Bobby K.B. Tay, MD
Brett A. Freedman, MD
John M. Rhee, MD
Scott D. Boden, MD
Harry B. Skinner, MD, PhD
![]() INFLAMMATORY DISEASES OF THE SPINE
INFLAMMATORY DISEASES OF THE SPINE
Bobby K.B. Tay, MD
RHEUMATOID ARTHRITIS (ICD-9 720.0)
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Up to 71% of patients with rheumatoid arthritis have C-spine involvement.
• C1-C2 instability, basilar invagination, and subaxial subluxation are common disease patterns.
• Inflammatory pannus causes synovial joint destruction.
• Eighty percent of patients are rheumatoid factor positive.
![]() General Considerations
General Considerations
Rheumatoid arthritis is the most common form of inflammatory arthritis. It affects 3% of women and 1% of men. The disease frequently affects the spine. Up to 71% of patients with rheumatoid arthritis show involvement of the cervical spine. The most common patterns of involvement are C1-C2 instability, basilar invagination, and subaxial subluxation (ICD-9 738.4). Of these patterns, both C1-C2 instability and basilar invagination have become less frequently encountered as a result of improvements in pharmacologic therapy. Sudden death associated with rheumatoid arthritis, most probably secondary to brainstem compression or vertebrobasilar insufficiency, is reported.
![]() Pathogenesis
Pathogenesis
The same inflammatory cells that destroy peripheral joints affect the synovium of apophyseal and uncovertebral joints of the spine, causing painful instability with or without neurologic compromise. The pannus, a conglomeration of hypertrophic synovium and inflammatory cells, often causes facet joint and transverse ligament destruction, leading to painful instability. The hypertrophic tissue can also cause direct compression of the spinal cord and nerve roots at the affected levels.
![]() Prevention
Prevention
Prevention of rheumatoid instability centers around control of the inflammatory component of the disease. The standard pharmacotherapeutic strategy initially involves the use of anti-inflammatory medication and ends the application of DMARDs.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
From 7 to 34% of patients present with neurologic problems. Documentation of neurologic function can be difficult because loss of joint mobility leads to general muscle weakness. Although many patients complain of nonspecific neck pain, atlantoaxial subluxation is the most common cause of pain in the upper neck, occiput, and forehead in patients with rheumatoid arthritis. Symptoms are aggravated by motion. Increasing compression of the spinal cord results in severe myelopathy with gait abnormalities, weakness, paresthesias, and loss of dexterity. Findings may also include Lhermitte sign (a tingling or electrical feeling that occurs in the arms, legs, or trunk when the neck is flexed), increased muscle tonus of the upper and lower extremities, and pathologic reflexes.
B. Imaging Studies
Instability of the upper cervical spine is determined on lateral flexion-extension radiographs. An atlantodens interval (ADI) that exceeds 3.5 mm is abnormal. Subluxation with an ADI of 10–12 mm indicates disruption of all supporting ligaments of the atlantoaxial complex (transverse and alar ligaments). The spinal cord in this position is compressed between the dens and the posterior arch of C1. Although the ADI is an important measurement for traumatic instability of the C1-C2 complex, the posterior atlantodens interval (PADI) is more prognostic to assess neurologic compromise. The PADI is a direct measure of the space available for the spinal cord at the C1-C2 level. The PADI is measured from the posterior aspect of the odontoid process to the nearest posterior structure (the foramen magnum or the posterior ring of the atlas). If the space available for the spinal cord is less than 13 mm, the likelihood that the patient will develop myelopathy is extremely high.
Cranial settling is present in 5–32% of patients. The odontoid process should not project more than 3 mm above the Chamberlain line, which is a line between the hard palate and the posterior rim of the foramen magnum. The tip of the dens should not project more than 4.5 mm above the McGregor line, which is a line connecting the posterior margin of the hard palate to the occiput. The Clark classification divides the axis into thirds in the sagittal plane. In severe cases of cranial settling, the anterior arch of C1 moves from station 1 (the upper third of C2) to station 3 (the lower third of C2). Neurologic compromise occurs as a result of impingement of the dens into the brainstem and the upper cervical spinal cord. The vertebral arteries can also become occluded as they course between the dens and the foramen magnum to enter the skull.
Lateral subluxation and posterior atlantoaxial instability are less frequent. From 10 to 20% of patients with rheumatoid arthritis present with subaxial subluxation. Erosion of the facet joints and narrowing of the disks leads to subtle anterior subluxations often found on several levels. This results in the characteristic so-called stepladder deformity that occurs most commonly at the C2-C3 and C3-C4 levels.
C. Laboratory Studies
Rheumatoid factor is positive in up to 80% of patients. The erythrocyte sedimentation rate (ESR) rate is elevated and the hemoglobin is decreased in the active phase of the disease. After plain radiographs, which should include lateral flexion-extension views, magnetic resonance imaging (MRI) is the study of choice to evaluate the degree of neural compression and deformity.
![]() Differential Diagnosis
Differential Diagnosis
• Osteoarthritis
• Other inflammatory arthritides
![]() Complications
Complications
Untreated instability can lead to loss of neurologic function, paralysis, and sudden death. Medical treatment using disease-modifying antirheumatoid drugs (DMARDs) can cause immunosuppression and lead to a higher risk of contracting infections. Complications of surgical treatment include a higher rate of infection, poor wound healing, lower fusion rate, and potentially a higher rate of instrumentation failure due to poor bone quality.
![]() Treatment
Treatment
Indications for surgery are severe neck pain and increasing loss of neurologic function. Most commonly, a posterior arthrodesis between C1 and C2 is performed (CPT 22590). A Gallie type or Brooks type of fusion can be done, or posterior transarticular screw fixation (CPT 22840) can be used (Figure 4–1). The latter obviates the need for postoperative halo immobilization. In cases of basilar invagination (cranial settling), extension of the fusion to the occiput (CPT 22590) is necessary. Preoperative halo traction (CPT 20661) is often required to reduce the subluxation or pull the odontoid process out of the foramen magnum. Often a suboccipital craniectomy (CPT 61343) is necessary to decompress the brainstem adequately. Good fixation can be obtained through the use of plate-screw and rod-screw constructs (CPT 22842). Subaxial subluxation with spinal cord compression should be treated with decompression and stabilization with spinal fusion. This can be done most easily via a posterior approach with laminectomy and posterior instrumented fusion or a combined anterior and posterior decompression and fusion for patients with poor bone stock or significant sagittal plane deformity (Figure 4–2).

![]() Figure 4–1. (A and B) Anteroposterior and lateral radiographic images of a 50-year-old woman with rheumatoid arthritis who suffered a fracture of the odontoid treated with posterior C1-C2 fusion with a transarticular screw construct on the right and a C1 lateral mass/C2 translaminar screw construct on the left.
Figure 4–1. (A and B) Anteroposterior and lateral radiographic images of a 50-year-old woman with rheumatoid arthritis who suffered a fracture of the odontoid treated with posterior C1-C2 fusion with a transarticular screw construct on the right and a C1 lateral mass/C2 translaminar screw construct on the left.


![]() Figure 4–2. (A and B) Preoperative anteroposterior (AP) and lateral images of a 58-year-old man with inflammatory arthritis demonstrating severe joint damage and subaxial subluxation. (C and D) Postoperative AP and lateral radiographic images after treatment with anterior and posterior decompression and reconstruction with fusion.
Figure 4–2. (A and B) Preoperative anteroposterior (AP) and lateral images of a 58-year-old man with inflammatory arthritis demonstrating severe joint damage and subaxial subluxation. (C and D) Postoperative AP and lateral radiographic images after treatment with anterior and posterior decompression and reconstruction with fusion.
Borenstein D: Inflammatory arthritides of the spine: surgical versus nonsurgical treatment. Clin Orthop Relat Res 2006;443:208. [PMID: 16462444]
Caird J, Bolger C: Preoperative cervical traction in cases of cranial settling with halo ring and Mayfield skull clamp. Br J Neurosurg 2005;19:488. [PMID: 16574561]
Gluf WM, Schmidt MH, Apfelbaum RI: Atlantoaxial transarticular screw fixation: a review of surgical indications, fusion rate, complications, and lessons learned in 191 adult patients. J Neurosurg Spine2005;2:155. [PMID: 15739527]
Higashino K, Sairyo K, Katoh S, Nakano S, Enishi T, Yasui N: The effect of rheumatoid arthritis on the anatomy of the female cervical spine: a radiological study. J Bone Joint Surg Br 2009;91:1058. [PMID: 19651834]
Kauppi MJ, Neva MH, Laiho K, et al: Rheumatoid atlantoaxial subluxation can be prevented by intensive use of traditional disease modifying antirheumatic drugs. J Rheumatol 2009;36:273. [PMID: 19132793]
Kim DH, Hilibrand AS: Rheumatoid arthritis in the cervical spine. J Am Acad Orthop Surg 2005;13:463. [PMID: 16272271]
Paus AC, Steen H, Rislien J, Mowinckel P, Teigland J: High mortality rate in rheumatoid arthritis with subluxation of the cervical spine: a cohort study of operated and nonoperated patients. Spine (Phila Pa 1976) 2008;33:2278. [PMID: 18784629]
Ronkainen A, Niskanen M, Auvinen A, Aalto J, Luosujrvi R: Cervical spine surgery in patients with rheumatoid arthritis: long-term mortality and its determinants. J Rheumatol 2006;33:517. [PMID: 16511921]
Wolfs JF, Kloppenburg M, Fehlings MG, van Tulder MW, Boers M, Peul WC: Neurologic outcome of surgical and conservative treatment of rheumatoid cervical spine subluxation: a systematic review. Arthritis Rheum2009;61:1743. [PMID: 19950322]
Wollowick AL, Casden AM, Kuflik PL, Neuwirth MJ: Rheumatoid arthritis in the cervical spine: what you need to know. Am J Orthop (Belle Mead NJ) 2007;36:400. [PMID: 17849024]
ANKYLOSING SPONDYLITIS (ICD-9 720.0)
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Seronegative spondyloarthropathy.
