Patrick J. McMahon, MD
Lee D. Kaplan, MD
Charles A. Popkin, MD
INTRODUCTION
Sports medicine developed in the 1970s as an orthopedic specialty focusing on competitive athletes. Today, sports medicine includes the overall care of athletes from many skill levels. Increasingly, care of recreational athletes has risen to that common for professional athletes. In addition to the musculoskeletal system, care includes the cardiovascular and pulmonary systems and also focuses on training techniques, nutrition, and women’s athletics. This wide range of care requires a multidisciplinary team of medical personnel, including athletic trainers, physical therapists, cardiologists, pulmonologists, orthopedic surgeons, and general practitioners.
![]() KNEE INJURIES
KNEE INJURIES
![]() Anatomy
Anatomy
The bones of the knee are the distal femur, the proximal tibia, and the patella. These bones depend on supporting ligaments, the joint capsule, and the menisci to provide stability for the joint.
A. Menisci and Joint Capsule
The menisci, or semilunar cartilages, are C-shaped fibrocartilaginous disks in the knee that provide shock absorption, allow for increased congruency between joint surfaces, enhance joint stability, and aid in distribution of synovial fluid.
The medial and lateral menisci provide a concave surface with which the convex femoral condyles can articulate. If the menisci are not present, the convex femoral condyles articulate with the relatively flat tibial plateaus, and the joint surfaces are not congruent. This decreases the surface area of contact and increases the pressure on the articular cartilage of the tibia and femur, which may lead to rapid deterioration of the joint surface. The medial meniscus is firmly attached to the joint capsule along its entire peripheral edge. The lateral meniscus is attached to the anterior and posterior capsule, but there is a region posterolaterally where it is not firmly attached (Figure 3–1). Therefore, the medial meniscus has less mobility than the lateral meniscus and is more susceptible to tearing when trapped between the femoral condyle and tibial plateau. The lateral meniscus is larger than the medial meniscus and carries a greater share of the lateral compartment pressure than the medial meniscus carries for the medial compartment.

![]() Figure 3–1. The medial and lateral menisci with their associated intermeniscal ligaments. Note: The lateral meniscus is not attached in the region of the popliteus tendon. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
Figure 3–1. The medial and lateral menisci with their associated intermeniscal ligaments. Note: The lateral meniscus is not attached in the region of the popliteus tendon. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
B. Ligaments
Within the knee, the anterior cruciate ligament (ACL) travels from the medial border of the lateral femoral condyle to its insertion site anterolateral to the medial tibial spine. This ligament prevents anterior translation and rotation of the tibia on the femur (Figure 3–2). The posterior cruciate ligament (PCL) prevents posterior subluxation of the tibia on the femur. It runs from the lateral aspect of the medial femoral condyle to the posterior aspect of the tibia, just below the joint line (Figure 3–3). On the medial side, the medial collateral ligament has superficial and deep portions (Figure 3–4), which stabilize the knee to valgus stresses. The lateral collateral or fibular collateral ligament runs from the lateral femoral condyle to the head of the fibula. It is the main stabilizer against varus stress (Figure 3–5). The lateral collateral ligament is part of the posterolateral “complex” or “corner” of the knee that also resists external rotation. An important component is the popliteofibular ligament, present in 90% of knees, that runs from the tendon of the popliteus muscle to the styloid on the posterior fibular head.

![]() Figure 3–2. Drawing of the anterior cruciate ligament with the knee in extension, showing the course of the ligament as it passes from the medial aspect of the lateral femoral condyle to the lateral portion of the medial tibial spine. (Reproduced, with permission, from Girgis FG, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)
Figure 3–2. Drawing of the anterior cruciate ligament with the knee in extension, showing the course of the ligament as it passes from the medial aspect of the lateral femoral condyle to the lateral portion of the medial tibial spine. (Reproduced, with permission, from Girgis FG, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)

![]() Figure 3–3. Drawing of the posterior cruciate ligament, showing the course of the ligament as it passes from the lateral aspect of the medial femoral condyle to the posterior surface of the tibia. (Adapted, with permission, from Girgis FG, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)
Figure 3–3. Drawing of the posterior cruciate ligament, showing the course of the ligament as it passes from the lateral aspect of the medial femoral condyle to the posterior surface of the tibia. (Adapted, with permission, from Girgis FG, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)

![]() Figure 3–4. Medial capsuloligamentous complex. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)
Figure 3–4. Medial capsuloligamentous complex. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)

![]() Figure 3–5. The lateral supporting structures of the knee. (Reproduced, with permission, from Rockwood CA Jr, Green DP, Bucholz RW, et al: Fractures in Adults, 2nd ed. New York: Lippincott; 1984.)
Figure 3–5. The lateral supporting structures of the knee. (Reproduced, with permission, from Rockwood CA Jr, Green DP, Bucholz RW, et al: Fractures in Adults, 2nd ed. New York: Lippincott; 1984.)
![]() History and Physical Examination
History and Physical Examination
A. General Approach
The history of a knee injury may be obtained by asking the patient the questions listed in Table 3–1. The physical examination begins with observation of the patient’s gait. The uninjured knee is then examined as a basis of comparison with the injured knee. Any swelling or effusion should be noted. A small effusion will cause obliteration of the recesses on the medial and lateral aspects of the patellar tendon; with a larger effusion, diffuse swelling is present in the region of the suprapatellar pouch. Then, a fluid wave can be palpated on the sides of the patella. Active and then passive range of motion is tested carefully. The knee is palpated to define areas of localized tenderness. The joint lines are located at the level of the inferior pole of the patella when the knee is flexed to 90 degrees.
Table 3–1. History of a knee injury.

B. Ligament Laxity Evaluation
To determine varus and valgus stability, the patient’s foot is held between the examiner’s elbow and hip. This leaves both hands free to palpate the joint (Figure 3–6). Stability should be determined at both full extension and 30 degrees of knee flexion. Grading of laxity is based on the amount of opening of the joint (grade 1, 0–5 mm; grade 2, 5–10 mm; and grade 3, 10–15 mm). Laxity in full extension to varus or valgus angulation is an ominous sign that indicates disruption of key ligamentous structures. If significant valgus laxity is present in full extension, the posteromedial capsule and medial collateral ligament are torn. With varus laxity in full extension, the posterolateral capsular complex is torn, in addition to the lateral collateral ligament. With either varus or valgus laxity at full extension, ACL and PCL tears are likely. At 30 degrees of flexion, the posterior capsule and cruciate ligaments are relaxed and the medial and lateral collateral ligaments are isolated. Pain with varus or valgus stress is more suggestive of ligament damage than a meniscus tear.

![]() Figure 3–6. The collateral ligaments being tested in extension and 30 degrees of flexion with the foot held between the examiner’s elbow and hip. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)
Figure 3–6. The collateral ligaments being tested in extension and 30 degrees of flexion with the foot held between the examiner’s elbow and hip. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)
C. Lachman Test
The Lachman test is the most sensitive test for ACL tears. It is done with the knee flexed at 20 degrees, stabilizing the distal femur with one hand and pulling forward on the proximal tibia with the other hand (Figure 3–7). With an intact ligament, minimal translation of the tibia occurs and a firm end point is felt. With a torn ACL, more translation is noted, and the end point is soft or mushy. The hamstring muscles must be relaxed during this maneuver to prevent false-negative findings. Comparison of the injured and uninjured knees is essential.
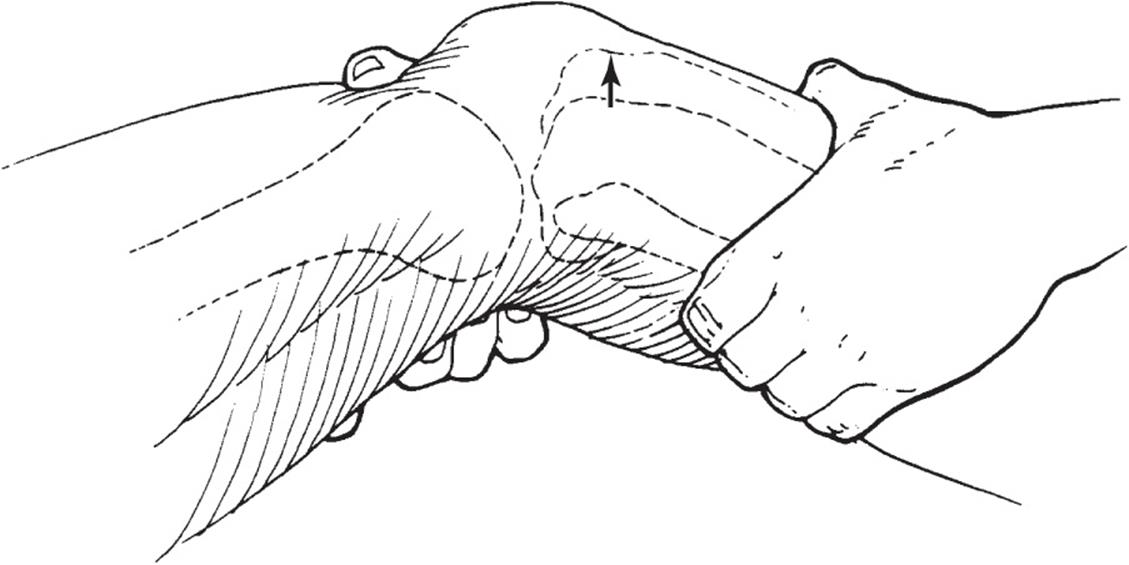
![]() Figure 3–7. Lachman test. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)
Figure 3–7. Lachman test. (Reproduced, with permission, from Feagin JA Jr: The Crucial Ligaments. New York: Churchill Livingstone; 1988.)
D. Anterior Drawer Test
The anterior drawer test is done with the knee at 90 degrees of flexion and is not as sensitive as the Lachman test but serves as an adjunct in the evaluation of ACL instability (Figure 3–8). With the patient supine and the knee flexed to 90 degrees (hip flexed to about 45 degrees), the foot is restrained by sitting on it and the examiner’s hands are placed around the proximal tibia. Then, while the hamstrings are felt to relax and the tibia is pulled forward, the displacement and the end point are evaluated.
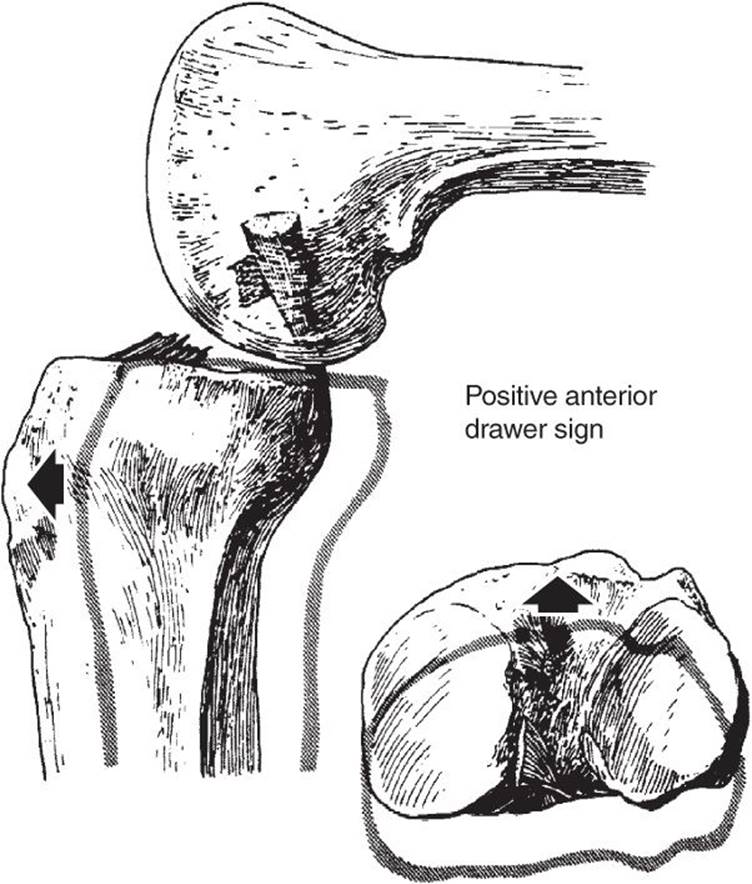
![]() Figure 3–8. A positive anterior drawer test signifying a tear of the anterior cruciate ligament. (Reproduced, with permission, from Insall JN: Surgery of the Knee. New York: Churchill Livingstone; 1984.)
Figure 3–8. A positive anterior drawer test signifying a tear of the anterior cruciate ligament. (Reproduced, with permission, from Insall JN: Surgery of the Knee. New York: Churchill Livingstone; 1984.)
E. Losee Test
The pivot shift phenomenon demonstrates the instability associated with an ACL tear. Once demonstrated, it is often difficult to repeat because the patient may find this maneuver uncomfortable and will guard against having it done again. As described by Losee, a valgus and internal rotation force is applied to the tibia (Figure 3–9). Starting at 45 degrees of flexion, the lateral tibial plateau is reduced. Extending the knee causes the lateral plateau to subluxate anteriorly with a thud at about 20 degrees of flexion. It reduces quietly at full extension. Many other ways of doing this test have been described, but the phenomenon and significance of the different tests are similar.

![]() Figure 3–9. The Losee pivot shift test. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
Figure 3–9. The Losee pivot shift test. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
F. Posterior Drawer Test
The posterior drawer test evaluates the integrity of the PCL. It is performed with posterior pressure on the proximal tibia with the knee flexed at 90 degrees and (Figure 3–10). Normally, the tibial plateau is anterior to the femoral condyles, and a “step-off” to the tibia is palpated when the thumb is slid down the femoral condyles. With a PCL injury, sagging of the tibial plateau may be appreciated, and no step-off is palpated (Figure 3–11). An associated contusion on the anterior tibia suggests a PCL injury.

![]() Figure 3–10. The posterior drawer test is done in the same fashion as the anterior drawer test, except that the examiner exerts a posterior force. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
Figure 3–10. The posterior drawer test is done in the same fashion as the anterior drawer test, except that the examiner exerts a posterior force. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)

![]() Figure 3–11. The posterior sag seen in posterior cruciate disruption. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
Figure 3–11. The posterior sag seen in posterior cruciate disruption. (Reproduced, with permission, from Scott WN: Ligament and Extensor Mechanism Injuries of the Knee: Diagnosis and Treatment. New York: Mosby-Year Book; 1991.)
G. McMurray Test
With the McMurray test, forced flexion and rotation of the knee will elicit a clunk along the joint line if there is a meniscus injury (see Figure 3–12). Found in less than 10% of patients with a meniscus injury, joint line pain with the McMurray test is much more common.

![]() Figure 3–12. The McMurray test to produce click. (Reproduced, with permission, from American Academy of Orthopaedic Surgeons: Athletic Training and Sports Medicine, 2nd ed. Burlington, MA: Jones and Bartlett; 1991.)
Figure 3–12. The McMurray test to produce click. (Reproduced, with permission, from American Academy of Orthopaedic Surgeons: Athletic Training and Sports Medicine, 2nd ed. Burlington, MA: Jones and Bartlett; 1991.)
![]() Arthroscopic Examination
Arthroscopic Examination
A. Indications for Knee Injuries
Indications for arthroscopic examination in the knee include the following:
1. Acute hemarthrosis
2. Meniscus injuries
3. Loose bodies
4. Selected tibial plateau fractures
5. Patellar chondromalacia and/or malalignment
6. Chronic synovitis
7. Knee instability
8. Recurrent effusions
9. Chondral and osteochondral fractures
Today, a specific diagnosis of the type of knee injury can usually be made with a history, physical examination, and appropriate imaging studies. With an examination under anesthesia and arthroscopic evaluation, a specific diagnosis can be confirmed, expanded, or revised, and treatment can be rendered as needed.
B. Technique
Examination under anesthesia is very helpful in diagnosing ligament injuries and instability. It should be performed before the beginning of the procedure, before preparing and draping the extremity. For diagnostic arthroscopy, the knee joint is distended with irrigating fluid (usually saline or lactated Ringer solution), which washes away blood and debris from the joint. A lateral portal for the arthroscope is placed about a thumb’s breadth above the joint line and just lateral to the patellar tendon. The medial portal is placed at about the same level, but just medial to the patellar tendon for introducing arthroscopic tools such as a probe. One approach to the general inspection of the joint is to start in the suprapatellar pouch. Loose bodies and plicas are sought. The patellofemoral joint is then inspected and observed for tracking problems and cartilage damage. The lateral gutter and the popliteus tendon are examined by flexion and valgus stress to the leg, prior to entering the medial compartment. The medial meniscus is probed using a nerve hook through the medial portal. The intercondylar notch, including the ACL, is inspected. The lateral compartment is then examined in a similar manner. Documentation of findings and procedures performed is important and may be done by videotape, photographs, and diagrammatic sketches. With assessment of the pathologic changes, treatment can be initiated, such as debridement and repair of meniscus tears, removal of loose bodies, or ACL reconstruction.
![]() Imaging and Other Studies
Imaging and Other Studies
A. Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) is a powerful technique for evaluation of the knee joint. While the diagnosis is usually evident from the history and physical examination, MRI can be used to confirm the suspected injury. Other times, when a physical examination is not possible because of pain or the diagnosis remains elusive, MRI can aid in proper diagnosis. The specificity, sensitivity, and accuracy of MRI are greater than 90% for the medial and lateral menisci and the ACL and PCL. Therefore, MRI is often appropriate for ruling out the need for diagnostic arthroscopic examination. It is less helpful for the diagnosis of problems in knees with previous surgery.
B. Imaging Studies
Roentgenographic examination of the knee is indicated in the evaluation of traumatic injury. In cases of minimal trauma, radiographs may not be needed if the injury proves to be self-limited. Arthrographic examination can be helpful in patients who are unable to undergo MRI because of claustrophobia, metal in the body that may be dislodged, or other contraindications.
C. Laboratory Tests
Laboratory tests may be helpful in ruling out nonmechanical disorders such as inflammatory arthritis, as described in Chapter 6.
Behairy NH, Dorgham MA, Khaled SA: Accuracy of routine magnetic resonance imaging in meniscal and ligamentous injuries of the knee: comparison with arthroscopy. Int Orthop 2009;33:961. [PMID: 18506445]
Kramer DE, Micheli LJ: Meniscal tears and discoid meniscus in children: diagnosis and treatment. J Am Acad Orthop Surg 2009;17:698. [PMID: 19880680]
Meserve BB, Cleland JA, Boucher TR: A meta-analysis examining clinical test utilities for assessing meniscal injury. Clin Rehabil 2008;22:143. [PMID 18212035]
Sanders TG, Miller MD: A systematic approach to magnetic resonance imaging interpretation of sports medicine injuries of the knee. Am J Sports Med 2005;33:131. [PMID: 15611010]
MENISCUS INJURY
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Acute tears occur after axial loading combined with rotation.
• Sensation of clicking or catching of the knee with motion.
• Positive joint line tenderness, effusion, and a positive McMurray test are important physical exam findings.
• MRI can help classify location and morphology.
Meniscal injuries are the most common reason for arthros-copy of the knee. The medial meniscus is more frequently torn than the lateral meniscus because the medial meniscus is securely attached around the entire periphery of the joint capsule, whereas the lateral meniscus has a mobile area where it is not attached. Meniscus injury is rare in childhood, occurs in the late teens, and peaks in the third and fourth decades. After the age of 50, meniscus tears are more often the result of arthritis than trauma.
![]() Clinical Findings
Clinical Findings
Acute traumatic tears of the menisci are often caused by axial loading combined with rotation. Patients typically report pain and swelling. Patients with smaller tears may have a sensation of clicking or catching in the knee. Patients with larger tears in the meniscus may complain of locking of the knee as the meniscus displaces into the joint and/or femoral notch. Loss of knee motion with a block to extension may result from a large bucket-handle tear. In acute tears involving an associated ACL injury, the swelling may be more significant and acute. ACL injuries often involve a lateral meniscus tear as the lateral compartment of the knee subluxates forward trapping the lateral meniscus between the femur and tibia.
Conversely, chronic or degenerative tears of the menisci often present in older patients (>40 years old) with the history of an insidious onset of pain and swelling with or without an acute increase superimposed. Often, no identifiable history of trauma is obtained, or the inciting event may be quite minor such as a bending or squatting motion.
The most important physical examination findings in the knee with a meniscus tear are joint line tenderness and an effusion. Other specialized tests include the McMurray, flexion McMurray, and Apley grind tests. The McMurray test is performed with the patient lying supine with the hip and knee flexed to about 90 degrees. While one hand holds the foot and twists it from external to internal rotation, the other hand holds the knee and applies compression (Figure 3–12). A positive test is one that elicits a pop or click that can be felt by the examiner when the torn meniscus is trapped between the femoral condyle and tibial plateau. A variation of this test is the flexion McMurray, in which the knee is held as for the McMurray test. To test the medial meniscus, the foot is externally rotated and the knee maximally flexed. A positive test occurs when the patient experiences pain over the posteromedial joint line as the knee is gradually extended. The Apley grind test requires placing the patient prone with the knee flexed to 90 degrees. The examiner applies downward pressure to the sole of the foot while twisting the lower leg in external and internal rotation. A positive test results in pain at either joint line.
In addition to the above, physical examination of the entire leg is essential. Assessing hip range of motion and irritability is useful, especially in children, as referred pain from the hip to the knee area is common. Examining for quadriceps atrophy and the presence of a knee effusion should also be done. Measurement of range of motion may reveal a loss of the normal knee extension. Assessing for tenderness of the femoral condyles, joint lines, tibial plateaus, and patellofemoral joint may give clues as to a possible osteochondral lesion, meniscus lesion, fracture, or chondrosis, respectively. Ligamentous testing including varus and valgus stress testing at full extension and 30 degrees of flexion and Lachman, anterior drawer, and posterior drawer testing should be done to assess stability.
![]() Tear Classification
Tear Classification
Meniscal tears can be classified either by etiology or by their arthroscopic and MRI appearance. Etiologic classification divides tears into either acute tears (excessive force applied to an otherwise normal meniscus) or degenerative tears (normal force applied to a degenerative structure).
Classification should describe the tear location and its associated vascularity, morphology, and stability. Tear location is described by its location in the anteroposterior plane (anterior, middle, or posterior) and its circumferential location with respect to its vascularity. The common vascular zones include the most peripheral red/red zone near the meniscocapsular junction, the intermediate red/white zone, and the most central white/white zone. As tears occur more centrally, the healing rate is lower because of a decreased blood supply. Tears can also occur at the meniscal root, which is the attachment of the meniscus to the tibia.
Tear morphology describes the orientation of the tear within the meniscus and includes vertical or horizontal longitudinal, radial (transverse), oblique, and complex (including degenerative) tears (Figure 3–13). Most acute tears in younger patients are vertical longitudinal or oblique tears, while complex and degenerative tears occur more commonly in older patients. Vertical longitudinal, or bucket-handle tears, can be complete or incomplete and usually start in the posterior horn and continue anteriorly a variable distance. Large tears can cause significant mobility of the torn meniscal fragment, allowing it to displace into the femoral notch and cause a locked knee (Figure 3–14). This more commonly occurs in the medial meniscus, possibly owing to its decreased mobility. Oblique tears commonly occur at the junction of the middle and posterior thirds. They are often smaller tears, but the free edge of the tear can catch in the joint and cause symptoms of catching. Complex or degenerative tears occur in multiple planes, are often located in or near the posterior horns, and are more common in older patients with degenerative menisci. Horizontal longitudinal tears are often associated with meniscal cysts. They usually start at the inner margin of the meniscus and extend toward the meniscocapsular junction. They are thought to result from shear stresses and, when associated with meniscal cysts, occur in the medial meniscus and cause localized swelling at the joint line.
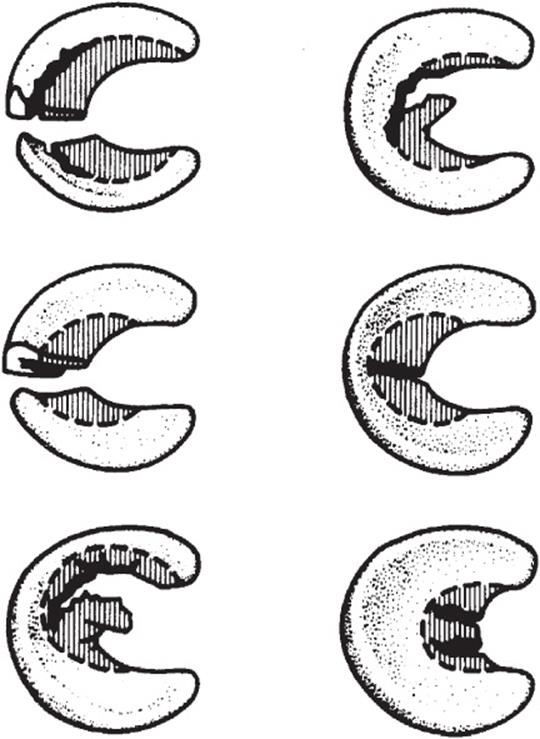
![]() Figure 3–13. Patterns of meniscal tears: bucket-handle, flap, horizontal cleavage, radial, degenerative, and double radial tear of a discoid meniscus. (Reproduced, with permission, from Scott WN: Arthroscopy of the Knee. New York: WB Saunders; 1990.)
Figure 3–13. Patterns of meniscal tears: bucket-handle, flap, horizontal cleavage, radial, degenerative, and double radial tear of a discoid meniscus. (Reproduced, with permission, from Scott WN: Arthroscopy of the Knee. New York: WB Saunders; 1990.)

