal tumor resection consistent with preservation of neurologic function, followed by limited-field radiation therapy (RT), and for most patients chemotherapy begun during or after RT. Several modern techniques facilitate the aggressive resection of gliomas and reduce the risk of neurologic morbidity for selected patients. Preoperative functional MRI, diffusion-tensor MRI, and intraoperative cortical and subcortical mapping can determine the tumor’s proximity to and involvement of motor and speech structures. Intraoperative MRI allows the surgeon to assess the degree of resection and possibly continue the resection to remove more residual tumor.
For patients with symptomatic tumor mass effect, aggressive surgery usually improves neurologic function. Whether the extent of initial resection of GBM/AA has a major impact on survival continues to be controversial. There has never been and probably never will be a prospective randomized study in which patients are randomized to undergo differing degrees of tumor resection. Most (not all) retrospective studies show a survival advantage for patients who undergo an “aggressive” resection. The cutoff value of how much of the enhancing tumor needs to be resected to impact survival ranges from 75% to almost 100% in various studies. In studies where multivariate analysis showed the extent of resection to be an independent prognostic factor, the impact on survival was nearly always less than that for patient age, tumor histology, and pretreatment performance status.
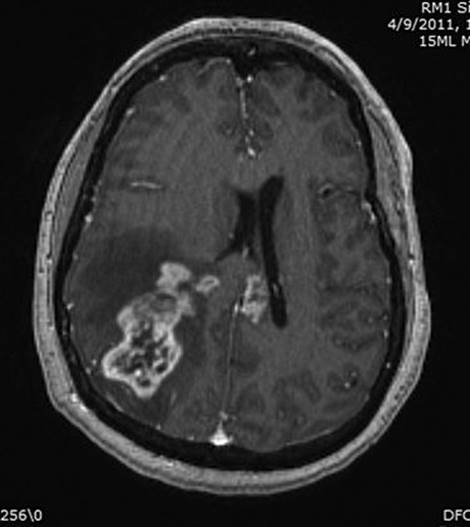
FIGURE 52.1 Axial T1-weighted MRI scan of a patient with a right parietal GBM, showing heterogeneous tumor enhancement, moderate surrounding cerebral edema and associated mass effect, and extension of tumor enhancement across the corpus callosum into the deep left hemisphere.
Standard postoperative RT for GBM/AA is 55 to 60 Gy “focal” or “limited-field” RT delivered to a target encompassing a 2 to 3 cm margin around the radiographically visible tumor area. GBM/AA occasionally spreads through the leptomeninges or recurs far from the initial tumor site, but for the vast majority of patients the ultimate cause of death is tumor recurrence within the initial RT target area. There is no evidence that higher doses of fractionated RT or a “boost” of stereotactic radiosurgery or brachytherapy in addition to conventional RT provide any survival advantage.
Based on a randomized prospective study, the current standard chemotherapy regimen for patients with newly diagnosed GBM is daily oral temozolomide taken concurrently during RT, followed by six or more monthly cycles of temozolomide after the completion of RT. Temozolomide is an alkylating agent that has excellent oral bioavailability and shows good penetration across the blood–brain barrier. Noncumulative myelosuppression is the dose-limiting toxicity. The efficacy of this chemotherapy regimen for patients with AA has not been definitively proven, but it is a common practice to administer the same treatment as for patients with GBM. For selected GBM/AA patients, another chemotherapy option is surgical implantation of BCNU-containing wafers at the time of initial resection.
Nearly all GBMs and AAs recur despite aggressive multimodality treatment. The median time to tumor progression after initial diagnosis of GBM is 6 to 9 months. For selected patients with relatively young age, good performance status, and accessible lesions, a second surgical resection can “set up” further chemotherapy, may improve neurologic function, and modestly prolongs survival. Depending on tumor size and location, some patients may benefit from single-dose or fractionated stereotactic radiosurgery. For many if not most patients with recurrent or progressive GBM or AA, further surgery or RT are judged not to be feasible or advisable. Systemic chemotherapy or other drug treatment is then the only option available. In the United States, the most commonly used treatment for recurrent GBM/AA is bevacizumab, a humanized monoclonal antibody against vascular endothelial growth factor. In addition to its antiangiogenic and antitumor effect, bevacizumab is a potent antivascular permeability and anticerebral edema agent.