• Juvenile ankylosing spondylitis has a predisposition to hip involvement.
• Unlike rheumatoid arthritis, males are more often affected than females.
• Eighty-eight percent to 96% of patients with ankylosing spondylitis are HLA-B27 positive.
![]() General Considerations
General Considerations
Ankylosing spondylitis is a chronic seronegative inflammatory disease that affects the axial skeleton, especially the sacroiliac joints, hip joints, and spine. Extraskeletal involvement is found in the aorta, lung, and uvea. The incidence of ankylosing spondylitis is 0.5–1 per 1000 people. Although males are affected more frequently than females, mild courses of ankylosing spondylitis are more common in the latter. The disease usually has its onset during early adulthood. However, juvenile ankylosing spondylitis affects adolescents (younger than 16 years) and has a predisposition to hip involvement.
![]() Pathogenesis
Pathogenesis
The human leukocyte antigen (HLA)-B27 surface antigen is found in 88–96% of patients, and investigators postulate that an endogenic component (ie, HLA-B27) and an exogenic component (eg, Klebsiellaor Chlamydia) are responsible for triggering of the disease process. The ESR is elevated in up to 80% of the cases but does not accurately reflect disease activity. The serum creatine phosphokinase (CPK), however, is a good indicator of the severity of the disease process.
![]() Prevention
Prevention
No preventive measures are available to avoid developing the disease. DMARDs may be useful in treating the pain that is associated with the inflammatory stage of the disease process. These include tumor necrosis factor-alpha (TNF-α) antagonists. Appropriate bracing prior to the onset of spinal ankylosis can minimize or prevent the development of spinal deformity.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
The onset is insidious, with early symptoms including pain in the buttocks, heels, and lower back. Patients complain typically of morning stiffness, the improvement of symptoms with activity during the day, and the return of symptoms in the evening. The earliest changes involve the sacroiliac joints and then extend upward into the spine. Spinal disease results in loss of motion and subsequent loss of lordosis in the cervical and lumbar spine. Synovitis in the early stages leads to progressive fibrosis and ankylosis of the joints during the reparative phase. Enthesitis occurs at the insertion of the annulus fibrosus on the vertebral body with eventual calcification that results in the characteristic “bamboo spine.” The pain from the inflammatory process subsides after full ankylosis of the affected joints occurs. Approximately 30% of patients develop uveitis, and 30% have chest tightness. Limited chest expansion indicates thoracic involvement. Fewer than 5% of patients have involvement of the aorta, characterized by dilation and possible conduction defects. In addition, patients may suffer from renal amyloidosis and pulmonary fibrosis.
B. Imaging Studies
The earliest radiographic changes are visible in the sacroiliac joints. Symmetric bilateral widening of the joint space is followed by subchondral erosions and ankylosis. Bony changes in the spine affect the vertebral body. Changes include loss of the anterior concavity of the vertebral body, squaring of the vertebra, and marginal syndesmophyte formation, which give the spine the appearance of bamboo. Ankylosis of the apophyseal joints also develops. The disease generally starts in the lumbar spine and migrates cephalad to the cervical spine. Atlantoaxial instability is seen occasionally.
![]() Differential Diagnosis
Differential Diagnosis
Early stages of the disease can appear to be similar to other inflammatory spondyloarthropathies.
![]() Complications
Complications
The main complications of untreated cervical spinal deformity include significant loss of functionality from inability to look forward (loss of horizontal gaze). Complications of surgical treatment include infection, paralysis, and C7 or T1 root injury leading to loss of triceps and intrinsic hand function, respectively. Because of medical treatments, these patients are at a higher risk for wound complications and postsurgical infections. TNF-α antagonists should be stopped for at least 2 weeks prior to surgical treatment to minimize the risk for infection. Prolonged use of a halo vest in the postoperative period (up to 6 months) can lead to halo-related complications, including pin site infection and intracranial perforation of the halo pins over time. The patient’s osteoporotic bone (especially in the cervical spine lateral masses) increases the risk of hardware pullout.
![]() Treatment
Treatment
The natural history of ankylosing spondylitis, with its slow progression over several decades, has to be considered in planning treatment. Initially, treatment consists of exercises and indomethacin. Approximately 10% of patients develop severe bony changes that eventually require surgical intervention. These changes characteristically include a fixed bony flexion deformity that limits their ambulatory potential. Hip disease should be addressed before correction of spinal deformities because correction of hip flexion deformities may allow significant compensation of the spinal kyphosis (ICD-9 737.9) to allow adequate horizontal gaze. When planning surgical treatment, it is important to stop treatment with TNF-α inhibitors for at least 2 weeks prior to surgery to minimize the risk for wound infection.
Loss of lumbar lordosis can be treated by multilevel V-shaped osteotomies posteriorly (the Smith-Petersen procedure; CPT 22214), by a decancellation procedure (the Heinig procedure; CPT 22207) of L3 or L4, or by pedicle subtraction osteotomy based at L3 or L4 (Thomassen osteotomy; CPT 22207) (Figure 4–3). The L3-L4 level is used because this correlates with the apex of the normal lumbar lordosis and allows for adequate distal fixation to hold the osteotomy in a stable configuration.

![]() Figure 4–3. Preoperative (A) and postoperative (B) lateral radiographs of a 38-year-old man with cervicothoracic kyphosis and loss of horizontal gaze who was treated with a posterior cervicothoracic osteotomy and instrumented fusion.
Figure 4–3. Preoperative (A) and postoperative (B) lateral radiographs of a 38-year-old man with cervicothoracic kyphosis and loss of horizontal gaze who was treated with a posterior cervicothoracic osteotomy and instrumented fusion.
The spine is then fused in the corrected position. Utilization of modern fixation systems such as a pedicle screw system allows for early mobilization of the patient. Thorough preoperative assessment of the deformity and measuring of the chin-eyebrow-to-floor angle are helpful for the exact planning of the corrective osteotomy. Relative contraindications to surgery are poor general health and significant scarring of the major vessels, which may be injured when the spine is extended.
The cervical osteotomy (CPT 22210) is performed between C7 and T1. This approach avoids injury to the vertebral artery that usually enters the transverse foramen at the C6 level. Historically the procedure was usually performed under local anesthesia in the semisitting position with facet wiring and halo application (CPT 20661) as the only forms of fixation. However, the evolution of somatosensory and transcranial motor evoked potential monitoring of the spinal cord permits the use of general anesthesia. After removal of the posterior elements and neural decompression, the kyphotic deformity is corrected with gentle extension of the head. The ossified disk space fractures under the extension force and hinges on the posterior longitudinal ligament. The head is held in the corrected position using internal fixation with rod-screw constructs or hook-rod constructs with adjunctive halo vest immobilization (see Figure 4–3). Long constructs with multiple levels of fixation often to C2 or C3 are necessary to obtain sufficient purchase in the ossified but osteoporotic bone to allow for adequate biomechanical stability. Other procedures, such as a decancellation wedge osteotomy of C7 (CPT 22206), have also been described. However, great care must be taken in these cases to avoid inadvertent spinal translation at the osteotomy site that can cause injury to the nerves or the spinal cord. More recently, the adoption of circumferential fusion and fixation techniques has allowed the potential to avoid halo vest immobilization.