![]() Figure 3–14. A: Diagram of a typical bucket-handle tear of the medial meniscus. B: Arthroscopic view of a bucket-handle fragment displaced into the intercondylar notch. (Reproduced, with permission, from McGinty JB: Operative Arthroscopy. Baltimore: Raven Press, 1991.)
Figure 3–14. A: Diagram of a typical bucket-handle tear of the medial meniscus. B: Arthroscopic view of a bucket-handle fragment displaced into the intercondylar notch. (Reproduced, with permission, from McGinty JB: Operative Arthroscopy. Baltimore: Raven Press, 1991.)
![]() Treatment and Prognosis
Treatment and Prognosis
Small stable meniscus tears often become asymptomatic and do not need to be treated surgically. Those causing persistent symptoms should be assessed with the arthroscope. Before the importance of the meniscus was understood and arthros-copy became available, the meniscus was often removed, even when normal. We now attempt to remove only the torn portion of the meniscus or repair the meniscus, if possible.
During arthroscopy, the meniscus can be visualized and palpated with a probe. The inner two thirds of the meniscus is avascular and often requires resection when torn. The remaining meniscus is smoothed and contoured to prevent further tearing from a jagged edge. Return to full function may be expected in 6–8 weeks.
Tears in the peripheral third of the meniscus, if small (<15 mm), may heal spontaneously because there is a blood supply in this portion of the adult meniscus. Larger tears need to be repaired because those who undergo meniscectomy at a young age are at risk of early osteoarthritis. These changes were first described by Fairbanks and include flattening of the femoral condyle, joint space narrowing, and osteophyte formation. Therefore, every effort should be made to preserve the meniscus.
A. Partial Meniscal Resection
Partial meniscectomy has a 90% rate of good or excellent results in patients without knee instability or osteoarthritis. A major advantage over meniscus repair is a short recovery period. However, results diminish over time, and osteoarthritis occurs with over 10 years of follow-up. Medial meniscus tears generally do better than lateral tears after partial resection, and an intact meniscal rim and those with normal articular cartilage and normal knee stability are associated with a better prognosis.
B. Meniscus Repair
Most surgeons will attempt a meniscus repair rather than a partial meniscectomy in young, active individuals. Other commonly accepted criteria for meniscus repair include a complete vertical longitudinal tear greater than 15 mm in length, a tear within the peripheral 10–30% of the meniscus (ie, within 3–4 mm of the meniscocapsular junction), a peripheral tear that can be displaced toward the center of the plateau with a probe, the absence of secondary meniscus degeneration, and a tear in a patient undergoing concurrent ligament or articular cartilage repair.
Multiple factors affect the success of meniscus repair. Although no absolute age limit exists, patients younger than 40 years are thought to have a better chance for healing. Knees with associated ligamentous instability, particularly ACL instability, have inferior rates of meniscus healing because of abnormal meniscus stresses from tibiofemoral instability. The location of the tear and the time lapsed from injury to treatment are also important. Acute tears located in the peripheral red/red or red/white zone have better healing capability than chronic tears located in the red/white or white/white zones. Tears 5 mm or more from the periphery are considered avascular (white zone), whereas those between 3 and 5 mm are variable in vascularity (red/white), and tears in the peripheral 3 mm are considered vascular (red). In areas with marginal vascularization, abrasion of the meniscocapsular junction or use of a fibrin clot may be performed. It is thought that a vascular pannus forms from the abraded tissue that aids in healing. Finally, the stability of the meniscus repair is a factor, with vertical mattress sutures generally considered the gold standard in meniscus repair. It is generally believed that the superiority of vertical mattress over horizontal mattress sutures is from the vertical mattress sutures capturing the strong peripheral, circumferential fibers of the meniscus.
Meniscus repair is more successful when done at the same time as ACL reconstruction. Then it is successful up to 90% of the time compared to approximately 50% success in patients with intact ACLs who had meniscus repairs. Many of the meniscus tears that occur with an ACL tear are amenable to repair. Then stabilizing the knee with ACL reconstruction protects the repaired meniscus from abnormal knee motion and has more success than if the knee is left unstable.
Types of repairs include the traditional open repair and arthroscopic repairs that can be done with inside-out, outside-in, or all-inside techniques. Inside-out and outside-in repairs are usually done with sutures and require a mini-incision and securing of the meniscus to the capsule with sutures. The all-inside technique has many device options, including sutures and various devices. Regardless of the type of repair chosen, adequate preparation of the tear site is required. The tear edges should be debrided or abraded with a shaver or rasp to stimulate bleeding. Restoration of biomechanical function is encouraged by anatomic apposition of the tear edges to ensure good healing potential.
1. Open meniscal repair—Open repair of meniscus tears has been shown to have successful long-term results. The technique involves making a small incision through the subcutaneous tissue, capsule, and synovium to directly visualize the tear. Open repair is most useful in peripheral or meniscocapsular tears, often occurring in conjunction with open repair of a collateral ligament injury or a tibial plateau fracture. Follow-up studies of 10 years or longer have shown survival rates of repaired menisci of 80–90%, in part influenced by the peripheral nature of the tear and the associated hemarthrosis present in ligament tears or fracture repair cases.
2. Arthroscopic meniscal repair
A. INSIDE-OUT MENISCAL REPAIR—Arthroscopic inside-out meniscus repairs are performed using long needles introduced through cannula systems with attached absorbable or nonabsorbable sutures passed perpendicularly across the tear from inside the knee to a protected area outside the joint capsule. These sutures are able to obtain consistent perpendicular placement across the meniscus tear, which gives this method an advantage over other repair techniques. Improved suture placement is gained at the expense of possible neurovascular injury from passing the needle from inside the knee to outside the joint. This technique requires a posteromedial or posterolateral incision to protect the neurovascular structures and safely retrieve the exiting needles. Because surgeons are able to place vertical mattress sutures, the best biomechanical construct for meniscus repair, this technique remains the gold standard for many surgeons. Numerous retrospective and prospective studies using second-look arthroscopy or arthrography to evaluate healing of the meniscus repairs have consistently shown rates of 70–90% in isolated repairs, and greater than 90% when done in conjunction with an ACL reconstruction. This technique is ideal for posterior and mid-posterior horn tears. There is difficulty in passing needles in mid to anterior horn meniscus tears.
B. OUTSIDE-IN MENISCAL REPAIR— The arthroscopic outside-in repair was developed in part to decrease the neurovascular risk associated with the inside-out technique. The outside-in technique involves passing a needle from outside the joint, across the tear, and into the joint. Two options then exist for repair of the meniscus tear. One option is then to retrieve the suture through an anterior portal, tie a knot outside the knee joint, and then bring the knot back in through the anterior portal placing the knot against the reduced meniscus body fragment. A second option is to use parallel needles and retrieve the suture through the second needle. This can be done using a suture relay. A knot is then tied outside the joint over the capsule. This method is useful for tears in the anterior horn or body of the menisci, but does not work for tears in or near the posterior horn. Results of the outside-in technique using MRI, arthrography, or second-look arthroscopy to assess healing have shown complete or partial healing; between 74% and 87% of meniscus repairs have been successful. As expected, more posterior horn tears and tears in unstable knees did worse.
C. ALL-INSIDE MENISCAL REPAIR— The popularity of the all-inside repairs has increased with the introduction of numerous devices and techniques to ease the technique. They do not require accessory incisions, thereby saving operative time, and they avoid more technical arthroscopic techniques required in other types of repairs. However, repairs with some devices have not been as successful as those with traditional techniques. Success rate is 60–90%, and some have found results comparable to traditional techniques, but there are complications including devices that have migrated from their original position, broken fragments, foreign-body reactions, inflammation, chronic effusions, and articular cartilage injuries.
Recent biomechanical studies have found repair with some of these devices to have properties equivalent to vertical mattress sutures. But there is considerable variation with the type of device. What remains to be known, however, is the meniscus repair strength needed for optimal meniscus healing.
C. Meniscal Transplantation
An alternative to leaving the patient with a meniscus-deficient knee, and almost certain early osteoarthrosis, is meniscus transplantation. This technique yields satisfactory results in about two thirds of patients. In the future, biologic scaffolds may enable menisci to be regenerated after meniscectomy.
Ahn JH, Wang JH, Yoo JC: Arthroscopic all-inside suture repair of medial meniscus lesion in anterior cruciate ligament–deficient knees: results of second-look arthroscopies in 39 cases. Arthroscopy2004;20:936. [PMID: 15525926]
Hommen JP, Applegate GR, Del Pizzo W: Meniscus allograft transplantation: ten-year results of cryopreserved allografts. Arthroscopy 2007;23:388. [PMID: 17418331]
Metcalf MH, Barrett GR: Prospective evaluation of 1485 meniscal tear patterns in patients with stable knees. Am J Sports Med 2004;32:675. [PMID: 15090384]
Salata MJ, Gibbs AE, Sekiya JK: A systematic review of clinical outcomes in patients undergoing meniscectomy. Am J Sports Med 2010;38:1907. [PMID: 20587698]
Shelbourne KD, Dersam MD: Comparison of partial meniscectomy versus meniscus repair for bucket-handle lateral meniscus tears in anterior cruciate ligament reconstructed knees. Arthroscopy 2004;20:581. [PMID: 15241307]
Steenbrugge F, Verstraete K, Verdonk R: Magnetic resonance imaging of the surgically repaired meniscus: a 13-year follow-up study of 13 knees. Acta Orthop Scand 2004;75:323. [PMID: 15260425]
Stone KR, Adelson WS, Pelsis JR, Walgenbach AW, Turek TJ: Long-term survival of concurrent meniscus allograft transplantation and repair of the articular cartilage: a prospective two- to 12-year follow-up report. J Bone Joint Surg Br 2010;92:941. [PMID: 20595111]
![]() CPT Codes for the Meniscus
CPT Codes for the Meniscus
27403 Arthrotomy with meniscus repair, knee
29868 Arthroscopy, knee, surgical; meniscal transplantation (includes arthrotomy for meniscal insertion), medial or lateral
29870 Arthroscopy, knee, diagnostic, with or without synovial biopsy (separate procedure)
29880 Arthroscopy, knee, surgical; with meniscectomy (medial and lateral, including any meniscal shaving)
29881 Arthroscopy, knee, surgical; with meniscectomy (medial or lateral, including any meniscal shaving)
29882 Arthroscopy, knee, surgical; with meniscus repair (medial or lateral)
29883 Arthroscopy, knee, surgical; with meniscus repair (medial and lateral)
KNEE FRACTURE
Articular cartilage injuries of the knee are infrequent, and there must be a high index of suspicion to detect them. MRI and arthroscopy are very helpful with these injuries, especially pure articular cartilage injuries, where radiographs will be normal.
1. Osteochondral Lesions
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Patients usually present with vague, poorly localized complaints of knee pain.
• Classic location is the posterolateral aspect of the medial femoral condyle.
• Involvement is bilateral in up to 25% of cases, so examine both knees.
• Effusion, crepitus, and an antalgic gait are possible findings on exam.
• Radiographs and MRI can be helpful in determining the location and size of the lesion.
![]() Osteochondral Fracture
Osteochondral Fracture
There is much confusion about the nomenclature and etiology of juvenile and adult osteochondral lesions (OCL) of the knee, also called osteochondritis dissecans. Inflammatory, ossification abnormalities and avascular necrosis have all been considered etiologies of this condition. However, basic science, histopathology, and vascular studies do not support any of them. The term “osteochondral injuries” has been used to describe injuries ranging from acute osteochondral fractures to pure chondral injuries. Currently, OCLs are defined as potentially reversible idiopathic lesions of subchondral bone, resulting in delamination or fragmentation with or without destruction of the overlying articular cartilage. OCLs are subdivided into juvenile and adult forms depending on the presence of an open distal femoral physis. In children, a combination of etiologies is now thought to be responsible for OCLs. For example, a stress fracture may develop in the subchondral bone of the distal femoral condyle. Such an injury may provoke further vascular compromise, which results in injury to the subchondral bone that was initially covered with normal articular cartilage. Loss of support from the subchondral bone may result in damage to the overlying articular cartilage. The vast majority of adult OCLs are thought to have arisen from a persistent juvenile OCL, although new lesions in adults are possible as well.
Both adult and juvenile OCLs that do not heal have the potential for further sequelae, including degenerative osteoarthritis. Juvenile OCLs, defined as knees with an open physes, generally have a better prognosis than adult lesions. The classic location of an OCL is the posterolateral aspect of the medial femoral condyle, which accounts for 70–80% of all OCLs. Lateral condyle OCLs are seen in 15–20% of patients, and patellar involvement ranges from 5–10%. The increased use of MRI and arthroscopy over the past decade may have resulted in greater recognition of OCLs.
![]() Clinical Findings
Clinical Findings
A common presentation of a patient with an OCL is aching and activity-related anterior knee pain that is poorly localized. Pain may worsen with stair climbing or running. Patients with stable OCLs do not have mechanical symptoms or knee instability. Mechanical symptoms are more common in patients with unstable or loose OCLs. Patients may limp, and knee swelling may be present. Tenderness with palpation of the femoral condyle may be observed with various degrees of knee flexion. Loss of range of motion or quadriceps atrophy may be noted in more long-standing cases.
It is important to identify patients with unstable lesions. There may be crepitus and pain with range of motion, and an effusion is typically present. Involvement is bilateral in up to 25% of cases, so both knees should be evaluated regardless of symptoms. Initial evaluation should include anteroposterior, lateral, and tunnel views of both knees. The goal of plain radiographs is to exclude any bony pathology, evaluate the physes, and localize the lesion. Lesion location and an estimation of size can be determined as described by Cahill. MRI may be helpful in diagnosis and can give an estimation of the size of the lesion (prognosis is better for small lesions), the condition of the overlying cartilage and underlying subchondral bone, the extent of bone edema, the presence of any loose bodies, and assessment of OCL stability. Four MRI criteria have been identified on T2-weighted images to assess OCL stability: a line of high signal intensity at least 5 mm in length between the OCL and underlying bone, an area of increased homogeneous signal at least 5 mm in diameter beneath the lesion, a focal defect of 5 mm or more in the articular surface, and a high signal line traversing the subchondral plate into the lesion. A high signal line is the most common sign in patients found to have unstable lesions that are most likely to fail nonoperative treatment. MRI is helpful with these injuries, especially pure articular cartilage injuries, where radiographs will be normal or may result in false-positive findings of fragment loosening. Arthroscopy remains the gold standard in evaluation of these lesions.
Equivocal prognostic value has been found in the use of intravenous gadolinium in OCLs. Technetium bone scans were initially proposed to monitor the presence of healing. However, because MRI eliminates the ionizing radiation and increased time required in bone scanning, bone scanning is not widely used.
![]() Treatment and Prognosis
Treatment and Prognosis
Prognosis is good for the immature child. Nonoperative management should be pursued in those with a stable OCL and open physes. The goal of nonoperative treatment is to obtain a healed lesion before physis closure so as to prevent early-onset osteoarthritis. Even if patients are within 6–12 months of phy-seal closure, a trial of nonoperative treatment is warranted.
Because failure of the subchondral bone precedes failure of the overlying articular cartilage, most orthopedists recommend some sort of activity modification. Debate exists whether activity modification should include the use of cast or brace immobilization. The tenet of nonoperative treatment is to reduce the activity level where pain-free activities of daily living are possible. However, there is no optimal immobilization protocol available in the literature.
Patients should be non–weight bearing or partial weight bearing with crutches for 3–6 weeks or until they are pain free. Repeat radiographs are obtained at approximately 6-week intervals. Physical therapy with full weight bearing may be initiated once patients are pain free. Physical therapy should focus on low-impact quadriceps and hamstring strengthening. If patients remain asymptomatic up to at least 3 months after the diagnosis was made, activity may be slowly advanced to higher impact activities such as running or jumping. Any recurrence of symptoms or any progression of the OCL on plain radiographs should prompt a return to non–weight bearing and possible immobilization for a longer period. Obvious patient frustration and lack of compliance (especially in adolescents) is common, and a full discussion of the risks and benefits of nonoperative or operative treatment is required.
Operative treatment should be considered in the following instances: (1) loose bodies, (2) an unstable OCL, (3) persistence of symptoms despite nonoperative treatment in a compliant patient, (4) worsening appearance on imaging studies, and (5) near or complete epiphyseal closure. Goals of operative treatment should include achievement of a stable osteochondral fragment that maintains joint congruity and allows early range of motion.
For stable lesions with an intact articular surface, arthroscopic drilling of the lesions is preferred. This creates channels for potential revascularization through the subchondral bone plate. Options include transarticular drilling and transepiphyseal drilling. Radiographic healing and relief of symptoms can be expected in 80–90% of patients with open physes. This decreases to 50–75% in those with closed physes.
Patients with partially unstable lesions such as a flap lesion should be managed by the status of the subchondral bone. If present, fibrous tissue between the lesion and subchondral bone should be debrided. If significant subchondral bone loss has occurred, it can be filled with autogenous bone graft prior to fixation of the OCL. If the OCL has sufficient bone such that an anatomic fit into its donor site is possible, fixation should be attempted. Various fixation methods have been described including Herbert or cannulated screws and bioabsorbable screws or pins, but there are complications with these treatments. Complications include devices that have migrated from their original position, broken fragments, foreign-body reactions, inflammation, chronic effusions, and articular cartilage injuries.
Simple excision of the larger fragments has shown poor results with more rapid progression of radiographic osteoarthritic changes. For lesions greater than 2 cm2, drilling or microfracture methods that depend on replacement of the defect with fibrocartilage have yielded poor results with worsening osteoarthritis over time. For these larger lesions, cartilage transplantation has been tried. Disadvantages of autologous osteochondral plugs or mosaicplasty include donor site morbidity and incongruent articular fit. Advantages include good fixation of the patient’s own tissue. Another option is autologous chondrocyte implantation, which involves harvesting of the patient’s chondrocytes, proliferating them over time, and then reimplanting the chondrocytes. Advantages include use of the patient’s own tissue and lessened donor site morbidity. Longer-term results in young adult patients show successful clinical results in up to 90% for both procedures. However, additional larger and longer-term follow-up studies are needed.
Cepero S, Ullot R, Sastre S: Osteochondritis of the femoral condyles in children and adolescents: our experience over the last 28 years. J Pediatr Orthop B 2005;14:24. [PMID: 15577303]
Crawford DC, Safran MR: Osteochondritis dissecans of the knee. J Am Acad Orthop Surg 2006;14:90. [PMID: 16467184]
Detterline AJ, Goldstein JL, Rue JP, et al: Evaluation and treatment of osteochondritis dissecans lesions of the knee. J Knee Surg 2008;21:106. [PMID: 18500061]
Gomoll AH, Farr J, Gillogly SD, Kercher J, Minas T: Surgical management of articular cartilage defects of the knee. J Bone Joint Surg Am 2010;92:2470. [PMID: 20962200]
Vasiliadis HS, Wasiak J: Autologous chondrocyte implantation for full thickness articular cartilage defects of the knee. Cochrane Database Syst Rev 2010;10:CD003323. [PMID: 20927732]
![]() CPT Codes for Osteochondral Lesions
CPT Codes for Osteochondral Lesions
27415 Osteochondral allograft, knee, open
29850 Arthroscopically aided treatment of intercondylar spine(s) and/or tuberosity fracture(s) of the knee, with or without manipulation; without internal or external fixation (includes arthroscopy)
29866 Arthroscopy, knee, surgical; osteochondral autograft(s) (eg, mosaicplasty) (includes harvesting of the autograft[s])
29867 Arthroscopy, knee, surgical; osteochondral allograft (eg, mosaicplasty)
29874 Arthroscopy, knee, surgical; for removal of loose body or foreign body (eg, osteochondritis dissecans fragmentation, chondral fragmentation)
29877 Arthroscopy, knee, surgical; debridement/shaving of articular cartilage (chondroplasty)
29879 Arthroscopy, knee, surgical; abrasion arthroplasty (includes chondroplasty where necessary) or multiple drilling or microfracture
29885 Arthroscopy, knee, surgical; drilling for osteochondritis dissecans with bone grafting, with or without internal fixation (including debridement of base of lesion)
29886 Arthroscopy, knee, surgical; drilling for intact osteochondritis dissecans lesion
29887 Arthroscopy, knee, surgical; drilling for intact osteochondritis dissecans lesion with internal fixation
KNEE LIGAMENT INJURY
Knee injuries occur during both contact and noncontact athletic activities. Advances in the diagnosis and treatment of ligament injuries have allowed athletes at all levels of ability to return to sports at their preinjury level of activity. The ligaments and menisci of the knee work in concert with one another, and frequently more than one structure is damaged when an acute injury occurs.
Ligament injuries are graded as follows: grade 1, stretching of the ligament with no detectable instability; grade 2, further stretching of the ligament with detectable instability, but with the fibers in continuity; and grade 3, complete disruption of the ligament.
![]() Anatomy
Anatomy
Knee stability requires proper functioning of four ligaments. These ligaments include the ACL, the PCL, the medial collateral ligament (MCL), and the lateral collateral ligament (LCL). There are also several accessory or secondary stabilizers of the knee. Secondary stabilizers of the knee include the menisci, iliotibial band, and biceps femoris. These secondary stabilizers become more important when a primary stabilizer is injured.
The MCL is the primary static stabilizer against valgus stress at the knee. The MCL originates from the central sulcus of the medial epicondyle. The sulcus of the C-shaped medial epicondyle is located anterior and distal to the adductor tubercle. The MCL is made up of three main static medial stabilizers of the knee. This includes the superficial MCL, the posterior oblique ligament, and the deep capsular ligament.
The LCL is the primary static stabilizer against varus stress at the knee. The LCL originates from the lateral epicondyle. This is the most prominent point of the lateral femoral condyle. The LCL insertion is on the styloid process of the fibular head, which projects superiorly from the posterolateral fibular head. The LCL joins with the arcuate ligament, the popliteus muscle, and the lateral head of the gastrocnemius to form a lateral arcuate complex to control statically and dynamically varus angulation and external tibial torsion. The iliotibial band and biceps femoris also contribute to stability on the lateral aspect of the knee.
The ACL is the primary static stabilizer of the knee against anterior translation of the tibia with respect to the femur. The ACL originates from the posteromedial surface of the lateral femoral condyle in the intercondylar notch. The ACL inserts on the tibial plateau just medial to the anterior horn of the lateral meniscus about 15 mm posterior to the anterior edge of the tibial articular surface. The blood supply to the ACL and PCL is the middle geniculate artery. Both the ACL and PCL are covered by a layer of synovium, making these ligaments intraarticular and extrasynovial.
The PCL is the primary static stabilizer of the knee against posterior translation of the tibia with respect to the femur. The PCL originates from the posterior aspect of the lateral surface of the medial femoral condyle in the intercondylar notch. The PCL inserts on the posterior aspect of the tibial plateau in a central depression just posterior to the articular surface. The insertion extends distally along the posterior aspect of the tibia for up to 1 cm in length. The PCL is a complex structure consisting of two major bands: the anterolateral and posteromedial bands. The anterolateral band is tight in flexion and loose in extension. The posteromedial band is loose in flexion and tight in extension. The cross-sectional area of the anterolateral band is twice as large as the posteromedial band. The meniscofemoral ligaments, the ligaments of Wrisberg and Humphrey, are the third component of the PCL. The meniscofemoral ligaments travel from the posterior horn of the lateral meniscus to the posteromedial femoral condyle.
![]() Differential Diagnosis of Knee Instability
Differential Diagnosis of Knee Instability
The differential diagnosis of acute or chronic knee instability can involve any of the knee ligaments and/or the structures of the posterolateral corner. There are often combinations of ligament injuries in addition to injuries of secondary stabilizing structures such as the menisci. The history and mechanism of injury are valuable information, if available. Similarly, the location of pain can help to narrow the diagnosis. Clearly, however, a thorough physical examination helps to distinguish which ligaments have been injured. Additionally, imaging studies are often obtained to confirm clinical suspicions and to evaluate for occult injuries.
Fanelli GC, Orcutt DR, Edson CJ: The multiple-ligament injured knee: evaluation, treatment and results. Arthroscopy 2005;21:471. [PMID: 15800529]
Micheo W, Hernández L, Seda C: Evaluation, management, rehabilitation, and prevention of anterior cruciate ligament injury: current concepts. PM R 2010;2:935. [PMID: 20970763]
1. Medial Collateral Ligament Injuries
![]() Essential of Diagnosis
Essential of Diagnosis
• Occurs after a valgus stress to the knee or noncontact rotational injury.
• Medial knee pain and instability at 30 degrees of flexion is diagnostic; consider ACL or PCL injuries in addition if opening at full extension with a valgus stress.
• Chronic injuries may have calcification at the insertion of the MCL on the medial femoral condyle.
• MRI can be helpful in confirming diagnosis and helping to rule out concomitant meniscal injury.
![]() Symptoms (History)
Symptoms (History)
How and when the patient was hurt are important parts of the history. Lower-grade MCL injuries typically occur in a noncontact external rotational injury, whereas higher-grade injuries generally involve lateral contact to the thigh or upper leg. Other important pieces of historical information include the location and presence of pain, instability, timing of swelling, and sensation of a “pop” or tear. Surprisingly, grade I and II injuries are often more painful than complete MCL rupture. Immediate swelling should make one suspicious for an associated cruciate ligament injury, fracture, and/or patellar dislocation.
A prior history of knee injuries or instability should always be sought when evaluating a new knee injury.
![]() Signs (Physical Examination)
Signs (Physical Examination)
MCL injuries are evaluated with a complete knee examination to evaluate for any other coexisting injuries. This is especially true with ACL and PCL evaluation because an injury to either of these ligaments would significantly change the treatment. Given the frequency of coexisting patellar dislocations in MCL injuries, palpation of the patella and the medial parapatellar stabilizing ligaments should be performed in addition to patellar apprehension testing.
Medial joint line tenderness along the course of the MCL is typical at the location of the tear. Laxity to valgus stresses is assessed by the amount of medial joint space opening that occurs at 30 degrees of flexion. It is important to stress the knee at 30 degrees of flexion because with the knee in full extension the posterior capsule and PCL will stabilize the knee to valgus stress. This stability to valgus stress in full extension could mislead the examiner to believe that the MCL is intact. Zero opening is considered normal, with 1-4 mm indicating a grade I injury, 5–9 mm indicating a grade II injury, and 10–15 mm indicating a complete or grade III injury. Additionally, grade I and II injuries typically have a firm end point, whereas a grade III injury tends to have a soft end point to valgus stress.
![]() Imaging Studies
Imaging Studies
A. Radiographs
A series of knee radiographs should be obtained in any patient with a suspected significant knee injury. Radiographs should be inspected for acute fracture, lateral capsular avulsion (Segond fracture; see section on ACL imaging), loose bodies, Pellegrini-Stieda lesion (MCL calcification), and evidence of patellar dislocation. Stress radiographs should be obtained in patients prior to skeletal maturity to rule out an epiphyseal fracture.
B. MRI
MRI is useful for confirming MCL injury and identifying the site of injury. It is also useful to detect the presence of meniscal and other injuries to the knee. Relative indications for an MRI include an uncertain ACL status despite multiple examinations, evaluation of a suspected meniscal tear, or preoperative evaluation for a planned MCL reconstruction or repair.
C. Special Tests
An examination under anesthesia can be valuable when physical examination is unreliable because of the patient guarding the knee. Diagnostic arthroscopy can also be used to evaluate for coexistant pathology. However, both of these diagnostic methods have largely been replaced by MRI.
![]() Treatment (Nonsurgical and Surgical)
Treatment (Nonsurgical and Surgical)
Treatment of an isolated MCL injury is generally nonoperative with protection against valgus stress and early motion. Grade I and grade II injuries can be placed in either a cast or a brace and bear weight as tolerated. Generally, knee motion is started within the first week or two, and full recovery is usually achieved more rapidly with early knee range of motion.
Grade III injuries are a bit more controversial. Several authors have shown increased instability in grade III tears treated nonsurgically, although most of these studies did not exclude knees with multiligamentous injuries. Comparison of isolated grade III MCL tears treated with surgical reconstruction versus nonsurgical management showed that the nonsurgical treatment group enjoyed better results in both subjective scoring and earlier return to activity.
The exception to the current trend of nonsurgical treatment of grade III injuries is in the setting of a multiligamentous knee injury. In this setting, particularly with a distal tibial avulsion of the MCL, nonsurgical treatment has not fared nearly as well as in isolated MCL injuries. MCL repair in the acute setting can include a primary repair, with shortening if needed, of the torn ligament. Similarly, avulsion fragments are treated with reduction and fixation in the acute setting. Primary repairs can be reinforced with auto-graft or allograft tissues if the remaining MCL is insufficient for a stand-alone repair. Chronic reconstructions also often include autograft or allograft tissue reconstruction.
Traditionally, casting or operative treatment of MCL injuries significantly limited an early return to range-of-motion exercises. With the addition of functional bracing and early motion to a nonsurgical treatment protocol, motion and strengthening of the knee can occur at an early stage while the ligament is protected from valgus stress. As knee motion improves, isotonic strengthening exercises are introduced. As the strength of the extremity improves, the intensity of functional rehabilitation increases accordingly.
![]() Complications
Complications
With nonsurgical treatment becoming the standard of care, complications associated with an MCL injury have decreased. The main complication of nonsurgical therapy is residual valgus laxity or medial knee pain. Radiographs may show residual calcification of the MCL (Pellegrini-Stieda lesion). Potential surgical complications include arthrofibrosis, infection, damage to the saphenous nerve or vein, or recurrent valgus laxity.
![]() Results/Return to Play
Results/Return to Play
In general, good outcomes can be achieved with nonsurgical treatment and rehabilitation of isolated MCL injuries. Return to professional football after nonsurgical treatment of isolated MCL injuries is 98%.
Azar FM: Evaluation and treatment of chronic medial collateral ligament injuries of the knee. Sports Med Arthrosc 2006;14:84. [PMID: 17135952]
Robinson JR, Bull A, Thomas R, et al: The role of the medial collateral ligament and posteromedial capsule in controlling knee laxity. Am J Sports Med 2006;34:1815. [PMID: 16816148]
Robinson JR, Sanchez-Ballester J, Bull AM, et al: The posterome-dial corner revisited. An anatomical description of the passive restraining structures of the medial aspect of the human knee. J Bone Joint Surg Br 2004;86:674. [PMID: 15274262]
Stannard JP: Medial and posteromedial instability of the knee: evaluation, treatment, and results. Sports Med Arthrosc 2010; 18:263. [PMID: 21079506]
2. Lateral Collateral Ligament Injuries
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Patients may complain of lateral knee pain and a varus thrust with daily activity.
• Varus stress to the knee with opening at 30 degrees of flexion is diagnostic for an isolated LCL injury.
• Frequently part of a multiligamentous injury to the knee.
• There is a high incidence of peroneal nerve injury; document neurovascular status to the involved extremity.
• MRI should be obtained as a useful adjunct to help diagnose posterolateral corner injuries.
![]() Symptoms (History)
Symptoms (History)
The most consistent symptom of an acute LCL injury is lateral knee pain. However, the symptoms of lateral and posterolateral instability are quite variable and depend on the severity of injury, patient activity level, overall limb alignment, and other associated knee injuries. For example, a sedentary individual with minimal laxity and overall valgus alignment will have few, if any, symptoms. However, if LCL laxity is combined with overall varus alignment, hyperextension, and an increased activity level, symptoms will be quite pronounced. These patients may complain of lateral joint line pain and a varus thrust of their leg with everyday activities. This is often described as the knee buckling into hyperextension with normal gait.
![]() Signs (Physical Examination)
Signs (Physical Examination)
Patients with an LCL and/or posterolateral corner injury often also have additional ligamentous injuries to the knee. Therefore, a thorough knee examination should be performed to evaluate for coexistant knee pathology. Additionally, a careful neurovascular examination should be performed as the incidence of neurovascular injury, particularly peroneal nerve injury, has been reported in 12–29% of posterolateral knee injuries.
The integrity of the LCL is assessed by placing a varus stress, with the knee in full extension and 30 degrees of flexion. Baseline varus opening is widely variable and should be compared to the contralateral leg. The average baseline for varus opening is 7 degrees. Exam findings with an isolated LCL injury should include varus laxity at 30 degrees of flexion and no instability in full extension. This is due to the stabilizing effect that the intact cruciate ligaments provide in full extension.
It is important to note that a significant posterolateral knee injury can be present without significant varus laxity. The most useful test to evaluate for posterolateral instability is the dial test. This is done by externally rotating each tibia and noting the angle subtended between the thigh and the foot. The dial test is performed at 30 and 90 degrees of flexion with a significant difference being an angle 5 degrees or greater than the contralateral leg. Injury to the posterolateral capsule alone is confirmed with greater external rotation at 30 degrees, an isolated PCL at 90 degrees, and to both structures when there is greater rotation at 30 and 90 degrees compared to the uninjured leg.
![]() Imaging Studies
Imaging Studies
A. Radiographs
A series of knee radiographs should be obtained in any patient with a suspected knee injury. Radiographs should be inspected for acute fractures, lateral capsular avulsion (Segond fracture; see section on ACL imaging), loose bodies, fibular head avulsions, and evidence of patellar dislocation. With chronic posterolateral instability, degenerative changes of the lateral compartment are often noted. Lateral joint space narrowing with osteophytes and subchondral sclerosis can be seen. Stress radiographs can help to better quantify the amount of varus angulation present.
B. MRI
MRI is often a useful adjunct for diagnosing posterolateral corner and LCL injuries in the severely injured knee. As mentioned earlier, this posterolateral injury can often go unnoticed during an initial evaluation, and MRI findings can refocus the examination to the posterolateral structures. Pain and guarding at the time of injury can often obscure posterolateral injury, and MRI can prove to be an extremely valuable adjunct in diagnosis.
C. Special Tests/Examinations
1. Reverse pivot shift test—This test involves starting with the knee flexed to 90 degrees. While the knee is extended, the leg is loaded axially with a valgus stress applied to the knee and the foot is held in external rotation. A palpable shift is noted as the tibia reduces from its posteriorly subluxed position as the knee is extended.
2. External rotation recurvatum test—This test is performed with the patient supine and the hip and knee fully extended. The leg is lifted off the bed by the toes. Hyperextension, varus instability, and external rotation of the tibial tubercle occurs with adequate quadriceps relaxation in a patient with posterolateral instability.
3. Posterolateral drawer test—A standard posterior drawer test (see section on PCL physical examination) is performed with the tibia in internal rotation, neutral, and externally rotated positions. With posterolateral injury, the magnitude of the posterior drawer displacement will be greatest with external tibial rotation.
4. Examination under anesthesia—An examination while the patient is relaxed under general anesthetic is extremely useful, particularly in the acute setting. If the patient with a multiligamentous knee injury is taken to the operating room, this is an excellent opportunity to examine the knee without guarding to improve the accuracy of the examination.
![]() Treatment
Treatment
A. Nonsurgical
Isolated LCL ligament injuries, as noted earlier, are rare injuries. However, in the case of an isolated LCL ligament injury with grade II or less magnitude, a period of immobilization from 2–4 weeks followed by a quadriceps strengthening program will usually yield good results. Grade III injuries often have better results with surgical treatment. The combination of delayed diagnosis along with an uncertain natural history of posterolateral instability makes the treatment of these injuries a challenge.
B. Surgical
LCL and posterolateral ligament injuries, as discussed earlier, rarely occur in isolation. Therefore, other injuries must also be considered in the treatment plan of the multiligamentous knee injury. Ideally, the posterolateral and LCL injuries are diagnosed in the acute setting. This allows the preferred surgical treatment of a primary repair of the injured structures with augmentation as needed. Primary repair is generally only feasible in the first few weeks following the knee injury.
The knee with chronic posterolateral instability will often require ligamentous reconstruction or advancement to reconstitute a static restraint to varus stresses. The key bio-mechanical concept of any lateral ligamentous reconstruction is that the isometric point of the LCL lies between the fibular head and the lateral epicondyle. Therefore, regardless of the graft material used to reconstruct the lateral ligamentous complex, a portion of the graft must pass between the lateral femoral epicondyle and the fibular head.
To improve the success rate of reconstruction of chronic lateral ligamentous instability, a proximal tibial valgus osteotomy may be performed to decrease the stress on the lateral structures of the knee.
![]() Rehabilitation
Rehabilitation
The rehabilitation of the knee after posterolateral reconstructions or repairs is largely guided by associated injuries to the ACL or PCL. It is generally necessary, however, to limit weight bearing for at least 6 weeks and protect the lateral structures with a brace for at least 3 months.
![]() Complications
Complications
The peroneal nerve runs just posterior to the fibular head. It is important to isolate the peroneal nerve prior to any lateral knee exposure to minimize the complication of a peroneal nerve injury.
![]() Results
Results
If injuries to the posterolateral corner of the knee are diagnosed and repaired acutely, the results are good for restoration of varus stability and return to play. Chronic posterolateral corner injury reconstructions also perform well when an isometric lateral reconstruction is achieved.
Laprade RF, Engebretsen L, Johansen S, et al: The effect of a proximal tibial medial opening wedge osteotomy on posterolateral knee instability. Am J Sports Med 2008;36:956. [PMID: 18227230]
Markolf KL, Graves BR, Sigward SM, et al: Effects of posterolateral reconstructions on external tibial rotation and forces in a posterior cruciate ligament graft. Bone Joint Surg Am 2007;89:2351. [PMID: 17974876]
Ranawat A, Baker C 3rd, Henry S, et al: Posterolateral corner injury of the knee: evaluation and management. J Am Acad Orthop Surg 2008;16:506. [PMID: 18768708]
Rios CG, Leger RR, Cote MP, Yang C, Arciero RA: Posterolateral corner reconstruction of the knee: evaluation of a technique with clinical outcomes and stress radiography. Am J Sports Med 2010;38:1564. [PMID: 20445013]
3. Anterior Cruciate Ligament Injuries
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Mechanism is either noncontact deceleration/rotation injury or contact injury with valgus force to an extended knee.
• Patients often hear a “pop.” They note feelings of instability and the knee giving out with twisting activities.
• Substantial knee effusion is present within first 12 hours after injury.
• There is a high incidence of associated injuries, including meniscus tears.
• Lachman is most sensitive test for diagnosis; pivot shift or Losee test helps evaluate rotational instability.
• Segond sign (avulsion of the anterolateral capsule of the tibia) may be seen on plain radiographs.
• MRI is helpful to confirm diagnosis and verify any additional concomitant injuries.
![]() Symptoms (History)
Symptoms (History)
The mechanism of injury should be elicited in any knee injury evaluation. This can guide the examination to additional structures that may also be injured. ACL injury can occur in a variety of ways; however, a few mechanisms predominate. The most common noncontact ACL injury mechanism involves a deceleration and rotational injury during running, cutting, or jumping activities. The most common contact injury involves either hyperextension and/or valgus forces to the knee by a direct blow.
ACL injury is often associated with a “pop” heard by the patient at the time of injury. This piece of history is not ACL specific, however. Upon return to competition, the patient will often notice instability of the knee or describe the knee “giving out” with twisting activities. Substantial knee swelling secondary to a hemarthrosis typically occurs within the first 4–12 hours following the injury.
![]() Signs (Physical Examination)
Signs (Physical Examination)
With the above history obtained and a proper physical examination, an ACL tear should be able to be diagnosed without any additional tests. A complete examination of the knee should be performed to evaluate for any other associated injuries. The uninjured knee is examined first to familiarize the patient with the knee examination.
The Lachman test is the most useful test for anterior laxity of the knee. The Lachman test is performed with the knee in 20–30 degrees of flexion as an anterior force is applied to the tibia while the other hand stabilizes the distal femur. The degree of anterior translation and the presence and character of an end point are assessed. The laxity is graded based on comparison to the uninjured contralateral knee. Grade 1 laxity is 1–4 mm of increased translation. Grade 2 laxity is 5–9 mm of increased translation. Grade 3 laxity is more than 10 mm of translation as compared to the injured contralateral knee.
The anterior drawer test is another test to evaluate anterior tibial translation. This is performed with the knee in 90 degrees of flexion as an anterior force is applied to the tibia. This test is less sensitive than the Lachman test.
In the acute setting of an ACL tear, there is often a window where an accurate examination can occur before extensive knee swelling and guarding inhibit examination. Aspiration of a hemarthrosis can help to decrease pain and improve the quality of the examination in the acute setting as well.
The pivot shift test (Losee test) is performed to test the rotational instability associated with an ACL tear. The test is based on the lateral tibial plateau subluxing anteriorly with extension and reduction of the lateral compartment with flexion. The most effective method of achieving this result is by flexing the knee with an axial load from full extension with valgus stress at the knee and internal rotation of the tibia. The reduction of the subluxation should occur at approximately 30 degrees of flexion. MCL injury and some meniscal tears may produce a false-negative test.
The pivot shift test is considered the most functional test to evaluate knee stability after ACL injury. An examination under anesthesia is also often useful in obtaining a more accurate pivot shift test. This can be useful in a patient with an unclear history of instability and an equivocal examination in the office.
![]() Imaging Studies
Imaging Studies
Plain radiographs of the knee should be obtained to rule out fractures about the knee. The Segond fracture, as discussed earlier, is an avulsion of the anterolateral capsule of the tibia. Before skeletal maturity, an avulsion of the tibial insertion of the ACL can also be seen radiographically. Following radiographs, an MRI is the most useful examination for an evaluation of associated injuries. Although generally not needed for diagnosis of an ACL tear, MRI can diagnose an ACL tear with 95% or better accuracy. Bone bruises of the lateral femoral condyle and lateral tibial plateau are noted in up to 80% of ACL injuries.
![]() Special Studies
Special Studies
Instrumented laxity evaluations can augment the physical examination and provide an objective baseline for future comparison. The most commonly used arthrometer, the KT-1000 (MEDmetric, San Diego, CA), uses a series of standard forces to measure anterior translation of the tibia with the knee in 20–30 degrees of flexion similar to the Lachman test.
![]() Treatment
Treatment
A. Nonsurgical
Rehabilitation following an isolated ACL injury should include an effort to regain knee motion and strengthen the muscles about the knee. Returning to activities that produce episodes of instability is discouraged. Once motion and strength have been restored, a gradual return to activities can be attempted to determine the functional level that can be attained without instability.
Nonoperative management with rehabilitation after an ACL injury generally yields poor results in patients who return to competitive activities. Significant episodes of instability resulting in pain, swelling, and disability occur in about 80% of individuals who participate in sporting activities such as tennis, football, and soccer. These episodes of instability are thought to place the menisci and articular cartilage of the knee at risk for further injury (Figure 3–15).