There has been intense interest in a growing number of drugs that have a cytostatic or cytotoxic effect on malignant gliomas by virtue of blocking or inhibiting growth factor receptors, intracellular or extracellular signaling pathways, angiogenesis, or tumor cell migration. To date, studies of these molecularly targeted agents for newly diagnosed or recurrent GBM/AA have unfortunately yielded disappointing results. Except for bevacizumab, none are currently used in standard therapy.
Supportive care for patients with GBM/AA includes varying doses of dexamethasone to reduce peritumoral edema and increase neurologic function, and aggressive treatment of pain and/or depression if they occur. The concepts and strategies for treating glioma-associated seizures are the same as those for treating localization-related epilepsy in general. There is no definite evidence that any particular antiepileptic drug is differentially effective for glioma-related seizures versus epilepsy caused by other structural brain lesions. For patients taking dexamethasone or receiving chemotherapy agents metabolized by the liver, the nonenzyme-inducing antiepileptic drugs (e.g., levetiracetam or valproate) may offer fewer drug interactions than enzyme-inducing drugs (e.g., phenytoin or carbamazepine). For patients who do not have seizures at initial presentation, there is no definite evidence to support long-term prophylactic antiepileptic drugs.
C. Prognosis. Patient age, tumor histology, and performance status are the most important prognostic factors for patients with malignant glioma. These are useful as predictors of individual patient outcome and are critically important in designing and interpreting clinical trials. With standard multimodality treatment, patients with GBM have a median survival of 12 to 18 months and only about 25% survive 24 months. Patients with AA have a median survival of 3 to 4 years.
Median survival is inversely proportional to age throughout all decades of adult life; older patients have a worse prognosis. Patients with a better performance status at the time of diagnosis have a better survival outlook than patients who present with severe neurologic impairment. Recent gene expression profiling studies have identified distinct molecular subclasses of GBM/AA, which may be associated with different survival outcome, but this is not yet part of standard clinical practice.
II. ASTROCYTOMA
A. Course of disease. Astrocytoma, oligodendroglioma, and mixed oligoastrocytoma together comprise 25% to 30% of all gliomas in adults. These “low-grade gliomas” should not be considered “benign” tumors, as they generally lead to a fatal outcome. Low-grade (WHO grade 2) astrocytomas are poorly circumscribed and are characterized by diffuse infiltration of atypical astrocytes with hyperchromatic nuclei. Gross and microscopic boundaries are difficult to define. Most tumors contain a mixture of cells with “fibrillary,” “protoplasmic,” or “gemistocytic” morphology. Expression of the intermediate filament glial fibrillary acidic protein (GFAP), as demonstrated by immunocytochemical staining, is a marker for astroglial derivation.
The median age at the diagnosis of supratentorial astrocytoma in adults is 35 to 40 years, which is significantly younger than for AA or GBM. There is a slight male predominance. Since the advent of MR scanning in the mid-1980s, at least 70% of patients with astrocytoma present with seizures and no headache or other neurologic symptoms. Astrocytoma usually appears as a poorly demarcated mass lesion hypointense on T1-weighted MR images and hyperintense on T2-weighted and FLAIR images. Gadolinium enhancement is present in 10% to 30% of cases. Infiltration of tumor cells nearly always extends beyond the margins of radiographically visible tumor.
B. Therapy. Few of the key issues regarding treatment for patients with low-grade astrocytoma have been studied in well-designed prospective or randomized clinical trials. It is therefore difficult to dogmatically state the “conventional” treatment for these tumors. The proper treatment for patients needs to be individualized and based on several factors, including patient’s age, clinical presentation, tumor size and location, and tumor histology.
The fact that MRI scanning identifies patients with astrocytoma early in their natural course raises the question of whether all patients require immediate treatment when the lesion is discovered. Currently, the unequivocal indications for early intervention for patients with a presumed or proven astrocytoma include neurologic signs and symptoms other than seizures; presence of significant mass effect on neuroimaging; growth of the lesion on serial scans; and patient age ≥50 years. It is unclear whether the presence of MRI contrast enhancement should be an indication of early treatment, assuming the area of enhancement is biopsied and shown to be histologically low grade. For younger patients with astrocytoma who have no neurologic symptoms other than seizures, it is reasonable to defer surgical resection or RT until clinical or radiographic tumor progression occurs. There is no clear evidence that deferring treatment in this subset of patients has a negative impact on overall survival or on the likelihood of malignant tumor transformation.