Baraliakos X, Listing J, von der Recke A, Braun J: The natural course of radiographic progression in ankylosing spondylitis—evidence for major individual variations in a large proportion of patients. J Rheumatol 2009;36:997. [PMID: 19332632]
Einsiedel T, Schmelz A, Arand M, et al: Injuries of the cervical spine in patients with ankylosing spondylitis: experience at two trauma centers. J Neurosurg Spine 2006;5:33. [PMID: 16850954]
Etame AB, Than KD, Wang AC, La Marca F, Park P: Surgical management of symptomatic cervical or cervicothoracic kyphosis due to ankylosing spondylitis. Spine (Phila Pa 1976) 2008;33:E559. [PMID: 18628698]
Gill JB, Levin A, Burd T, Longley M: Corrective osteotomies in spine surgery. J Bone Joint Surg Am 2008;90:2509. [PMID: 18978421]
Hoh DJ, Khoueir P, Wang MY: Management of cervical deformity in ankylosing spondylitis. Neurosurg Focus 2008;24:E9. [PMID: 18290747]
Kanter AS, Wang MY, Mummaneni PV: A treatment algorithm for the management of cervical spine fractures and deformity in patients with ankylosing spondylitis. Neurosurg Focus 2008;24:E11. [PMID: 18290737]
Kelleher MO, Tan G, Sarjeant R, Fehlings MG: Predictive value of intraoperative neurophysiological monitoring during cervical spine surgery: a prospective analysis of 1055 consecutive patients. J Neurosurg Spine 2008;8:215. [PMID: 18312072]
Kubiak EN, Moskovich R, Errico TJ, Di Cesare PE: Orthopaedic management of ankylosing spondylitis. J Am Acad Orthop Surg 2005;13:267. [PMID: 16112983]
Maksymowych WP: Disease modification in ankylosing spondylitis. Nat Rev Rheumatol 2010;6:75. [PMID: 20125174]
Simmons ED, DiStefano RJ, Zheng Y, Simmons EH: Thirty-six years experience of cervical extension osteotomy in ankylosing spondylitis: techniques and outcomes. Spine (Phila Pa 1976) 2006;31:3006. [PMID: 17172997]
Smith MD, Scott JM, Murali R, Sander HW: Minor neck trauma in chronic ankylosing spondylitis: a potentially fatal combination. J Clin Rheumatol 2007;13:81. [PMID: 17414535]
Thumbikat P, Hariharan RP, Ravichandran G, McClelland MR, Mathew KM: Spinal cord injury in patients with ankylosing spondylitis: a 10-year review. Spine (Phila Pa 1976) 2007;32:2989. [PMID: 18091492]
Tokala DP, Lam KS, Freeman BJ, Webb JK: C7 decancellisation closing wedge osteotomy for the correction of fixed cervicothoracic kyphosis. Eur Spine J 2007;16:1471. [PMID: 17334795]
van der Heijde D, Landew R, Einstein S, et al: Radiographic progression of ankylosing spondylitis after up to two years of treatment with etanercept. Arthritis Rheum 2008;58:1324. [PMID: 18438853]
Vosse D, van der Heijde D, Landew R, et al: Determinants of hyperkyphosis in patients with ankylosing spondylitis. Ann Rheum Dis 2006;65:770. [PMID: 16219704]
Whang PG, Goldberg G, Lawrence JP, et al: The management of spinal injuries in patients with ankylosing spondylitis or diffuse idiopathic skeletal hyperostosis: a comparison of treatment methods and clinical outcomes. J Spinal Disord Tech 2009;22:77. [PMID: 19342927]
Woodward LJ, Kam PC: Ankylosing spondylitis: recent developments and anaesthetic implications. Anaesthesia 2009;64:540. [PMID: 19413825]
![]() DISEASES AND DISORDERS OF THE CERVICAL SPINE
DISEASES AND DISORDERS OF THE CERVICAL SPINE
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Adequate imaging is essential to diagnosis.
• Imaging must include the entire cervical spine and the occiptocervical and cervicothoracic junctions.
• Lateral cervical spine view is the most important view in radiographic imaging of the cervical spine.
• Inadequate imaging will miss over 20% of cervical injuries.
![]() General Considerations
General Considerations
In evaluating the cervical spine, the use of appropriate imaging studies is critical to a timely and precise diagnosis. Available imaging techniques include plain radiography, tomography, myelography, computed tomography (CT), CT with myelography, three-dimensional reconstruction CT, MRI, and scintigraphy. An understanding of the advantages and disadvantages of each technique is necessary for the proper selection of imaging studies and interpretation of results.
A. Plain Radiography
In evaluating the patient with neck pain, cervical spine radiographs are important in the initial search for a possible lesion. In the trauma setting, when a head or neck injury is suspected, radiographic studies must be carried out appropriately, or a life-threatening lesion may be overlooked. The trauma series includes anteroposterior (AP), right oblique, left oblique, and open-mouth (odontoid) views in addition to an initial cross-table lateral view. When all five views are taken, sensitivity is 92%. Cervical spine precautions must be implemented throughout the radiographic evaluation (see the section on injuries of the cervical spine later in the chapter). In the absence of a history of trauma, the oblique and odontoid views are not always required.
The lateral view reveals the majority of traumatic lesions if performed correctly. Inadequate views can miss more than 20% of cervical spine injuries, however. All seven vertebrae should be clearly visible. Gentle traction on the upper extremities may be necessary to view C7. If this is unsuccessful, a swimmer’s view may be necessary. Careful scrutiny of the prevertebral soft tissue, the anterior border of the vertebral bodies, the vertebral bodies themselves, the posterior border of the bodies, the spinal canal proper, and the posterior elements must be done.
The prevertebral region may reveal swelling consistent with a hematoma, and this may serve as the only clue to a traumatic lesion. The upper limits for the prevertebral space are 10 mm at C1; 5 mm at C2; 7 mm at C3 and C4; and 20 mm at C5, C6, and C7. The contours of the cervical bony structures are regular, and subtle incongruities may indicate significant instability. Variations in normal cervical anatomy do exist, however, and a familiarity with them may prevent an overzealous workup. The ADI normally measures less than 3 mm in adults and less than 4 mm in children.
In reviewing the AP radiograph, careful assessment of the interspinous distance must be undertaken. Vertical widening at a given level greater than 1.5 times the level above and below indicates a hyperflexion injury with posterior instability or interlocking of the posterior facets. Traumatic tilting may also be noted in the AP plane while not appreciated on the lateral view.
Oblique views taken at 45 degrees allow visualization of the articulations of the facet joints. The open-mouth view permits evaluation of the odontoid process, the lateral masses, and the articulations of the lateral masses, and it also permits assessment of the distance between each lateral mass and the odontoid process. In atlantoaxial rotatory subluxation, the lateral mass of the atlas that is rotated forward is closer to the midline (medial offset); the opposite mass is farther away from the midline (lateral offset). Burst fractures of the C1 ring cause overhang of the C1 lateral masses on C2. A combined overhang exceeding 6.9 mm is highly correlated with insufficiency of the transverse ligament and C1-C2 sagittal instability.
This radiographic series is equally important in evaluating infants and children with suspected congenital or developmental defects and adults with insidious neck pain. Arthritic changes may be subtle or readily apparent with osteophytes, disk space narrowing, and facet sclerosis. Bone quality can also be assessed on plain radiographs.
B. Computed Tomography
CT scans allow excellent visualization of the bony architecture and the paravertebral soft tissues of the cervical spine. The pedicles, laminae, spinous processes, and bony spinal canal can be examined with significantly better resolution when CT is used than when conventional radiographs are taken (Figure 4–4). CT with myelography or intrathecal contrast enhancement permits visualization of the spinal canal contents.

![]() Figure 4–4. Sagittal CT (A) and MRI (B) images of a patient with cervical spondylotic myelopathy and spinal stenosis. CT demonstrates excellent bony detail, and the MRI allows assessment of the spinal cord and disks.
Figure 4–4. Sagittal CT (A) and MRI (B) images of a patient with cervical spondylotic myelopathy and spinal stenosis. CT demonstrates excellent bony detail, and the MRI allows assessment of the spinal cord and disks.
CT is an appropriate modality for evaluating congenital variations and malformations, including spinal canal stenosis and spina bifida. Pars defects, atlantoaxial joint diseases, inflammatory changes, primary tumors, and metastatic carcinoma are well appreciated with CT. Although cervical disk disease is detectable when thin cuts and contrast enhancement are used with CT, it is better visualized with MRI.
In the trauma patient with questionable findings on plain radiographs, CT is integral in evaluating possible fractures or instability. Atrophy, deformity, and displacement of the spinal cord from acute or chronic injury are all appreciable with the use of intrathecal contrast. With the advent of MRI, however, CT is now reserved for the assessment of the bony architecture, which it does better than MRI.
Three-dimensional reconstruction of CT images gained wide clinical acceptance with the advancement of computer graphics. The reconstructions can be rotated in space to evaluate the anatomy from almost any perspective. This technique is valuable in the understanding of atlantoaxial rotatory subluxations or complex fractures of the spinal column.
C. Magnetic Resonance Imaging
MRI permits axial, sagittal, coronal, or oblique plane analysis of the anatomy. It is routinely noninvasive, requiring contrast material in only selected cases.
MRI is the standard for assessing cervical spinal cord damage. Spinal cord tumors and trauma, as well as central disk herniation, can be easily visualized. In the preoperative evaluation of patients with spondylosis or disk herniation, MRI is the neuroimaging test of choice (see Figure 4–4).
Intravenous paramagnetic agent gadolinium is commonly used to differentiate tissues receiving higher blood flow. This is helpful in the diagnosis of infection, tumor, and postsurgical scar.
High-resolution dynamic (flexion/extension/upright) MRIs have allowed the diagnosis of more subtle patterns of spinal impingement that may not be apparent in a supine (nonloaded) MRI study with the neck in a neutral position. These can allow imaging to be done in a neck position that reproduces the patient’s symptoms.