![]() Figure 3–15. Flow chart that summarizes the current management of acute anterior cruciate ligament (ACL) injuries. MRI, magnetic resonance imaging. (Reproduced, with permission, from Marzo JM, Warren RF: Results of nonoperative treatment of anterior cruciate ligament injury: changing perspectives. Adv Orthop Surg 1991;15:59.)
Figure 3–15. Flow chart that summarizes the current management of acute anterior cruciate ligament (ACL) injuries. MRI, magnetic resonance imaging. (Reproduced, with permission, from Marzo JM, Warren RF: Results of nonoperative treatment of anterior cruciate ligament injury: changing perspectives. Adv Orthop Surg 1991;15:59.)
B. Surgical
The decision to surgically reconstruct an ACL tear is individualized and based on the patient’s desire to return to competition, age, accompanying degenerative changes, and objective and subjective knee instability. For example, a young, active patient with continued desire to compete in cutting and jumping sports with both objective and subjective knee instability may be best treated with surgical reconstruction. On the other hand, an older patient with some degenerative arthritis of the knee and minimal desire for continued competitive athletics and no subjective instability would be much more suited to nonsurgical care.
Early in the history of ACL surgery, primary repairs of the ligament were found to do poorly. This gave way to ligament reconstruction using a variety of graft materials. Everything from synthetics to autograft and allograft tissues has been used for reconstruction of the ACL. Over time, autograft bone-patellar tendon-bone, semitendinosus/gracilis hamstring autograft, and allograft bone-patellar tendon-bone constructs have proven to be the most commonly used grafts and have been successful for ACL reconstructions.
The goal of ACL reconstruction is to reproduce the strength, function, and location of the intact ACL. Recently, there have been some articles challenging the results seen after single-bundle reconstruction. They point to instability in up to 30% of patients and only a 60–70% return to sport. Therefore, in an effort to replicate the normal anatomy and try to improve outcomes after ACL surgery, the double-bundle reconstruction has been advocated. This technique attempts to take advantage of the anatomy of the native ACL, which is composed of two bundles: anteromedial (AM) and posterolateral. The AM bundle is thought to provide stability to anteroposterior movement, and the posterolateral bundle provides rotational control. Advocates of the double-bundle reconstruction point to its ability to resist rotatory loads and mimic normal knee kinematics more closely. Biomechanical and some level I studies have demonstrated a benefit in objective rotational stability, but a clear clinical improvement has not been proven versus traditional single-bundle reconstruction. Double-bundle versus single-bundle reconstruction of the ACL remains a controversial and highly debated topic. Regardless of whether single-bundle or double-bundle reconstruction is used, the focus should be on attempting to restore the normal anatomy of the ACL with the position and placement of the tunnels.
1. Single bundle—Once a graft of adequate strength is selected, the location of placement of the graft is of utmost importance. The graft is generally passed through a bone tunnel in the tibia and a bone tunnel through the femur. The intraarticular placement of the tibial tunnel is generally in the center of the native ACL stump just in front of the PCL origin and just medial to the center of the notch in the coronal plane for a single-bundle reconstruction (Figures 3–16 and 3–17).

![]() Figure 3–16. Drawing of the medial surface of the right lateral femoral condyle showing the average measurements and body relations of the femoral attachment of the anterior cruciate ligament. (Reproduced, with permission, from Arnoczky SP: Anatomy of the anterior cruciate ligament. Clin Orthop Relat Res 1983;172:19.)
Figure 3–16. Drawing of the medial surface of the right lateral femoral condyle showing the average measurements and body relations of the femoral attachment of the anterior cruciate ligament. (Reproduced, with permission, from Arnoczky SP: Anatomy of the anterior cruciate ligament. Clin Orthop Relat Res 1983;172:19.)

![]() Figure 3–17. The upper surface of the tibial plateau to show average measurements and relations of the tibial attachments of the anterior cruciate ligament. (Reproduced, with permission, from Girgis FC, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)
Figure 3–17. The upper surface of the tibial plateau to show average measurements and relations of the tibial attachments of the anterior cruciate ligament. (Reproduced, with permission, from Girgis FC, Marshall JL, Monajem A: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis. Clin Orthop Relat Res 1975;106:216.)
Once the graft is in place, the proper tension and fixation of the graft must occur to achieve a successful ACL reconstruction. Establishing proper tension in the graft is important. A lax ACL graft may not restore stability to the knee, and an overtightened graft may cause failure of the graft or limit knee range of motion. Fixation of the graft is achieved through a variety of measures. The most common method involves placing an interference screw up the bone tunnel that captures the graft in the tunnel. The graft can also be fixed via sutures tied over various devices located on the outer cortex of the tunnels.
2. Double bundle—There are a couple differences to point out with the double-bundle reconstruction. First, an accessory AM portal is required in addition to the AM and anterolateral portals normally required for a knee arthros-copy. This portal becomes crucial for drilling accurately the femoral-sided tunnels, especially the AM femoral tunnel. Furthermore, special attention is taken to examine the tear pattern, which helps in locating the native locations of the AM and posterolateral bundles. Measuring the width/length of the insertions is also important because an ACL insertion less than 12 mm is extremely difficult technically to perform. Care must also be taken to ensure that there is at least a 2-mm bridge of bone between the two tunnels or the risk of convergence of the tunnels becomes very high. The recommended grafts by the authors at Pittsburgh are two tibialis anterior or posterior allograft tendons. Fixation on the femoral side is done with EndoButtons (Smith and Nephew endoscopy) and on the tibial side with interference screws. The grafts are tensioned at 0–15 degrees for the posterolateral bundle and 45–60 degrees for the AM bundle.
![]() Complications
Complications
Although ACL reconstruction often results in a successful outcome, there are several complications that can occur. One of the most common complications is a loss of knee motion. This is minimized by obtaining and maintaining full knee extension immediately following surgery. Knee flexion exercises are begun as soon as possible postoperatively, with a goal of 90 degrees by 1 week after surgery. Additionally, patellar mobilization is performed in an attempt to minimize patellofemoral scarring. Another common complication of ACL reconstruction is anterior knee pain. The exact etiology of this pain is unclear. However, it is thought that patellar tendon autograft harvest may increase the incidence of patellofemoral pain. Less common complications (<1%) include patellar fracture, patellar tendon rupture, and quadriceps tendon rupture depending on the graft harvest site.
![]() Results/Return to Play
Results/Return to Play
The goal of any rehabilitation protocol for an ACL reconstruction is to return the patient to the full desired level of activity in as short amount of time as possible while avoiding any complications or setbacks. Through improved surgical techniques and accelerated rehabilitation protocols, most studies have shown a 90% or better return to play and patient satisfaction. Patients generally are able to return between 4 and 6 months postoperatively, with some professional athletes returning successfully to competition in 3 months. Specific criteria for return to sports vary from institution to institution, with a combination of functional testing, subjective reporting, and clinical examination contributing to the decision. In general, the criteria for return to sports include full range of motion, KT1000 testing within 2–3 mm of the uninjured knee, >85% quadriceps strength and full hamstring strength, and functional testing within 85% of the contralateral leg.
Herrington L, Wrapson C, Matthews M, et al: Anterior cruciate ligament reconstruction, hamstring versus bone-patella tendon-bone grafts: a systematic literature review of outcome from surgery. Knee2005;12:41. [PMID: 15664877]
Järvelä T, Moisala AS, Sihvonen R, et al: Double-bundle anterior cruciate ligament reconstruction using hamstring autografts and bioabsorbable interference screw fixation: prospective, randomized clinical study with 2 year results. Am J Sports Med 2008;36:290. [PMID: 17940145]
Laxdal G, Kartus J, Hansson L, et al: A prospective randomized comparison of bone-patellar tendon-bone and hamstring grafts for anterior cruciate ligament reconstruction. Arthroscopy 2005;21:34. [PMID: 15650664]
Prodromos CC, Fu FH, Howell SM, et al: Controversies in soft-tissue anterior cruciate ligament reconstruction: grafts, bundles, tunnels, fixation and harvest. J Am Acad Orthop Surg 2008;16:376. [PMID: 18611995]
4. Posterior Cruciate Ligament Injuries
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Most common mechanisms of injury are a direct blow to the anterior tibia with the knee flexed or a fall into the ground with the foot plantar flexed.
• Patients complain of knee pain, swelling, and stiffness.
• Physical examination may show positive posterior drawer, Godfrey, and reverse pivot shift tests.
• Must perform thorough knee exam because concomitant injuries are common with PCL injury (posterolateral corner, meniscus).
• Imaging studies should include plain radiographs as well as confirmatory MRI. Radiographs are helpful in chronic setting of PCL to assess patellofemoral and medial compartment arthritis.
![]() Symptoms (History)
Symptoms (History)
When evaluating a patient for a PCL injury, it is important to obtain the mechanism of injury, the severity of the injury, and any potential associated injuries. In contrast to an ACL tear, it is rare for patients with PCL injuries to report hearing a “pop” or report any feelings of subjective instability. More commonly, patients will complain of knee pain, swelling, and stiffness.
The presentation of a patient with a subacute or chronically injured PCL can range from asymptomatic to significant instability and pain. Patients with significant varus alignment or injury to the lateral structures of the knee will often complain of feelings of instability and giving way. There are a few characteristic mechanisms of PCL injury that differ significantly from the mechanism of ACL injuries. One of the most common mechanisms of PCL injury is the “dashboard” injury during which the anterior tibia sustains a posteriorly directed force from the dashboard with the knee in 90 degrees of flexion. Sports injuries to the PCL result from an outside force or blow, in contrast to the typical deceleration twisting mechanism of an ACL injury. The most common methods of a sports PCL injury include a direct blow to the anterior tibia or via a fall onto the flexed knee with the foot in plantar flexion. The most common mechanism for isolated PCL injury in the athlete is a partial tear associated with hyperflexion of the knee. Additionally, significant multiligamentous knee injuries with PCL tears can be seen after a varus or valgus stress is applied to the hyperextended knee.
![]() Signs (Physical Examination)
Signs (Physical Examination)
As with other ligamentous injuries, a thorough knee examination is necessary. Specific cues to injury to the PCL on initial inspection include abrasions or ecchymosis around the proximal anterior tibia and ecchymosis in the popliteal fossa. Assessment for meniscal damage and associated ligamentous injury should be performed. Evaluation of ACL laxity in the presence of an acute PCL injury is challenging due to the lack of a stable reference point to perform a Lachman or anterior drawer test.
Examination of the PCL in the acutely injured knee can be challenging. Despite increased awareness of the injury, many PCL injuries go undiagnosed in the acute setting. The most accurate clinical test of PCL integrity is the posterior drawer test. The knee is flexed to 90 degrees with the patient supine and a posteriorly directed force is applied to the anterior tibia. The amount of posterior translation and the presence and character of the end point are noted. The extent of translation is assessed by noting the change in the distance of the step-off between the AM tibial plateau and the medial femoral condyle. The tibial plateau is approximately 1 cm anterior to the medial femoral condyle on average. However, the contralateral knee must be examined to establish a baseline.
Another test for examination of the PCL is the posterior sag or Godfrey test. This test involves flexing the knee and hip and noting the posterior pull of gravity creating posterior “sag” of the tibia on the femur. An adjunct to this test involves watching for a reduction of this subluxation with active quadriceps contraction.
The reverse pivot shift is the analog to the pivot shift in the evaluation of an ACL injury. This is performed by placing a valgus stress on the knee with the foot externally rotated. The knee is then extended from 90 degrees of flexion, and a palpable reduction of the posterolateral tibial plateau is noted between 20 and 30 degrees of flexion.
It is extremely important to evaluate the posterolateral structures of the knee in the setting of a suspected PCL injury. Injury to the posterolateral structures has been reported to occur in up to 60% of PCL injuries.
![]() Imaging Studies
Imaging Studies
A. Radiographs
Given the magnitude of the forces required to injure the PCL, plain radiographs of the knee are essential to evaluate for bony injuries, dislocation, or evidence of other associated injuries. Subtle posterior subluxation on the lateral radiograph may also indicate PCL injury. Stress posterior drawer radiographs and contralateral comparisons may also increase the sensitivity for detecting PCL injuries with plain radiographs. In the chronic setting of PCL injury, radiographs are useful to assess for patellofemoral and medial compartment degenerative changes that can occur over time.
B. MRI
Although plain films are necessary and useful in the evaluations of these injuries, MRI has become the diagnostic study of choice for the knee with a presumed PCL injury. MRI has been reported to be 96–100% sensitive at diagnosing PCL tears. Equally or more importantly, MRI is extremely valuable in its ability to detect associated injuries. This is particularly important in diagnosing posterolateral corner injuries because these can often be missed on the initial clinical examination. In multiligamentous knee injuries, MRI can also be of use in assessing the ACL as clinical examination of the ACL is challenging in the setting of a complete PCL tear.
C. Special Studies
In the setting of a chronic isolated PCL tear, pain in the medial and patellofemoral compartments is generally evaluated with radiographs. If these are normal, some surgeons will proceed with a bone scan to evaluate for increased uptake in these areas. Areas under increased stress demonstrate increased uptake on the bone scan before signs of advanced arthritis occur on radiographs. This subset of patients may benefit from a PCL reconstruction to decrease the stress and delay osteoarthritis.
![]() Treatment
Treatment
There is significant controversy in the treatment of isolated PCL injuries. There are multiple factors that must be evaluated in the decision to treat a complete PCL rupture. The patient’s age, activity level, expectations, and associated injuries must be taken into account. The literature on operative versus nonsurgical treatment of these injuries can be difficult to interpret, and there are no long-term follow-up studies of randomized patient groups.
A. Nonsurgical
Rehabilitation of the PCL injured knee is often largely dependent on the associated injuries sustained by the knee. This is particularly true with the commonly associated posterolateral corner injury. Therefore, we will focus on the rehabilitation of the isolated PCL injured knee. Regaining motion and strength are the two key objectives of a rehabilitation program. Obtaining full quadriceps strength is essential for achieving the optimal result with nonsurgical treatment. The initial treatment is aimed at keeping the tibia reduced under the femur and minimizing tension on the injured PCL. With partial injuries (grade I and II), the prognosis is quite good, and early motion with weight bearing is the usual course of therapy. In a complete PCL tear, most will keep the knee immobilized in extension to protect the posterolateral structures. Early strengthening exercises focus on quadriceps strength with quadriceps sets, straight leg raises, and partial weight bearing in extension.
Overall, most patients benefit from nonsurgical treatment of a PCL tear. Despite objective findings of instability that are often noted on examination, most patients subjectively are satisfied with the function of the knee. Bracing is generally ineffective in controlling PCL laxity clinically.
The main subjective complaint with chronic PCL insufficiency, however, is pain rather than instability. A PCL-deficient knee with posterior tibial subluxation places significantly increased stresses on the patellofemoral and medial compartments of the knee. In one series where patients with PCL injuries were followed with serial radiographs, 60% of patients displayed some degenerative changes of the medial compartment.
B. Surgical
Surgical management of PCL injuries are broken down into avulsion fractures, isolated acute PCL injuries, multiligamentous injuries, and chronic PCL insufficiency. Avulsion fractures of the PCL are rare fractures. If nondisplaced, these injuries are treated nonsurgically. If significantly displaced, these fractures are generally treated with open reduction and internal fixation.
Isolated PCL injuries are generally still treated with nonsurgical care by the majority of surgeons at this time. However, it has been shown that nonoperative care of these injuries is not without consequences. Although subjective results in these patients are good in the short term, many continue to have objective instability and display degenerative arthritic changes over time. A follow-up of PCL-deficient knees at an average of 15 years after injury found that 89% of patients had persistent pain and half had chronic effusions. All patients in this group showed degenerative changes when followed for 25 years. Therefore, given the risks of continued instability and the potential of an increased chance of arthritic changes, surgical reconstruction of the PCL is a reasonable choice.
Initially, surgical care of complete PCL tears consisted of a primary repair of midsubstance tears. The objective stability of these repairs was generally disappointing. Current reconstruction methods generally involve routing either autograft or allograft tendons through bone tunnels to reconstruct the PCL in an anatomic fashion. Although there are several different methods of reconstructing the PCL, the two main categories of PCL reconstruction consist of single-and double-bundle repairs. Classically, reconstructions of the PCL anatomically replicated the anterolateral bundle of the native PCL with a single-bundle reconstruction. As problems were noted with recurrence of posterior laxity in the postoperative period, a double-bundle technique was derived to reconstruct both the anterolateral and posteromedial bundles of the native PCL. The advantages of the double-bundle technique are thus far theoretical, and there is no long-term clinical follow-up demonstrating the superiority of a double-bundle reconstruction at this time.
The severe instability noted with PCL injuries associated with multiligamentous knee injuries makes the argument for ligament reconstruction more compelling in this patient population. Many of the studies involving PCL reconstruction in these complex knee injuries have involved primary repair attempts. Although subjective results were generally good, residual excessive, objective laxity was very common following repairs. More recently, ligament reconstructions with allograft and autograft have become the dominant method of PCL reconstruction in this challenging patient population.
![]() Complications
Complications
The most common complication following PCL reconstruction is the return of objective posterior laxity on physical examination. This does not present as subjective laxity, however, and patient satisfaction remains high despite objective laxity. Acute PCL reconstructions in the setting of a multi-ligamentous knee repair/reconstruction can result in arthrofibrosis with extensive postoperative scarring.
![]() Results/Return to Play
Results/Return to Play
Even with nonsurgical management of a PCL injury, the prognosis for a functional recovery and return to competition is very good. A strong quadriceps muscle and extensor mechanism can significantly compensate for PCL laxity. Athletes should spend a minimum of 3 months in rehabilitation before attempting a return to competition. However, a subset of patients experience significant instability with a grade III PCL injury that does not allow a return to competition. This subset of patients may benefit from PCL reconstruction.
On the other hand, the prognosis for a PCL tear associated with a multiligamentous knee injury is guarded with respect to return to play. Although prompt recognition of a multiligamentous injury and appropriately timed treatment, reconstruction, and rehabilitation are essential for optimal recovery, these injuries are such that a significant percentage of patients will not be able to return to full competition.
Jung TM, Lubowicki A, Wienand A, Wagner M, Weiler A: Knee stability after posterior cruciate ligament reconstruction in female versus male patients: a prospective matched-group analysis. Arthroscopy2011;27:399. [PMID: 21168303]
Li G, Papannagari R, Li M, et al: Effect of posterior cruciate ligament deficiency on in vivo translation and rotation of the knee during weightbearing flexion. Am J Sports Med 2008;36:474. [PMID: 18057390]
Lien OA, Aas EJ, Johansen S, Ludvigsen TC, Figved W, Engebretsen L: Clinical outcome after reconstruction for isolated posterior cruciate ligament injury. Knee Surg Sports Traumatol Arthrosc2010;18:1568. [PMID: 20571763]
McAllister DR, Petrigliano FA: Diagnosis and treatment of posterior cruciate ligament injuries. Curr Sports Med Rep 2007;6:293. [PMID: 17883964]
![]() CPT Codes for Ligament Injuries to the Knee
CPT Codes for Ligament Injuries to the Knee
27405 Repair, primary, torn ligament and/or capsule, knee; collateral
27407 Repair, primary, torn ligament and/or capsule, knee; cruciate
27409 Repair, primary, torn ligament and/or capsule, knee; collateral and cruciate ligaments
27427 Ligamentous reconstruction (augmentation), knee; extraarticular
27428 Ligamentous reconstruction (augmentation), knee; intraarticular (open)
27429 Ligamentous reconstruction (augmentation), knee; intraarticular (open) and extraarticular
27552 Closed treatment of knee dislocation; requiring anesthesia
27556 Open treatment of knee dislocation, includes internal fixation, when performed; without primary ligamentous repair or augmentation/reconstruction
27557 Open treatment of knee dislocation, includes internal fixation, when performed; with primary ligamentous repair
27558 Open treatment of knee dislocation, includes internal fixation, when performed; with primary ligamentous repair, with augmentation/reconstruction
27570 Manipulation of knee joint under general anesthesia (includes application of traction or other fixation devices)
29850 Arthroscopically aided treatment of intercondylar spine(s) and/or tuberosity fracture(s) of the knee, with or without manipulation; without internal or external fixation (includes arthroscopy)
29875 Arthroscopy, knee, surgical; synovectomy, limited (eg, plica or shelf resection) (separate procedure)
29876 Arthroscopy, knee, surgical; synovectomy, major, two or more compartments (eg, medial or lateral)
29884 Arthroscopy, knee, surgical; with lysis of adhesions, with or without manipulation (separate procedure)
29888 Arthroscopically aided anterior cruciate ligament repair/augmentation or reconstruction
29889 Arthroscopically aided posterior cruciate ligament repair/augmentation or reconstruction
5. Patella Dislocation
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Almost always a lateral dislocation.
• Pain, swelling, and tenderness over medial border of the patella and apprehension with knee flexed and patella pushed laterally.
• Check hypermobility of the contralateral knee for comparison.
• Look for osteochondral fragment on radiographs.
Dislocation of the patella is a potential cause of acute hemarthrosis and must be considered when evaluating a patient with an acute knee injury. The injury occurs when valgus force and external rotation of the tibia are applied to a flexed leg. It is most common in females in the second decade of life.
![]() Clinical Findings
Clinical Findings
The patella almost always dislocates laterally. The patient may notice the patella sitting laterally or might say that the rest of the knee has shifted medially. It is unusual to see actual dislocation of the patella except at the time of injury. Reduction occurs when the knee is extended.
Examination will demonstrate tenderness over the medial retinaculum and adductor tubercle, which is the origin of the medial patellofemoral ligament. The patient will also have pain and apprehension when the patella is pushed laterally with the knee slightly bent. Radiographs, including an axial patellar view, should be obtained to determine whether there are osteochondral fractures. Often, a small fleck of bone is avulsed by the capsule on the medial aspect of the patella. This is not intraarticular and does not require removal. A displaced osteochondral fracture will require excision or internal fixation. Examination of the uninjured knee is recommended to determine whether there are predisposing factors for dislocation, such as patella alta, genu recurvatum, increased Q angle, and patellar hypermobility. Patella alta, or high-riding patella, is identified by measuring the length of the patellar tendon and dividing by the length of the patella. The upper limit of normal is 1.2. The Q angle is formed by a line through the patellar tendon intersecting a line from the anterior superior iliac spine in the center of the patella. A normal Q angle is about 10 degrees, with a range of about plus or minus 5 degrees. Patients with generalized hypermobility have increased extension of the knee, or genu recurvatum, which in effect gives them patella alta. They also often have hypermobility of all the capsular ligamentous structures, including the static stabilizers of the kneecap, giving them significant patellar hypermobility.
![]() Treatment and Prognosis
Treatment and Prognosis
A wide variety of treatment options have been recommended for patellar dislocations, including immediate mobilization and strengthening exercises, immobilization in a cylinder cast for 6 weeks followed by rehabilitation, arthroscopy with or without retinacular repair, surgical repair of the torn retinaculum, or immediate patellar realignment.
Treatment is based on which predisposing factors are present. Little is lost by functional treatment, similar to the treatment of isolated MCL sprains, which is often successful. If dislocation recurs, realignment may be performed. A long-term study showed that patients treated surgically for patellar malalignment problems had a higher incidence of osteoarthritis than those treated nonoperatively.
Buchner M, Baudendistel B, Sabo D, et al: Acute traumatic primary patellar dislocation: long-term results comparing conservative and surgical treatment. Clin J Sport Med 2005;15:62. [PMID: 15782048]
Gerbino PG, Zurakowski D, Soto R, et al: Long-term functional outcome after lateral patellar retinacular release in adolescents: an observational cohort study with minimum 5 year follow-up. J Pediatr Orthop 2008;28:118. [PMID: 18157056]
Smith TO, Davies L, Chester R, Clark A, Donell ST: Clinical outcomes of rehabilitation for patients following lateral patellar dislocation: a systematic review. Physiotherapy 2010;96:269. PMID: 21056161]
![]() CPT Codes for Patellar Dislocations
CPT Codes for Patellar Dislocations
27340 Excision, prepatellar bursa
27420 Reconstruction of dislocating patella (eg, Hauser-type procedure)
27422 Reconstruction of dislocating patella, with extensor realignment and/or muscle advancement or release (eg, Campbell, Goldwaite-type procedure)
27524 Open treatment of patellar fracture, with internal fixation and/or partial or complete patellectomy and soft-tissue repair
27562 Closed treatment of patellar dislocation; requiring anesthesia
27566 Open treatment of patellar dislocation, with or without partial or total patellectomy
27570 Manipulation of knee joint under general anesthesia (includes application of traction or other fixation devices)
29435 Application of patellar tendon bearing cast
29873 Arthroscopy, knee, surgical; with lateral release
29874 Arthroscopy, knee, surgical; for removal of loose body or foreign body (eg, osteochondritis dissecans fragmentation, chondral fragmentation)
KNEE TENDON INJURY
Ruptures of the quadriceps and patellar tendons usually result from a tremendous eccentric contraction of the quadriceps muscle, as may occur when an athlete stumbles and tries not to fall. Both types of ruptures are more common in patients with underlying disorders of the tendon.
1. Rupture of the Quadriceps Tendon
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Occur in patients older than 40 years.
• Patient will be unable to extend the knee.
• The quadriceps will retract proximally if left untreated.
Quadriceps tendon ruptures occur most frequently in patients over 40 years old. Biopsies of fresh rupture sites showed local degenerative changes already present, consistent with the theory that normal tendons do not rupture. Rarely the injury occurs bilaterally and is often associated with gout, diabetes, or steroid use. When it does occur bilaterally with only a small amount of trauma, the diagnosis may be difficult to make because of the small amount of swelling or symptoms of injury.
The cardinal symptom is inability to extend the knee. When extension is attempted, a gap develops in the suprapatellar region. The patella rides at a slightly lower level, and the anterior border of the femoral condyles may be palpated.
Acute complete quadriceps tendon ruptures should be paired surgically. If left untreated, proximal migration and scarring of the quadriceps muscle will occur. Direct-end repair produces excellent results. Neutralizing the forces across the repair is difficult, and immobilization in extension is recommended. Repair of ruptures more than 2 weeks old may be difficult and may require quadriceps lengthening, muscle or tendon transfers, or a combination of these procedures.
West JL, Keene JS, Kaplan LD: Early motion after quadriceps and patellar tendon repairs: outcomes with single-suture augmentation. Am J Sports Med 2008;36:316. [PMID: 17932403]
2. Rupture of the Patellar Tendon
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Typically occurs in patients younger than 40.
• Patella alta on radiographs.
• Inability to extend the knee.
Rupture of the patella tendon occurs more frequently in patients less than 40 years old. The patient cannot actively extend the knee, the patella is high-riding, and a defect is palpable beneath the patella. Surgical repair is the treatment of choice. The tendon, along with the medial and lateral retinaculum, should be sewn end to end. A stress-relieving wire may be placed around the patella and through the tibial tubercle. The wire should be removed in 6–8 weeks. Chronic patellar tendon ruptures are very hard to treat. The quadriceps must be freed up from the femur and the patella pulled down to the proper location. The gracilis and semitendinosus tendons can be used to substitute for the patellar tendon.
The extensor mechanism may also be disrupted at the inferior pole of the patella where the patellar tendon originates. This usually occurs in a child aged between 8 and 12 years. The distal pole of the patella plus a large sleeve of articular cartilage is pulled off (Figure 3–18). This may be easily misdiagnosed if the fragment of bone is small. Reestablishment of an intact extensor mechanism is necessary. With displaced fractures, open reduction and internal fixation with tension band wiring are recommended.