Surgical resection or RT are the main treatment options for astrocytoma. Surgery is rarely curative. The impact of the extent of surgical resection on patients’ ultimate survival remains a matter of controversy. There has never been a prospective study in which “ideal candidates” with astrocytoma were randomly assigned to undergo varying degrees of surgical resection. Several retrospective series indicate a survival advantage for patients who underwent extensive surgical resection as compared with those who had only biopsy or “partial” tumor excision. In other series, the extent of surgery was not an independent predictor of survival. It is difficult to interpret these retrospective series because of a considerable selection bias: patients who have extensive resection are more likely to be younger, have better performance status, and have small, unilateral, relatively well-circumscribed tumors in noncritical locations than patients who have more conservative surgery.
There is also uncertainty whether all patients with astrocytoma should receive RT early in their course. In a large prospective multicenter study by the European Organization for Research and Treatment of Cancer (EORTC), patients with newly diagnosed astrocytoma (or oligodendroglioma) were randomized to either receive 54 Gy RT immediately after initial biopsy or resection, or to receive no RT until tumor progression. There was no difference in overall survival of patients who received early RT compared with patients in whom RT was deferred. There is no definite evidence that early RT benefits the subset of astrocytoma patients who undergo only biopsy versus surgical resection. Subsequent analysis of the EORTC study data showed a statistically significant prolongation in the time from initial diagnosis to tumor progression among patients who received early RT. These findings have been criticized for methodologic shortcomings because the study was not adequately designed to assess time to tumor progression as an endpoint.
If early RT for astrocytoma does not prolong survival but actually does delay tumor progression, it might be argued that early RT would delay tumor-related decline in patients’ neurologic function. The counter argument is that RT itself can cause neurotoxicity and should therefore be delayed as long as possible. Long-term neurocognitive toxicity of RT is a significant concern for patients with low-grade gliomas, because at the time of initial diagnosis most patients are young, have mild or no neurologic deficits, and have an anticipated survival of at least several years. The few published studies of serial neuropsychological testing of “long-term” survivors show conflicting results as to whether RT causes significant neurocognitive deficits. Recent evidence suggests worsening neurocognitive function as patients are followed for many years after RT. The question of whether early RT is more likely to have a positive or a negative effect on patients’ long-term neurologic function and quality of life is still unanswered.
C.Prognosis. In series published since the mid-1980s, the median survival of adults with supratentorial astrocytoma is 5 to 9 years. Patient’s age at diagnosis is a strong independent predictor of outcome: time to tumor progression and overall survival are significantly shorter for older patients, particularly those over 50 years of age. Significant neurologic impairment at diagnosis has a negative impact on survival. Patients who present with a long history of seizures and no other neurologic deficits have a relatively favorable prognosis. In some series, the presence of contrast enhancement on initial CT or MRI scans was predictive of shorter survival.
At the time of tumor recurrence or progression, 50% to 75% of initially low-grade astrocytomas will have undergone “malignant transformation” to AA or GBM. Malignant transformation of astrocytoma tends to occur sooner and more frequently among older patients than in younger patients. This histologic and phenotypic transformation reflects the acquisition of several genetic abnormalities. Unfortunately, it is not rare for astrocytomas that have remained stable for several years to eventually show progression leading to a fatal outcome. There does not seem to be a time point beyond which patients with astrocytoma can be confidently declared to be “cured” of their tumor.
III. OLIGODENDROGLIOMA
A. Course of disease. As with tumors of astrocytic derivation, oligodendrogliomas exhibit a spectrum of histologic malignancy. The WHO classification divides oligodendrogliomas and mixed oligoastrocytomas into two groups, low grade (grade 2) and anaplastic (grade 3). The classic histologic features of oligodendrogliomas are tumor cells with uniform round nuclei and clear perinuclear halos (“fried egg appearance”), with a “chicken wire” network of branching capillaries. Microcalcifications and microcystic spaces are common. The morphologic spectrum of oligodendrogliomas has been expanded to include tumors containing microgemistocytes or GFAP-expressing gliofibrillary oligodendrocytes. The presence of “significant” mitotic activity, microvascular proliferation, or necrosis defines an anaplastic oligodendroglioma. Median age of onset is 35 to 40 years for low-grade oligodendroglioma and around 50 years for anaplastic tumors. Mixed oligoastrocytomas contain some tumor cells with astrocytic morphology and other cells with oligodendrocytic morphology. The two elements are either spatially separate, or more often intermingled. Overall, low-grade and anaplastic oligodendrogliomas and oligoastrocytomas comprise about one-third of gliomas in adults.