D. Scintigraphy
Bone scans that employ technetium-99m phosphate permit assessment of physiologic processes within the musculoskeletal system. Metabolic, metastatic, and inflammatory abnormalities can be detected. Technetium-99m phosphate is a bisphosphonate. Its chemical similarity to pyrophosphate promotes its incorporation into bone hydroxyapatite and accumulates in areas of increased osteogenesis. Early-phase imaging with technetium-99m gives blood flow information. Accordingly, subtle fractures, avascular necrosis, and osteomyelitis can be detected. Other radioisotopes used in scintigraphy include gallium-67 citrate, which labels serum proteins, and indium-111, which labels white blood cells. These labeling techniques are helpful in discerning areas of neoplasia or acute infection.
Currently, positron emission scintigraphy combined with high-resolution CT provides a more precise image of the affected areas.
Anderson PA, Muchow RD, Munoz A, Tontz WL, Resnick DK: Clearance of the asymptomatic cervical spine: a meta-analysis. J Orthop Trauma 2010;24:100. [PMID: 20101134]
Bailitz J, Starr F, Beecroft M, et al: CT should replace three-view radiographs as the initial screening test in patients at high, moderate, and low risk for blunt cervical spine injury: a prospective comparison. J Trauma2009;66:1605. [PMID: 19509621]
Barrett TW, Schierling M, Zhou C, et al: Prevalence of incidental findings in trauma patients detected by computed tomography imaging. Am J Emerg Med 2009;27:428. [PMID: 19555613]
Brandenstein D, Molinari RW, Rubery PT, Rechtine GR 2nd. Unstable subaxial cervical spine injury with normal computed tomography and magnetic resonance initial imaging studies: a report of four cases and review of the literature. Spine (Phila Pa 1976) 2009;34:E743. [PMID: 19752695]
Como JJ, Diaz JJ, Dunham CM, et al: Practice management guidelines for identification of cervical spine injuries following trauma: update from the Eastern Association for the Surgery of Trauma Practice Management Guidelines Committee. J Trauma 2009;67:651. [PMID: 19741415]
Gonzalez RP, Cummings GR, Phelan HA, Bosarge PL, Rodning CB: Clinical examination in complement with computed tomography scan: an effective method for identification of cervical spine injury. J Trauma 2009;67:1297. [PMID: 20009681]
Gore PA, Chang S, Theodore N: Cervical spine injuries in children: attention to radiographic differences and stability compared to those in the adult patient. Semin Pediatr Neurol 2009;16:42. [PMID: 19410157]
Grauer JN, Vaccaro AR, Lee JY, et al: The timing and influence of MRI on the management of patients with cervical facet dislocations remains highly variable: a survey of members of the Spine Trauma Study Group. J Spinal Disord Tech 2009;22:96. [PMID: 19342930]
Hashem R, Evans CC, Farrokhyar F, Kahnamoui K: Plain radiography does not add any clinically significant advantage to multidetector row computed tomography in diagnosing cervical spine injuries in blunt trauma patients. J Trauma 2009;66:423. [PMID: 19204517]
Lehman RA Jr, Helgeson MD, Keeler KA, Bunmaprasert T, Riew KD: Comparison of magnetic resonance imaging and computed tomography in predicting facet arthrosis in the cervical spine. Spine (Phila Pa 1976) 2009;34:65. [PMID: 19127162]
Manchikanti L, Dunbar EE, Wargo BW, Shah RV, Derby R, Cohen SP: Systematic review of cervical discography as a diagnostic test for chronic spinal pain. Pain Physician 2009;12:305. [PMID: 19305482]
Mummaneni PV, Kaiser MG, Matz PG, et al: Preoperative patient selection with magnetic resonance imaging, computed tomography, and electroencephalography: does the test predict outcome after cervical surgery? J Neurosurg Spine 2009;11:119. [PMID: 19769491]
Pieretti-Vanmarcke R, Velmahos GC, Nance ML, et al: Clinical clearance of the cervical spine in blunt trauma patients younger than 3 years: a multi-center study of the American Association for the Surgery of Trauma. J Trauma2009;67:543. [PMID: 19741398]
Richards PJ, George J, Metelko M, Brown M: Spine computed tomography doses and cancer induction. Spine (Phila Pa 1976) 2010;35:430. [PMID: 20081559]
Saltzherr TP, Beenen LF, Reitsma JB, Luitse JS, Vandertop WP, Goslings JC: Frequent computed tomography scanning due to incomplete three-view x-ray imaging of the cervical spine. J Trauma2009;68:1213. [PMID: 20016389]
Schoenfeld AJ, Bono CM, McGuire KJ, Warholic N, Harris MB: Computed tomography alone versus computed tomography and magnetic resonance imaging in the identification of occult injuries to the cervical spine: a meta-analysis. J Trauma 2010;68:109. [PMID: 20065765]
Simon JB, Schoenfeld AJ, Katz JN, et al: Are “normal” multidetector computed tomographic scans sufficient to allow collar removal in the trauma patient? J Trauma 2010;68:103. [PMID: 20065764]
Song KJ, Choi BW, Kim GH, Kim JR: Clinical usefulness of CT-myelogram comparing with the MRI in degenerative cervical spinal disorders: is CTM still useful for primary diagnostic tool? J Spinal Disord Tech 2009;22:353. [PMID: 19525791]
Xu-hui Z, Jia-hu F, Lian-shun J, et al: Clinical significance of cervical vertebral flexion and extension spatial alignment changes. Spine (Phila Pa 1976) 2009;34:E21. [PMID: 19127144]
CONGENITAL MALFORMATIONS (ICD-9 756.10)
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Os odontoideum (ICD-9 756.10) is a congenital nonunion of the dens that can lead to significant C1-C2 instability.
• Injuries can occur after minimal trauma.
• Klippel-Feil syndrome (ICD-9 756.16) exhibits a triad of clinical features: a short “web” neck, a low posterior hairline, and limited cervical neck motion.
• Syndromic conditions such as VATER (vertebrae, anus, trachea, esophagus, and renal abnormalities) must be ruled out in the presence of congenital failure of formation or segmentation in the cervical spine.
![]() General Considerations
General Considerations
The atlanto-occipital region is a frequent location for abnormalities. Various combinations involving bone and nervous structures are possible. During embryologic development, 42 somites are formed from the paraxial mesoderm. The somites divide into sclerotomes, which form the vertebral bodies after separation into a caudal and cephalad portion. The middle portion builds the intervertebral disk. The second, third, and fourth somites fuse and become the occiput and posterior part of the foramen magnum. The fate of the first somite is unclear. The development of the neural tube progresses simultaneously with that of the cartilaginous skeleton.
Disturbances of embryologic development can result in incomplete development or absence of a tissue or part, as found in dysraphism, aplasia of the odontoid process, incomplete closure of the atlas, or absence of the atlas facet. Lack of segmentation results in atlanto-occipital fusion, block vertebrae, and possible instability at adjacent cervical levels. A disturbance of neurologic development, alone or in combination with bony defects, can lead to basilar impression, Arnold-Chiari malformation, and syringomyelia, all of which manifest in various states of spinal cord dysfunction (myelopathy).
1. Os Odontoideum
![]() Pathogenesis
Pathogenesis
Os odontoideum is an uncommon type of pseudarthrosis between the odontoid process and the body of the axis (Figure 4–5). It can cause significant atlantoaxial instability and myelopathy and can result in sudden death. Gross instability at the C1-C2 level can lead to spinal cord impingement or injury as it is compressed against the anterior portion of the axis or the posterior ring of the atlas. In some cases, extrinsic compression of the vertebral arteries results in ischemic insult to the brain.