![]() Figure 3–18. Sleeve fracture of the patella. A: A small segment of the distal pole of the patella is avulsed with a relatively large portion of the articular surface. B: Lateral radiograph of the knee with a displaced sleeve fracture of the patella. Note that the small osseous portion of the displaced fragment is visible, but the cartilaginous portion is not seen. (Reproduced, with permission, from Rockwood CA Jr, ed: Fractures in Children, 3rd ed. Philadelphia: Lippincott; 1991.)
Figure 3–18. Sleeve fracture of the patella. A: A small segment of the distal pole of the patella is avulsed with a relatively large portion of the articular surface. B: Lateral radiograph of the knee with a displaced sleeve fracture of the patella. Note that the small osseous portion of the displaced fragment is visible, but the cartilaginous portion is not seen. (Reproduced, with permission, from Rockwood CA Jr, ed: Fractures in Children, 3rd ed. Philadelphia: Lippincott; 1991.)
Brooks P: Extensor mechanism ruptures. Orthopedics 2009;32:9. [PMID: 19751001]
![]() CPT Codes for Tendon Ruptures
CPT Codes for Tendon Ruptures
27380 Suture of infrapatellar tendon; primary
27381 Suture of infrapatellar tendon; secondary reconstruction, including fascial or tendon graft
27385 Suture of quadriceps or hamstring muscle rupture; primary
27386 Suture of quadriceps or hamstring muscle rupture; secondary reconstruction, including fascial or tendon graft
27430 Quadricepsplasty
KNEE PAIN
Pain in the knee region is a very common complaint of athletes. If there is no history of an acute injury, then overuse is commonly the cause. The patient is often able to point to the area of pain. The history of activity must be obtained as well as overall evaluation of the extremities.
1. Anterior Knee Pain
![]() Patellofemoral Disorders
Patellofemoral Disorders
A. Essentials of Diagnosis
• Pain with activity involving stairs or hills.
• Commonly involves young females.
• Check Q angle, femoral anteversion, patellar mobility, and quadriceps strength and tone.
• On radiographs, look for valgus alignment of knee, OCLs, and patella alta.
B. Clinical Findings
1. Symptoms and signs—This is a common complaint and is frequently bilateral. It is most common in females during the second decade of life. The patellofemoral joint is often the source of pain. Entities such as chondromalacia patella, patellofemoral arthralgia, and lateral patellofemoral compression syndrome are diagnostic considerations.
Patellar pain is often felt when going up or down hills or stairs, and there may be complaints of instability during walking, running, or other sports activities. These activities may create a joint reaction force of several times the body weight on the patella with each step. Swelling is seldom a complaint. If the pain is in one knee only, the patient may alter the way of climbing and descending stairs so that the affected leg is kept straight and each step leads with the same foot. This strategy significantly decreases the joint reaction force on the patellofemoral joint.
Many of these problems arise because the patellofemoral joint is semiconstrained, especially in the range of 0–20 degrees of flexion, and the constraint increases as flexion increases. The degree of constraint is also dependent on a number of other factors, including the angle of the sulcus of the femur, the presence or absence of patella alta, and the generalized ligamentous laxity of the patient. In addition, femoral anteversion and increased Q angle (Figure 3–19) may lead to increased instability of the patellofemoral joint. This lack of constraint may predispose the patella to frank dislocation, although subluxation is a much more common finding. The degree of congruity is anatomically variable and may lead to high-contact stresses caused by anatomic configuration and static and dynamic constraints on the patella. Increased pressure may cause pain and patellofemoral osteoarthritis.

![]() Figure 3–19. Q angle and valgus angulation. (Reproduced, with permission, from American Academy of Orthopaedic Surgeons: Athletic Training and Sports Medicine, 2nd ed. Burlington: Jones and Bartlett; 1991.)
Figure 3–19. Q angle and valgus angulation. (Reproduced, with permission, from American Academy of Orthopaedic Surgeons: Athletic Training and Sports Medicine, 2nd ed. Burlington: Jones and Bartlett; 1991.)
On physical examination of the patient with patellofemoral subluxation, minimal findings in relation to complaints may be present. Occasionally, crepitance, a crackling or clicking sound, is found with flexion and extension. Quadriceps strength, tone, and bulk are usually reduced. Pain may be elicited at a particular angle of flexion by putting the knee through its range of motion with resistance. Subluxation may often be diagnosed with the apprehension sign, a rapid contraction of the quadriceps when the patella is passively moved laterally.
2. Imaging studies—Roentgenographic examination will frequently show a valgus angulation of the knee on anteroposterior views. Occasionally, patella alta may be identified on the lateral view, and tangential views of the patella at various knee flexion angles will reveal a lack of contact of the medial facet of the patella with the medial facet of the trochlear groove of the femur. Lateral subluxation of the patellofemoral joint may also be observed.
This syndrome with a normal roentgenographic examination is frequently called chondromalacia patellae, or with subluxation identified on radiograph, it is referred to as patellofemoral subluxation. A more accurate term would be patellofemoral arthralgia, because patellofemoral subluxation was probably present prior to the onset of pain and because chondromalacia patellae (softening of the patellar cartilage) is an arthroscopic or pathologic diagnosis. Patellofemoral arthralgia is a clinical diagnosis.
C. Treatment
1. Chondromalacia patellae—Initially, treatment is conservative, with the intent of improving quadriceps strength and stamina to stabilize the patellofemoral joint. Weight loss is prescribed to decrease the stress on the patellofemoral joint; reduction in loading the knee in the flexed position also accomplishes pressure reduction. Knee orthotics may be beneficial. When subluxation and fear of dislocation are major concerns, an orthotic that limits extension of the knee may be beneficial because the patella becomes inherently more stable with knee flexion. Nonsteroidal anti-inflammatory medication may be beneficial.
2. Patellofemoral arthralgia—Only when conservative treatment has been exhausted is surgical treatment considered. Alteration in the alignment of the patellofemoral joint may be beneficial in patellofemoral arthralgia. Lateral retinacular release followed by a period of conservative treatment will be beneficial in some cases. Distal realignment may be necessary to achieve appropriate alignment and reduction in pain in those cases with an abnormality such as valgus knee or increased femoral anteversion.
3. Patellofemoral compression syndrome—With lateral patellofemoral compression syndrome, there is tenderness along the lateral facet of the patella or along the femoral condyle. Without cartilage damage, an effusion is rarely present. Treatment includes decreasing the activity level, including avoiding hills or step aerobics. Ice massage, quadriceps and hamstring stretching, and short-arc quadriceps exercises against resistance are recommended to strengthen the vastus medialis obliquus muscle without aggravating the pain. Patellar supports or neoprene sleeves may also be helpful. Most patients will respond to this regimen and gradually resume their activities. The role of releasing a contracted lateral patellofemoral retinaculum is controversial.
4. Patellar tendinitis—Patellar tendinitis, or jumper’s knee, is seen in basketball and volleyball players. Tenderness along the tendon, usually at the inferior pole of the patella, is noted. Treatment with ice and avoiding jumping usually suffice. In refractory cases, debridement of mucinous degenerative material from the tendon may be successful.
D. Prognosis
The prognosis for jumper’s knee is quite good. The condition is often persistent but self-limiting. The patient can always alleviate the symptoms by avoiding the activities that cause the problem.
Collado H, Fredericson M: Patellofemoral pain syndrome. Clin Sports Med 2010;29:379. [PMID: 20610028]
2. Lateral Knee Pain
![]() Iliotibial Band Friction Syndrome
Iliotibial Band Friction Syndrome
A. Essentials of Diagnosis
• Lateral knee pain.
• Commonly affects runners and cyclists.
• Tenderness over lateral epicondyle and positive Ober test.
Lateral knee pain that is not located on the joint line may result from iliotibial band friction syndrome. This is a form of bursitis caused by rubbing of the iliotibial band against the lateral epicondyle. Tenderness over the lateral epicondyle at about 30 degrees of flexion when the knee is being extended is indicative of this diagnosis. The Ober test, with abduction and then adduction of the leg, can also demonstrate the tightness of the iliotibial band when the patient is in a lateral decubitus position and the hip is hyperextended. Runners and cyclists are commonly afflicted. Crossover gait or running on banked terrain is thought to be a causative factor.
Treatment involves decreasing the athlete’s activities, ice massage, stretching of the iliotibial tract, and use of a lateral wedge orthotic in patients with heel varus. Running on flat terrain and changing the gait pattern may be helpful. In cyclists, lowering the seat height so the full extension of the knee is not reached and adjusting the pedals so that the toes are not internally rotated should help. Steroid injections are infrequently needed, and release of the inflamed portion of the iliotibial band is seldom necessary. As for other overuse syndromes of the knee, the prognosis is good.
Hariri S, Savidge ET, Reinold MM, Zachazewski J, Gill TJ: Treatment of recalcitrant iliotibial band friction syndrome with open iliotibial band bursectomy: indications, technique, and clinical outcomes. Am J Sports Med2009;37:1417. [PMID: 19286912]
Lavine R: Iliotibial band friction syndrome. Curr Rev Musculoskelet Med 2010;3:18. [PMID: 21063495]
![]() Other CPT Codes for the Knee
Other CPT Codes for the Knee
27305 Fasciotomy iliotibial band
27310 Arthrotomy, knee, with exploration, drainage, or removal of foreign body
27412 Autologous chondrocyte implantation, knee
27552 Closed treatment of knee dislocation; requiring anesthesia
27570 Manipulation of knee joint under general anesthesia (includes application of traction or other fixation devices)
29870 Arthroscopy, knee, diagnostic, with or without synovial biopsy (separate procedure)
29871 Arthroscopy, knee, surgical; for infection, lavage and drainage
29874 Arthroscopy, knee, surgical; for removal of loose body or foreign body (eg, osteochondritis dissecans fragmentation, chondral fragmentation)
29875 Arthroscopy, knee, surgical; synovectomy, limited (eg, plica or shelf resection) (separate procedure)
29876 Arthroscopy, knee, surgical; synovectomy, major, two or more compartments (eg, medial or lateral)
29877 Arthroscopy, knee, surgical; debridement/shaving of articular cartilage (chondroplasty)
29884 Arthroscopy, knee, surgical; with lysis of adhesions, with or without manipulation (separate procedure)
![]() ANKLE OR FOOT PAIN
ANKLE OR FOOT PAIN
Evaluation of foot and ankle injuries is described in Chapter 8. Injury specific to athletics includes chronic Achilles tendonitis, heel pain, plantar fasciitis, and posterior tibial syndrome.
![]() Clinical Findings
Clinical Findings
Achilles tendonitis is a frequent complaint in runners. This may result from a contracted gastrocsoleus, or hyperpronation may cause overpulling of the medial insertion. Additionally, there may be a bony prominence on the superior-posterior aspect of the calcaneus, causing retrocalcaneal bursitis.
Heel pain is a common problem in runners and is difficult to treat because of the uncertainty as to cause. Theories include painful heel spurs, bursitis, fat-pad atrophy, stress fracture, plantar fasciitis, or entrapment of the terminal branches of the posterior tibial nerve.
Many patients have pain localized in the posteromedial surface of the foot just distal to the attachment of the plantar fascia to the calcaneus (plantar fasciitis). This pain is often most severe on initially getting up in the morning and decreases as the day goes on.
Posterior tibial syndrome occurs in runners with hyperpronation. As the longitudinal arch flattens out, the posterior tibial musculotendinosis unit elevates the flattened arch and has abnormal strain placed upon it.
![]() Treatment
Treatment
Treatment depends on the cause of the injury but includes decreasing running activities, using a heel lift, and performing stretching exercises. If hyperpronation is thought to be the cause, an orthotic may be used. Steroid injections are not recommended as they could lead to weakening and subsequent rupture of the tendon.
Surgical intervention for chronic Achilles tendinitis or retrocalcaneal bursitis is rarely necessary. This would be done to remove areas of fibrosis or calcium within the tendon and possibly some bone from the posterior process of the calcaneus. The treatment for plantar fasciitis includes rest, ice massage, and possibly anti-inflammatory medications. A small shock-absorbing type of heel cup often is helpful, and a steroid injection may be given in recalcitrant cases. Acute rupture of the plantar fascia may occur. The pain is usually quite sharp and may cause significant disability for 6–12 weeks.
Hyperpronation may also cause fibular stress fractures. A semirigid orthosis may be recommended for this to decrease the amount and angular velocity of pronation. Using an orthosis while running actually increases the work of running, but if it decreases abnormal stresses in those who hyperpronate, it may be quite helpful.
Hanlon DP: Leg, ankle, and foot injuries. Emerg Med Clin North Am 2010;28:885. [PMID: 20971396]
Mizel MS, Hecht PJ, Marymont JV, et al: Evaluation and treatment of chronic ankle pain. Instr Course Lect 2004;53:311. [PMID: 15116624]
Simpson MR, Howard TM: Tendinopathies of the foot and ankle. Am Fam Physician 2009;80:1107. [PMID: 19904895]
![]() CPT Codes for the Ankle and Foot
CPT Codes for the Ankle and Foot
27650 Repair, primary, open or percutaneous, ruptured Achilles tendon
27652 Repair, primary, open or percutaneous, ruptured Achilles tendon; with graft (includes obtaining graft)
27654 Repair, secondary, Achilles tendon, with or without graft
27675 Repair, dislocating peroneal tendons; without fibular osteotomy
27676 Repair, dislocating peroneal tendons; with fibular osteotomy
27810 Closed treatment of bimalleolar ankle fracture (eg, lateral and medial malleoli, or lateral and posterior malleoli or medial and posterior malleoli); with manipulation
27814 Open treatment of bimalleolar ankle fracture (eg, lateral and medial malleoli, or lateral and posterior malleoli, or medial and posterior malleoli), includes internal fixation, when performed
28119 Ostectomy, calcaneus; for spur, with or without plantar fascial release
28445 Open treatment of talus fracture, includes internal fixation, when performed
29894 Arthroscopy, ankle surgical; with removal of loose body
29895 Arthroscopy, ankle surgical; synovectomy, partial
29897 Arthroscopy, ankle surgical; debridement, limited
29898 Arthroscopy, ankle surgical; debridement, extensive
![]() OTHER INJURIES OF THE LOWER BODY
OTHER INJURIES OF THE LOWER BODY
Many disorders seen while caring for athletes may be difficult to diagnose with certainty. The differential diagnosis must be carefully made to rule out more severe injuries. Often, a period of rest followed by gradual return to activities is the best treatment. During convalescence, application of ice packs, stretching exercises, and gradual strengthening of the injured limb will facilitate return to sports activities.
OVERUSE SYNDROMES OF THE LOWER EXTREMITIES
Many athletes such as runners, cyclists, aerobics enthusiasts, volleyball players, and basketball players have developed painful disorders of the lower extremities without an acute injury. History taking is very important, and the examiner should ask specific questions about the circumstances in which the discomfort occurs. In a runner, for example, the examiner should ask whether there was an increase in the distance run or a change in the running surface, at what point the pain was felt, and what home remedies have been tried before the runner sought advice from a physician.
The physical examination should include not only the affected area but also evaluation of the back, pelvis, leg lengths, genu varum or valgum, femoral and tibial torsion, and cavus or flatfoot deformities. The presence of hamstring and heel cord contracture should be determined, and the gait pattern should be observed. Running shoes should be inspected for wear patterns, which may be quite helpful.
1. Muscle Strains
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Classically involves muscles that span two joints (gastrocnemius).
• Patient will feel muscle “grab,” localized pain in the involved area.
• Eccentric forces thought to be causative factors.
Muscle strains of the lower extremity are frequent and disabling muscle injuries, with strain of the distal muscle tendon junction being most common. Muscles may stretch to about 125% of their resting length before tearing. Strains are graded as mild, moderate, and severe, based on the degree of pain, spasm, and disability that the strain causes. A severe strain would be complete disruption of the muscle, with a palpable defect and balling up of the muscle proximally.
Despite the frequency of muscle strains and the disability they produce, there is little scientific information on their pathologic basis. Muscles susceptible to more stretching are more susceptible to strains. In the lower extremity, the muscles most frequently injured are the hamstring, quadriceps, and gastrocnemius muscles. These muscles all cross two joints, and they may be unable to resist full stretching across both joints. The most powerful muscles are more likely to be strained, and strains are more common in “explosive” type athletics. Eccentric contraction (muscle contraction while the muscle is lengthening) is often thought to be causative in muscle strains.
![]() Clinical Findings
Clinical Findings
The diagnosis is relatively easy. Often the athlete will feel the muscle “grab” while he or she is accelerating. There is localized tenderness over the muscle and pain on stretching of the muscle. Because the two joint muscles are most frequently involved, the muscles should be stretched over both of the joints during examination.
![]() Treatment and Prognosis
Treatment and Prognosis
The treatment of muscle strains should begin with ice in the immediate postinjury period. Flexibility and strength should be regained prior to return to activity. This may take many months, and if the patient returns to activity too early, there may be a setback to the level of the original injury.
Strengthening of the muscles might make them less susceptible to being torn. It is commonly believed that flexibility will help prevent muscle strains, but there are conflicting reports regarding this.
Askling CM, Tengvar M, Saartok T, et al: Proximal hamstring strains of stretching type in different sports: injury situations, clinical and magnetic imaging characteristics and return to sport. Am J Sport Med2008;36:1799. [PMID: 18448581]
Fousekis K, Tsepis E, Poulmedis P, Athanasopoulos S, Vagenas G: Intrinsic risk factors of non-contact quadriceps and hamstring strains in soccer: a prospective study of 100 professional players. Br J Sports Med 2011;45:709. [PMID: 21119022]
2. Shin Pain
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Pain over the anterior tibia.
• Associated with increase in training or activity level.
• Radiographs will be negative for fracture.
![]() Clinical Findings
Clinical Findings
A. Shin Splints
The term “shin splints” is widely used for shin pain, but it is not a diagnostic term. A more specific diagnosis should be made if possible. Shin splints are usually defined as pain associated with activity in the beginning of training after a relatively inactive period. The pain and tenderness are usually located over the anterior compartment and disappear in 1–2 weeks as the athlete becomes conditioned to the exercise. Care must be taken to differentiate shin splints from stress fractures of the tibia, which cause more localized pain and have many more potential complications if not cared for properly.
B. Medial Tibial Syndrome
Medial tibial syndrome is also seen in runners, occurring along the medial border of the distal tibia. After 3–4 weeks, some hypertrophy of the cortical bone and periosteal new bone formation may be seen on radiograph. It is thought to be either a periosteitis or possibly an incomplete stress fracture. The pull of the tibialis posterior muscle from its origin on the tibia and posterior tibial tendinitis are also thought to be possible causes.
![]() Treatment
Treatment
Treatment for shin splints and medial tibial syndrome is rest and resumption of athletic activities in a graduated fashion.
3. Stress Fractures
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Localized pain after increase in training or activity.
• Plain radiographs often normal initially; MRI and bone scan are better diagnostic tests.
Stress fractures may occur in the pelvis, femoral neck, tibia, navicular, and metatarsals. They are usually the result of a significant increase in training and activity. In the female athlete, a triad of eating disorders resulting in poor nutrition, osteoporosis, and amenorrhea are associated with a higher prevalence of stress fractures.
The history is important in differentiating stress fractures from infection or neoplasm, particularly when there is a finding on radiographs. Plain radiographs are normal at first. MRI and technetium bone scans are the best diagnostic tests. If symptoms persist for over a month, radiographs may become positive.
Treatment of stress fractures involves rest and avoidance of high-impact activities until healing has occurred. This includes resolution of the tenderness and signs of fracture healing on plain radiographs. Continuous activity with stress fractures may lead to complete fractures. Patients must be made aware of this and all the complications that may develop with a complete fracture.
Feingold D, Hame SL: Female athlete triad and stress fractures. Orthop Clin North Am 2006;37:575. [PMID: 17141015]
Fredericson M, Jennings F, Beaulieu C, et al: Stress fractures in athletes. Top Magn Reson Imaging 2006;17:309. [PMID: 17414993]
Rauh MJ, Macera CA, Trone DW, et al: Epidemiology of stress fracture and lower-extremity overuse injury in female recruits. Med Sci Sports Exerc 2006;38:1571. [PMID: 16960517]
4. Exertional Compartment Syndromes
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Recurrent claudication/pain during exertional activity and relieved by rest.
• Measure compartment pressures while exercising on treadmill (pressures >30 mm Hg 1 minute after exercise or >20 mm Hg 5 minutes after exercise, or absolute values >15 mm Hg while resting are consistent with this diagnosis).
Exertional compartment syndromes may result from muscle hypertrophy within the confining osseofascial compartment. As the muscles hypertrophy and the amount of edema within the compartment increases, the blood supply to the nerves and muscles within the involved compartment is diminished, and the pressure continues to increase.
The syndrome presents as recurrent claudication during exertional activity and is relieved by rest. After exercise, the findings of localized pain, pain on passive motion, and hypesthesia are indicative.
Treatment consists of activity modification including gradual onset of training. If unsuccessful, compartment pressures may be measured while the patient is exercising on a treadmill, and if the pressures are elevated, surgical fasciotomy is usually effective.
Shah SN, Miller BS, Kuhn JE: Chronic exertional compartment syndrome. Am J Orthop 2004;33:335. [PMID: 15344575]
Tucker AK: Chronic exertional compartment syndrome of the leg. Curr Rev Musculoskelet Med 2010;3:32. [PMID: 21063498]
![]() CPT Codes for Overuse Injuries
CPT Codes for Overuse Injuries
20950 Monitoring of interstitial fluid pressure (includes insertion of device, eg, wick catheter technique, needle manometer technique) in detection of muscle compartment syndrome
27187 Prophylactic treatment (nailing, pinning, plating or wiring) with or without methylmethacrylate, femoral neck and proximal femur
27600 Decompression fasciotomy, leg; anterior and/or lateral compartments only
27601 Decompression fasciotomy, leg; posterior compartment(s) only
27602 Decompression fasciotomy, leg; anterior and/or lateral, and posterior compartment(s)
27759 Open treatment of tibia fracture by intramedullary implant
27892 Decompression fasciotomy, leg; anterior and/or lateral compartments only, with debridement of nonviable muscle and/or nerve
27893 Decompression fasciotomy, leg; posterior compartment(s) only, with debridement of nonviable muscle and/or nerve
27894 Decompression fasciotomy, leg; anterior and/or lateral, and posterior compartment(s), with debridement of nonviable muscle and/or nerve
28485 Open treatment of metatarsal fracture, includes internal fixation, when performed, each
CONTUSIONS AND AVULSIONS OF THE LOWER BODY
1. Contusion to the Quadriceps Muscle
![]() Essentials of Diagnosis
Essentials of Diagnosis
• History of direct trauma.
• Peripheral calcification on radiographs (as opposed to parosteal osteosarcoma, which has central calcification).
![]() Clinical Findings
Clinical Findings
A severe contusion to the quadriceps muscle (charley horse) is disabling, results in prolonged inactivity, and frequently occurs in football players. With significant bleeding into the muscle, there is inhibition of movement. Rarely, a compartment syndrome will occur.
Myositis ossificans may occur after these injuries. It may be apparent 2–4 weeks after the injury. Radiographically and histologically, myositis ossificans may be similar to parosteal osteosarcoma; therefore, the history of contusion is very important. Radiographs should be obtained after such a contusion to minimize myositis ossificans being confused with cancer.
![]() Treatment and Prognosis
Treatment and Prognosis
Quadriceps contusions should be treated with elevation of the leg and the hip and knee flexed to tolerance to minimize bleeding. After a few days, the knee can be moved with continuous passive motion or “drop-and-dangle,” gravity-assisted exercises. For the latter, the patient is seated on a table high enough to keep the feet off the floor. The patient then hooks the uninjured foot behind the ankle of the injured leg. The uninjured leg extends the knee of the injured leg, and gravity flexes the injured knee. Average length of disability for mild contusions is 2 weeks, and length for severe contusions is 3 weeks.
If heterotopic ossification is present, no specific treatment is recommended other than treatment for the contusion. Normal function may be obtained, but the recovery period is longer. Because early surgery may cause exacerbation of the heterotopic ossification, it should be avoided. There have also been recent animal studies examining the benefit of suramin, an antifibrotic agent that has been shown to help muscle regeneration and improved healing. While these initial results are encouraging, further studies are needed before suramin gains widespread clinical use after contusions.
Cooper DE: Severe quadriceps muscle contusions in athletes. Am J Sports Med 2004;32:820. [PMID: 15090402]
Kary JM: Diagnosis and management of quadriceps strains and contusions. Curr Rev Musculoskelet Med 2010;3:26. [PMID: 21063497]
CONTUSIONS ABOUT THE HIP AND PELVIS
![]() Clinical Findings
Clinical Findings
Contusions about the pelvis and hip region may be very painful and disabling. Because of the subcutaneous location of the iliac crests and the greater trochanters, these regions are at risk in contact sports.
A contusion over the greater trochanter may cause persistent bursitis, tenderness directly over the greater trochanter, and increased pain with adduction of the leg. Females are more prone to trochanteric bursitis because of their broader pelvis.
A hip pointer is a very painful contusion over the iliac crest that occurs from many contact sports. It must be differentiated from an avulsion fracture in a child, and a tear of the muscle aponeurosis in an adult. Profuse bleeding may occur and can be very painful.
![]() Treatment and Prognosis
Treatment and Prognosis
For contusion over the greater trochanter, treatment consists of ice applications and decreased activities. Padding may be helpful to prevent recurrent injuries. The prognosis is good. For hip pointer injuries, initial treatment with ice is helpful. Protective pads are useful in preventing these injuries and returning the athlete to activities sooner.
AVULSION OF THE TIBIAL TUBERCLE
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Adolescent athletes between 14 and 16 years of age.
• Result of a forceful contraction by quadriceps against a fixed tibia.
• Significant swelling and tenderness over the tubercle; palpable defect with substantial displacement.
• Patient will not be able to actively extend the knee.
• Radiographs will be helpful to determine amount of displacement and guide treatment course.
![]() Clinical Findings
Clinical Findings
Tibial tubercle avulsions occur in adolescent athletes, most often in males aged between 14 and 16 years. They result from a powerful contraction of the quadriceps muscle against a fixed tibia. This can occur by forced passive flexion of the knee against a powerful quadriceps contraction, as in an awkward landing at the end of a jump or fall. Avulsion of the tubercle may occur with either a sudden acceleration or deceleration of the knee extensor mechanism. The patellar tendon must pull hard enough to overcome the strength of the growth plate, the surrounding perichondrium, and the adjacent periosteum.
Swelling and tenderness are located over the proximal anterior tibia. A tense hemarthrosis may be present. A palpable defect in the anterior tibia is associated with a much-displaced avulsion. Proximal migration of the patella occurs, and the patella may seem to float off the anterior aspect of the femur. The knee is held flexed; with displaced fractures, the patient is unable actively to extend the knee.
Watson-Jones defined three types of avulsion fractures, which were subsequently refined as the following three types (Figure 3–20): type 1 fracture, in which the fracture line lies across the secondary center of ossification at the level of the posterior border of the patellar ligament; type 2 fracture, in which a separation breaks out at the primary and secondary ossification centers of epiphysis; and type 3 fracture, in which the separation propagates upward through the main portion of the proximal tibial epiphysis. The degree of displacement depends on the severity of injury to the surrounding soft-tissue moorings. A lateral radiograph with the tibia slightly internally rotated is the best view to see the fracture and the degree of displacement.

![]() Figure 3–20. Classification of avulsion fractures of the tibial tubercle. Type 1 fracture (left) across the secondary ossification center at level with the posterior border of the inserting patellar ligament. Type 2 fracture (center) at the junction of the primary and secondary ossification centers of the proximal tibial epiphysis. Type 3 fracture (right) propagates upward across the primary ossification center of the proximal tibial epiphysis into the knee joint. This fracture is a variant of the Salter-Harris III separation and is analogous to the fracture of Tillaux at the ankle because the posterior portion of the physis of the proximal tibia is closing. (Reproduced, with permission, from Odgen JA, Tross RB, Murphy MJ: Fractures of the tibial tuberosity in adolescents. J Bone Joint Surg Am 1980;62:205.)
Figure 3–20. Classification of avulsion fractures of the tibial tubercle. Type 1 fracture (left) across the secondary ossification center at level with the posterior border of the inserting patellar ligament. Type 2 fracture (center) at the junction of the primary and secondary ossification centers of the proximal tibial epiphysis. Type 3 fracture (right) propagates upward across the primary ossification center of the proximal tibial epiphysis into the knee joint. This fracture is a variant of the Salter-Harris III separation and is analogous to the fracture of Tillaux at the ankle because the posterior portion of the physis of the proximal tibia is closing. (Reproduced, with permission, from Odgen JA, Tross RB, Murphy MJ: Fractures of the tibial tuberosity in adolescents. J Bone Joint Surg Am 1980;62:205.)
![]() Differential Diagnosis
Differential Diagnosis
Osgood-Schlatter disease, or osteochondrosis of the tuberosity of the tibia, should not be confused with acute avulsion of the tibial tubercle. In the former, the patient is usually between 11 and 15 years of age and is involved in athletics. Pain is located at the tibial tubercle, and it has usually been present intermittently over a period of several months. Walking on a flat surface is not difficult, but ascending or descending stairs causes difficulty. Radiograph examination shows slight separation of the tibial tubercle with new bone formation beneath it (Figure 3–21).