As with astrocytomas, low-grade oligodendrogliomas present as seizures without other neurologic symptoms in at least 70% of patients. Anaplastic oligodendrogliomas or oligoastrocytomas present with some combination of seizures, headache, and focal neurologic symptoms. On MRI scans, low-grade oligodendrogliomas generally appear as an ill-defined nonenhancing lesion (Fig. 52.2A and B), whereas anaplastic tumors usually show heterogeneous or ring enhancement. Tumor calcifications are more frequent among oligodendroglial than among astrocytic tumors. Neither low-grade nor anaplastic oligodendrogliomas can be absolutely distinguished from astrocytic tumors based solely on neuroimaging characteristics.

FIGURE 52.2 Axial MRI scan from a patient with a left temporal low-grade oligodendroglioma, showing no contrast enhancement on T1-weighted images (A) and diffuse hyperintensity on FLAIR images (B).
B. Therapy. Treatment options for anaplastic oligodendroglioma or anaplastic oligoastrocytoma are similar to those for AA or GBM, including the maximal safe surgical resection followed by RT. As a group, oligodendrogliomas are more sensitive to chemotherapy than astrocytic tumors. Two prospective randomized studies of patients with newly diagnosed anaplastic oligodendroglioma showed that adding procarbazine/CCNU/vincristine chemotherapy (the PCV regimen) to surgery and RT significantly prolonged time to tumor progression but did not significantly prolong overall survival. Most patients in these studies who did not receive upfront PCV eventually received PCV or temozolomide at the time of tumor recurrence. Alternative options for patients with newly diagnosed anaplastic oligodendroglioma after biopsy or resection are RT only, temozolomide chemotherapy only, or combined RT plus temozolomide as is done for patients with GBM.
Many of the issues regarding management of patients with low-grade oligodendroglioma are similar to those for astrocytoma (see above). Low-grade oligodendrogliomas are generally more indolent than astrocytomas; for young patients who present with seizures and no other neurologic symptoms, it is reasonable to defer intervention until there is radiographic or clinical tumor progression. When intervention is deemed, necessary, patients should probably undergo the maximal surgical resection consistent with improved or preserved neurologic function. Early (vs. deferred) RT for low-grade oligodendroglioma does not prolong overall survival. Several nonrandomized studies have shown that chemotherapy (usually temozolomide) is effective in many patients with newly diagnosed or recurrent oligodendroglioma or oligoastrocytoma. The maximum radiographic response of oligodendroglioma to chemotherapy may not be seen for 8 to 12 months. Favorable response is associated with chromosome 1p/19q deletion, and with lower expression of the methylguanine methyltransferase DNA repair protein. There is no direct randomized comparison of chemotherapy versus radiation for low-grade oligodendroglioma, but chemotherapy is a reasonable alternative to RT in the hope of being able to defer RT and its risk of neurocognitive toxicity for as long as possible.
C. Prognosis. As with astrocytic tumors, younger age and good performance status are predictors of better outcome for patients with oligodendroglioma, independent of tumor grade. In recent published series, the median survival is 8 to 12 years for adults with low-grade oligodendroglioma and is 4 to 6 years for persons with anaplastic oligodendroglioma. Mixed oligoastrocytomas generally have a survival outcome intermediate between astrocytomas and “pure” oligodendrogliomas.
Two genetic changes in oligodendrogliomas have known prognostic significance. A chromosomal translocation resulting in codeletion of one copy of 1p and 19q is present in 60% to 75% of newly diagnosed low-grade oligodendrogliomas and about 50% of anaplastic oligodendrogliomas. A point mutation of the IDH1 gene encoding isocitrate dehydrogenase occurs in at least two-thirds of low-grade and anaplastic oligodendrogliomas and oligoastrocytomas. The 1p/19q co-deletion and IDH1 mutation usually coexist in the same tumor. Each is associated with improved survival independent of other prognostic factors.
IV. MENINGIOMA
A. Course of disease. The majority of meningiomas are asymptomatic and discovered incidentally by neuroimaging studies or at autopsy (see below). Symptomatic meningiomas are twice as common among women than men and account for approximately 20% of primary brain tumors in adults. There is a steadily increasing incidence above 20 years of age, with peak incidence during the seventh decade.
The WHO classification uses hypercellularity, nuclear pleomorphism, mitotic rate, focal necrosis, and infiltration of brain parenchyma to divide meningiomas into “benign” (grade 1, accounting for 85% of cases), “atypical” (grade 2, 5% to 15% of cases), and “malignant” or “anaplastic” (grade 3, 1% to 5% of cases). There is a variety of histologic subtypes of meningiomas, including meningothelial, transitional, and fibrous; these subtypes generally do not have prognostic significance, except for more aggressive clinical behavior among the clear-cell, chordoid, rhabdoid, and papillary subtypes. Brain invasion is associated with a higher rate of tumor recurrence after therapy.