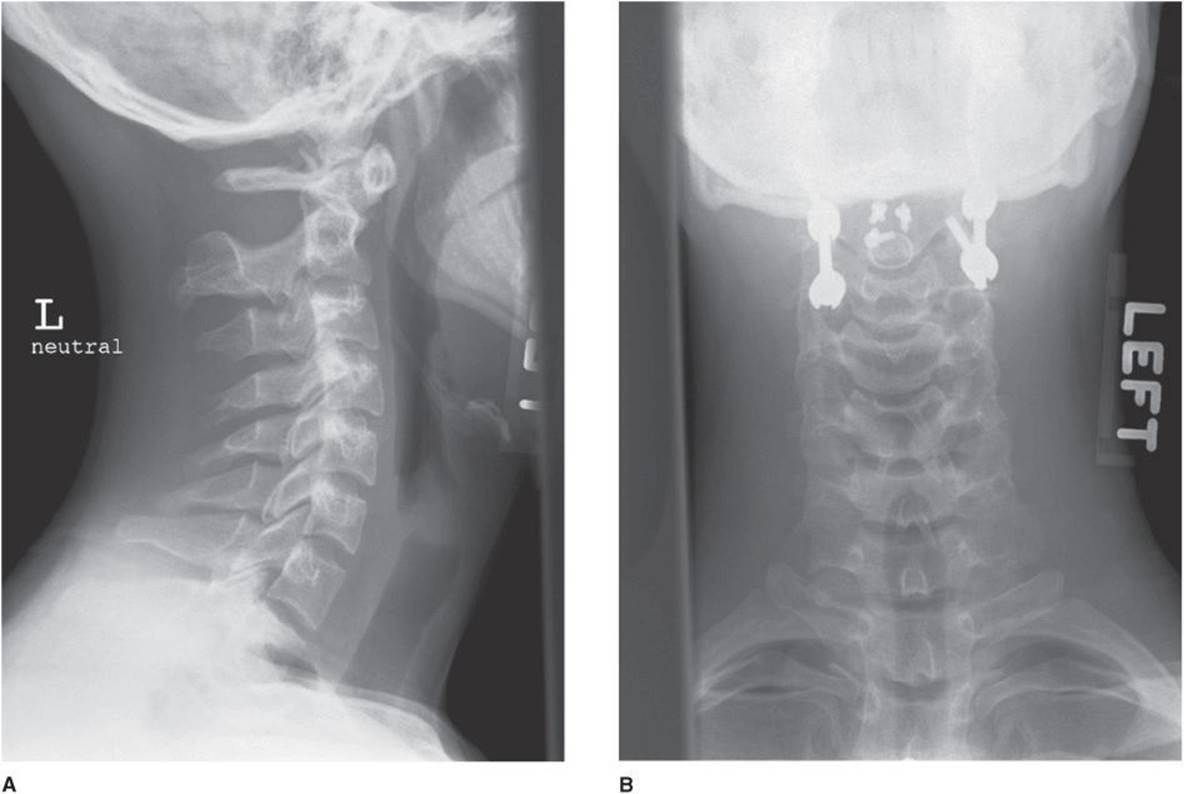

![]() Figure 4–5. (A) Lateral radiograph of a 24-year-old man with a symptomatic os odontoideum. (B and C) Anteroposterior and lateral radiographs after stabilization and fusion of C1-C2 to stabilize the os odontoideum.
Figure 4–5. (A) Lateral radiograph of a 24-year-old man with a symptomatic os odontoideum. (B and C) Anteroposterior and lateral radiographs after stabilization and fusion of C1-C2 to stabilize the os odontoideum.
![]() Prevention
Prevention
There is no preventive measure to avoid this congenital anomaly. However, a stable os odontoideum (no motion on flexion and extension) may be treated without surgical stabilization. In this case, the patient should be counseled about the risks of neurologic injury with potentially minor trauma.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Patients with os odontoideum may be asymptomatic or may present with symptoms and signs that relate to atlantoaxial instability, such as ill-defined neck complaints or focal or diffuse neurologic deficits. A careful history may be needed to rule out trauma, although congenital os odontoideum may come to the attention of the surgeon secondary to a reported but inconsequential neck injury.
B. Imaging Studies
The radiographic findings may be extremely subtle and difficult to distinguish. In the mature skeleton, os odontoideum appears as a radiographic lucency. In children younger than 5 years, however, an anomalous gap may be confused with a normal neural synchondrosis. Flexion-extension views must therefore be obtained to demonstrate motion between the odontoid process and the body of the axis. The ossicle in os odontoideum is either round or ovoid, with a smooth surface and uniform cortical thickness. It is usually approximately half the size of the normal odontoid process. In traumatic nonunion, the edge is irregular with a narrow gap. The fracture line may involve the body of C2 as well. An additional radiologic finding in os odontoideum is hypertrophy of the anterior ring of the atlas with a corresponding hypoplastic posterior ring. In flexion-extension views, the ossicle travels with the anterior ring of the atlas (see Figure 4–5). In cases that are difficult to diagnose, further studies include open-mouth views, tomograms, and CT reconstructions.
![]() Differential Diagnosis
Differential Diagnosis
Dens fractures (ICD-9 805.02) may appear similar to an os odontoideum. However, these fractures are often associated with more significant trauma (ie, motor vehicle accident).
![]() Complications
Complications
Complications of nonsurgical treatment include neurologic injury, chronic neck pain, and sudden death. Complications of surgical treatment include paralysis, infection, stroke, or death from vertebral artery injury.
![]() Treatment
Treatment
Patients diagnosed with os odontoideum must be warned of the gravity of the situation because minimal trauma can be fatal. Patients with cervical myelopathy can be treated with traction, immobilization, or both, but they often require subsequent posterior fusion. Direct osteosynthesis of the os odontoideum fragment is often not possible due to its small size. Sometimes symptoms are reversible with or without intervention. Management of asymptomatic patients with instability is controversial. The benefits of surgical stabilization in an attempt to avoid potentially lethal injury from relatively minor trauma are counterbalanced by the possible complications of surgery. Improvements in image-guided surgery using systems such as STEALTH or BrainLAB have improved the accuracy and safety of placing internal fixation devices in this anatomically unique area. Alternative fixation techniques such as C1 lateral mass fixation combined with C2 translaminar or pars/pedicle fixation have minimized but not eliminated the potential for vertebral artery injury.
If fusion is indicated, usually a posterior fusion of C1-C2 (CPT 22595) is adequate. Different fusion techniques are available. Most surgeons use internal fixation with transarticular screws or C1 lateral mass/C2 screw fixation with rods (CPT 22840) combined with the Gallie or Brooks technique of structural bone grafting (CPT 20931, 20938). The Gallie technique involves the use of a single block-shaped bone graft between the posterior ring of C1 and the spinous process of C2. A single sublaminar wire holds the graft in place. The Brooks technique uses from two to four sublaminar wires, and two bone grafts are wedged between the laminae of C1 and C2. The loss of motion between atlas and axis results in an overall decrease of 50% of cervical rotation. Use of transarticular screws or screw-rod constructs that purchase into the lateral masses of C1 and the pedicle of C2 are rigid enough to allow the patient to mobilize without a soft cervical collar.
2. Klippel-Feil Syndrome (ICD-9 756.16)
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Syndrome associated with congenital fusion of cervical vertebrae.
• “Classic triad.”
• Look for associated anomalies including scoliosis, renal disorders, deafness, and Sprengel deformity.
![]() Pathogenesis
Pathogenesis
Klippel-Feil syndrome refers to an array of clinical disorders associated with congenital fusion of one or more cervical vertebrae. The fusion, which may be multilevel, results from a failure of the normal division of the cervical somites during the third through eighth weeks of embryogenesis. The cause of this failure is unknown. The syndrome was first described in 1912 by M. Klippel and A. Feil as a triad of clinical features: a short “web” neck, a low posterior hairline, and limited cervical neck motion. Interestingly, only 50% of patients with the syndrome that now bears the names of Klippel and Feil present with this classic triad.
Various conditions were subsequently seen in association with congenitally fused cervical vertebrae. These include scoliosis (seen in approximately 60% of cases), renal abnormalities (in 35%), deafness (in 30%), Sprengel deformity (in 30%), synkinesis or mirror movement (in 20%), congenital heart defects (in 14%), brainstem anomalies, congenital cervical stenosis, adrenal aplasia, ptosis, Duane contracture, lateral rectus palsy, facial nerve palsy, syndactyly, and upper extremity diffuse or focal hypoplasia.
![]() Prevention
Prevention
There is no preventive measure to avoid this congenital anomaly. Children with mild involvement can be expected to grow up to lead healthy, normal lives. Patients with more severe involvement can do comparably well if the associated conditions are successfully treated at an early age.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Decreased range of motion is the most frequent finding in patients with cervical spine involvement. Involvement of only the lower cervical spine or fusion of fewer than three vertebrae results in minimal loss of motion, however. Patients may also be able to compensate at other cervical interspaces, masking any loss of motion.
Neck shortening is difficult to detect unless extreme. Webbing of the neck (pterygium colli), facial asymmetry, or torticollis is seen in fewer than 20% of patients. Webbing of the neck can nevertheless be dramatic, with underlying muscle involvement extending from the mastoid to the acromion. Sprengel deformity, which results from a failure of either or both scapulae to descend from their embryologic origin at C4, is seen in approximately 30% of patients. Sometimes an omovertebral bone bridges the cervical spine to the scapulae and limits the neck and shoulder motion.
Cervical spine symptoms in Klippel-Feil syndrome are related to the secondary hypermobility of the unfused vertebrae. Except for atlantoaxial joint involvement, resulting in a significant decrease in occipital rotation, the fused joints at a given level are asymptomatic. Because of the increased mechanical demands placed on the uninvolved joints, secondary osteoarthritis, disk degeneration, spinal stenosis, and instability may result at these levels. Neurologic sequelae, usually confined to the head, neck, and upper extremities, result from impingement of the cervical nerve roots. With progressive cervical instability, the spinal cord may become involved, leading to spasticity, weakness, hyperreflexia, and even quadriplegia or sudden death from minor trauma.
B. Imaging Studies
Radiographic findings of congenital cervical vertebral fusion are diagnostic of Klippel-Feil syndrome (Figure 4–6). This may present as synostosis of two vertebral bodies or as a multilevel fusion, as originally described in 1912. Other noteworthy findings are flattening of the involved vertebral bodies and the absence of disk spaces. Hypoplastic cervical disks in a child are often hard to appreciate radiographically. If suspected, flexion-extension views can be taken. CT scanning and MRI have improved the assessment of bony and nerve root involvement.