![]() Figure 3–21. Development of Osgood-Schlatter lesion. (Left) Avulsion of osteochondral fragment that includes surface cartilage and a portion of the secondary ossification center of the tibial tubercle. (Right) New bone fills in the gap between the avulsed osteochondral fragment and the tibial tubercle. (Reproduced, with permission, from Rockwood CA Jr, ed: Fractures in Children, 3rd ed. Philadelphia: Lippincott; 1991.)
Figure 3–21. Development of Osgood-Schlatter lesion. (Left) Avulsion of osteochondral fragment that includes surface cartilage and a portion of the secondary ossification center of the tibial tubercle. (Right) New bone fills in the gap between the avulsed osteochondral fragment and the tibial tubercle. (Reproduced, with permission, from Rockwood CA Jr, ed: Fractures in Children, 3rd ed. Philadelphia: Lippincott; 1991.)
Treatment recommendations vary from decreasing the amount of running and jumping, but continuing participation in athletics, to cylinder cast immobilization for a short period of time. The long-term prognosis is excellent. While symptoms are often present for 2 years, early short-term cast immobilization may shorten this period of discomfort to 9 months. In most children, casting is not necessary. Explaining the benign nature of the problem to both the patient and the parents, reassuring them that the long-term prognosis is good, and modifying activities usually allow continued participation in athletics. Hamstring stretching and ice massage will hopefully decrease symptoms during the time needed for maturation of the tibial tubercle. The pain will go away when the tubercle unites with the tibia. In a very small number of cases, chronic pain will be present if the ossicle fails to unite. Painful ossicles in the adult are successfully treated with simple excision.
![]() Treatment
Treatment
Full function of the extensor mechanism is necessary, and therefore, treatment of tibial tubercle avulsion fractures is aimed at this goal. If the fracture is minimally displaced and the patient is able to fully extend the knee against gravity, nonoperative treatment is acceptable. A cylinder cast should be applied with the knee extended and worn for 4 weeks. Active range of motion and strengthening exercises should then commence. At 6 weeks, quadriceps exercises against resistance are initiated. For displaced fractures, open reduction and internal fixation are recommended, with screws if the piece or pieces are large enough. If rigid fixation of large fragments is obtained, early active flexion and passive extension may be initiated. If a tenuous repair is obtained, protection in a cast is advisable.
![]() Prognosis
Prognosis
Because the injury occurs in children who are close to skeletal maturity, meaningful growth abnormalities at the proximal tibial physis do not occur. Return to activities is allowed after the athlete develops quadriceps mass and strength equal to the contralateral side.
Abalo A, Akakpo-numado KG, Dossim A, Walla A, Gnassingbe K, Tekou AH: Avulsion fractures of the tibial tubercle. J Orthop Surg 2008;16:308. [PMID: 19126896]
![]() CPT Codes for Tibial Tubercle Avulsion
CPT Codes for Tibial Tubercle Avulsion
27418 Anterior tibial tubercleplasty
27530 Closed treatment of proximal tibial fracture (including tubercle)
27535 Open treatment of tibial fracture, proximal (plateau); unicondylar, includes internal fixation, when performed
AVULSIONS ABOUT THE PELVIS
![]() Clinical Findings
Clinical Findings
In the skeletally immature athlete, the apophysis, or growth plate where the muscle attaches to bone, is the weak link in the bone-muscle-tendon unit. Therefore, just as the growth plate is prone to breaking in children’s fractures, the bony origin of muscles may be pulled off. This most commonly occurs in athletes between 14 and 25 years of age. Comparison radiographs may be helpful to make sure the avulsion fracture is not just a normal anatomic variant. In the pelvis, this may occur at the iliac crest (abdominal muscles), anterior superior iliac spine (sartorius origin), anterior inferior iliac spine (rectus femoris origin), ischial tuberosity (hamstring origin), and lesser trochanter of the femur (iliopsoas insertion).
![]() Treatment and Prognosis
Treatment and Prognosis
Symptomatic care with a few days of rest followed by ambulation with crutches for about a month is recommended. It is usually 6–10 weeks before athletic activities may be resumed. Long-term athletic activity will probably not be affected. Open reduction and internal fixation have not shown superior results and therefore are usually not warranted. Abundant calcification may occur in the ischial tuberosity region and may be the cause of chronic bursitis and pain. Excision of the exuberant callous should cure this problem. Another indication for surgery is a painful fibrous nonunion, which also may be cured with excision of the fragment.
Sanders TG, Zlatkin MB: Avulsion injuries of the pelvis. Semin Musculoskelet Radiol 2008;12:42. [PMID: 18382943]
![]() SHOULDER INJURIES
SHOULDER INJURIES
The shoulder is the third most commonly injured joint during athletic activities, after the knee and the ankle. Sports-related injuries of the shoulder may result from a direct traumatic event or repetitive overuse. Any activity that requires arm motion, particularly overhead arm motion such as throwing, may stress the soft tissues surrounding the glenohumeral joint to the point of injury. The shoulder is the most mobile joint in the body partly, as a result of minimal containment of the large humeral head by the shallow and smaller glenoid fossa. The tradeoff for this mobility is less structural restraint to undesirable and potentially damaging movements. Thus, a fine balance must be struck to maintain full range of shoulder motion and normal glenohumeral joint stability.
![]() Anatomy
Anatomy
A. The Bony Articulation of the Glenohumeral Joint
The glenohumeral joint is a modified ball-and-socket joint. The glenoid fossa is a shallow inverted, comma-shaped, articular surface one fourth the size of the humeral head. The articular surface of the humeral head is retroverted approximately 30 degrees relative to the transverse axis of the elbow. Because the scapula is oriented anterolaterally about 30 degrees on the thorax, relative to the coronal plane of the body, the glenoid fossa matches the humeral head retroversion. So with the arm relaxed at the side, the glenoid fossa matches the humeral head retroversion. With shoulder motion, the scapula also moves so that the glenoid accommodates changing humeral head positions. As a result, the humeral head is centered in the glenoid throughout most shoulder motions. When this centered position is disturbed, instability may result.
B. The Clavicle and Its Articulations
The clavicle articulates medially with the sternum at the sternoclavicular joint and laterally with the acromion of the scapula at the acromioclavicular joint. The clavicle rotates on its long axis and acts as a strut, serving as the only bone connecting the appendicular upper extremity to the axial skeleton.
C. The Glenohumeral Joint Capsule, Ligaments, and Labrum
The thin redundant joint capsule has almost twice the surface area of the humeral head to allow large range of joint motion. Different regions of the joint capsule provide stability at different joint positions. With the arm at the side, the superior portion of the capsule is taut and the inferior portion is lax. With overhead elevation, this relationship reverses.
There are folds or thickenings visible on the inside of the capsule with the shoulder at the side, which have been termed glenohumeral ligaments. Traditionally the anterior capsule has been described as being composed of the superior, middle, and inferior glenohumeral ligaments (Figure 3–22). While use of the term “ligament” is generally accepted, it needs some clarification. Ligaments are soft-tissue structures that connect bones. They are most commonly band-like with parallel collagen fibers running between their insertion sites and have clearly defined edges, such as the MCL of the knee. The glenohumeral capsule as a whole may be considered a sheetlike ligament connecting the humerus and the scapula. The collagen fibers are not organized in a parallel fashion, the margins of the folds are indistinct, and functional study does not indicate it to have “band-like” properties. This may be the reason that the “ligaments” of the anterior capsule have been described with variable prevalence; different authors have had varying success in identifying them. Also, with the shoulder in abduction and external rotation, even the most consistently reported fold in the anteroinferior capsule, the anterior band of the inferior glenohumeral ligament, is often indistinct. While terminology may be currently causing confusion in anatomic, biomechanical, and clinical studies, there is little doubt that different regions of the capsule have differing roles in joint function, and this understanding has resulted in improved outcomes for shoulder injuries.
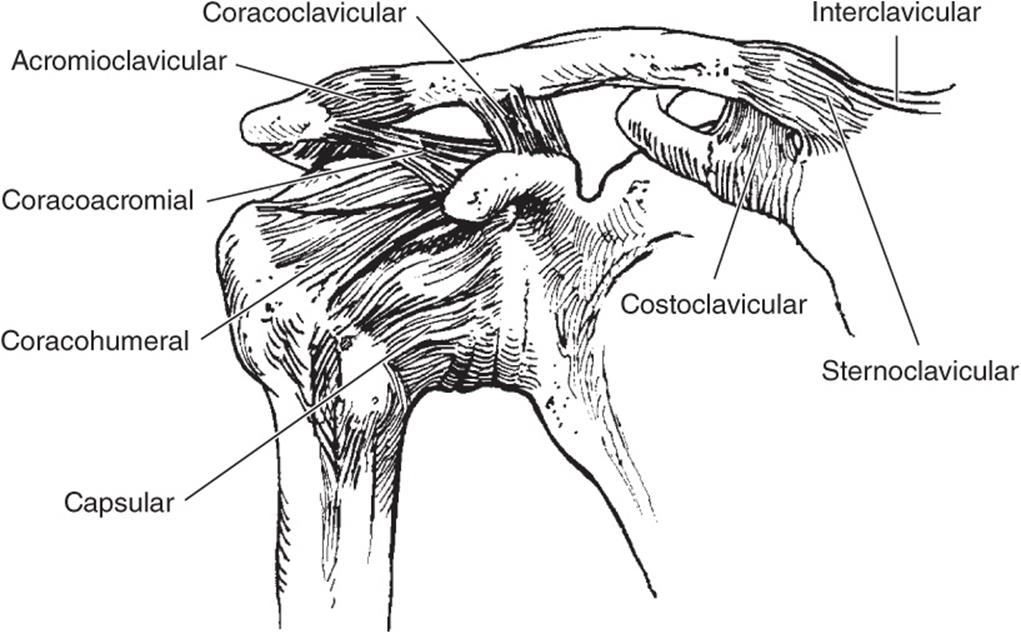
![]() Figure 3–22. Ligaments about the shoulder girdle.
Figure 3–22. Ligaments about the shoulder girdle.
The capsule inserts into the glenoid labrum and onto the glenoid bone. The glenoid labrum acts not only as an attachment site for the capsuloligamentous structures but also as an extension of the articular cavity. Its presence deepens the glenoid socket by nearly 50%, and the triangular cross-section of the labrum acts as a chock-block to help prevent subluxation.
D. The Shoulder Musculature
The muscles around the shoulder may be divided into three functional groups: glenohumeral, thoracohumeral, and those that cross both the shoulder and elbow.
1. Glenohumeral muscles—Four muscles compose the rotator cuff: the supraspinatus, subscapularis, infraspinatus, and teres minor. The supraspinatus has its origin on the posterosuperior scapula, superior to the scapular spine. It passes under the acromion, through the supraspinatus fossa, and inserts on the greater tuberosity with an extended attachment of fibrocartilage. The supraspinatus is active during the entire arc of scapular plane abduction; paralysis of the suprascapular nerve results in an approximately 50% loss of abduction torque. The infraspinatus and the teres minor muscles originate on the posterior scapula, inferior to the scapular spine, and insert on the posterior aspect of the greater tuberosity. Despite their origin below the scapular spine, their tendinous insertions are not separate from the supraspinatus tendon. These muscles function together to externally rotate and extend the humerus. Both account for approximately 80% of external rotation strength in the adducted position. The infraspinatus is more active with the arm at the side, while the teres minor activates mainly with the shoulder in 90 degrees of elevation. The subscapularis muscle arises from the anterior scapula and is the only muscle to insert on the lesser tuberosity. The subscapularis is the sole anterior component of the rotator cuff and functions to internally rotate and flex the humerus. The tendinous insertion of the subscapularis is continuous with the anterior capsule so that both provide anterior glenohumeral stability.
The deltoid is the largest of the glenohumeral muscles. It covers the proximal humerus on a path from its tripen-nate origin at the clavicle, acromion, and scapular spine to its insertion midway on the humerus at the deltoid tubercle. Abduction of the joint results from activity of the anterior and middle portions. The anterior portion is also a forward flexor. The posterior portion does not abduct the joint, but instead adducts and extends the humerus. The deltoid is active throughout the entire arc of glenohumeral abduction; paralysis of the deltoid results in a 50% loss of abduction torque. The deltoid muscle can fully abduct the glenohumeral joint with the supraspinatus muscle inactive.
The teres major muscle originates from the inferior angle of the scapula and inserts on the medial lip of the bicipital groove of the humerus, posterior to the insertion of the latissimus dorsi. The axillary nerve and the posterior humeral circumflex artery pass inferior to the subscapularis muscle and the inferior glenohumeral joint capsule, and then inferior to the teres minor muscle through the quadrilateral space also bordered by the teres major, the triceps, and the humerus. The teres major muscle contracts with the latissimus dorsi muscle, and the two muscles function as a unit in humeral extension, internal rotation, and adduction.
2. Thoracohumeral muscles—The pectoralis major and the latissimus dorsi muscles are powerful movers of the shoulder and, hence, contribute to the joint force that in turn usually stabilizes the glenohumeral joint. The pectoralis major muscle arises as a broad sheet of two distinct heads with the lowermost fibers of the sternal head inserting most proximally on the humerus.
Muscles that have origin on the thorax contribute to glenohumeral stability and may have roles in instability as well. When the shoulder is placed in horizontal abduction, similar to the apprehension position, the lowermost fibers of the sternal head of the pectoralis major muscle are stretched to an extreme. Because anterior instability also occurs from forcible horizontal abduction of the shoulder, the humeral head can be pulled out of the glenoid by passive tension in the pectoralis major and latissimus dorsi muscles.
3. Biceps brachii muscle—Both heads of the biceps brachii muscle have their origin on the scapula. The short head originates from the coracoid and, with the coracobrachialis muscle, forms the conjoined tendon. The long head of the biceps has its origin just superior to the articular margin of the glenoid from the posterosuperior labrum and the supraglenoid tubercle and is inside the synovial sheath of the glenohumeral joint. It traverses the glenohumeral joint, passing over the anterior aspect of the humeral head to the bicipital groove where it exits the joint under the transverse humeral ligament.
Its origin on the scapula and insertion of the radius leaves the long head of the biceps brachii muscle with potential for function at both the shoulder and the elbow. Its function at the elbow has been well established to include both flexion and supination. Long considered a depressor of the humeral head, the role of the active biceps has been recently questioned as electromyographic studies have shown that there was little or no activity of the biceps when elbow motion was controlled. This does not preclude a passive role or an active role associated with elbow motion, as tension in the tendon may then contribute to glenohumeral joint stability.
E. The Neurovascular Supply
The axillary artery traverses the axilla, extending from the outer border of the first rib to the lower border of the teres minor muscle, forming the brachial artery. The axillary artery lies deep to the pectoralis muscle, but is crossed in its midregion by the pectoralis minor tendon, just before the tendon inserts on the coracoid process. The axillary vein travels with the axillary artery, and branches of the axillary artery supply most of the shoulder girdle. The brachial plexus consists of the ventral rami of the fifth through eighth cervical nerves and the first thoracic nerve. This network of nerve fibers begins with the joining of the ventral rami proximally in the neck and continues anteriorly and distally, crossing into the axillary region obliquely underneath the clavicle at about the junction area of the distal one third and proximal two thirds. Clavicular fractures in this area have the potential of injuring the brachial plexus. The plexus then lies inferior to the coracoid process, where its cords form the peripheral nerves that continue down the arm. Muscles of the shoulder girdle are supplied by the nerves arising at all levels of the brachial plexus.
Moore SM, Stehle JH, Rainis EJ, McMahon PJ, Debski RE: The current anatomical description of the inferior glenohumeral ligament does not correlate with its functional role in positions of external rotation. J Orthop Res2008;26:1598. [PMID: 18524007]
Rispoli DM, Athwal GS, Sperling JW, Cofield RH: The anatomy of the deltoid insertion. J Shoulder Elbow Surg 2009;18:386. [PMID: 19186076]
![]() History and Physical Examination
History and Physical Examination
A. General Approach
The history of shoulder complaints must include age, arm dominance, location, intensity, duration, temporal occurrence, aggravating and alleviating factors, radiation of discomfort, physical activity level, occupation, and the mechanism of injury. Previous responses to treatments will help to characterize their efficacy and establish a pattern of disease or injury progression. The physical examination begins with the patient undressing so that both shoulders are fully exposed. Patients should be examined first in the standing position. The surface anatomy should be checked for asymmetry, atrophy, or external lesions. The supraspinatus and infraspinatus fossae are especially important to examine for atrophy. The area of pain should be pointed out by the patient prior to the physician manipulating the shoulder to avoid hurting the patient unnecessarily. A thorough neurovascular examination of the upper extremity should be performed.
B. Shoulder Range of Motion
1. Types of movement—Many terms may be used to describe movements of the shoulder joint (Figure 3–23). Flexion occurs when the arm begins at the side and elevates in the sagittal plane of the body anteriorly. Extension occurs when the arm starts at the side and elevates in the sagittal plane of the body posteriorly. Adduction occurs when the arm moves toward the midline of the body, with abduction occurring as the arm moves away from the midline of the body. Internal rotation occurs when the arm rotates medially, inward toward the body, and external rotation occurs as the arm rotates laterally or outward from the body. Horizontal adduction occurs as the arm starts at 90 degrees of abduction and adducts forward and medially toward the center of the body, and horizontal abduction happens as the arm starts at 90 degrees of abduction and moves outward, away from the body. Elevation is the angle made between the thorax and arm, regardless if it is in the abduction plane, flexion plane, or in between.

![]() Figure 3–23. Descriptions of shoulder motion.
Figure 3–23. Descriptions of shoulder motion.
2. Evaluation of movement—Rangeof motion of the injured shoulder should be compared with the opposite shoulder, along with the strength during abduction and rotation. This should be done both passively and actively. The shoulder should be inspected for any changes in synchrony, such as scapular winging, elevation of the scapula, muscle fasciculations indicating abnormal function, and any other irregular or asymmetric movements of the scapula. Information may be gained on loss of flexibility and instability resulting from muscle imbalance, fibrosis, and tendon, capsular, or ligament contractures. Loss of flexibility usually occurs in the capsular tissues of the glenohumeral joint. Sudden pain or clicking may indicate an intraarticular problem. Loss of motion in either internal or external rotation is suggestive of a chronic anterior or posterior dislocation, respectively.
3. Provocative tests—Specific tests are then performed that aid in making the correct diagnosis. The specific tests for instability, impingement syndrome, bicipital tendonitis, and superior capsulolabral/biceps anchor lesions are discussed as follows.
![]() Imaging and Other Studies
Imaging and Other Studies
Many varieties of radiologic views and projections are available to examine shoulder injuries. An initial radiographic evaluation of the shoulder should consist of an anteroposterior view of the glenohumeral joint in both internal and external rotation and an axillary lateral view. Additional plain radiographic views depend on the underlying pathology. MRI may be indicated in evaluation of rotator cuff disorders recalcitrant to conservative treatment. A magnetic resonance arthrogram may be useful in detecting labral pathology. Ultrasonography is also useful in diagnosis of rotator cuff tendon injury, but it is operator dependent. Electromyographic examination can be useful in identifying shoulder pain of cervical origin.
![]() Arthroscopic Evaluation
Arthroscopic Evaluation
A. Indications for Arthroscopic Evaluation of Shoulder Injuries
Indications for arthroscopic examination of the shoulder include the following:
1. Impingement syndrome, including subacromial bursitis, rotator cuff tendonitis, and rotator cuff tears
2. Acromioclavicular joint osteoarthritis
3. Loose bodies
4. Chronic synovitis
5. Glenohumeral instability
6. Superior capsulolabral/biceps anchor (ie, SLAP) lesions
7. Adhesive capsulitis (frozen shoulder)
B. Technique
With the patient either in the lateral decubitus or the beach chair position, the arthroscope is inserted into the posterior shoulder. With visualization of the glenohumeral joint, an anterior portal immediately lateral to the coracoid allows additional inflow and entrance of additional instruments. Additional portals may be used; for example, an additional anterior portal inferior to the first may be used for instability repair. The arthroscope is then removed from the joint and placed into the subacromial bursa. A portal lateral to the acromion allows subacromial decompression and rotator cuff repair.
C. Steps in Evaluation
Examination of shoulder range of motion and stability with the patient under anesthesia is helpful in the diagnosis and treatment of shoulder injuries. This should be performed in the operating room prior to arthroscopy. The steps in arthroscopic examination should then include the following:
1. Glenohumeral articular surfaces
2. Rotator cuff from inside the joint
3. Labrum including the biceps anchor
4. Anterior capsule
5. Rotator cuff from the subacromial bursal space
6. Coracoacromial ligament
7. Acromion
8. Acromioclavicular joint
Ludewig PM, Phadke V, Braman JP, Hassett DR, Cieminski CJ, LaPrade RF: Motion of the shoulder complex during multi-planar humeral elevation. J Bone Joint Surg Am 2009;91:378. [PMID: 19181982]
Saupe N, Zanetti M, Pfirrmann CW, Wels T, Schwenke C, Hodler J: Pain and other side effects after MR arthrography: prospective evaluation in 1085 patients. Radiology 2009;250:830. [PMID: 19164115]
Vlychou M, Dailiana Z, Fotiadou A, Papanagiotou M, Fezoulidis IV, Malizos K: Symptomatic partial rotator cuff tears: diagnostic performance of ultrasound and magnetic resonance imaging with surgical correlation. Acta Radiol2009;50:101. [PMID: 19052931]
SHOULDER TENDON AND MUSCLE INJURY
![]() Rotator Cuff Tendon Injuries
Rotator Cuff Tendon Injuries
Injury to the rotator cuff is the most common cause of shoulder pain and disability. Although shoulder weakness and decreased range of motion are associated with a rotator cuff tendon tear, pain from subacromial bursitis or rotator cuff tendinosis may also be the cause. Symptoms are often worsened by activity, especially with overhead activity. Night pain is also common, and many patients complain of awakening after rolling onto the affected shoulder.
Any prolonged repetitive activity involving overhead motion such as tennis, pitching, golf, or swimming may cause rotator cuff injury. Injury, whether repetitive or acute, can produce a continuous vicious cycle (Figure 3–24). Blood supply to this tendon is precarious, thus decreasing its capacity for healing.

![]() Figure 3–24. The cycle of injury and reinjury resulting from rotator cuff impingement.
Figure 3–24. The cycle of injury and reinjury resulting from rotator cuff impingement.
1. Subacromial Bursitis and Rotator Cuff Tendinosis
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Mild or moderate pain with overhead shoulder motion.
• Occasional night pain.
• History of repetitive overhead activity.
• No muscle atrophy.
• No weakness or mild weakness from the shoulder pain.
• Pain is relieved with a subacromial lidocaine injection.
![]() Prevention
Prevention
Limiting repetitive overhead activities and maintenance of good rotator cuff strength are keys to prevention. Additionally, overall conditioning and stretching and strengthening with careful attention paid to technique can be helpful in minimizing many injuries resulting from overuse.
![]() Clinical Findings
Clinical Findings
Bursitis of the shoulder refers to an inflammation of the subacromial bursa. It may or may not be present along with rotator cuff tendinosis, and the two entities are similar. Pain is present with activity involving overhead motion, and there is usually no pain or only mild pain with the arm at the side.
Of the four rotator cuff muscles, the supraspinatus tendon is most often initially involved. Rotator cuff tendinosis also results from impingement syndrome and is characterized by pain with activity involving overhead motion. The patient may occasionally be awakened by pain at night.
Active range of shoulder motion may be limited by pain. No atrophy of the shoulder muscles is present, and manual muscle testing demonstrates mild weakness. Passively, when the internally rotated shoulder is moved into forward flexion, the patient will experience discomfort. This is called the Neer impingement sign (Figure 3–25). This pain then resolves, and there is a dramatic increase in strength and range of motion with the Neer impingement test (10 mL of lidocaine is injected into the subacromial space).

![]() Figure 3–25. Evaluating for impingement of the supraspinatus tendon with the “empty can” test.
Figure 3–25. Evaluating for impingement of the supraspinatus tendon with the “empty can” test.
Radiographic views of the subacromial space such as the supraspinatus outlet view may show a spur on the undersur-face of the acromion, causing narrowing of the subacromial space. In recent years, advances in imaging methods such as ultrasonography and MRI have aided in the diagnosis of subacromial bursitis, rotator cuff tendinosis, and rotator cuff tendon tear (Figure 3–26).

![]() Figure 3–26. MRI demonstrating (A) normal shoulder anatomy and (B) cystic changes at the greater tuberosity with rotator cuff tear (arrow).
Figure 3–26. MRI demonstrating (A) normal shoulder anatomy and (B) cystic changes at the greater tuberosity with rotator cuff tear (arrow).
![]() Treatment
Treatment
Treatment starts with conservative measures such as activity modification, physical therapy, and oral nonsteroidal anti-inflammatory drugs (NSAIDs). Only with normal function of the rotator cuff tendons will glenohumeral mechanics be improved and the impingement syndrome cease. If this treatment fails, a subacromial injection of corticosteroids may be helpful. Most patients respond well to these nonoperative treatments.
Surgical intervention is indicated if symptoms do not resolve after a few months of such treatment. Then, acromioplasty, also called subacromial decompression, which is shaving the under-surface of the acromion, usually results in relief of symptoms. An exception is the young athlete with glenohumeral instability and secondary tendinosis. In this case, the instability should be treated first, and the rotator cuff tendinosis will then resolve. This procedure can be done arthroscopically to decrease postoperative discomfort and minimize the complication of deltoid muscle rupture from the acromion. Those who require surgical treatment are usually able to return to pain-free activities.
2. Rotator Cuff Tendon Tear
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Moderate or severe pain with activity involving overhead motion.
• Persistent night pain.
• History of repetitive overhead activity.
• Weakness with moderate and severe tears.
• Atrophy of rotator cuff muscles with severe tears.
• Pain is relieved with a subacromial lidocaine injection.
![]() Prevention
Prevention
Maintenance of overall body conditioning with regular stretching and strengthening of the rotator cuff and scapular stabilizing muscles can help prevent rotator cuff injuries.
![]() Clinical Findings
Clinical Findings
A rotator cuff tendon tear is characterized by pain with activity involving overhead motion. However, the patient is often awakened at night with pain as well. The athlete with a chronic rotator cuff tear may experience a gradual loss of strength. Pain may be persistent, occurring even when the arm is at the side. Active range of shoulder motion is limited, and if the tear is severe, there will be atrophy of the shoulder muscles. Manual muscle testing demonstrates weakness. The Neer impingement sign is positive, and the pain resolves with a subacromial injection of lidocaine. Radiographic evaluation is similar to that for subacromial bursitis and rotator cuff tendinosis.
![]() Treatment
Treatment
Radiographic evaluation and treatment are similar to subacromial bursitis management. Unlike acute tears, chronic rotator cuff tears often present insidiously, with slow progression from subacromial bursitis to rotator cuff tendinosis and eventual tendon tear. Differentiating severe rotator cuff tendinosis from partial or small full-thickness chronic rotator cuff tears may be difficult.
There are two important considerations in treating an individual with a rotator cuff tear—the current symptoms and the risk of the tear progressing. Although the lesion location and size are helpful in describing a rotator cuff tear, symptoms do not correlate with these factors alone. Some individuals are able to cope with the symptoms of a rotator cuff tear, and some may be completely asymptomatic. The severity of symptoms is influenced by a number of other factors including pain tolerance, the acute or chronic nature of the injury, the age and activity level of the individual, humeral head superior migration, shoulder muscle strength, muscle atrophy, fatty changes in the muscle, arthritis, and workman’s compensation status.
Rest, rehabilitation, and taking NSAIDs, sometimes for as long as 4–9 months, may relieve symptoms. Range-of-motion and strengthening exercises are recommended, unless they cause significant discomfort. Strengthening the other shoulder muscles may increase the individual’s ability to cope with the rotator cuff tear. Avoidance of activities that exacerbate the symptoms, such as activities involving overhead motion, is also recommended. Symptoms of pain, weakness, or decreased range of motion that persist after a nonoperative treatment program has been tried indicate the need for surgical intervention.
Because rotator cuff tears may progress in size over time, immediate repair may be warranted in some at-risk individuals. Both epidemiologic and imaging studies of the general population indicate a high incidence of partial-thickness rotator cuff tears at younger ages and full-thickness rotator cuff tears at older ages. The increasing prevalence of rotator cuff injuries in older individuals may be the best evidence that rotator cuff tears progress in severity. Specifically, about 25% of individuals over 60 years of age have a tear, and in those over 80 years of age, there is a full-thickness rotator cuff tear in about 50% of individuals. The risk of a rotator cuff tear progressing to a more severe tear cannot currently be predicted, but it is thought to be higher in young, active individuals, partly because they have many more years to sustain an injury.
The thin degenerated tissue of a chronic rotator cuff tear makes surgical repair more difficult than repair of an acute tear. The repair can be accomplished with either an arthroscopic or an open technique. For years, surgical decompression has been routine with rotator cuff repair, but recently some have questioned whether it needs to be done. The arthroscopic technique yields results comparable to the open technique. There have been many recent changes to the arthroscopic technique such as using a double row of fixation rather than a single row and using novel methods of securing the sutures.
Some severe tears may be impossible to repair. This includes many of the tears that are large to massive in size or that involve two or more rotator cuff tendons; when the humeral head is positioned superior, against the acromion; and when there is meaningful atrophy of the rotator cuff muscles or fatty changes. Debridement of the rotator cuff and the subacromial spurs may diminish pain in such instances.
Rehabilitation after a repair lasts from 3 months to 1 year with gradual exercise progression needed to restore normal, or near-normal, function and strength. This varies with the size of the tear that was repaired and the type of surgery performed. Typically, immediately after the procedure, passive motion and isometric strengthening exercises start, along with elbow-, hand-, and grip-strengthening exercises. At 6 weeks, the athlete may be able to begin low-intensity active strengthening exercises against gravity. The goals are to bring the athlete to normal strength with a functional, pain-free range of motion.
![]() Prognosis
Prognosis
The prognosis following a rotator cuff tear depends on many factors, as described earlier. There are few specific criteria governing return to sports following rotator cuff injuries. Determining factors must be individualized to the athlete, considering the nature and treatment of the rotator cuff injury as well as the desired sport. Patients must be pain free and have attained full range of motion with near full strength prior to returning to their sport to minimize reinjury.
3. Partial-Thickness Rotator Cuff Tendon Tear
A partial articular-sided tendon avulsion is much more common than a bursal side tear of the rotator cuff. As with other rotator cuff injuries, symptoms may resolve with appropriate physical therapy and analgesics. Yet, some individuals with a partial-thickness tear have persistent or recurrent symptoms. If a conservative program of exercises and gradual return to activity do not lead to steady improvement, then further diagnostic evaluation with ultrasonography, MRI, or arthros-copy may be helpful. Whereas repair of the partial-thickness rotator cuff tear may be best in some, debridement of the abnormal cuff may diminish or relieve symptoms in others. Some clinicians use involvement of greater than 50% of the tendon thickness as an indication for repair. Repair necessitates a rehabilitation program similar to that described earlier for full-thickness rotator cuff tears. Following debridement, immediate resumption of range-of-motion and muscle-strengthening exercises begins. Typically, it requires 6–12 months for an athlete whose sport involves throwing to return to athletics following arthroscopic debridement of a partial-thickness rotator cuff tear.
4. Cuff “Arthropathy”
Severe rotator cuff tears may lead to the humeral head being positioned superior against the undersurface of the acromion. Because of the tear of the rotator cuff, the humeral head no longer stays centered on the glenoid and the pull of the deltoid is unopposed. Over time, the humeral head and the glenoid wear from the abnormal contact. Most individuals have severe shoulder dysfunction, sometimes described as pseudoparalysis. They are able to move their shoulder only the smallest amounts. Interestingly, others are able to lift their arms and complain only of pain and weakness. This is a difficulty in caring for rotator cuff injuries. Symptoms may not correlate with the severity; some are able to cope with the injury, while others cannot. Nonoperative treatment including rehabilitation, NSAIDs, and steroid injection is usually effective in diminishing symptoms. In the elderly, shoulder hemiarthroplasty diminishes pain but improves function mildly. More pain relief and improvement in function is possible with a shoulder replacement with reverse ball-and-socket prosthesis. But complications such as loosening are more prevalent, so it is reserved for the elderly.
Feeley BT, Gallo RA, Craig EV: Cuff tear arthropathy: current trends in diagnosis and surgical management. J Shoulder Elbow Surg 2009;18:484. [PMID: 19208484]
Levy O, Venkateswaran B, Even T, Ravenscroft M, Copeland S: Mid-term clinical and sonographic outcome of arthroscopic repair of the rotator cuff. J Bone Joint Surg Br 2008;90:1341. [PMID: 18827245]
Mall NA, Kim HM, Keener JD, et al: Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am 2010;92:2623. [PMID: 21084574]
Matsen FA 3rd: Open rotator cuff repair without acromioplasty. J Bone Joint Surg Am 2009;91:487. [PMID: 19182000]
Pennington WT, Gibbons DJ, Bartz BA, et al: Comparative analysis of single-row versus double-row repair of rotator cuff tears. Arthroscopy 2010;26:1419. [PMID: 20875720]
Zumstein MA, Jost B, Hempel J, Hodler J, Gerber C: The clinical and structural long-term results of open repair of massive tears of the rotator cuff. J Bone Joint Surg Am 2008;90:2423. [PMID: 18978411]
![]() CPT Codes for Rotator Cuff Injuries
CPT Codes for Rotator Cuff Injuries
23130 Acromioplasty or acromionectomy, partial, with or without coracoacromial ligament release
23410 Repair of ruptured musculotendinous cuff (eg, rotator cuff) open; acute
23412 Reconstruction of complete shoulder (rotator) cuff avulsion, chronic (includes acromioplasty)
23415 Coracoacromial ligament release, with or without acromioplasty
23420 Reconstruction of complete shoulder (rotator) cuff avulsion, chronic (includes acromioplasty)
28926 Arthroscopy, shoulder, surgical; decompression of subacromial space with partial acromioplasty, with or without coracoacromial release
29827 Arthroscopy, shoulder, surgical; with rotator cuff repair
GLENOHUMERAL JOINT INSTABILITY
Distinguishing between shoulder laxity and instability is difficult, both because of the wide variability of normal joint laxity and the absence of biomechanical studies defining laxity and instability. In contrast, other joints such as the knee have precise definitions of instability. A Lachman test revealing greater than 5 mm of translation compared to the contralateral side is widely accepted as being associated with an ACL rupture and resulting knee instability. At the shoulder, instability is often defined as translations that result in symptoms. This poor definition of shoulder instability hampers proper diagnosis and intra- and interobserver classification and makes comparison of research studies difficult. Future study of normal and abnormal joint kinematics is needed to aid clinicians in diagnosis and classification of shoulder instability.
To make the correct diagnosis, the glenohumeral joint must be tested for anterior, posterior, and inferior instability. Different classifications of glenohumeral joint instability have been proposed, based on various characteristics. TUBS and AMBRI have been used to distinguish the two main types of instability. TUBS is an acronym describing instability caused by a traumatic event, which is unidirectional, is associated with a Bankart lesion, and often requires surgical treatment. AMBRI refers to atraumatic, multidirectional instability that may be bilateral and is best treated by rehabilitation. In this classification, the etiology of multidirectional instability is thought to be enlargement of the capsule from either a genetic or microtraumatic origin. TUBS and AMBRI are of historic interest as clinicians have recognized more types of instability.
FEDS is an acronym describing the four most important characteristics of shoulder instability (frequency, etiology, direction, and severity) and can be obtained by history and physical examination. Frequency is classified as solitary, occasional (two to five episodes) or frequent (more than five episodes). Etiology is classified as traumatic or atraumatic. Direction is classified as anterior, inferior, or posterior. Lastly, severity is classified as subluxation or dislocation. Easy to remember, a weakness of the FEDS classification is that it does not distinguish unidirectional from multidirectional instability.
The positive sulcus sign has been used as the diagnostic hallmark for multidirectional instability, but we now know that the sulcus sign is sometimes found in shoulders of asymptomatic individuals with increased laxity. Laxity or joint play is a trait of body constitution that differs from one individual to another. Individuals may be loose or tight jointed. A shoulder is hyperlax if the examiner can easily subluxate the humeral head out of the glenoid in the anterior, posterior, and inferior directions without eliciting symptoms. Unfortunately, this makes classification of instability based on etiology, or direction alone, extremely difficult. Instead, classification is best based on the direction of instability that elicits symptoms and the presence or absence of hyperlaxity (Table 3–2). Multidirectional instability is defined as instability in both the anterior and posterior directions, which is most often subluxation rather than dislocation.
Table 3–2. Classification of glenohumeral instability based on the direction of instability and the presence or absence of hyperlaxity.
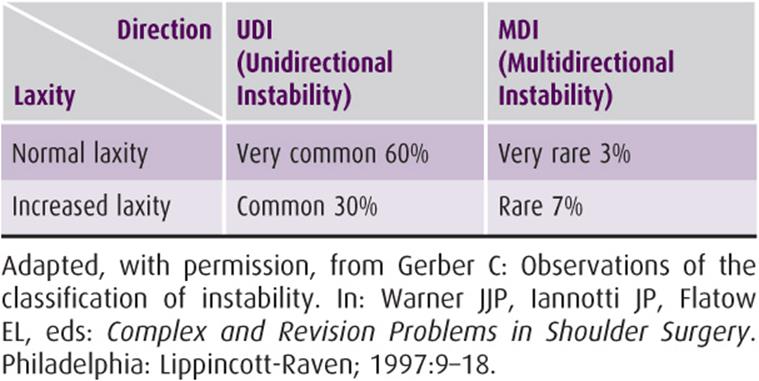
Often there is a tear of the glenoid labrum, the fibrocartilaginous rim around the glenoid fossa that deepens the socket and provides stability for the humeral head. It also is a connection for the surrounding capsuloligamentous structures. Glenoid labrum tears may occur from repetitive shoulder motion or acute trauma. In the athlete with repeated anterior subluxation of the shoulder, tears of the anteroinferior labrum may occur and lead to progressive instability. Patients with glenoid labrum injuries may describe their pain as interrupting smooth functioning of the shoulder during their specific activity. A labrum tear may be felt as a “pop” or “click” with motion of the shoulder. MRI arthrogram is useful for detection of these lesions.
Kuhn JE: A new classification system for shoulder instability. Br J Sports Med 2010;44:341. [PMID: 20371559]
![]() Glenohumeral Joint Instability Evaluation
Glenohumeral Joint Instability Evaluation
A. Anterior Instability
The apprehension test is performed to assess anterior instability. The test applies an anterior-directed force to the humeral head from the back with the arm in abduction and external rotation (Figure 3–27). A positive test results from the patient’s apprehension that the joint will dislocate. This maneuver mimics the position of subluxation, or dislocation, and causes reflex guarding. Conversely, the relocation test is positive if relief is obtained by applying a posterior-directed force to the humeral head (Figure 3–28).