The clinical presentation of meningiomas is determined by their anatomic site. The most common sites of origin are along the cerebral convexity, parasagittal area and falx, and along the sphenoid ridge; together these sites account for at least two-thirds of cases. The slow growth of these tumors is reflected in the slow progression of signs and symptoms.
Meningiomas arise adjacent to dural surfaces and have a characteristic diffuse, homogeneous contrast enhancement on CT, or on MRI scans (Fig. 52.3). Calcification is present in at least one-third of cases. Peritumoral edema is variable and in some cases dramatic. Approximately 20% of patients have hyperostosis in the skull adjacent to the tumor; this bone is usually invaded by tumor cells. Approximately 5% of patients have two or more meningiomas at separate sites. The neuroimaging features of atypical or malignant meningiomas do not differ reliably from those of benign tumors. Arteriography or MR angiography/venography are frequently required to delineate the tumor’s blood supply in consideration of surgery or preoperative embolization.
B. Therapy. Modern neuroimaging techniques have led to increased detection of incidentally discovered, asymptomatic meningiomas. Serial MRI scans in these patients usually show slow or no growth over several years. It is reasonable to defer surgery or other intervention, especially in elderly patients, unless symptoms develop or the tumor clearly enlarges.
The optimum treatment for symptomatic meningiomas is total surgical resection, if it can be done safely. The success (and morbidity) of aggressive surgery depends mainly on tumor location. Overall, gross total resection can be achieved in approximately 75% of patients. Tumors along the hemispheric convexity, anterior falx, or olfactory groove are most amenable to complete excision. For meningiomas arising in some anatomic locations such as petroclival, parasellar, cavernous sinus, or orbital tumors, gross total resection is often not possible without causing unacceptable neurologic morbidity. Some patients with recurrent meningioma are candidates for a second surgical resection, depending on tumor size and location.

FIGURE 52.3 Axial T1-weighted MRI scan from a patient with a right parasellar meningioma, showing homogeneous contrast enhancement extending into the adjacent cavernous sinus and orbit.
RT is not given following gross total resection of benign meningioma, but is generally recommended for patients with (1) symptomatic benign meningioma not amenable to aggressive surgical resection; (2) significant residual benign meningioma following attempted resection; (3) recurrent tumor following surgery; and (4) newly diagnosed atypical or anaplastic meningioma, regardless of the extent of initial surgical resection. Of these indications for RT, perhaps the most controversial is the group of patients with subtotally resected benign meningioma. The weight of retrospective data suggests a significantly lower long-term recurrence rate for these patients if they receive RT shortly after surgery.
RT options for meningioma include “standard” fractionated conformal RT, intensity-modulated RT, single-dose or fractionated stereotactic radiosurgery, interstitial brachytherapy, and proton beam therapy. Intensity-modulated RT, stereotactic radiosurgery, and proton beam therapy offer the theoretical advantage of being able to deliver a therapeutic dose to an irregularly shaped target, with reduced risk to nearby normal structures such as the optic pathways or brainstem. Published series report partial tumor shrinkage in 30% to 50% of patients and “tumor control” (stable or improved MRI scans) in up to 90% of patients, at least over a 5-year follow-up period. There are only a few studies that have determined control rates over 10 years or longer. Despite the common occurrence of meningiomas, there are virtually no prospective or controlled comparative studies of any RT approach or modality.
A significant proportion of patients with recurrent meningioma has tumors, which are not surgically resectable, have exhausted options for RT, and would therefore benefit from effective systemic treatment. Unfortunately, meningiomas are generally not sensitive to currently available chemotherapy agents. There are a few reports of partial response or prolonged tumor stabilization in patients with recurrent or anaplastic meningiomas treated with hydroxyurea, temozolomide, other chemotherapy regimens, tamoxifen, antiprogesterone agents, interferon-alpha, or somatostatin. To date, molecularly targeted agents such as the tyrosine kinase inhibitors imatinib or erlotinib have not shown significant efficacy.
C. Prognosis. The most important prognostic factors for meningioma are the extent of initial resection and the histologic tumor grade. Following gross total resection of benign meningioma, recurrence-free survival rates are close to 90% at 5 years, declining to 75% at 10 years, and 65% at 15 years. A high percentage of these patients never have tumor recurrence during their lifetime. Following subtotal resection alone, tumor recurrence rates at various time points are at least twice as high as for patients with gross total resection. Outcome is improved for patients who receive postoperative RT after incomplete resection.