![]() Figure 4–6. (A and B) Anteroposterior (AP) and lateral radiographs of a 60-year-old man with Klippel-Feil demonstrating the congenitally fused cervical vertebrae that have led to deterioration of the adjacent disk segments, leading to severe spinal stenosis. (C and D) AP and lateral radiographs after posterior laminectomy and fusion with instrumentation.
Figure 4–6. (A and B) Anteroposterior (AP) and lateral radiographs of a 60-year-old man with Klippel-Feil demonstrating the congenitally fused cervical vertebrae that have led to deterioration of the adjacent disk segments, leading to severe spinal stenosis. (C and D) AP and lateral radiographs after posterior laminectomy and fusion with instrumentation.
Spinal canal stenosis is not usually seen until adulthood. Although anterior spina bifida is infrequent, the posterior form is not. Enlargement of the foramen magnum with fixed hyperextension often accompanies the cervical spina bifida. Hemivertebrae can also occur in this syndrome.
Involvement of the upper thoracic spine may be the first sign of an undiagnosed cervical synostosis. Because of the potential for multiorgan involvement in patients with Klippel-Feil syndrome, an electrocardiogram and renal ultrasound are also recommended.
![]() Differential Diagnosis
Differential Diagnosis
In the presence of associated congenital abnormalities such as hemivertebra, other syndromic conditions such as VATER should be ruled out.
![]() Complications
Complications
Complications are directly related to the treatment of the specific symptomatic conditions. The complications of surgical treatment include nerve injury and paralysis. Nonfusion-type procedures can lead to kyphosis because the levels adjacent to the congenitally fused levels are often degenerated at the time of presentation. Anterior fusion surgery can often span more than one motion segment and can lead to loss of range of motion (from fusion) and can potentially accelerate the rate of wear of the segments adjacent to the surgical fusion. Extensile anterior approaches can be complicated by postoperative swallowing disorders or unilateral vocal cord paralysis.
![]() Treatment
Treatment
Treatment of cervical spine abnormalities is limited. Multilevel involvement leads to hypermobility at uninvolved joints, so affected patients should be cautious in their activities. Prophylactic surgical stabilization is not routinely performed in asymptomatic patients because the risk-benefit ratio has not been well defined. In some cases, however, surgical fusion is performed.
Secondary osteoarthritis may be treated in the usual manner, including use of a cervical collar, traction, and anti-inflammatory agents. Nerve root impingement requires careful evaluation before surgical decompression because more than one level may be involved and there may also be central abnormalities.
Surgical correction of the aesthetic deformities is only moderately successful. Carefully selected candidates may benefit from soft-tissue Z-plasty or tenotomies. This may improve the appearance of the patient but does not affect cervical motion.
Campbell RM Jr: Spine deformities in rare congenital syndromes: clinical issues. Spine (Phila Pa 1976) 2009;34:1815. [PMID: 19644333]
Grob D: Fusion in craniocervical malformation. Eur Spine J 2009; 18:1241. [PMID: 19693545]
Klimo P Jr, Kan P, Rao G, Apfelbaum R, Brockmeyer D: Os odontoideum: presentation, diagnosis, and treatment in a series of 78 patients. J Neurosurg Spine 2008;9:332. [PMID: 18939918]
Menezes AH: Pathogenesis, dynamics, and management of os odontoideum. Neurosurg Focus 1999;6:e2. [PMID: 16972748]
Samartzis D, Kalluri P, Herman J, Lubicky JP, Shen FH: The extent of fusion within the congenital Klippel-Feil segment. Spine (Phila Pa 1976) 2008;33:1637. [PMID: 18594455]
Samartzis D, Lubicky JP, Herman J, Shen FH: Faces of spine care: from the clinic and imaging suite. Klippel-Feil syndrome and associated abnormalities: the necessity for a multidisciplinary approach in patient management. Spine J 2007;7:135. [PMID: 17269206]
Sankar WN, Wills BP, Dormans JP, Drummond DS: Os odontoideum revisited: the case for a multifactorial etiology. Spine (Phila Pa 1976) 2006;31:979. [PMID: 16641773]
Shen FH, Samartzis D, Herman J, Lubicky JP: Radiographic assessment of segmental motion at the atlantoaxial junction in the Klippel-Feil patient. Spine (Phila Pa 1976) 2006;31:171. [PMID: 16418636]
Tracy MR, Dormans JP, Kusumi K: Klippel-Feil syndrome: clinical features and current understanding of etiology. Clin Orthop Relat Res 2004;424:183. [PMID: 15241163]
CERVICAL SPONDYLOSIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Cervical spondylosis is directly associated with disk degeneration.
• The most frequently involved levels are the more mobile segments: C5-C6, C6-C7, and C4-C5.
• Spinal canal stenosis is present when the canal diameter becomes less than 13 mm.
• Extension of the cervical spine often exacerbates the symptoms of spinal cord and nerve root compression.
![]() General Considerations
General Considerations
Cervical spondylosis (ICD-9 721.0, 721.1) is defined as a generalized disease process affecting the entire cervical spine and related to chronic disk degeneration. In approximately 90% of men older than 50 years and 90% of women older than 60 years, degeneration of the cervical spine can be demonstrated by radiographs. Initial disk changes are followed by facet arthropathy, osteophyte formation, and ligamentous instability. Myelopathy, radiculopathy, or both may be seen secondarily. Cervical myelopathy is the most common form of spinal cord dysfunction in people older than 55 years. People older than 60 years are more likely to have multi-segmental disease. The incidence of cervical myelopathy (ICD-9 721.1) is twice as great in men as in women.
![]() Pathogenesis
Pathogenesis
The relationship between the spinal cord and its bony arcade has been studied extensively. The first publication on the subject was written in the early 1800s and gave the first account of a “spondylotic bar,” which was actually a thickened posterior longitudinal ligament protruding into the canal secondary to disk degeneration. Subsequent work revealed that disk degeneration and osteoarthritis could lead to spinal cord and nerve root impingement.
Acute traumatic disk herniation (ICD-9 722.0) was distinguished from the chronic spondylotic process in the mid-1950s. Concurrently, anterior spinal artery impingement by the disk or osteophyte was proposed as part of the pathogenesis. As indicated in these studies, disk degeneration starts with tears in the posterolateral region of the annulus. The subsequent loss of water content and proteoglycans in the nucleus then leads to a decrease of disk height. The longitudinal ligaments degenerate and form bony spurs at their insertion into the vertebral body. These so-called hard disks have to be distinguished from soft disks, which represent acute herniation of disk material into the spinal canal or into the neural foramen. The most frequently involved levels are the more mobile segments: C5-C6, C6-C7, and C4-C5. The converging of the cervical disk space may result in buckling of the ligamentum flavum, with further narrowing of the spinal canal. Segmental instability results in hypertrophic formation of osteophytes by the uncovertebral joint of Luschka and by the facet joints. These prominent spurs result in compression of both the exiting nerve roots and the spinal cord (ICD-9 722.71).
Further work revealed that the sagittal cervical canal diameter was appreciably smaller (3 mm on average) in the myelopathic spondylotic spine than in the normal spine. The anterior-posterior dimensions of the cervical spinal canal measure between 17 and 18 mm in normal individuals. Spinal canal stenosis is present when the canal diameter becomes less than 13 mm. With extension of the neck, both the spinal canal diameter and the neuroforaminal diameter decrease.
![]() Prevention
Prevention
Cervical spondylosis (ICD-9 721.0, 721.1, 721.90) is generally a progressive, chronic disease process (Figure 4–7). In a study of 205 patients with neck pain, Gore et al (2009) found that many patients had decreased pain at the 10-year follow-up, but those with the most severe involvement did not improve. Conservative measures may retard the disease process in its early stages. Maintaining overall aerobic conditioning and fitness and awareness, early diagnosis, and appropriate treatment of spinal cord compression can reduce or prevent functional losses.

![]() Figure 4–7. (A and B) Anteroposterior and lateral radiographs of a 53-year-old woman with multilevel cervical spondylosis.
Figure 4–7. (A and B) Anteroposterior and lateral radiographs of a 53-year-old woman with multilevel cervical spondylosis.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Headache (ICD-9 784.0) may be the presenting symptom of cervical spondylosis. Usually, the headache is worse in the morning and improves throughout the day. It is commonly located in the occipital region and radiates toward the frontal area. Infrequently, patients complain of a painful, stiff neck. Signs include decreased range of motion, crepitus, or both. With more advanced cases, radicular or myelopathic symptoms may be present.
1. Cervical spondylotic radiculopathy (ICD-9 722.0)—Cervical radiculopathy in spondylosis can be quite complex, with nerve root involvement seen at one or more levels and occurring either unilaterally or bilaterally. The onset may be acute, subacute, or chronic, and impingement on the nerve roots may be from either osteophytes or disk herniation. With radiculopathy, sensory involvement in the form of paresthesias or hyperesthesia is more common than motor or reflex changes. Several dermatomal levels may be involved, with radiation into the anterior chest and back. The chief complaint is radiation of pain into the interscapular area and into the arm. Typically, patients have proximal arm pain and distal paresthesias. Extension of the neck with rotation toward the side of neural impingement (Spurling sign) can reproduce the patient’s pain pattern.