![]() Figure 3–27. The apprehension test for anterior instability.
Figure 3–27. The apprehension test for anterior instability.

![]() Figure 3–28. The relocation test is positive if relief is obtained by applying a posterior-directed force to the humeral head.
Figure 3–28. The relocation test is positive if relief is obtained by applying a posterior-directed force to the humeral head.
B. Posterior Instability
No single test has high sensitivity and specificity for posterior instability. There is no posterior apprehension test, similar as that for anterior instability, that is helpful. The Jahnke test, sometimes called the jerk test, is performed by applying a posteriorly directed force to the forward flexed and internally rotated shoulder. The shoulder is then moved in horizontal abduction, into the coronal plane, as an anterior-directed force is applied to the humeral head. A clunk occurs as the humeral head reduces from the subluxed position (Figure 3–29). To perform the circumduction test, the patient is instructed to actively move the shoulder in a large circle starting from a flexed, internally rotated, and cross-body position, then to forward flexion, then to an abducted and externally rotated position, and lastly to the arm at the side. The examiner stands behind the patient and palpates the posterior shoulder. If positive, the joint subluxes in the flexed, internally rotated, and cross-body position and reduces as the shoulder is moved. In the Kim test, with the patient in a sitting position and the arm at 90 degrees of abduction, the examiner holds the elbow and lateral aspect of the proximal arm, and a strong axial loading force is applied. Then, while the arm is further elevated 45 degrees diagonally upward, downward and backward force is applied to the proximal arm. A sudden onset of posterior shoulder pain indicates a positive test result, regardless of accompanying posterior clunk of the humeral head.

![]() Figure 3–29. The Jahnke test for posterior instability. A: A posterior-directed force applied to the forward flexed shoulder (in the upper left column). B: The shoulder is then moved into the coronal plane as an anterior-directed force is applied to the humeral head (in the lower left column). A clunk occurs as the humeral head reduces from the subluxed position. (Reprinted, with permission, from Hawkins RJ, Bokor DJ: Clinical evaluation of shoulder problems. In: Rockwood CA, Matsen F III, eds: The Shoulder. New York: WB Saunders; 1998, p. 186.)
Figure 3–29. The Jahnke test for posterior instability. A: A posterior-directed force applied to the forward flexed shoulder (in the upper left column). B: The shoulder is then moved into the coronal plane as an anterior-directed force is applied to the humeral head (in the lower left column). A clunk occurs as the humeral head reduces from the subluxed position. (Reprinted, with permission, from Hawkins RJ, Bokor DJ: Clinical evaluation of shoulder problems. In: Rockwood CA, Matsen F III, eds: The Shoulder. New York: WB Saunders; 1998, p. 186.)
C. Inferior Instability
The sulcus sign is used to evaluate laxity and inferior instability. The test is performed with the athlete in a sitting position with the arm at the side. A distraction force is applied longitudinally along the humerus. If positive, discomfort or apprehension of instability is experienced as the skin just distal to the lateral acromion hollows out (Figure 3–30).

![]() Figure 3–30. The sulcus test for inferior instability. (Reprinted, with permission, from Hawkins RJ, Bokor DJ: Clinical evaluation of shoulder problems. In: Rockwood CA, Matsen F III, eds: The Shoulder. New York: WB Saunders; 1998, p. 189.)
Figure 3–30. The sulcus test for inferior instability. (Reprinted, with permission, from Hawkins RJ, Bokor DJ: Clinical evaluation of shoulder problems. In: Rockwood CA, Matsen F III, eds: The Shoulder. New York: WB Saunders; 1998, p. 189.)
GLENOHUMERAL DISLOCATION
When the shoulder is forced beyond the limit of its normal range of motion, the articular surface of the humeral head may be displaced from the glenoid to varying degrees. The majority of glenohumeral dislocations, or subluxations, are in the anteroinferior direction.
1. Anterior Dislocation
![]() Essentials of Diagnosis
Essentials of Diagnosis
• The arm is typically held supported at the side.
• A visible dimple may be seen under the acromion due to the absence of the humeral head.
• The humeral head may be palpable under the coracoid or in the axilla.
• Range of motion will be extremely painful and limited.
• Appropriate radiographs will confirm the direction of dislocation and possible associated injuries.
![]() Prevention
Prevention
Shoulder dislocations are typically the result of an acute traumatic injury. Therefore, although avoiding injury to the shoulder is the best form of prevention, minimizing the risk of dislocation following a blow can be achieved with regular stretching and strengthening of the rotator cuff musculature.
![]() Clinical Findings
Clinical Findings
Anterior glenohumeral dislocation occurs from either exceeding the normal range of motion in external rotation and/or abduction or a direct posterior or posterolateral blow on the shoulder strong enough to displace the humeral head. The anterior capsule is either stretched or torn within its attachment to the anterior glenoid. The head may be displaced into a subcoracoid, subglenoid, or rarely subclavicular or intrathoracic position. Two major lesions are typically seen in patients with recurrent anterior dislocations (Figure 3–31). First is the Bankart lesion, an anterior capsular injury associated with a tear of the glenoid labrum off the anterior glenoid rim. The Bankart lesion may occur with fractures of the glenoid rim. Such fractures are often minimally displaced, and treatment is usually dictated by the joint instability. The second major lesion associated with recurrent anterior dislocations is the Hill-Sachs lesion, a compression fracture of the posterolateral articular surface of the humeral head. It is created by the sharp edge of the anterior glenoid as the humeral head dislocates over it. When large, both the Bankart and the Hill-Sachs lesions predispose to recurrent dislocations when the arm is placed in abduction and external rotation. If the glenoid rim fracture involves more than 20% of the glenoid diameter, then the joint becomes prone to instability, and treatment with open reduction and internal fixation is best. If the fracture is old, or the glenoid rim is worn to a similar level, then corticocancellous bone grafting of the glenoid rim is indicated.

![]() Figure 3–31. Anatomic lesions producing shoulder instability.
Figure 3–31. Anatomic lesions producing shoulder instability.
Other injuries associated with anterior dislocation may occur. These include avulsion of the greater tuberosity from the humerus, caused by traction from the rotator cuff, and injury to the axillary nerve, which may be stretched or torn. Loss of axillary nerve function results in denervation of the deltoid and teres minor muscles and loss of sensation over the proximal lateral aspect of the arm. Axillary nerve palsy may also occur during reduction of the dislocation and therefore should be tested both before and after reduction. The deltoid extension lag sign, described in the section on axillary nerve injury, may be the best way to assess function of this nerve. Lastly, the dead arm syndrome may occur after anterior joint instability. For example, a pitcher may report a sudden inability to throw, with the arm going numb and becoming extremely weak after ball release. The symptoms are transient, resolving within a few seconds to minutes.
Athletes who sustain a shoulder dislocation will try to hold the injured extremity at their side, gripping the forearm with the opposite hand. Most athletes know their shoulder is dislocated and will immediately seek help. On physical examination of an anterior dislocation, the examiner will note a space underneath the acromion where the humeral head should lie and a palpable anterior mass representing the humeral head in the anterior axilla.
![]() Treatment
Treatment
Acute and recurrent anterior glenohumeral dislocations must be distinguished, as an acute dislocation sustains severe trauma with the increased probability of associated injuries. The recurrent dislocation may occur with minimal trauma, and reduction may be accomplished with much less effort. Anterior dislocations may be reduced by one of several techniques. Longitudinal traction may be exerted on the affected arm with external rotation, followed by internal rotation of the arm. Care must be taken to avoid direct pressure on the neurovascular structures. Another method is to have the patient lie face down on the table and tie or tape a bucket to the injured arm and slowly fill it with water. This allows the musculature around the shoulder to relax from the force of the weight and effect a spontaneous reduction.
Following reduction of an initial dislocation, the shoulder should be immobilized in internal rotation for 2–6 weeks. Healing will generally take at least 6 weeks. Before returning to athletics, the patient should have normal range of motion without pain and normal strength in the shoulder. Emphasis must be placed on strengthening the rotator cuff muscles to compensate for the laxity of the ligamentous support. When weight training is begun, military press, fly exercises, a narrow grip while bench pressing, and deep shoulder dips must be excluded until considerable time has elapsed and healing is complete.
Recurrent dislocations should be treated with minimal immobilization until the pain subsides, followed by range-of-motion and muscle-strengthening exercises. Many restraining devices are available to help prevent recurrent dislocations during sporting activities, focusing on keeping the arm from going into abduction and external rotation. These orthotics may be effective, but because they limit the athlete’s range of shoulder motion, their use is limited for certain competitive activities.
If an athlete has sustained multiple dislocations and is unresponsive to conservative treatment, surgical reconstruction of the shoulder joint may be indicated. There is a wide variety of repair procedures with either open or arthroscopic techniques. Repair of the labral lesion (ie, Bankart repair) and tightening of the anteroinferior capsule are integral to all of these procedures (Table 3–3), and success is frequent. When present, large Hill-Sachs and bony Bankart lesions should be treated as well.
Table 3–3. Repair of capsule and labrum back to the glenoid rim.

For most surgical procedures, aggressive range-of-motion exercises do not start until at least 3 weeks postoperatively. The goal is to have full abduction and 90 degrees of external rotation. By 12 weeks, patients have often progressed well into their initial programs and may begin a variety of weight-training exercises, avoiding exercises that strain the anterior capsule.
![]() Prognosis
Prognosis
Young patients are at a high risk for redislocation after a primary traumatic anterior shoulder dislocation if treated conservatively with rehabilitation. Surgical stabilization should be considered in these cases. In general, despite surgical stabilization, patients have up to a 10% chance of redislocation if returning to play in contact sports.
2. Posterior Dislocation
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Posterior dislocations are more difficult to diagnose than anterior dislocations.
• The arm is typically held in internal rotation and is not able to be externally rotated.
• Appropriate radiographs will confirm the direction of dislocation and possible associated injuries.
![]() Prevention
Prevention
Shoulder dislocations are typically the result of an acute traumatic injury. Therefore, while avoiding injury to the shoulder is the best form of prevention, the risk of dislocation following a blow can be minimized with regular stretching and strengthening of the rotator cuff musculature.
![]() Clinical Findings
Clinical Findings
Posterior glenohumeral dislocations result from the posterior capsule being torn, stretched, or disrupted from the posterior glenoid. A reverse Hill-Sachs lesion (see Figure 3–31) may appear on the anterior articular surface of the humerus. With a posterior dislocation, the subscapularis, or its insertion on the lesser tuberosity, may be injured. Posterior dislocations are often difficult to diagnose, as the patient may have a normal contour to the shoulder or the deltoid of a well-developed athlete may mask signs of a displaced humeral head. The patient holds the injured shoulder in internal rotation and the examiner cannot externally rotate it. Anteroposterior and axillary radiographs must be obtained to diagnose a posterior dislocation.
![]() Treatment
Treatment
Applying traction in the line of the adducted humerus, with an anterior directed force to the humeral head, reduces a posterior dislocation. Anesthesia often helps decrease the trauma of reduction. Following reduction, the shoulder is immobilized for 2–6 weeks in external rotation and a small amount of abduction. Surgical treatment should be considered if these measures fail to provide the desired results.
![]() Prognosis
Prognosis
Patients with an acute posterior dislocation are often able to return to their sport following a course of rehabilitation emphasizing range-of-motion and rotator cuff strengthening.
3. Multidirectional Instability
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Multidirectional instability is often difficult to diagnose.
• Shoulder pain is not specific to overhead activities or associated with other shoulder injuries.
• Fatigue or paresthesias may be present.
• Evidence of both anterior and posterior instability with the history and physical examination.
• Positive sulcus sign.
• Must be evaluated for hyperlaxity that must then be distinguished from instability.
![]() Clinical Findings
Clinical Findings
Some patients will have instability in both the anterior and posterior directions, which is most often subluxation rather than dislocation. This may result in a painful shoulder, especially if rotator cuff strength decreases. The pain is often primarily a result of rotator cuff inflammation, likely from attempts to stabilize the humeral head during activity. Patients may complain of vague symptoms, including upper extremity fatigue, discomfort, pain, apprehension, and paresthesias. They may describe frank episodes of instability. Physical examination should include evaluation for signs of generalized hyperlaxity, which include hyperextension of the metacarpophalangeal joints, elbows, and knees and the ability to adduct the thumb to the ipsilateral wrist. Generalized hyperlaxity does not necessarily indicate symptomatic instability of the shoulder. The shoulder examination should include tests for anterior, posterior, and inferior instability as described earlier. MRI can be a useful adjunct to plain radiographs and may reveal an enlarged axillary pouch and labral or rotator cuff pathology.
![]() Treatment and Prognosis
Treatment and Prognosis
Initial treatment for multidirectional instability is nonoperative and leads to successful results in the vast majority of cases. This includes patient education, modification of activity, and a strengthening program for the rotator cuff and scapular stabilizing muscles. When this fails, surgery is often effective in relieving symptoms. Because of differences in classifying multidirectional instability, results of treatment vary as well. For those with multidirectional instability classified as instability in both the anterior and posterior directions, about two thirds of patients, less than that with unidirectional anterior instability, will have relief after surgery.
Bahu MJ, Trentacosta N, Vorys GC, Covey AS, Ahmad CS: Multidirectional instability: evaluation and treatment options. Clin Sports Med 2008;27:671. [PMID: 19064150]
Barchilon VS, Kotz E, Barchilon Ben-Av M, Glazer E, Nyska M: A simple method for quantitative evaluation of the missing area of the anterior glenoid in anterior instability of the glenohumeral joint. Skeletal Radiol2008;37:731. [PMID: 18523766]
Bartl C, Schumann K, Vogt S, Paul J, Imhoff AB: Arthroscopic capsulolabral revision repair for recurrent anterior shoulder instability. Am J Sports Med. 2011;39:511. [PMID: 21212311]
Bradley JP, Forsythe B, Mascarenhas R: Arthroscopic management of posterior shoulder instability: diagnosis, indications, and technique. Clin Sports Med 2008;27:649. [PMID: 19064149]
DiPaola MJ, Jazrawi LM, Rokito AS, et al: Management of humeral and glenoid bone loss—associated with glenohumeral instability. Bull NYU Hosp Jt Dis 2010;68:245. [PMID: 21162700]
Hovelius L, Olofsson A, Sandström B, et al: Nonoperative treatment of primary anterior shoulder dislocation in patients forty years of age and younger. A prospective twenty-five-year follow-up. J Bone Joint Surg Am2008;90:945. [PMID: 18451384]
Purchase RJ, Wolf EM, Hobgood ER, Pollock ME, Smalley CC: Hill-Sachs “remplissage”: an arthroscopic solution for the engaging Hill-Sachs lesion. Arthroscopy 2008;24:723. [PMID: 18514117]
![]() CPT Codes for Shoulder Instability
CPT Codes for Shoulder Instability
23650 Closed treatment of shoulder dislocation, with manipulation; without anesthesia
23655 Closed treatment of shoulder dislocation, with manipulation; requiring anesthesia
23600 Open treatment of acute shoulder dislocation
23450 Capsulorrhaphy, anterior; Putti-Platt procedure or Magnuson type operation
23455 Capsulorrhaphy, anterior; with labral repair (eg, Bankart procedure)
23460 Capsulorrhaphy, anterior, any type; with bone block
23462 Capsulorrhaphy, anterior, any type; with coracoid process transfer
23465 Capsulorrhaphy, glenohumeral joint, posterior, with or without bone block
23466 Capsulorrhaphy, glenohumeral joint, any type multidirectional instability
29806 Arthroscopy, shoulder, surgical; capsulorrhaphy
SLAP LESIONS
SLAP lesions involve the origin of the long head of the biceps brachii (biceps anchor) and the superior capsulolabral structures. The acronym SLAP is for superior labrum anterior and posterior. A type I lesion is fraying of the labrum. Type II lesions are most common, accounting for over 50% of patients with a SLAP lesion, and are detachment of the superior labrum from the glenoid. A type III lesion is a bucket-handle tear of the superior labrum with firm attachment of the remainder of the labrum. In a type IV lesion, there is a tear of the labrum that extends into the biceps tendon (Figure 3–32).

![]() Figure 3–32. The five types of the SLAP lesion include fraying of the superior capsulolabrum (type I), detachment of the superior capsulolabrum and the biceps anchor (type II), bucket-handle tearing of the superior capsulolabrum (type III), detachment of the superior capsulolabrum and tearing into the biceps anchor (type IV), and combinations of these (type V).
Figure 3–32. The five types of the SLAP lesion include fraying of the superior capsulolabrum (type I), detachment of the superior capsulolabrum and the biceps anchor (type II), bucket-handle tearing of the superior capsulolabrum (type III), detachment of the superior capsulolabrum and tearing into the biceps anchor (type IV), and combinations of these (type V).
Types V to VII SLAP lesions were later added to this initial four-part classification. A type V lesion is an anterior-inferior Bankart lesion that continues superiorly to include separation of the biceps tendon. A type VI lesion includes a biceps separation with an unstable flap tear of the labrum. Finally, a type VII lesion involves a superior labrum-biceps tendon separation that extends anteriorly beneath the middle glenohumeral ligament.
Proposed mechanisms of injury include a fall onto an outstretched arm, with the shoulder positioned in abduction and slight forward flexion at the time of the impact, and in overhead athletes, posteroinferior capsular tightness that results in diminished internal rotation of the abducted shoulder, diagnosed with comparison to the contralateral side. With overhead throwing, this results in posterosuperior translation of the humeral head, accompanied by pain in the cocking phase of throwing and demonstrated on clinical examination by pain in the posterior shoulder when placed in the apprehension position of abduction and external rotation. A peel-back mechanism is proposed as being responsible for the SLAP lesion. Mild anterior shoulder instability also occurs due to these alterations in the glenohumeral joint and aberrations of scapulothoracic motion. However, SLAP lesions may most commonly be found incidentally in elderly individuals undergoing surgical treatment of a rotator cuff tear.
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Common clinical complaints are shoulder pain greater with activities involving overhead motion and a painful “catching” or “popping.”
• Pain with resisted forward flexion with the arm in the internally rotated and slightly adducted position, relieved by externally rotating the arm.
• Magnetic resonance arthrography can aid in the diagnosis.
![]() Prevention
Prevention
Because labral injuries can result from repetitive activity or an acute traumatic event, it is important to maintain good strength and flexibility of the shoulder to minimize these injuries.
![]() Clinical Findings
Clinical Findings
SLAP lesions cause shoulder pain both from mechanical symptoms and mild shoulder instability. Yet, they remain difficult to diagnose despite efforts to develop specific tests. No single test is both sensitive and specific for the diagnosis of SLAP lesions. Magnetic resonance arthrography can be helpful. However, diagnostic arthroscopy remains the best means to definitively diagnose SLAP lesions. The active compression test may prove to be the most useful single provocative maneuver. The internally rotated shoulder is forward flexed to 90 degrees and is then brought across the body in horizontal abduction about 10 degrees. The test is positive if the patient has pain with resisted forward flexion that is relieved by external rotation of the shoulder.
A complicating factor in making the diagnosis is that the majority of SLAP lesions are associated with other shoulder pathology such as rotator cuff tears, acromioclavicular joint pathology, and instability. Less than 28% of SLAP lesions are isolated.
![]() Treatment
Treatment
Although the history, physical examination, and imaging, specifically magnetic resonance arthrography, are helpful, definitive diagnosis of SLAP lesions is best made with diagnostic arthroscopy. Treatment can be simplified by noting whether the lesion would contribute to detachment of either the biceps anchor or the anterosuperior capsulolabrum. Lesions producing meaningful detachment of the anterior capsuloligamentous structures generally require repair of these structures back to the bony glenoid rim. Lesions extending into the biceps tendon may require debridement, biceps tenotomy, or tenodesis.
Alpert JM, Wuerz TH, O’Donnell TF, Carroll KM, Brucker NN, Gill TJ: The effect of age on the outcomes of arthroscopic repair of type II superior labral anterior and posterior lesions. Am J Sports Med2010;38:2299. [PMID: 20739578]
Barber FA, Field LD, Ryu RK: Biceps tendon and superior labrum injuries: decision making. Instr Course Lect 2008;57:527. [PMID: 18399607]
Boileau P, Parratte S, Chuinard C, Roussanne Y, Shia D, Bicknell R: Arthroscopic treatment of isolated type II SLAP lesions: biceps tenodesis as an alternative to reinsertion. Am J Sports Med 2009;37:929. [PMID: 19229046]
Franceschi F, Longo UG, Ruzzini L, Rizzello G, Maffulli N, Denaro V: No advantages in repairing a type II superior labrum anterior and posterior (SLAP) lesion when associated with rotator cuff repair in patients over age 50: a randomized controlled trial. Am J Sports Med 2008;36:247. [PMID: 17940144]
Kanatli U, Ozturk BY, Bolukbasi S: Anatomical variations of the anterosuperior labrum: prevalence and association with type II superior labrum anterior-posterior (SLAP) lesions. J Shoulder Elbow Surg2010;19:1199. [PMID: 21070956]
Meserve BB, Cleland JA, Boucher TR: A meta-analysis examining clinical test utility for assessing superior labral anterior posterior lesions. Am J Sports Med 2009;37:2252. [PMID: 19095895]
![]() CPT Code for SLAP Lesions
CPT Code for SLAP Lesions
29807 Arthroscopy, shoulder, surgical; repair of SLAP lesion
SHOULDER STIFFNESS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Very painful and/or limited range of motion of the shoulder.
• May be idiopathic or posttraumatic.
• Loss of active and passive range of motion, most notably internal rotation.
• Arthrography can aid in the diagnosis.
![]() Prevention
Prevention
Most patients have some sort of antecedent trauma to their shoulder, be it minimal or severe. Initiating gentle range-of-motion and strengthening exercises immediately after the traumatic event are essential to minimizing the likelihood of developing shoulder stiffness.
![]() Clinical Findings
Clinical Findings
Often called adhesive capsulitis or frozen shoulder, shoulder stiffness is a painful condition characterized by significant restriction in both active and passive range of motion. With shoulder stiffness, the articular surfaces are normal and the joint is stable, yet there is a restriction in range of motion. Stiffness usually results from soft-tissue contracture but can also occur from malaligned articular surfaces, bursal adhesions, or a shortened muscle-tendon unit. Often of uncertain etiology, the restrictions of shoulder motion are global. That is, none of the shoulder planes of motion is spared.
Shoulder stiffness may be separated into idiopathic and posttraumatic etiologies. Idiopathic shoulder stiffness is most common in older individuals, especially women between 40 and 60 years of age. Other factors that predispose to idiopathic shoulder stiffness include cervical, cardiac, pulmonary, neoplastic, neurologic, and personality disorders. Patients with diabetes mellitus are also at a high risk of developing shoulder stiffness, with 10–35% of diabetics having restriction of shoulder motion. Diabetics who have been insulin dependent for many years have the greatest incidence and bilateral involvement. Because of this close association, clinicians should ask their patients with shoulder stiffness about symptoms of diabetes; 70% of individuals with shoulder stiffness may have diabetes or a prediabetes condition. The pathophysiology of idiopathic shoulder stiffness remains uncertain, but the pathoanatomy is commonly limited to contracture of the glenohumeral capsule (Figure 3–33). Most prominently involved is the rotator interval, which includes the superior glenohumeral ligament and coracohumeral ligament.
![]() Figure 3–33. Adhesive capsulitis of the shoulder. Note the small irregular joint capsule with addition of contrast material.
Figure 3–33. Adhesive capsulitis of the shoulder. Note the small irregular joint capsule with addition of contrast material.
Although all patients can recall some traumatic event that preceded their shoulder stiffness, those with distinct trauma such as a prior fracture, rotator cuff tear, or surgical procedure have a posttraumatic etiology. Stiffness after shoulder surgery is typical and usually resolves with time and appropriate rehabilitation. The shoulder should not be neglected after any surgery about the shoulder girdle. This includes axillary or cervical lymph node dissections, especially when combined with radiation therapy, cardiac catheterization in the axilla, and coronary artery bypass grafting with sternotomy and thoracotomy. All surgeons should be aware that these procedures may result in restricted shoulder motion.
The clinical presentation of idiopathic shoulder stiffness is classically described as having three phases. The first phase is the painful, freezing phase. The pain is typically achy in nature, and sudden jolts or attempts at rapid motion exacerbate the chronic discomfort. The pain may begin at night, and shoulder motion becomes progressively limited. Patients often hold their arm at their side and in internal rotation with the forearm across the belly. They may also be treated for nonspecific shoulder pain with a sling in this position. This inflammatory phase often lasts between 2 and 9 months.
The second phase of progressive stiffness lasts between 3 and 12 months. Stiffness progresses to a point at which shoulder motion is restricted in all planes. Essentially, the shoulder has undergone fibrous arthrodesis. Fortunately, pain progressively decreases from the initial inflammatory phase. With time, patients are able to use the shoulder with little or no pain, within the restricted range of motion, but attempts to exceed this range are accompanied by pain. The patient’s symptoms then plateau. Unfortunately, this phase may be persistent, with symptoms lasting for extended periods. In the resolution, or thawing phase, the shoulder slowly and progressively becomes more supple. It can be as short as a month, but typically lasts 1–3 years.
On clinical examination, there is loss of both active and passive range of shoulder motion. Often the first motion to be affected is internal rotation, demonstrated by an inability to bring the arm up the back to the same level as the normal shoulder. MRI reveals decreased rotator interval size, and arthrography demonstrates marked reduction in joint capacity; often the affected shoulder will not take more than a few milliliters of dye, whereas the normal capacity is 20–30 mL.
![]() Treatment
Treatment
Treatment varies, but conservative modalities and progressive range-of-motion exercises seem effective. Range-of-motion exercises for external rotation and abduction will help minimize the length of restriction in motion and dysfunction. Nonoperative treatments are successful for most. When operative treatments are chosen, manipulation under anesthesia and capsular distension, along the mainstays of intervention, are replaced by selective arthroscopic capsular release.
![]() Prognosis
Prognosis
Whether treated nonoperatively or operatively, a return of about 80% shoulder range of motion is usual.
Blanchard V, Barr S, Cerisola FL: The effectiveness of corticosteroid injections compared with physiotherapeutic interventions for adhesive capsulitis: a systematic review. Physiotherapy 2010;96:95. [PMID: 20420956]
Hand C, Clipsham K, Rees JL, Carr AJ: Long-term outcome of frozen shoulder. J Shoulder Elbow Surg 2008;17:231. [PMID: 17993282]
Hand GC, Athanasou NA, Matthews T, Carr AJ: The pathology of frozen shoulder. J Bone Joint Surg Br 2007;89:928. [PMID: 17673588]
Milgrom C, Novack V, Weil Y, Jaber S, Radeva-Petrova DR, Finestone A: Risk factors for idiopathic frozen shoulder. Isr Med Assoc J 2008;10:361. [PMID: 18605360]
Rill BK, Fleckenstein CM, Levy MS, Nagesh V, Hasan SS: Predictors of outcome after nonoperative and operative treatment of adhesive capsulitis. Am J Sports Med 2011;39:567. [PMID: 21160014]
Saccomanni B: Inflammation and shoulder pain: a perspective on rotator cuff disease, adhesive capsulitis, and osteoarthritis: conservative treatment. Clin Rheumatol 2009;28:495. [PMID: 19224130]
Tauro JC, Paulson M: Shoulder stiffness. Arthroscopy 2008;24:949. [PMID: 18657745]
Thomas SJ, McDougall C, Brown ID, et al: Prevalence of symptoms and signs of shoulder problems in people with diabetes mellitus. J Shoulder Elbow Surg 2007;16:748. [PMID: 18061115]
![]() CPT Codes for Shoulder Stiffness
CPT Codes for Shoulder Stiffness
23020 Capsular contracture release (eg, Sever-type procedure)
23700 Manipulation under anesthesia, shoulder joint, including application of fixation apparatus (dislocation excluded)
29825 Arthroscopy, shoulder, surgical; with lysis and resection of adhesions, with or without manipulation
FRACTURES ABOUT THE SHOULDER
1. Clavicular Fracture
The clavicle is one of the most commonly fractured bones in the body, with direct trauma being the usual cause in athletic events (Figure 3–34). Football, wrestling, and ice hockey are the sports most commonly involved in clavicular fractures, which is not surprising as all three are associated with high-speed contact between players.