Patients with atypical and malignant meningiomas clearly have a higher tumor recurrence rate and a shorter survival outlook than benign tumors. Approximately 50% of patients with atypical or malignant meningioma have tumor recurrence within 5 years of initial treatment. Reported median overall survival times vary between 2 and 10 years.
V. PRIMARY CNS LYMPHOMA
A. Course of disease. The great majority of primary CNS lymphomas (PCNSLs) occur sporadically in persons with no apparent immune deficiency. The incidence of PCNSL among immunocompetent persons has been increasing over the past 25 years, with a peak incidence between age 60 and 65 years. PCNSL accounts for about 3% of all primary brain tumors in adults. It is disproportionately common among patients with HIV infection, though the incidence has dropped dramatically with the advent of modern antiretroviral therapy. It may also occur in recipients of organ transplants or in persons with other iatrogenic immunodeficiency states.
More than 90% of PCNSLs are classified as diffuse large-cell B-lymphocyte tumors. The Epstein–Barr virus is detected in 90% of PCNSLs associated with HIV infection or organ transplants, but only rarely in sporadically occurring tumors. The origin and pathophysiology of PCNSL in immunocompetent persons is poorly understood.
Patients generally present with a combination of altered mental status and focal neurologic symptoms. Neurologic deficits often progress rapidly and the diagnosis is usually made within 2 to 3 months. Seizures are less common than among patients with gliomas. PCNSL has a predilection for arising in deep or midline brain structures. On MRI scans, PCNSL characteristically appears as a bright fairly homogeneously enhancing mass lesion (Fig. 52.4). About one-half of patients have multifocal lesions. Lymphomatous infiltration of the posterior vitreous and/or retina (often asymptomatic) occurs in 10% to 20% of patients. Leptomeningeal dissemination occurs at some time in 10% to 40% of patients and is usually asymptomatic when present at the time of initial diagnosis.
B. Therapy. PCNSL is unique among primary brain tumors in that corticosteroids not only reduce peritumoral cerebral edema, but also have a direct oncolytic effect and can produce significant (but temporary) clinical and radiographic improvement. Whenever possible, steroids should be withheld prior to biopsy of patients with suspected PCNSL because their oncolytic effect may render the biopsy nondiagnostic.
Attempted surgical resection is of no benefit for patients with PCNSL. The only role for surgery is to provide diagnostic biopsy.
Patients with newly diagnosed PCNSL should have a staging workup including MRI of the brain and total spine, CSF exam (if safe) for cytology and flow cytometry studies, slit lamp ophthalmologic exam, CT scans of the chest and abdomen, serum lactate dehydrogenase, and HIV serology. IgH gene rearrangement analysis of CSF may show evidence for leptomeningeal tumor when other studies are negative or equivocal. It is not clear whether all patients should have a bone marrow biopsy and/or total-body FDG-PET scan.
High-dose intravenous (IV) methotrexate is the mainstay of treatment for PCNSL. When given in sufficient doses, methotrexate penetrates into the brain parenchyma regardless of the state of the blood–brain barrier and also produces cytotoxic concentrations in the CSF. The optimal methotrexate dose, schedule, and number of treatment cycles are not clearly known. For patients treated with high-dose IV methotrexate, upfront intrathecal chemotherapy is generally not recommended in the absence of definite radiographic or CSF evidence for leptomeningeal tumor. Several nonrandomized studies of high-dose methotrexate-based chemotherapy for PCNSL have shown radiographic complete responses in the majority of patients, and median survival of 3 years or more.

FIGURE 52.4 Coronal T1-weighted MRI scan from a patient with PCNSL, showing a large area of bright homogeneously enhancing tumor centered in the left thalamus, and a smaller focus of enhancing tumor in the superficial right frontal lobe.
High-dose IV cytarabine also achieves good brain and CSF concentrations. There are numerous published studies of multiagent regimens in which high-dose cytarabine, other chemotherapy drugs, or rituximab (an anti-CD20 monoclonal antibody) are added to high-dose methotrexate. Multiagent methotrexate-based regimens may yield longer progression-free survival than single-agent methotrexate therapy, but are generally more toxic, and have not been clearly shown to prolong overall survival. Other chemotherapy options for newly diagnosed PCNSL include high-dose chemotherapy with autologous stem cell rescue, or hyperosmolar disruption of the blood–brain barrier followed by a combination of intra-arterially and systemically administered drugs.