2. Cervical spondylotic myelopathy (ICD-9 722.1)—Cervical myelopathy has a variable clinical presentation, given the complex pathogenic mechanisms involved. These include static or dynamic canal impingement, facet arthropathy, vascular ischemia, and the presence of spondylotic transverse bars. In addition, given its neuronal topography, the cord may be affected in dramatically different ways by relatively minor differences in anatomic regions of compression. The clinical course of myelopathy is usually progressive, leading to complete disability over a period of months to years with stepwise deteriorations in function.
Patients often present with paresthesias, dyskinesias, or weakness of the hand, the entire upper extremity, or the lower extremity. Deep aching pain of the extremity, broad-based gait, loss of balance, loss of hand dexterity, and general muscle wasting are found in patients with advanced myelopathy. Impotence is not uncommon in these patients. Patients with severe myelopathy may exhibit a positive Lhermitte sign. In this situation, dropping the head rapidly into flexion elicits a “lightning/electrical” sensation that radiates into the arms and down the thoracic spine. Lhermitte sign is also positive in patients suffering from multiple sclerosis. Appropriate imaging studies, including a brain MRI, are necessary to rule this diagnosis out.
Hyperextension injuries of the spondylotic cervical spine can precipitate a central cord syndrome in which motor and sensory involvement is typically greater in the upper extremities than the lower extremities. Recovery from this injury is usually incomplete. Complete quadriplegia can also occur if the preexisting stenosis is severe. In this setting, the 1-year mortality approaches 80%.
Deep tendon reflexes can be either hyporeflexic or hyperreflexic, with the former seen in anterior horn cell (upper extremity) involvement and the latter seen in corticospinal tract (lower extremity) involvement. Hyporeflexia is found at the level of compression; hyperreflexia occurs on the level below. Long-tract signs, such as the presence of the Hoffmann reflex or Babinski reflex, indicate an upper motor neuron lesion. Clonus is often present although asymmetric. Upper extremity involvement is often unilateral, whereas lower extremities are affected bilaterally. High cervical spondylosis (C3-C5) leads to complaints of numb and clumsy hands; myelopathy of the lower cervical spine (C5-C8) presents with spasticity and loss of proprioception in the legs.
Abdominal reflexes are usually intact, enabling the clinician to differentiate spondylosis from amyotrophic lateral sclerosis, in which reflexes are often absent. Multiple compressions of the spinal cord cause more severe deterioration functionally and electrophysiologically than does a single-level compression.
B. Imaging Studies
Although spondylosis results from cervical spine degeneration, not every patient with radiographic evidence of cervical disk degeneration has symptoms. Furthermore, patients with all the radiographic stigmas of cervical spondylosis may be asymptomatic, and others with clinical evidence of myelopathy may show only modest radiographic changes. This paradox is explainable by canal size differences, with the smaller-diameter canal having less space to buffer the degenerative lesion.
The average AP diameter of the spinal canal measures 17 mm from C3 to C7. The space required by the spinal cord averages 10 mm. The dural diameter increases by 2–3 mm in extension. The smallest sagittal AP diameter is measured between an osteophyte on the inferior aspect of the vertebral body to the base of the spinous process of the next vertebra below. An absolute spinal canal stenosis exists with a sagittal diameter of less than 10 mm. The stenosis is relative if the diameter measures 10–13 mm.
Plain film findings also vary according to the stage of spondylosis at which they were taken. Radiographs may appear normal in early disk disease. Alternatively, they may show single or multilevel disk space narrowing with or without osteophytes. C5-C6 and C6-C7 are the two most commonly involved segments. Vertebral body sclerosis at the adjacent base plates may also be seen. Cortical erosion is uncommon and indicates an inflammatory process such as rheumatoid arthritis.
Oblique views permit evaluation of the facet joints and detection of osteophytosis and sclerosis. The superior facets undergo degeneration more frequently than their inferior counterparts. The superior joints may then subluxate posteriorly and erode into the lamina below. Inferior osteophytes, however, may prevent significant slippage. If instability seen on flexion-extension views is significant (>3.5 mm when measured at the posteroinferior corner of the vertebral body), foraminal stenosis and vertebral artery impingement may result.
MRI permits visualization of the entire cervical canal and spinal cord by showing the spinal cord and nerve roots in two planes (Figure 4–8). The use of a contrast-enhanced CT scan is occasionally required in elderly (more than 60 years) patients with advanced degenerative bony changes of the cervical spine. Accurate identification of the location and extent of pathologic changes is necessary to determine the optimal approach for decompression. Selective nerve root blocks and electromyography may be useful to identify the level of involvement.
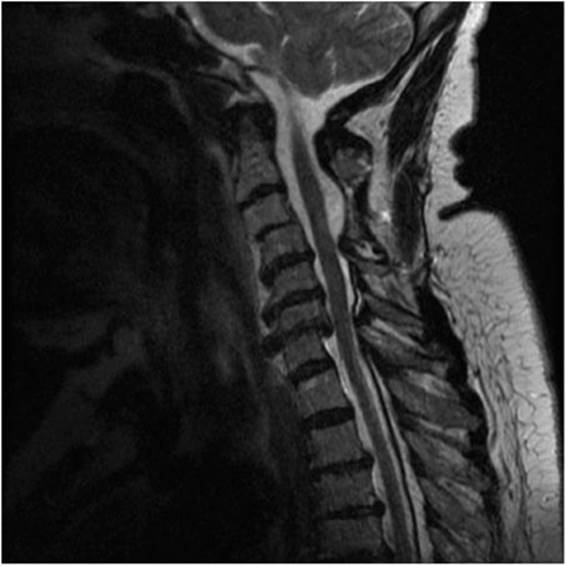
![]() Figure 4–8. Sagittal T2-weighted MRI image of a patient with cervical spondylotic myelopathy demonstrating severe multisegmental spinal stenosis.
Figure 4–8. Sagittal T2-weighted MRI image of a patient with cervical spondylotic myelopathy demonstrating severe multisegmental spinal stenosis.
![]() Differential Diagnosis
Differential Diagnosis
Inflammatory, neoplastic, and infectious conditions can mimic cervical spondylotic radiculopathy and myelopathy.
The cervical spine is affected in most rheumatoid arthritis patients. Atlantoaxial subluxation or subaxial instability can cause symptoms similar to those seen in degenerative cervical myelopathy. A primary tumor or metastatic disease can present with unremitting neck pain, often more intense at night. MRI can distinguish neoplastic conditions from degenerative disorders. Infections of the cervical spine occur in children and in elderly (more than 60 years) or immunocompromised individuals. Multiple sclerosis should be considered in the differential diagnosis. It occurs in younger patients but can present with similar motor signs. Pancoast tumors may invade the brachial plexus, resulting in upper extremity symptoms. Syringomyelia presents with tingling sensations plus motor weakness. A low protein concentration in the cerebrospinal fluid and characteristic changes on MRI are found. Disorders of the shoulder, especially rotator cuff tendinitis, can imitate cervical radiculopathy. Compressive peripheral neuropathies, such as thoracic outlet syndrome, also have to be ruled out.
![]() Treatment
Treatment
Patients should be divided into three groups, according to the predominance of their symptoms: neck pain alone, radiculopathy, and myelopathy. The duration and progression of symptoms need to be considered in the planning of treatment. Several studies suggest that patients with cervical radiculopathy or myelopathy have better long-term results from surgery if symptoms are of short duration.
![]() Prevention
Prevention
Initial management of patients with cervical spondylosis may involve a soft collar, anti-inflammatory agents, and physical therapy consisting of mild traction and the use of isometric strengthening and range-of-motion exercises. The soft cervical collar should be worn only briefly, until the acute symptoms subside. Analgesics are important in the acute phase, and muscle relaxants are helpful in breaking the cycle of muscle spasm and pain. Diazepam should be avoided because of its side effects as a clinical depressant. Epidural corticosteroid injections may be efficacious in patients with radicular pain. Trigger point injections are an empirical form of therapy that seems to work well in patients with chronic neck pain.
The value of cervical traction remains unclear. It is contraindicated in patients with cord compression, rheumatoid arthritis, infection, or osteoporosis. A careful screening of roentgenograms before treatment is mandatory. No evidence indicates that home traction is more effective than manual traction. Isometric strengthening exercises of the paravertebral musculature should be started after the acute symptoms resolve. The patient should be instructed to start a home exercise program early to avoid long-term dependency on passive therapy modalities. Although ice, moist heat, ultrasound, transcutaneous electrical nerve stimulation (TENS), and interferential stimulation are safe to use, there is no scientific proof of their efficacy.
![]() Complications
Complications
Complications of surgical treatment include nerve injury (ICD-9 953.0), paralysis (ICD-9 952.00, 952.05), and infection (ICD-9 998.59). If anterior approaches are employed, dysphagia and vocal cord paralysis can occur. Postsurgical kyphotic deformity (ICD-9 737.10) can develop after multilevel laminectomy. If fusions are performed, failure of fusion (nonunions) that will require surgical augmentation/repair can occur.