![]() Figure 3–34. Analysis of 1603 shoulder girdle injuries, showing the frequency and location of fractures and dislocations.
Figure 3–34. Analysis of 1603 shoulder girdle injuries, showing the frequency and location of fractures and dislocations.
![]() Essentials of Diagnosis
Essentials of Diagnosis
• History of injury to the shoulder.
• Swelling and ecchymosis overlying the injured clavicle.
• Pain and crepitation upon palpation of the fracture site.
• Pain and limited range of motion of the arm, specifically in forward flexion and abduction.
• Appropriate radiographs will define the location and severity of the fracture.
![]() Clinical Findings
Clinical Findings
Despite the proximity of vital structures, clavicular fractures that occur during athletic activities are rarely associated with neurovascular damage, and accompanying soft-tissue disorders are uncommon. The patient will usually give a history of falling in the area of the shoulder or receiving a blow to the clavicle, experiencing immediate pain and an inability to raise the arm. Radiography will usually confirm the clinical impression and must show the entire clavicle, including the shoulder girdle, upper third of the humerus, and sternal end of the clavicle.
Of clavicular fractures, midclavicular fractures account for 80%, distal fractures for 15%, and proximal fractures for 5%. Most fractures of the shaft of the clavicle heal well. However, some neurovascular complications, such as a tear of the subclavian artery or a brachial plexus injury, are serious, although rare. Therefore, when evaluating and treating clavicular fractures, an initial neurovascular examination is very important. Pulses in the distal part of the upper extremity, strength, and sensation must be carefully evaluated.
Because the clavicle is the only bone structure that fixes the shoulder girdle to the thorax, a fracture through the clavicle causes the shoulder to sag forward and downward. The pull of the sternocleidomastoid muscle may displace the proximal fragment superiorly. These forces tend to hinder the initial reduction and maintenance of reduction. In addition, distal fractures, which are more common in older age groups, may involve tears in the coracoclavicular ligament, which allows the proximal clavicle to ride up superiorly, mimicking an acromioclavicular dislocation. Delayed union is much more common in this type of fracture than in other clavicular fractures.
![]() Treatment
Treatment
Mid and proximal clavicular fractures are usually treated with a short period of rest, with a sling on the affected side to support the extremity. Immobilization is usually discontinued at 3–4 weeks, and once the clavicular fracture has healed, range-of-motion and strengthening exercises should begin. Comminuted mid and proximal clavicular fractures with meaningful displacement and especially when it is shortening may be best treated with open reduction and internal fixation. Distal fractures with tears in the coracoclavicular ligament, mimicking an acromioclavicular dislocation, are best treated with open reduction and internal fixation.
![]() Prognosis
Prognosis
Onset of exercises prior to healing may contribute to nonunion. Athletes should not be allowed to return to play until shoulder strength and range of motion return to preinjury levels. Generally, no special braces or pads are required when the athlete returns to play.
Khan LA, Bradnock TJ, Scott C, Robinson CM: Fractures of the clavicle. J Bone Joint Surg Am 2009;91:447. [PMID: 19181992]
Kulshrestha V, Roy T, Audige L: Operative versus nonoperative management of displaced midshaft clavicle fractures: a prospective cohort study. J Orthop Trauma 2011;25:31. [PMID: 21164305]
Robinson CM, Court-Brown CM, McQueen MM, et al: Estimating the risk of nonunion following nonoperative treatment of a clavicular fracture. J Bone Joint Surg Am 2004;86-A:1359. [PMID: 15252081]
![]() CPT Codes for Clavicle Fractures
CPT Codes for Clavicle Fractures
23500 Closed treatment of clavicular fracture; without manipulation
23505 Closed treatment of clavicular fracture; with manipulation
23515 Open treatment of clavicular fracture, includes internal fixation, when performed
2. Proximal Humerus Fracture
![]() Essentials of Diagnosis
Essentials of Diagnosis
• History of trauma to the shoulder.
• Swelling and ecchymosis overlying the shoulder that may extend down to the elbow.
• Tenderness and crepitation over the fracture site.
• Pain with attempted range of motion of the shoulder.
• Appropriate radiographs will define the location and severity of the fracture.
Fractures of the proximal humerus, which represent approximately 4–5% of all fractures, are a relatively uncommon sports injury. They most often present in young adolescents with open growth plates or in elderly osteoporotic patients. When they do occur in the athlete, they are typically the result of a high-energy impact injury or are secondary to an underlying pathologic bone condition.
![]() Clinical Findings
Clinical Findings
The proximal humerus consists of four major bony components: the humeral head, the greater tuberosity, the lesser tuberosity, and the humeral shaft. Fractures, which can occur between any or all of these regions, are traditionally defined by the location and displacement of the fracture fragments (Figure 3–35). The patient with a proximal humerus fracture will usually be able to report the mechanism of injury and will complain of pain, swelling, and an inability to use the shoulder. A physical examination will often reveal loss of the normal contour of the shoulder, tenderness about the shoulder, ecchymosis that may extend down to the elbow, and crepitus on attempted range of motion. A thorough neurovascular examination is essential, as brachial plexus and axillary nerve injuries have been reported in association with proximal humerus fractures. Because the axillary nerve is the nerve most commonly injured in these cases, sensation to light touch and pin-prick over the lateral aspect of the upper arm and deltoid muscle function must be tested. An accurate radiographic evaluation is necessary to confirm the type and severity of the fracture and is essential in determining the treatment plan. Anteroposterior and lateral views in the plane of the scapula, as well as an axillary view to rule out an associated glenohumeral dislocation, are necessary.

![]() Figure 3–35. Four-part classification for fractures of the proximal humerus. AN, anatomic neck; GT, greater tuberosity; LT, lesser tuberosity; SN, surgical neck. [Reprinted with permission from Norris TR, Green A: Proximal humerus fractures and fracture-dislocations. In: Browner BD, et al, eds: Skeletal Trauma: Fractures, Dislocation and Ligamentous Injuries. Elsevier; 1998.]
Figure 3–35. Four-part classification for fractures of the proximal humerus. AN, anatomic neck; GT, greater tuberosity; LT, lesser tuberosity; SN, surgical neck. [Reprinted with permission from Norris TR, Green A: Proximal humerus fractures and fracture-dislocations. In: Browner BD, et al, eds: Skeletal Trauma: Fractures, Dislocation and Ligamentous Injuries. Elsevier; 1998.]
![]() Treatment
Treatment
Most proximal humerus fractures are minimally displaced and can be treated nonoperatively with sling immobilization and early passive range of motion. However, about 20% need to be treated operatively. Many factors contribute to this decision-making process, including fracture type and degree of displacement, bone quality, activity level, and associated injuries. Surgical options range from closed reduction and percutaneous pinning to open reduction with internal fixation to humeral head replacement.
![]() Prognosis
Prognosis
For minimally displaced fractures, the prognosis is generally good. Loss of motion is the most common complication. It can take 12–18 months to attain the maximal result, so range-of-motion exercises should be continued for an extended period of time.
Cannon CP, Paraliticci GU, Lin PP, Lewis VO, Yasko AW: Functional outcome following endoprosthetic reconstruction of the proximal humerus. J Shoulder Elbow Surg 2009;18:705. [PMID: 19186077]
Zhu Y, Lu Y, Shen J, Zhang J, Jiang C: Locking intramedullary nails and locking plates in the treatment of two-part proximal humeral surgical neck fractures: a prospective randomized trial with a minimum of three years of follow-up. J Bone Joint Surg Am 2011;93:159. [PMID: 21248213]
PROXIMAL HUMERAL EPIPHYSEAL FRACTURE
In young athletes, epiphyseal fractures of the proximal humerus may occur. The separate growth centers of the articular surface, greater tuberosity, and lesser tuberosity coalesce at approximately age 7 years, with the remaining growth plates closing at 20–22 years of age. Therefore, fracture separations may occur at any age until the growth plates have closed. Fortunately, fractures in this area usually do not arrest growth.
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Proximal humerus pain.
• Widening of the proximal humeral physis on radiographs.
![]() Clinical Findings
Clinical Findings
Injury can occur to the shoulder in the growing musculoskeletal system of young athletes engaged in sports that involve overhead throwing. Proximal humerus pain associated with widening of the proximal humerus epiphysis, especially while throwing, has been termed “little league shoulder.” Although widening of the proximal humerus epiphysis can be an adaptive change to throwing, when painful, it may represent a fracture resulting from overuse.
![]() Treatment
Treatment
Cessation of throwing is the first step in treatment. Once pain has resolved, range-of-motion and strengthening exercises can be initiated. Ultimately, throwing can be resumed as long as the patient is pain free.
Bahrs C, Zipplies S, Ochs BG, et al: Proximal humeral fractures in children and adolescents. J Pediatr Orthop 2009;29:238. [PMID: 19305272]
![]() CPT Codes for Proximal Humerus Fractures
CPT Codes for Proximal Humerus Fractures
23600 Closed treatment of proximal humeral (surgical or anatomic neck) fracture; without manipulation
23605 Closed treatment of proximal humeral (surgical or anatomic neck) fracture; with manipulation, with or without skeletal traction
23615 Open treatment of proximal humeral (surgical or anatomic neck) fracture, includes internal fixation, when performed, includes repair of tuberosity(s), when performed
23616 Open treatment of proximal humeral (surgical or anatomic neck) fracture, includes internal fixation, when performed, includes repair of tuberosity(s), when performed; with proximal humeral prosthetic replacement
23620 Closed treatment of greater humeral tuberosity fracture; without manipulation
23525 Closed treatment of greater humeral tuberosity fracture; with manipulation
23630 Open treatment of greater humeral tuberosity fracture, includes internal fixation, when performed
GLENOHUMERAL JOINT OSTEOARTHRITIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Constant achy pain in all shoulder positions, even with the arm at the side.
• Pain worsened with activity.
• Crepitus with motion.
• Pain is relieved with lidocaine injection into glenohumeral joint.
![]() Prevention
Prevention
It is best to curtail activity once the articular surfaces of the glenohumeral joint have been injured to minimize progression of osteoarthritis. Even rigorous activity in the uninjured shoulder is unlikely to result in osteoarthritis.
![]() Clinical Findings
Clinical Findings
Achy, persistent pain and limitation of motion are often initial symptoms of osteoarthritis of the glenohumeral joint. Most common after the sixth decade, osteoarthritis can present at earlier ages in those having suffered injury earlier in life. Pain is exacerbated by activity and often persists for hours after the activity ceases. In mild to moderate cases, the pain diminishes with NSAIDs. Shoulder motion is associated with crepitus, and active and passive range of motion are the same, similar to adhesive capsulitis. Shoulder weakness and muscle atrophy are secondary to the pain and disuse of the shoulder that occur when the osteoarthritis is severe.
Radiographs of the shoulder reveal decreased joint space, osteophytes, subchondral sclerosis, and subchondral cysts of the glenohumeral joint. The prominence of these findings is associated with the severity of the disease but is only loosely associated with complaints of pain. Like the hip, knee, and other joints, some have severe pain with mild radiographic findings, whereas others cope well with severe findings. MRI may be useful to evaluate the shoulder for other injuries such as a rotator cuff tear.
![]() Treatment
Treatment
Rest, rehabilitation, and taking NSAIDs may diminish symptoms in those with mild to moderate osteoarthritis. Range-of-motion and strengthening exercises are recommended, unless they cause significant discomfort. Activities that exacerbate the symptoms should be avoided, and strengthening of the shoulder muscles should be done as long as they do not exacerbate symptoms. Injection of corticosteroids may relieve symptoms for a period of time, and repeated injections at intervals of a few months are sometimes effective. Individuals with pain, weakness, or decreased range of motion that persist after nonoperative treatment may elect surgical intervention with glenohumeral joint arthroplasty.
![]() Prognosis
Prognosis
Glenohumeral joint arthroplasty is effective in diminishing shoulder pain when both the humeral head and the glenoid are resurfaced (total joint arthroplasty). Another option is hemiarthroplasty of the humeral head only, which decreases pain, on average, by about two thirds. The latter may yield a more durable arthroplasty as the glenoid component loosens more often over long periods of time than the humeral component. About 90% of total joint arthroplasties will last 10 years, and nearly 75% will last 20 years. Neither procedure yields normal joint function; patients have only partial improvement in strength and range of motion.
Hambright D, Henderson RA, Cook C, Worrell T, Moorman CT, Bolognesi MP: A comparison of perioperative outcomes in patients with and without rheumatoid arthritis after receiving a total shoulder replacement arthroplasty. J Shoulder Elbow Surg 2011;20:77. [PMID: 20655764]
Millett PJ, Gobezie R, Boykin RE: Shoulder osteoarthritis: diagnosis and management. Am Fam Physician 2008;78:605. [PMID: 18788237]
Saltzman MD, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd: Comparison of patients undergoing primary shoulder arthroplasty before and after the age of fifty. J Bone Joint Surg Am 2010;92:42. [PMID: 20048094]
Singh JA, Sperling J, Buchbinder R, McMaken K: Surgery for shoulder osteoarthritis: a Cochrane systematic review. J Rheumatol 2011;38:598. [PMID: 21239751]
![]() CPT Codes for Glenohumeral Joint
CPT Codes for Glenohumeral Joint
Osteoarthritis
23470 Arthroplasty, glenohumeral joint; hemiarthroplasty
23472 Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement [eg, total shoulder])
29822 Arthroscopy, shoulder, surgical; debridement, limited
29823 Arthroscopy, shoulder, surgical; debridement, extensive
ACROMIOCLAVICULAR JOINT INJURY
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Pain and swelling over the acromioclavicular joint.
• May have visible elevation or displacement of the clavicle relative to the acromion (asymmetric to the contralateral shoulder).
• Pain with forward elevation of the arm.
• Appropriate radiographs can be confirmatory.
![]() Prevention
Prevention
Avoiding activities that may result in a downward blow to the tip of the shoulder is the best way to prevent these injuries.
![]() Clinical Findings
Clinical Findings
Acromioclavicular dislocations or subluxations, commonly referred to as separations, vary in severity depending on the extent of injury to the stabilizing ligaments and capsule. The typical mechanism of injury is a direct downward blow to the tip of the shoulder. Clinically, pain at the top of the shoulder over the acromioclavicular joint is the predominant symptom, with varying decreases in motion depending on the severity of the injury. The athlete who has sustained this type of injury will typically leave the field holding the arm close to the side.
When checking for instability of the acromioclavicular joint, the examiner should manipulate the midshaft of the clavicle, rather than the acromioclavicular joint to rule out pain from contusion to the acromioclavicular area. For milder acromioclavicular injuries, the patient should put the hand of the affected arm on the opposite shoulder, and the examiner may then gently apply downward pressure at the patient’s affected elbow, noting if this maneuver causes pain at the acromioclavicular joint.
Acromioclavicular joint injuries were initially divided into grades I to III (Figure 3–36). Grade I injuries are typically produced by a mild blow causing a partial tear of the acromioclavicular ligament. When the acromioclavicular ligament is completely torn, but the coracoclavicular ligament remains intact, a grade II injury that involves subluxation or partial displacement results. When the force of injury is severe enough to tear the coracoclavicular and acromioclavicular ligaments in addition to the capsule, a grade III injury occurs.

![]() Figure 3–36. Grades of acromioclavicular joint separations.
Figure 3–36. Grades of acromioclavicular joint separations.
Three additional injuries were later added to the classification. In grade IV injuries, the clavicle is displaced posterior and buttonholed through the fascia of the trapezius muscle. Grade V injuries demonstrate severe inferior displacement of the glenohumeral joint, with the clavicle often 300% superior to the acromion. Lastly, in grade VI injuries, the distal end of the clavicle is locked inferior to the coracoid.
Acromioclavicular joint displacement is often obvious on physical examination, but it is best classified by radiography. An anteroposterior radiograph that is aimed 10 degrees cephalad allows visualization of the acromioclavicular joint. A radiograph of the entire upper thorax allows the vertical distance between the coracoid and the clavicle on both the involved and uninvolved sides to be compared. Anteroposterior radiographs with weights applied to the upper extremities are usually unnecessary. An axillary lateral radiograph is also essential for proper classification.
![]() Treatment
Treatment
Management of acromioclavicular joint injuries depends on their severity. Grade I and II injuries may be treated with a sling until discomfort dissipates, usually within 2–4 weeks. Next a rehabilitation program starts, and normal range of motion and strength to the upper extremity begin to be restored. Treatment of acute grade III injuries or complete dislocations in athletes is controversial; most believe grade III injuries are best managed nonoperatively, but others advocate operative treatment. Grade IV to VI injuries are best treated with open reduction and internal fixation along with reconstruction of the coracoclavicular ligament.
Nonsurgical treatment usually includes a sling for comfort. Ice and other modalities are used for an acute acromioclavicular injury to reduce soreness and swelling. Pain is the limiting factor in beginning range-of-motion and isometric muscle-strengthening exercises. It should be used as a guide for initiation and progress through rehabilitation. Range-of-motion exercises can be begun rather quickly and strengthening added when pain permits.
Before resuming athletic activities, the patient should have full, painless range of motion, no tenderness on palpation, and sufficient strength.
![]() Prognosis
Prognosis
Athletes who do not need to elevate their arms, such as soccer or football players, tend to return to sports earlier than players engaged in sports that require overhead arm activity, such as tennis, baseball, and swimming.
Johansen JA, Grutter PW, McFarland EG, Petersen SA: Acromioclavicular joint injuries: indications for treatment and treatment options. J Shoulder Elbow Surg 2011;20(2 Suppl):S70. [PMID: 21195634]
Rios CG, Mazzocca AD: Acromioclavicular joint problems in athletes and new methods of management. Clin Sports Med 2008;27:763. [PMID: 19064155]
CORACOID FRACTURE
Fractures of the coracoid process are rare; they are usually seen in professional riflemen and skeet shooters, although they have also been reported in baseball and tennis players. They are identified on radiographs, and conservative treatment, including cessation of activity, usually results in uncomplicated healing after 6–8 weeks.
STERNOCLAVICULAR JOINT INJURY
In the skeletally mature adult athlete, injury to the sterno-clavicular joint usually involves the surrounding soft tissue and capsule tearing, leading to subluxation or dislocation. The mechanism of injury is either a blow to the point of the shoulder, which predisposes the athlete to anterior dislocation, or a direct blow to the clavicle or chest with the shoulder in extension, which predisposes the athlete to posterior dislocation. The injury may range from a symptomatic sprain to a complete sternoclavicular dislocation with disruption of the capsule and its restraining ligaments.
1. Anterior Dislocation
![]() Essentials of Diagnosis
Essentials of Diagnosis
• History of trauma to the upper chest wall.
• Painful prominence overlying the proximal end of the clavicle.
• Appropriate radiographs or computed tomography (CT) scan can be diagnostic.
![]() Clinical Findings
Clinical Findings
The most common type of sternoclavicular dislocation is anterior dislocation. This is recognized clinically by an anterior prominence of the proximal clavicle on the involved side. Radiographic documentation of an anterior sternoclavicular dislocation is difficult because the rib, sternum, and clavicle overlap at the joint, but may be confirmed by oblique views. A CT scan is very sensitive and should be done if the radiograph appears normal but a dislocation is suspected.
![]() Treatment
Treatment
Although dislocation of the anterior sternoclavicular joint may cause considerable distress initially, the symptoms usually subside rapidly, with no loss of shoulder function. A variety of surgical and nonsurgical approaches have been advocated, but surgery for anterior dislocations often results in significant complications. Closed treatment modalities vary from using a sling to attempted closed reduction, which may be successful initially but is difficult to maintain.
2. Posterior Dislocation
![]() Essentials of Diagnosis
Essentials of Diagnosis
• History of trauma to the upper chest wall.
• Pain in the region of the proximal end of the clavicle.
• Patient may present with hoarseness, dysphagia, or severe respiratory distress.
• Appropriate radiographs or CT scan can be diagnostic.
![]() Clinical Findings
Clinical Findings
Posterior sternoclavicular dislocation is much less common but is associated with more complications because of the potential for injury to the esophagus, great vessels, and trachea. Presenting symptoms range from mild to moderate pain in the sternoclavicular region to hoarseness, dysphagia, severe respiratory distress, and subcutaneous emphysema from tracheal injury.
![]() Treatment
Treatment
In most instances, closed reduction of posterior dislocations, if performed early, is successful and stable. To effect reduction, a pillow is placed under the upper back of the supine patient, and gentle traction is applied with the shoulder held in 90 degrees of abduction and at maximum extension (Figure 3–37). Rarely, closed reduction under general anesthesia or open reduction is required.

![]() Figure 3–37. Method for reducing (A) anterior sternoclavicular dislocation and (B) posterior sternoclavicular dislocation.
Figure 3–37. Method for reducing (A) anterior sternoclavicular dislocation and (B) posterior sternoclavicular dislocation.
After reduction, the patient is put in an immobilization splint and is instructed to use ice and oral NSAIDs. Once the joint has healed sufficiently, usually within 2–3 weeks, range-of-motion exercises may begin. Elevation of the arm should not be attempted until 3 weeks after injury.
3. Medial Clavicular Epiphyseal Fracture
In athletes younger than 25 years of age, sternoclavicular injuries may not result in true dislocations, but rather in fractures through the growth plate of the proximal clavicle. These clavicular epiphyseal fractures may appear clinically as dislocations, particularly if some displacement is present, and may be treated conservatively. Typically, these are not associated with growth deformities, and reduction of the fracture is not needed unless there is severe displacement. Symptomatic treatment for pain will usually suffice. Sometimes an adolescent presents with an enlarging mass at the sternoclavicular joint, accompanied by parents with worries of cancer. A careful history reveals trauma several weeks earlier, and the mass represents the callus of a healing clavicular epiphyseal fracture that can be demonstrated radiographically.
Jaggard MK, Gupte CM, Gulati V, Reilly P: A comprehensive review of trauma and disruption to the sternoclavicular joint with the proposal of a new classification system. J Trauma 2009;66:576. [PMID: 19204537]
![]() CPT Codes for Acromioclavicular and Sternoclavicular Joint Injuries
CPT Codes for Acromioclavicular and Sternoclavicular Joint Injuries
23101 Arthrotomy, acromioclavicular joint or sternoclavicular joint, including biopsy and/or excision of torn cartilage
23120 Claviculectomy; partial
23520 Closed treatment of sternoclavicular dislocation; without manipulation
23525 Closed treatment of sternoclavicular dislocation; with manipulation
23530 Open treatment of sternoclavicular dislocation, acute or chronic
23532 Open treatment of sternoclavicular dislocation, acute or chronic; with fascial graft (includes obtaining graft)
23540 Closed treatment of acromioclavicular dislocation; without manipulation
23545 Closed treatment of acromioclavicular dislocation; with manipulation
24550 Open treatment of acromioclavicular dislocation, acute or chronic
23552 Open treatment of acromioclavicular dislocation, acute or chronic; with fascial graft (includes obtaining graft)
![]() OTHER SHOULDER TENDON AND MUSCLE
OTHER SHOULDER TENDON AND MUSCLE
BICEPS TENDON INJURIES
1. Bicipital Tendinosis
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Pain localized to the anterior proximal humerus and shoulder joint.
• Pain with resisted forward flexion and supination.
• Pain may be relieved by a steroid injection into the sheath of the biceps tendon.
![]() Prevention
Prevention
Similar to the prevention of rotator cuff injuries, general conditioning and stretching and strengthening before activities can help minimize injury to the biceps tendon.
![]() Clinical Findings
Clinical Findings
The long head of the biceps muscle is an intraarticular structure deep in the rotator cuff tendon as it passes under the acromion to its insertion at the top of the glenoid. The same mechanism that initiates symptoms of impingement syndrome in rotator cuff injuries may inflame the tendon of the biceps in its subacromial position, causing bicipital tendinosis. Tendinosis may also result from subluxation of the tendon out of its groove in the proximal humerus and can occur under, into, or on top of the subscapular tendon. Subluxation of the biceps tendon is almost always associated with a subscapularis tendon tear. The symptoms of bicipital tendinosis, whether the result of impingement or tendon subluxation, are essentially the same. Pain is localized to the proximal humerus and shoulder joint, with resisted supination of the forearm aggravating the pain. Pain may also occur on manual testing of the elbow flexors and on palpation of the tendon itself. The Yergason test is used to determine instability of the long head of the biceps in its groove.
![]() Treatment
Treatment
If the biceps tendinosis is associated with shoulder impingement, then therapy aimed at treating the impingement syndrome will relieve the bicipital tendinosis. If subluxation of the tendon within its groove is the cause of the irritation, conservative therapy includes NSAIDs and restriction of activities, followed by a slow resumption of activities after a period of rest. Strengthening of the muscles that assist the biceps in elbow flexion and forearm supination is also beneficial. Steroid injections into the sheath of the biceps tendon are helpful, but they may be hazardous if placed into the substance of the tendon because they will promote tendon degeneration. Persistent symptoms may warrant tenodesis of the biceps tendon directly into the humerus.
![]() Prognosis
Prognosis
Recovery from biceps tenodesis is difficult, and it is doubtful if a competitive athlete could return to peak performance after treatment.
2. Biceps Tendon Tears at the Shoulder
![]() Essentials of Diagnosis
Essentials of Diagnosis
• “Popeye” appearance of the upper arm from distal retraction of the biceps muscle.
• May or may not be painful and ecchymotic depending on the chronicity of the injury.
![]() Prevention
Prevention
Similar to the prevention of rotator cuff injuries, general conditioning and stretching and strengthening before activities can help minimize injury to the biceps tendon.
![]() Clinical Findings
Clinical Findings
The long head of the biceps tendon may rupture proximally, either from the supraglenoid tubercle of the scapula at the entrance of the bicipital groove proximally or at the exit of the tunnel at the musculotendinous junction. The muscle mass moves distally, producing a bulging or “Popeye” appearance to the arm, but the short head remains intact; rupture of the biceps tendon distally involves both heads, and the muscle mass moves proximally. Rupture of the long head of the biceps may be predictive of a rotator cuff tear. The mechanism is usually a forceful flexion of the arm and is more common in older athletes or following direct trauma. Microtears probably serve to render the tendon vulnerable to an acute tearing event. The degree of ecchymosis is dependent on the location of the tear, with avascular areas having less ecchymosis and the musculotendinous junction producing quite a noticeable amount of ecchymosis. Diagnosis is usually easily accomplished, as the deformity is obvious.
![]() Treatment
Treatment
Surgical treatment of proximal ruptures, if indicated, is usually reserved for younger patients. The proximal end of the tendon is usually found beneath the attachment of the pectoralis major. Proximal biceps tendon rupture usually occurs with rotator cuff tears in middle-aged and older athletes.
![]() Prognosis
Prognosis
Athletes are permitted to return to full contact play once they have achieved maximal functional strength and range of elbow motion, which typically occurs 4–6 months following a proximal biceps repair.
Nho SJ, Strauss EJ, Lenart BA, et al: Long head of the biceps tendinopathy: diagnosis and management. J Am Acad Orthop Surg 2010;18:645. [PMID: 21041799]
![]() CPT Codes for Biceps Tendon Injuries at the Shoulder
CPT Codes for Biceps Tendon Injuries at the Shoulder
23430 Tenodesis of long tendon of biceps
23440 Resection or transplantation of long tendon of biceps
29828 Arthroscopy, shoulder, surgical; biceps tenodesis
PECTORALIS MAJOR RUPTURE
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Sudden pain.
• Ecchymosis and swelling along the pectoralis major muscle.
![]() Prevention
Prevention
Similar to the prevention of rotator cuff injuries, general conditioning and stretching and strengthening before activities can help minimize injury to the pectoralis major.
![]() Clinical Findings
Clinical Findings
Rupture of the pectoralis major tendon is an uncommon injury, usually occurring during bench press exercises in weight lifting and caused by sudden unexpected muscle contraction during pulling or lifting. The athlete usually experiences sudden pain and develops local ecchymosis and swelling. As the swelling subsides, a sulcus and deformity may be visible, and the patient notices weakness of the arm in adduction and internal rotation.
![]() Treatment
Treatment
The rupture may be partial or complete, and nonoperative treatment usually results in satisfactory function for the activities of daily life. Surgery may be considered if the athlete wishes to return to heavy weight lifting.
![]() Prognosis
Prognosis
Athletes are permitted to return to contact sports once they have achieved full strength and range of motion, which typically occurs 6 months following a pectoralis major repair.
Antosh IJ, Grassbaugh JA, Parada SA, Arrington ED: Pectoralis major tendon repairs in the active-duty population. Am J Orthop 2009;38:26. [PMID: 19238264]
Provencher MT, Handfield K, Boniquit NT, Reiff SN, Sekiya JK, Romeo AA: Injuries to the pectoralis major muscle: diagnosis and management. Am J Sports Med 2010;38:1693. [PMID: 20675652]
SHOULDER NEUROVASCULAR INJURY
1. Brachial Plexus Injury
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Often preceded by a fall onto the shoulder.
• Paresthesias and/or motor loss in the affected extremity that can be transient or permanent.
• EMG can help localize the lesion and aid in prognosis.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
Brachial plexus injuries are typically caused by a fall on the shoulder as seen in acromioclavicular joint injuries. Most brachial plexus injuries do not involve motor loss and exhibit paresthesias, which resolve in a period of minutes to weeks, although some cases may persist for months or years. Early in the course of the injury, a transient slowing of conduction across the plexus or a mild prolongation of nerve latency may be seen. The “burner” or “stinger” is one of the most common brachial plexus injuries encountered in athletes. The key to diagnosis is a short duration of upper extremity paresthesias and shoulder weakness, with pain-free range of motion of the cervical spine. Players may return to competition after shoulder strength and full, pain-free range of motion have returned.
Rarely, a severe injury will occur (eg, from motorcycle racing). Chronic injuries result in instability of the shoulder that may be treated with trapezius transfer. Arthrodesis is an alternative, initially or after failed muscle transfer.
Safran MR: Nerve injury about the shoulder in athletes. Part 2: long thoracic nerve, spinal accessory nerve, burners/stingers, thoracic outlet syndrome. Am J Sports Med 2004;32:1063. [PMID: 15150060]
2. Long Thoracic Nerve Injury
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Palsy of the serratus anterior results in medial winging of the scapula.
• May be painless, but often there is medial scapula pain.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
Traction incidents may cause a long thoracic nerve palsy, with subsequent serratus anterior paralysis and winging of the scapula. Traction and blunt trauma may also cause injury to the spinal accessory nerve, another cause of winging of the scapula. These can be differentiated on physical examination by the position of the scapula. With serratus anterior palsy, the inferior portion of the scapula tends to go medially, whereas the opposite occurs with spinal accessory nerve palsy. Treatment is usually conservative, with return of function in weeks if the nerve has not been divided.
3. Suprascapular Nerve Injury
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Poorly localized pain and weakness in the posterolateral shoulder.
• Weakness and atrophy of the supraspinatus and/or infra-spinatus muscles.
• MRI may reveal the presence of a cyst in the suprascapular or spinoglenoid notch.
• Electromyography/nerve conduction velocity (EMG/NCV) can aid in the diagnosis.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
Entrapment of the suprascapular nerve is often associated with activities such as weight lifting, baseball pitching, volleyball, and backpacking. Traction and repetitive shoulder use are the mechanisms of injury. Compression of the nerve may occur from entrapment at the anterior suprascapular notch of the scapula or at the level of the spinoglenoid notch. The latter occurs in volleyball players and baseball players and is likely caused by rapid overhead acceleration of the arm. Compression is associated with poorly localized pain and weakness in the posterolateral aspect of the shoulder girdle. This may be followed by atrophy of the supraspinatus or infraspinatus muscles. Eventually, there is weakness of forward flexion and external rotation of the shoulder. The diagnosis is confirmed by EMG and nerve conduction studies.
Conservative therapy consists of rest, NSAIDs, and physical therapy designed to increase muscular tone and strength. If this is unsuccessful, then surgical exploration is indicated, which may reveal hypertrophy of the transverse scapular ligament, anomalies of the suprascapular notch, and ganglion cysts. Results of surgery vary with the lesion discovered, but many patients return to full function postoperatively.
4. Musculocutaneous Nerve Injury
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Weak or absent biceps muscle function with sensory loss in the lateral forearm.
• EMG/NCV may aid in the diagnosis and prognosis.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
This nerve is susceptible to direct frontal blows or surgical procedures. Injury is associated with numbness in the lateral forearm to the base of the thumb and weak to absent biceps muscle function. Most injuries seen in sports are transient and respond to conservative treatment in a matter of days to weeks.
5. Axillary Nerve Injury
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Present following a shoulder dislocation or proximal humerus fracture.
• Weakness or absence of deltoid muscle function.
• Positive deltoid extension lag sign.
• EMG/NCV can aid in diagnosis and prognosis.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
The mechanism of injury may be trauma by direct blow to the posterior aspect of the shoulder, following dislocation or fracture of the proximal humerus, and following surgery. The axillary nerve can be injured especially during arthroscopic capsular release procedures and is most prone to be injured with release of the anteroinferior capsule. The axillary nerve passes caudal to the glenohumeral joint capsule, and its position relative to the capsule varies with shoulder position; its distance from the capsule diminishes with abduction of the shoulder. With the arm at the side, it courses 1–1.5 cm lateral to the anteroinferior glenoid rim at the 5 o’clock position (right shoulder). As the nerve courses posterior, it is located increasing more lateral to the glenoid rim. It is 2–2.5 cm from the posteroinferior glenoid rim at the 7 o’clock position (right shoulder). The degree of injury to the nerve varies because the initial presentation may be mild weakness during elevation and abduction of the arm with or without numbness of the lateral arm. The deltoid extension lag sign is indicative of axillary nerve injury. To perform this test, the examiner elevates the arm into a position of near full extension, asks the patient to hold the arm in this position, and then releases the arm. If there is complete deltoid paralysis, the arm will drop. For partial nerve injuries, the magnitude of the angular drop, or lag, is an indicator of deltoid strength. Approximately 25% of all dislocated shoulder injuries are associated with axillary nerve traction injuries, which respond well to rest, physical therapy, and time. If recovery is not complete within 3–6 months, surgical intervention is recommended with exploration, using neurolysis or grafting, or both, as necessary. Results of surgery are usually favorable, with sensory recovery occurring before motor recovery.
Zarkadas PC, Throckmorton TW, Steinmann SP: Neurovascular injuries in shoulder trauma. Orthop Clin North Am 2008;39:483. [PMID: 18803978]
THORACIC OUTLET SYNDROME
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Symptoms are often nonspecific; may be neurologic, venous, or arterial; and may include edema, pallor, or coolness as well as paresthesias.
• Doppler examination and EMG/NVC studies can assist in the diagnosis.
![]() Clinical Findings, Treatment, and Prognosis
Clinical Findings, Treatment, and Prognosis
The symptoms resulting from thoracic outlet compression may be neurologic, venous, or arterial in nature. Obstruction of the subclavian vein may lead to stiffness, edema, and even thrombosis of the limb. Arterial obstruction may be the result of direct compression and manifests with pallor, coolness, and forearm claudication. Doppler examination reveals changes in arterial and venous flow. EMG and nerve conduction studies are also helpful in diagnosis.
Nonoperative treatment is recommended for less severe forms of this syndrome, and once the pain subsides, an exercise program to strengthen the pectoral girdle muscles is beneficial. Special exercises to strengthen the upper and lower trapezius, along with the erector spinae and serratus anterior muscles, yield good results. Correcting poor posture and an ongoing maintenance program are mandatory once improvement is reached. Progression of symptoms or failure of non-operative treatment is an indication for surgical exploration and correction of the pathologic factors encountered.
Laulan J, Fouquet B, Rodaix C, Jauffret P, Roquelaure Y, Descatha A: Thoracic outlet syndrome: definition, aetiological factors, diagnosis, management and occupational impact. J Occup Rehabil. 2011;21:366. [Epub ahead of print][PMID: 21193950]
![]() ELBOW INJURIES
ELBOW INJURIES
EPICONDYLITIS (TENNIS ELBOW)
Tennis elbow is an eponym given to many painful conditions about the elbow. An anatomic location may usually be found and specific diagnosis made.
1. LATERAL EPICONDYLITIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• A history of repetitive activity or overuse.
• Pain localized to the lateral elbow that may radiate to the forearm.
• Tenderness at the extensor carpi radialis brevis origin, just anterior and distal to the center of the lateral epicondyle.
• Normal elbow range of motion.
• Usually normal radiographs and rarely (<10%) calcification adjacent to the lateral epicondyle.
Lateral epicondylitis is commonly known as tennis elbow and involves the tendons of the extensor muscles of the wrist and hand. Patients who perform repetitive wrist extension against resistance (such as the backhand stroke in tennis) are at risk. The pain they have is usually chronic in nature and more bothersome than disabling. Tenderness is located over the lateral humeral epicondyle, and pain is produced by extending the wrist against resistance. This is most marked when done with the elbow extended. The tendon of the extensor carpi radialis brevis is the most common site of the lesion. Other causes for lateral elbow pain should be considered, including radiocapitellar arthritis and posterior inter-osseous nerve compression. Radiographs only rarely reveal soft-tissue calcification near the lateral humeral epicondyle, and MRI is of questionable aid in making the diagnosis.
Treatment includes decreasing specific activities and using a counterforce brace that theoretically distributes the tension of the muscular pull over a larger area. A lighter racquet, proper grip size on the racquet, and proper technique are also helpful. Stretching the extensor and supinator muscles is done by flexing the wrist of the upper extremity with the elbow extended. It is often helpful to pronate the forearm. Exercises to strengthen the wrist extensor muscles should be included. If this approach fails, an injection of local anesthetic and cortisone into the most tender region is often curative. Surgical treatment yields satisfactory outcomes in recalcitrant cases, and multiple procedures, including arthroscopic techniques, have been described. Commonplace in all procedures is release of the common extensor origin. Histologic studies of the afflicted tendon show degenerative changes with angiofibroblastic proliferation. These are thought to be similar to the pathologic changes of the torn rotator cuff, with diminished vascularity, an altered nutritional state, and tearing of the susceptible tendon.
2. MEDIAL EPICONDYLITIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• A history of repetitive activity or overuse, often from activities that result in valgus elbow forces, such as occurs with baseball throwing.
• Pain localized to the medial elbow that may radiate to the forearm
• Tenderness at the flexor-pronator origin, most commonly of the pronator teres and the flexor carpi ulnaris.
• Normal elbow range of motion.
• Usually normal radiographs and rarely (<10%) calcification adjacent to the medial epicondyle.
Medial epicondylitis involves the common flexor pronator origin and is commonly known as golfer’s elbow. Treatment is similar to the management of lateral epicondylitis, although with emphasis on the wrist flexors and the forearm pronators. Ulnar nerve compression at the elbow may occur in conjunction with medial tennis elbow. In about 60% of the cases treated surgically, ulnar nerve compression was present. The common flexor origin is an important medial stabilizer of the elbow, so if surgical treatment is indicated, the debrided tendon should be reattached rather than released from the medial epicondyle.
Baker CL Jr, Baker CL 3rd: Long-term follow-up of arthroscopic treatment of lateral epicondylitis. Am J Sports Med 2008;36:254. [PMID: 18202296]
Calfee RP, Patel A, DaSilva MF, Akelman E: Management of lateral epicondylitis: current concepts. J Am Acad Orthop Surg 2008;16:19. [PMID: 18180389]
Coombes BK, Bisset L, Vicenzino B: Efficacy and safety of corticosteroid injections and other injections for management of tendinopathy: a systematic review of randomised controlled trials. Lancet2010;376:1751. [PMID: 20970844]
![]() CPT Codes for Lateral and Medial Epicondylitis
CPT Codes for Lateral and Medial Epicondylitis
24357 Tenotomy, elbow, lateral or medial (eg, epicondylitis, tennis elbow, golfer’s elbow); percutaneous
24358 Tenotomy, elbow, lateral or medial (eg, epicondylitis, tennis elbow, golfer’s elbow); debridement, soft tissue and/or bone, open
24359 Tenotomy, elbow, lateral or medial (eg, epicondylitis, tennis elbow, golfer’s elbow); debridement, soft tissue and/or bone, open with tendon repair or reattachment
ELBOW INSTABILITY
Rupture of the collateral ligaments of the elbow occurs most commonly from elbow dislocation. This can result from excessive valgus force, and initially, the ulnar collateral ligament ruptures. Excessive posterolateral rotatory force may also result in rupture of the lateral ulnar collateral ligament. In either case, the elbow may dislocate, and typically the direction is posterior. Treatment after relocation and brief immobilization consists of active range of motion exercises. Recurrent instability is rare; instead, a small loss of elbow extension, usually less than 10 degrees, commonly results.
1. Valgus Instability
![]() Essential of Diagnosis
Essential of Diagnosis
• Sudden or gradual onset of medial elbow pain after throwing.
• Pain greatest at late cocking and acceleration phase of throwing.
• Maximum tenderness about 1 cm distal to the medial epicondyle.
• Provocative maneuvers that place valgus stress on elbow reproduce symptoms.
• There may also be ulnar neuropathy and posteromedial elbow impingement, medial epicondylitis, and cubital tunnel syndrome.
Valgus instability may result from overuse in overhead throwing sports such as baseball, football, and javelin throwing. With acute MCL rupture, a pop may be felt during a throw. Tenderness is present on the medial side of the elbow, usually just distal to the medial epicondyle. Instability can then be appreciated when a valgus force is applied to the elbow. This must be done with the elbow flexed 20 degrees, as failure to unlock the olecranon from within the olecranon fossa in full extension creates a false sense of stability. Comparison to the contralateral side aids in making the correct diagnosis. If the ulnar collateral ligament has been injured but remains intact, then the valgus stress test may elicit pain, but no instability. Then the “milking maneuver” (Figure 3–38) will also elicit pain along the medial side of the elbow. Eliciting pain while moving the elbow in flexion and extension with valgus stress during the milking maneuver may be the best test for diagnosing MCL injuries in the elbow.