PCNSL is highly responsive to RT, but following RT alone the tumor recurs quickly and the median survival is only 12 to 18 months. Whole-brain RT (35 to 50 Gy) is generally given after the “induction” methotrexate-based chemotherapy. Whole-brain RT should be given to all patients who do not attain a complete radiographic remission (no contrast-enhancing tumor) following induction chemotherapy. It is less clear whether all patients who achieve a complete radiographic remission to initial methotrexate-based chemotherapy should then receive “consolidation” whole-brain RT. There are conflicting data whether consolidation RT necessarily extends overall survival in these patients versus deferring RT until the time of tumor recurrence. There is also concern over delayed neurotoxicity of PCNSL treatment: the risk of severe neurocognitive decline and leukoencephalopathy in survivors increases with patient age and among patients who receive methotrexate plus RT.
C. Prognosis. Younger age and good initial performance status are the two most important prognostic factors for survival outcome in PCNSL. There is a discrepancy between published median survivals of 3 to 4 years or more in studies of methotrexate-based chemotherapy versus median survivals of 12 to 18 months in population-based series. This may reflect a selection bias in which patients with better prognostic factors are entered into chemotherapy trials, and/or a less aggressive treatment approach in general community practice.
VI. MEDULLOBLASTOMA
A. Course of disease. Medulloblastoma is the most common malignant brain tumor of childhood, comprising 20% of brain tumors occurring before age 18 years. There is a bimodal incidence peak, at 3 to 4 years and 8 to 10 years of age, with a slight male predominance. About 20% of all medulloblastomas occur in adults, usually before age 30 years.
The WHO classification divides medulloblastomas into three main histologic groups. At least two-thirds of tumors have a “classic” histology, with sheets of “small, round, blue” tumor cells with hyperchromatic nuclei. The nodular/desmoplastic histologic pattern is present in 10% to 20% of cases and is disproportionately common in adults and in children under 3 years of age. The third histologic group shows severe anaplasia and/or large tumor cells, accounting for 5% to 20% of cases.
Medulloblastomas in children usually arise in or near the cerebellar vermis and fourth ventricle, and thus present with a combination of headache, vomiting (often occurring in the morning), lethargy, and gait ataxia. Tumors arising more laterally in the cerebellar hemisphere present with ipsilateral ataxia, with or without signs and symptoms of increased intracranial pressure. Tumor enlargement or invasion into the brainstem cause cranial nerve palsies and long-tract findings. The diagnosis is usually made within 2 to 3 months of symptom onset.
Medulloblastoma is the primary brain tumor most likely to disseminate in the subarachnoid space and is also the brain tumor most likely to metastasize outside the CNS, usually in the setting of recurrent disease at the primary site.
On MRI scans, medulloblastoma usually appears as a homogeneously or heterogeneously enhancing mass filling and distorting the fourth ventricle. The tumor may be centered more laterally in the cerebellar hemisphere (Fig. 52.5). Calcifications or hemorrhage may be present. Hydrocephalus is present in at least 75% of cases at diagnosis.
B. Therapy. Multimodality treatment for medulloblastoma includes aggressive surgical resection, followed by “risk-adapted” RT and chemotherapy. Gross total resection can be performed in approximately 75% of patients. One-half of patients require placement of a permanent CSF shunt. Postoperatively, children are clinically stratified into “average-risk” and “high-risk” prognostic subgroups based the extent of initial surgical resection and the presence or absence of leptomeningeal dissemination. Average-risk patients (about two-thirds of the total) have a gross total or nearly gross total tumor resection, negative CSF cytology, and no leptomeningeal spread on brain and total spine MRI scanning. Patients are high-risk if they have a less complete tumor resection, and/or evidence for leptomeningeal dissemination at diagnosis.
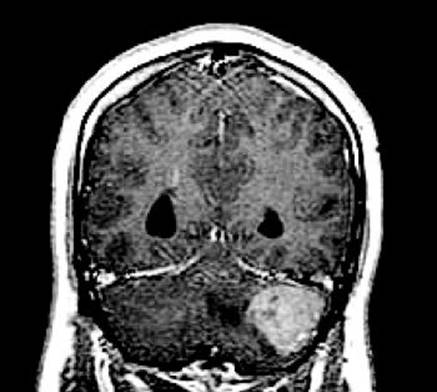
FIGURE 52.5 Coronal contast-enhanced T1-weighted MRI scan from a young man with a desmoplastic medulloblastoma in the left cerebellar hemisphere.
Average-risk children older than 3 years of age receive RT to the craniospinal axis (23 to 25 Gy), plus a higher RT dose to the tumor site (55 Gy), and posterior fossa (36 Gy). Multiagent chemotherapy (usually including cisplatin, cyclophosphamide, and vincristine) is given for several months after RT.
Children over the age 3 years with high-risk disease also receive postoperative RT, usually at a higher craniospinal axis dose than average-risk patients. There is a general strategy to give more intensified chemotherapy regimens to high-risk than to average-risk children, though there is no clear evidence establishing the best approach.
Children < 3 years of age have a higher incidence of leptomeningeal dissemination at diagnosis. The risk of severe neurocognitive toxicity in survivors is much higher if RT is given early in childhood, so these patients generally receive intensive chemotherapy regimens after surgery with the goal of obviating or at least deferring RT for as long as possible.
Following tumor resection, adults with medulloblastoma are stratified into average-risk and high-risk groups as are older children, and treated with RT and chemotherapy accordingly. Adults tend to tolerate the multiagent “childhood medulloblastoma” chemotherapy regimens less well than do children.
C. Prognosis. The most frequent mode of treatment failure is recurrence in the posterior fossa, with or without leptomeningeal dissemination. In modern series which include chemotherapy as part of multimodality treatment, the 5-year progression-free survival rate for older children with average-risk disease is 80% or more. The majority of these children are cured. The 5-year progression-free survival rate is 40% to 70% for older children with high-risk disease, and 30% to 50% for children < 3 years of age. Large cell/anaplastic medulloblastomas generally carry a worse outcome. The nodular/desmoplastic variant in young children is associated with better survival outcome. Metastatic disease at diagnosis carries a worse prognosis at any age. The present clinical staging scheme is not able to identify which average-risk patients are more likely to develop tumor recurrence and therefore require more aggressive initial treatment, nor to identify which patients could be cured with less aggressive treatment (especially the dose of craniospinal RT) so as to reduce the incidence and severity of long-term treatment sequelae. In recent gene expression profiling studies, medulloblastomas fall into 4 or 5 groups with differing molecular genetics and somewhat different (though overlapping) representation of patient age, gender, and tumor histology. It is hoped that molecular subtyping will further refine the current clinical patient staging and help to individualize therapy.
The relatively favorable survival outcome and potential curability of medulloblastoma are tempered by significant treatment sequelae in the majority of survivors. About 25% of children develop the “cerebellar mutism syndrome” within a few days after surgery. This consists of mutism, axial hypotonia, ataxia, and irritability. Patients eventually recover, but one-half of severely affected children have long-term speech, motor, and cognitive deficits. Survivors of childhood medulloblastoma have a high incidence of other long-term sequelae, including growth failure and other neuroendocrine dysfunction, neurocognitive deficits which may be progressive, requirement for special education, behavioral disorders, and the risk of second neoplasms including meningioma and glioma. Toxicities are more common and more severe among children treated at a younger age.
![]()
Recommended Readings
Bromberg JE, van den Bent MJ. Oligodendrogliomas: molecular biology and treatment. Oncologist. 2009;14:155–163.
Campbell BA, Jhamb A, Maguire JA, et al. Meningiomas in 2009: controversies and future challenges. Am J Clin Oncol. 2009;32:73–85.
Crawford JR, MacDonald TJ, Packer RJ. Medulloblastoma in childhood: new biological advances. Lancet Neurol. 2007;6:1073–1085.
Morris PG, Abrey LE. Therapeutic challenges in primary CNS lymphoma. Lancet Neurol. 2009;8:581–592.
Pfister SM, Korshunov A, Kool M, et al. Molecular diagnosis of CNS embryonal tumors. J Neuro-oncol. 2010;120:553–566.
Schiff D, Brown PD, Giannini C. Outcome in adult low-grade glioma: the impact of prognostic factors and treatment. Neurology. 2007;69:1366–1373.
Stupp R, Hegi ME, Mason WP, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466.
van den Bent MJ, Afra D, de Witte O, et al. Long-term efficacy of early versus delayed radiotherapy for low-grade astrocytoma and oligodendroglioma in adults: the EORTC 22845 randomised trial. Lancet. 2005;366:985–990.
Wen PY, Kesari S. Malignant gliomas in adults. N Engl J Med. 2008;359:492–507.