![]() Figure 3–38. The valgus stress and milking maneuver tests for medial ulnar collateral ligament injury. (Reprinted, with permission, from Chen FS, Rokito AS, Jobe FW: Medial elbow problems in the overhead-throwing athlete. J Am Acad Orthop Surg 2001;9:102.)
Figure 3–38. The valgus stress and milking maneuver tests for medial ulnar collateral ligament injury. (Reprinted, with permission, from Chen FS, Rokito AS, Jobe FW: Medial elbow problems in the overhead-throwing athlete. J Am Acad Orthop Surg 2001;9:102.)
A stress radiograph may aid in making the diagnosis. An anteroposterior radiograph can be taken while the examiner performs the valgus stress test. Alternatively, gravity can be used to apply the valgus stress. For this, an anteroposterior radiograph of the elbow is taken with the shoulder externally rotated at 90 degrees with the elbow flexed at approximately 20 degrees. When instability is present, there will be a wider medial opening than on the contralateral normal side. MRI may also be useful, especially if an arthrogram is performed concurrently, as dye leaking through the ulnar collateral ligament is diagnostic of a rupture.
Surgical repair may be indicated in overhead throwing athletes who suffer an acute rupture of their ulnar collateral ligament and still want to continue to participate in their sport. Soccer, basketball players, and other athletes participating in nonoverhead throwing sports may be treated with a program of early active range-of-motion exercises with expectation of full return to their sport. Chronic ulnar collateral ligament injuries resulting from overuse are best treated with rehabilitation, NSAIDs, and avoidance of throwing for as long as 3 months. Only those with residual pain and instability after participation in such a program should undergo reconstruction of the anterior band of the ulnar collateral ligament. In this surgery pioneered by Dr Frank Jobe, a tendon graft, usually the palmaris longus, is used to reconstruct the ligament from the anterior and distal aspect of the medial humeral epicondyle to the sublime tubercle of the ulna. Up to 85% of athletes are able to return to highly competitive throwing after such surgery.
Cain EL Jr, Andrews JR, Dugas JR, et al: Outcome of ulnar collateral ligament reconstruction of the elbow in 1281 athletes: results in 743 athletes with minimum 2-year follow-up. Am J Sports Med2010;38:2426. [PMID: 20929932 ]
Murthi AM, Keener JD, Armstrong AD, Getz CL: The recurrent unstable elbow: diagnosis and treatment. J Bone Joint Surg Am 2010;92:1794. [PMID: 20660245]
2. Posterolateral Rotatory Instability
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Sudden or gradual onset of lateral elbow pain.
• There may be snapping, catching, locking, or a sensation of elbow instability.
• A history of tennis elbow surgery.
• The provocative maneuver of the posterolateral rotator instability test reproduces symptoms.
Posterolateral rotatory instability of the elbow may result from a fall on the outstretched upper extremity, surgery of the lateral side of the elbow, or chronic varus stress as may occur in long-term crutch walkers. The instability covers a spectrum of severity from mild subluxation to recurrent dislocation. Those with mild forms complain of intermittent symptoms on the lateral side of the elbow associated with supination of the forearm such as pain, snapping, or catching. More severe symptoms include locking or sensations of elbow instability. To perform the posterolateral rotatory instability test, a valgus stress is applied to the supinated elbow with the patient supine and the upper extremity over the head (Figure 3–39). Subluxation of the radial head occurs with the elbow in extension and resolves when the elbow is flexed. This maneuver also reproduces the patient’s symptoms. A lateral stress radiograph, done with the elbow in extension as described for the posterolateral rotatory instability test, may also demonstrate the instability (see Figure 3–39). Treatment for acute cases consists of an elbow brace to hold the forearm in pronation and restrict terminal elbow extension for a 6-week period of time. Chronic cases are best treated with reconstruction of the lateral ulnar collateral ligament. Postoperatively, the patient is put in the same brace as used for acute posterolateral rotatory instability for 6–12 weeks.

![]() Figure 3–39. The posterolateral rotatory instability test reproduces the patient’s symptoms. At right is a lateral stress radiograph.
Figure 3–39. The posterolateral rotatory instability test reproduces the patient’s symptoms. At right is a lateral stress radiograph.
Charalambous CP, Stanley JK: Posterolateral rotatory instability of the elbow. J Bone Joint Surg Br 2008;90:272. [PMID: 18310745]
![]() CPT Codes for Elbow Instability
CPT Codes for Elbow Instability
24343 Repair lateral collateral ligament, elbow, with local tissue
24344 Reconstruction lateral collateral ligament, elbow, with tendon graft (includes harvesting of graft)
24345 Repair medial collateral ligament, elbow, with local tissue
24346 Reconstruction medial collateral ligament, elbow, with tendon graft (includes harvesting of graft)
CUBITAL TUNNEL SYNDROME
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Medial elbow pain exacerbated by throwing and paresthesias in ring and little fingers.
• Positive Tinel sign over cubital tunnel and positive elbow flexion test.
• Associated with both medial epicondylitis and MCL injury.
Prevention relies on sound throwing techniques, which minimize valgus load on elbow. Correction of known causes of nerve irritation including valgus instability of the elbow may also prevent the syndrome. The cubital tunnel is formed by the medial epicondyle, the elbow joint, and the two heads of the flexor carpi ulnaris. Structures proximal to, within, and distal to the tunnel can cause compression, entrapment, traction, subluxation, or irritation of the ulnar nerve. Proximally, these structures include the arcade of Struthers (not to be confused with the ligament of Struthers, which is associated with median neuropathy) and medial head of the triceps. Within the groove, they include the medial epicondyle, the epicondylar groove, anconeus epitrochlearis, the two heads of the flexi carpi ulnaris, and their interconnecting ligament of Osborne. Distally, an offending structure may be the deep flexor-pronator fascia. Regardless of cause or site, the final common pathway of cubital tunnel syndrome is the onset of nerve ischemia and fibrosis.
Initial symptoms may be medial elbow pain with occasional radiation to the medial forearm. Paresthesias may occur in the ulnar two fingers. Athletes will often present before onset of weakness. Mechanical complaints such as snapping may occur with nerve subluxation. Diagnosis is chiefly clinical and relies on two provocative tests: a positive Tinel sign over the nerve and a positive elbow flexion test. This test is performed by placing the elbow in full flexion and the wrist in maximal extension. A test is positive if pain or paresthesias are elicited within 1 minute. Sensory changes can be detected with Semmes-Weinstein monofilament testing and, in more advanced cases, with two-point discrimination tests. Motor deficits often occur late, and athletes usually complain before asymmetric hypothenar atrophy, decreased pinch and grip strength, abducted small finger or Wartenberg sign, Froment sign, and clawing of the ulnar two fingers occur. Motor deficits may not be present, even in late cases, if the intrinsic muscles of the hand receive innervation from the median nerve—the result of an anatomic variant known as the Martin-Gruber anastomosis.
A thorough exam of the neck and proximal upper extremity is performed to eliminate neuropathic etiologies with similar manifestations such as cervical radiculopathy, brachial plexopathy (of the medial cord), and thoracic outlet syndrome.
Plain radiographs including special views such as cubital tunnel view may reveal bony abnormalities causing ulnar nerve compression. Similarly, MRI may identify soft-tissue abnormalities having the same effect. EMG and NCV studies will be negative in more than 50% of patients with the syndrome. Slowing of conduction velocities to less than 50 m/s when the elbow is flexed is indicative of disease. Reduction in sensory nerve action potential also confirms early neuropathy.
Treatment of ulnar neuropathy at the cubital tunnel is initially nonoperative: rest, ice, anti-inflammatory medication, and padded splinting at 30–45 degrees of elbow flexion. Nighttime extension splinting is often quite helpful in reducing symptoms early in the disease process. Corticosteroid injections are not recommended due to the superficial position of the nerve. Conservative management will often fail in athletes due to the high demands, especially if there is a subluxing ulnar nerve. Surgical indications include failed non-operative management and ulnar nerve subluxation. Several techniques have been used, including simple decompression, medial epicondylectomy, subcutaneous transposition, and submuscular transposition. With each technique, the ulnar nerve should be released at all possible sites of compression, from the ligament of Struthers proximally, through the cubital tunnel, and past the two heads of the flexor carpi ulnaris. Each technique has potential complications and approximately an 85% success rate. After brief postoperative immobilization, passive and then active range of motion is begun (by 4 weeks). Strengthening and throwing exercises are started by the eighth postoperative week. Complications are uncommon but include injury to the medial antebrachial cutaneous nerve, injury to the MCL complex, and perineural scarring. Unrecognized medial elbow pathology will also limit success.
Conservative therapy has excellent results except in high-demand athletes. Results of surgical intervention vary inversely with degree of preoperative nerve involvement. Many patients with good to excellent results return to unrestricted play by 6 months postoperatively. This can be a career-ending insult to a throwing athlete if the pathology has been present for a prolonged time prior to treatment.
Gellman H: Compression of the ulnar nerve at the elbow: cubital tunnel syndrome. Instr Course Lect 2008;57:187. [PMID: 18399580]
![]() CPT Codes for Cubital Tunnel Syndrome
CPT Codes for Cubital Tunnel Syndrome
64718 Neuroplasty and/or transposition; ulnar nerve at elbow
BICEPS TENDON TEARS AT THE ELBOW
![]() Essentials of Diagnosis
Essentials of Diagnosis
• “Popeye” appearance of the upper arm from proximal retraction of the biceps muscle.
• May or may not be painful and ecchymotic depending on the chronicity of the injury.
• Weakness of elbow flexion and forearm supination.
![]() Prevention
Prevention
General conditioning and stretching and strengthening before activities can help minimize injury to the biceps tendon.
![]() Clinical Findings
Clinical Findings
The long head of the biceps tendon may rupture distally at the insertion into the radial tuberosity, at the myotendinous junction, or within the muscle itself. The muscle mass moves proximally, producing a bulging or “Popeye” appearance to the arm. Rupture of the biceps tendon distally involves both heads. The mechanism is usually a forceful flexion of the arm and is more common in older athletes or following direct trauma. Microtears probably serve to render the tendon vulnerable to an acute tearing event. The degree of ecchymosis is dependent on the location of the tear, with avascular areas having less ecchymosis and the musculotendinous junction producing quite a noticeable amount of ecchymosis. Diagnosis is usually easily accomplished, as the deformity is obvious.
![]() Treatment
Treatment
In older individuals, nonoperative treatment is an option, although elbow flexion and forearm supination remain weak. In others, rupture of the distal biceps tendon often warrants surgical repair and is effective in restoring strength of elbow flexion and forearm supination. The tendon is easily found after acute injury, about 6 cm above the elbow joint, and care must be taken to avoid damage to the lateral antebrachial cutaneous nerve. After approximately 3 months, the tendon may be coiled up and scarred, and restoration of normal muscle may be very difficult. Surgical repair has traditionally been done with a twoincision technique, and the tendon that avulsed from bone is sewed back though drill holes. Newer, one-incision techniques using suture anchors are equally effective.
![]() Prognosis
Prognosis
Athletes are permitted to return to full-contact play once healing is sufficient and they have achieved maximal functional strength and range of elbow motion, which typically occurs 4–6 months following a distal biceps repair. Complications include loss of elbow range of motion, heterotopic bone, rerupture, nerve injuries (particularly the posterior interosseous portion of the radial nerve), and synostosis of the radius and ulna.
Frazier MS, Boardman MJ, Westland M, Imbriglia JE: Surgical treatment of partial distal biceps tendon ruptures. J Hand Surg Am 2010;35:1111. [PMID: 20610056]
Vidal AF, Drakos MC, Allen AA: Biceps tendon and triceps tendon injuries. Clin Sports Med 2004;23:707. [PMID: 15474231]
![]() CPT Codes for Biceps Tendon Tears at the Elbow
CPT Codes for Biceps Tendon Tears at the Elbow
24340 Tenodesis of biceps tendon at elbow (separate procedure)
24342 Reinsertion of ruptured biceps or triceps tendon, distal, with or without tendon graft
OTHER ELBOW OVERUSE INJURIES
1. Posterior and Posteromedial Elbow Impingement
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Posterior elbow pain during terminal extension of the throwing arm or posteromedial elbow pain during the acceleration phase of throwing.
• Loss of elbow extension and tenderness along posterior or posteromedial olecranon.
• Pain elicited with forced rapid elbow extension in presence of valgus load.
• Posterior and posteromedial olecranon osteophyte sometimes visible on plain films.
• Associated with valgus extension overload and valgus instability.
Impingement may result from mechanical abutment of bone and soft tissues in the posterior elbow. This may or may not be associated with injury of the ulnar collateral ligament.
Hyperextension injuries with an intact ulnar collateral ligament occur in gymnasts, football lineman, weightlifters, and others. The lesion is usually located in the center of the posterior elbow, and the pain is reproduced by forcible extension of the elbow. If there is insufficiency of the ulnar collateral ligament, as is often the case when there is posterior elbow impingement in overhead athletes, the lesion is posteromedial. In this case, the impingement is between the medial aspect of the olecranon and the lateral side of the medial wall of the olecranon fossa (Figure 3–40). Pain may be reproduced with the valgus stress test, as described earlier for valgus instability, but the pain is posteromedial and medial. Radiographs may demonstrate osteophytes of the olecranon fossa.

![]() Figure 3–40. Mechanism of posteromedial impingement between the medial aspect of the olecranon and the lateral side of the medial wall of the olecranon fossa. (Reprinted, with permission, from Chen FS, Rokito AS, Jobe FW: Medial elbow problems in the overhead-throwing athlete. J Am Acad Orthop Surg 2001;9:105.)
Figure 3–40. Mechanism of posteromedial impingement between the medial aspect of the olecranon and the lateral side of the medial wall of the olecranon fossa. (Reprinted, with permission, from Chen FS, Rokito AS, Jobe FW: Medial elbow problems in the overhead-throwing athlete. J Am Acad Orthop Surg 2001;9:105.)
As with most injuries caused by repetitive trauma, treatment begins with prevention. The number of innings pitched is probably the most important factor relating to injury. If symptoms persist, removal of osteophytes is successful treatment, provided that no ulnar collateral ligament injury is present. Treatment of the valgus instability is also required for successful outcome.
Moskal MJ, Savoie III FH, Field LD: Arthroscopic treatment of posterior elbow impingement. Instr Course Lect 1999;48:399. [PMID: 10098066]
Sellards R, Kuebrich C: The elbow: diagnosis and treatment of common injuries. Prim Care 2005;32:1. [PMID: 15831310]
2. Fatigue Fracture of the Medial Epicondyle
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Sudden onset of medial epicondyle pain and swelling from throwing.
In children, fatigue fractures of the medial epicondyle cause pain and swelling. This has been blamed on throwing curve balls, but some studies have shown that a properly thrown curve ball causes no more injuries than the traditional fast-ball. Prevention or minimization of damage involves several steps. First, it is important to maintain proper conditioning by continuing pitching practice in the off season or beginning the baseball season in a slow progressive fashion. Second, pain and inflammation should be avoided, and if the elbow becomes painful, the athlete should stop throwing immediately. An accurate pitching count should be kept during a game, and a stopping point should be planned in advance. If the pitcher begins having pain or shows loss of control, pitching should be temporarily terminated, and treatment to decrease the swelling and inflammation should begin. No competitive throwing is allowed until full range of motion returns and no pain or tenderness is associated with throwing.
3. Osteochondritis Dissecans of the Capitellum
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Gradual onset of lateral elbow pain in overhead athletes and gymnasts.
• Catching or locking of the elbow.
• Grinding of the lateral elbow.
• There may be loss of range of motion, especially if there is an effusion or there are associated loose bodies.
Osteochondritis dissecans of the capitellum affects pitchers over 10 years of age (Figure 3–41) and gymnasts sometimes at an earlier age. Changes in the radiocapitellar joint are very worrisome because of possible permanent loss of function. Many surgical procedures have been described, but if fragmentation occurs, loose bodies may require excision. Recently, some have tried using osteochondral transplants to “repair” the lesion.

![]() Figure 3–41. Anteroposterior view of an elbow with osteochondritis dissecans of the capitellum.
Figure 3–41. Anteroposterior view of an elbow with osteochondritis dissecans of the capitellum.
Rahusen FT, Brinkman JM, Eygendaal D: Arthroscopic treatment of posterior impingement of the elbow in athletes: a medium-term follow-up in sixteen cases. J Shoulder Elbow Surg 2009;18:279. [PMID: 19218052]
Ruchelsman DE, Hall MP, Youm T: Osteochondritis dissecans of the capitellum: current concepts. J Am Acad Orthop Surg 2010;18:557. [PMID: 20810937]
![]() CPT Codes for Posterior and Posteromedial Elbow Impingement and Osteochondritis Dissecans of the Capitellum
CPT Codes for Posterior and Posteromedial Elbow Impingement and Osteochondritis Dissecans of the Capitellum
29834 Arthroscopy, elbow, surgical; with removal of loose body or foreign body
29837 Arthroscopy, elbow, surgical; debridement, limited
29838 Arthroscopy, elbow, surgical; debridement, extensive
![]() SPINE INJURIES
SPINE INJURIES
CERVICAL SPINE INJURY
Cervical spine injuries in athletes are relatively infrequent, but the potential for serious injury to the nervous system exists. If spine injury is suspected, it is wise to be extremely cautious until a proper diagnosis can be made. This is the best way to prevent a repairable injury from becoming a catastrophe. Most often, a spine injury results from a collision and sometimes includes associated head injuries. The head and neck must be immobilized immediately, and ease of breathing and level of consciousness ascertained immediately.
1. Brachial Plexus Neuropraxia
The most common cervical injury is pinching or stretching neuropraxia of the nerve root and brachial plexus. The injury is of short duration, and the patient has a full pain-free range of motion of the neck. These injuries are commonly called “stinger” or “burner” injuries. They result from lateral impact of the head and neck with simultaneous depression of the shoulder. This may cause stretching and pinching of the nerves of the brachial plexus, with burning pain, numbness, and tingling extending from the shoulder down into the hand and arms. Symptoms frequently involve the C5 and C6 root levels. Usually, recovery is spontaneous within a few minutes after the acute episode.
Patients who demonstrate full muscle strength of the intrinsic muscles of the shoulder and upper extremity and have full pain-free range of motion of the cervical spine may return to their activities. If they have residual weakness or numbness, they should not be allowed to reenter the game. Absence of neck pain should alert one to the possibility of a cervical spine injury, as neck pain is not part of the syndrome.
Persistence of paresthesia or weakness requires further evaluation before returning to play. This includes neurologic, electromyographic, and radiographic evaluation. The athlete should not participate in contact sports until full muscle strength has been achieved and a repeat electromyogram shows evidence of axonal regeneration, usually at least 4–6 weeks.
Prevention of “stinger” injuries is chiefly through correct head and neck techniques and strengthening of the neck musculature. Additionally, the use of cervical rolls may eliminate extremes of motion during impact.
2. Cervical Strain
Acute strains of the muscles of the neck are probably the most frequent cervical injuries in athletes. The word strain implies injury to a muscle, whereas a sprain is a ligamentous injury. A strain happens when a muscle tendon unit is overloaded or stretched. The clinical picture is common to all musculotendinous injuries. Motion of the neck becomes painful, reaching a peak after several hours or the next day. Anti-inflammatory medication, heat, massage, and other modalities are beneficial.
3. Cervical Sprain
With cervical sprain, there has been damage to the ligamentous and capsular structure connecting the facet joints and vertebra. It is often difficult to differentiate from a strain. There is limited motion and pain in the area of the injury and along the muscle groups overlying the area of the injury. Ligamentous disruption may be extensive enough to result in instability with associated neurologic involvement. Routine cervical spine radiographs are indicated. In athletes with diminished motion as well as pain, stability of the cervical spine should be documented. This may be done with flexion and extension radiographs.
Treatment of a cervical sprain consists of immobilization, rest, support, and anti-inflammatory therapy. Return to participation is permitted when motion and muscle strength normalize.
4. Cervical Spinal Cord Neuropraxia With Transient Tetraplegia
The phenomenon of cervical spinal cord neuropraxia with transient tetraplegia is a distinct clinical entity. Sensory changes include a burning pain, numbness, tingling, or loss of sensation. Motor changes include weakness or complete paralysis, which is usually transient, with complete recovery occurring in 10–15 minutes, although in some cases, gradual resolution occurs over 36–48 hours. Complete motor function and full pain-free cervical motion return. Routine radiographs of the cervical spine are negative for fractures or dislocations. Some radiographic findings include spinal stenosis, congenital fusions, cervical instability, and intervertebral disk disease.
The risks of permanent neurologic injury from cervical stenosis are thought to be significant. The Torg ratio was previously used for diagnosis of cervical stenosis. It is defined as ratio of the anteroposterior diameter of the spinal canal divided by the anteroposterior diameter of the vertebral body (Figure 3–42) being less than 0.80. But this ratio has recently been thought to be of low predictive value. Current methods for diagnosis of cervical spinal stenosis rely on MRI and CT. A cervical canal diameter of less than 13 mm is considered stenotic and less than 10 mm is considered absolute stenosis. Patients who have had neurologic symptoms and are found to have cervical stenosis should not be cleared for contact sports. Those found to have spinal stenosis who have not had symptoms should be treated on an individual basis.

![]() Figure 3–42. The ratio of the spinal canal to the vertebral body is the distance from the midpoint of the posterior aspect of the vertebral body to the nearest point on the corresponding spinolaminar line (A) divided by the anteroposterior width of the vertebral body (B). (Reproduced, with permission, from Torg JS, Pavlov H, Genuario SE, et al: Neurapraxia of the cervical spinal cord with transient quadriplegia. J Bone Joint Surg Am 1986;68:1354.)
Figure 3–42. The ratio of the spinal canal to the vertebral body is the distance from the midpoint of the posterior aspect of the vertebral body to the nearest point on the corresponding spinolaminar line (A) divided by the anteroposterior width of the vertebral body (B). (Reproduced, with permission, from Torg JS, Pavlov H, Genuario SE, et al: Neurapraxia of the cervical spinal cord with transient quadriplegia. J Bone Joint Surg Am 1986;68:1354.)
Athletes who have suffered transient tetraplegia are not known to be at any greater risk for permanent tetraplegia. Patients who have this syndrome and associated instability of the cervical spine or cervical disk disease should be precluded from further participation in contact sports.
More severe injuries, including fractures and dislocation of the cervical spine, may occur. Treatment of these begins on the playing field, with immobilization of the spine. A face mask, if worn, may be cut off with bolt cutters. After thoroughly stabilizing the spine, the patient is moved to a spine board. Sandbags are used to immobilize the head and neck. The patient may then be transported to a local emergency room for further evaluation and treatment. Fractures and dislocations with or without permanent neurologic injury are treated like other spine injuries.
Crowl AC, Kong JF: Cervical spine. In: Johnson DL, Mair SD, eds: Clinical Sports Medicine. Philadelphia: Mosby Elsevier; 2006:143-149.
Dailey A, Harrop JS, France JC: High-energy contact sports and cervical spine neuropraxia injuries: what are the criteria for return to participation? Spine (Phila Pa 1976) 2010;35(21 Suppl): S193. [PMID: 20881462]
Torg JS, Corcoran TA, Thibault LE, et al: Cervical cord neurapraxia: classification, pathomechanics, morbidity, and management guidelines. J Neurosurg 1997;87:843. [PMID: 9384393]
LUMBAR SPINE INJURY
![]() Clinical Findings
Clinical Findings
Spondylolysis is a disruption of the pars interarticularis, while spondylolisthesis involves anterior slippage of one vertebral body over the next. Spondylolysis is most often found at L5 and L4, but may occasionally be seen at L3 and L2. It is believed to result from repeated stress around the pars inter-articularis during hyperextension of the lumbar spine. If continued hyperextension activity occurs, spondylolysis may become spondylolisthesis. Sports in which spondylolisthesis is commonly found include gymnastics, football, and weight lifting. Female teenage gymnasts, for example, often have back pain but normal early radiographs. Approximately 3–6 weeks later, a stress response may be seen around the pars interarticularis, with increased density developing. At this time, the bone scan will be positive, indicating an impending stress fracture that will show up on plain radiographs in 2–4 weeks. A physician who is aware of which sports put stress on the pars interarticularis should consider a bone scan to rule out spondylolisthesis.
![]() Treatment and Prognosis
Treatment and Prognosis
The treatment of spondylolisthesis involves cessation of all aggravating sports and other actions producing spinal hyperextension. A certain percentage of these fractures will heal spontaneously. Healing time for spondylolysis of the lumbar spine is usually about 6 months. If after that period of time no significant signs of healing are apparent, it is unlikely that spontaneous healing will take place. At this point, spinal fusion should be considered, or the patient should be willing to confine his activities to less stressful, pain-free sports.
Many patients with spondylolisthesis engage in high-level sporting activities without significant pain or neurologic deficit. Only a small percentage actually present for evaluation and care. Complete evaluation and treatment recommendations for spondylolisthesis and spondylolysis are found in the section on the spine.
Leone A, Cianfoni A, Cerase A, Magarelli N, Bonomo L: Lumbar spondylolysis: a review. Skeletal Radiol 2011;40:683. [PMID: 20440613]
Milanese S, Grimmer-Somers K: What is adolescent low back pain? Current definitions used to define the adolescent with low back pain. J Pain Res 2010;3:57. [PMID: 21197310]
Purcell L: Causes and prevention of low back pain in young athletes. Paediatr Child Health 2009;14:533. [PMID: 20885805]