![]() Surgical Treatment
Surgical Treatment
Surgical intervention should be considered if the patient does not respond to a conservative treatment protocol or shows evidence of deteriorating myelopathy or radiculopathy. The spinal cord can be effectively decompressed by anterior, posterior, or combined approaches.
The anterior approach allows for multilevel diskectomy (CPT 63075, 63076), vertebrectomy (CPT 63081, 63082), foraminotomy, preparation of the interspaces (CPT 22554) and fusion with structural autograft (CPT 20938) taken from the iliac crest bone, structural allografts (CPT 20931), or synthetic fusion cages supplemented with autograft, allograft, or synthetic matrices. Cervical plating improves fusion rate, decreases the potential for bone graft dislodgement, and helps to maintain cervical alignment during the healing process. However, supplemental posterior fixation and fusion should be considered after two-level vertebrectomy and should be definitely performed after a three-level corpectomy. Posterior fixation minimizes the risk of anterior dislodgement of the graft even in the presence of solid anterior fixation. Alternatively, short (one-level) corpectomy combined with diskectomy for long fusions (three or more levels) improves fixation and thus decreases the potential for graft dislodgement (Figure 4–9). Anterior interbody fusion (CPT 22554) after decompression for a herniated cervical disk has a high success rate. However, fusion does lead to increased biomechanical stresses and intradiskal pressures at the adjacent unfused disk spaces. This may lead to premature degeneration of those adjacent levels.

![]() Figure 4–9. (A and B) Anteroposterior and lateral radiographs of a patient treated with an anterior decompression and fusion with a corpectomy of C5 and diskectomy of C6-C7 and anterior plating.
Figure 4–9. (A and B) Anteroposterior and lateral radiographs of a patient treated with an anterior decompression and fusion with a corpectomy of C5 and diskectomy of C6-C7 and anterior plating.
Cervical disk replacement prostheses were also developed to provide a motion-sparing alternative to anterior cervical diskectomy and fusion (Figure 4–10). By maintaining existing motion or restoring motion to a diseased motion segment, these prostheses have the potential to decrease the rate of symptomatic adjacent segment degeneration. Currently, 5-year data from clinical trials approved by the Food and Drug Administration (FDA) provide evidence that the disk prosthesis for one-level cervical disk disease achieves neurologic and neck pain relief comparable to anterior cervical diskectomy and fusion while maintaining near physiologic motion at the treated level. Function and segmental motion are also maintained at 5-year follow-up.

![]() Figure 4–10. (A and B) Anteroposterior and lateral radiographs of a 45-year-old woman with a C5-C6 herniated disk treated with a diskectomy and reconstruction with a cervical disk replacement.
Figure 4–10. (A and B) Anteroposterior and lateral radiographs of a 45-year-old woman with a C5-C6 herniated disk treated with a diskectomy and reconstruction with a cervical disk replacement.
The number of involved levels may be important in deciding which of the surgical approaches to use. Patients with cervical myelopathy and involvement of more than three vertebral body levels may be best managed by a posterior approach. Multilevel laminectomy (CPT 63015) or laminoplasty (CPT 63050, 63051) shows excellent results. If laminectomies are performed, the facet joints and capsules should be preserved to minimize the chance of postlaminectomy deformity. Late swan-neck deformities after laminectomy can be avoided with simultaneous posterior fusion using lateral mass fixation (Figure 4–11). Laminoplasty is advantageous in that the cervical spinal cord can be decompressed while minimizing the development of late deformity (Figure 4–12). In addition, the morbidity associated with instrumentation and fusion can be avoided and some cervical motion can be preserved.

![]() Figure 4–11. Intraoperative image of the decompressed dura and stabilized spine after a posterior C3 to C7 laminectomy and fusion with instrumentation.
Figure 4–11. Intraoperative image of the decompressed dura and stabilized spine after a posterior C3 to C7 laminectomy and fusion with instrumentation.


![]() Figure 4–12. (A) Preoperative sagittal T2-weighted MRI image of a patient with multilevel cervical spinal stenosis and myelopathy. (B and C) Lateral flexion and extension radiographs after decompression with a posterior C3 to C7 laminoplasty demonstrating excellent cervical motion.
Figure 4–12. (A) Preoperative sagittal T2-weighted MRI image of a patient with multilevel cervical spinal stenosis and myelopathy. (B and C) Lateral flexion and extension radiographs after decompression with a posterior C3 to C7 laminoplasty demonstrating excellent cervical motion.
Operative treatment in cases of cervical spondylotic radiculopathy and myelopathy must be individualized for every patient.
Anderson PA, Matz PG, Groff MW, et al: Laminectomy and fusion for the treatment of cervical degenerative myelopathy. J Neurosurg Spine 2009;11:150. [PMID: 19769494]
Boakye M, Patil CG, Santarelli J, Ho C, Tian W, Lad SP: Cervical spondylotic myelopathy: complications and outcomes after spinal fusion. Neurosurgery 2008;62:455. [PMID: 18382324]
Buchowski JM, Anderson PA, Sekhon L, Riew KD: Cervical disc arthroplasty compared with arthrodesis for the treatment of myelopathy. Surgical technique. J Bone Joint Surg Am 2009;91(Suppl 2):223. [PMID: 19805586]
Dimar JR 2nd, Bratcher KR, Brock DC, Glassman SD, Campbell MJ, Carreon LY: Instrumented open-door laminoplasty as treatment for cervical myelopathy in 104 patients. Am J Orthop (Belle Mead NJ)2009;38:E123. [PMID: 19714281]
Fehlings MG, Arvin B: Surgical management of cervical degenerative disease: the evidence related to indications, impact, and outcome. J Neurosurg Spine 2009;11:97. [PMID: 19769487]
Fehlings MG, Gray R: Importance of sagittal balance in determining the outcome of anterior versus posterior surgery for cervical spondylotic myelopathy. J Neurosurg Spine 2009;11:518. [PMID: 19929352]
Gwinn DE, Iannotti CA, Benzel EC, Steinmetz MP: Effective lordosis: analysis of sagittal spinal canal alignment in cervical spondylotic myelopathy. J Neurosurg Spine 2009;11:667. [PMID: 19951018]
Harrop JS, Naroji S, Maltenfort M, et al: Cervical myelopathy: a clinical and radiographic evaluation and correlation to cervical spondylotic myelopathy. Spine (Phila Pa 1976) 2010 Feb 10. [Epub ahead of print] [PMID: 20150835]
Holly LT, Matz PG, Anderson PA, et al: Clinical prognostic indicators of surgical outcome in cervical spondylotic myelopathy. J Neurosurg Spine 2009;11:112. [PMID: 19769490]
Holly LT, Matz PG, Anderson PA, et al: Functional outcomes assessment for cervical degenerative disease. J Neurosurg Spine 2009;11:238. [PMID: 19769503]
Holly LT, Moftakhar P, Khoo LT, Shamie AN, Wang JC: Surgical outcomes of elderly patients with cervical spondylotic myelopathy. Surg Neurol 2008;69:233. [PMID: 18325426]
Hyun SJ, Rhim SC, Roh SW, Kang SH, Riew KD: The time course of range of motion loss after cervical laminoplasty: a prospective study with minimum two-year follow-up. Spine (Phila Pa 1976)2009;34:1134. [PMID: 19444059]
Matz PG, Anderson PA, Holly LT, et al: The natural history of cervical spondylotic myelopathy. J Neurosurg Spine 2009;11:104. [PMID: 19769489]
Matz PG, Anderson PA, Groff MW, et al: Cervical laminoplasty for the treatment of cervical degenerative myelopathy. J Neurosurg Spine 2009;11:157. [PMID: 19769495]
Matz PG, Holly LT, Mummaneni PV, et al: Anterior cervical surgery for the treatment of cervical degenerative myelopathy. J Neurosurg Spine 2009;11:170. [PMID: 19769496]
Mummaneni PV, Kaiser MG, Matz PG, et al: Cervical surgical techniques for the treatment of cervical spondylotic myelopathy. J Neurosurg Spine 2009;11:130. [PMID: 19769492]
Nikolaidis I, Fouyas IP, Sandercock PA, Statham PF: Surgery for cervical radiculopathy or myelopathy. Cochrane Database Syst Rev 2010;1:CD001466. [PMID: 20091520]
O’Shaughnessy BA, Liu JC, Hsieh PC, Koski TR, Ganju A, Ondra SL: Surgical treatment of fixed cervical kyphosis with myelopathy. Spine (Phila Pa 1976) 2008;33:771. [PMID: 18379404]
Pimenta L, McAfee PC, Cappuccino A, Cunningham BW, Diaz R, Coutinho E: Superiority of multilevel cervical arthroplasty outcomes versus single-level outcomes. Spine (Phila Pa 1976) 2007;32:1337. [PMID: 17515823]
Rao RD, Currier BL, Albert TJ, et al: Degenerative cervical spondylosis: clinical syndromes, pathogenesis, and management. J Bone Joint Surg Am 2007;89:1360. [PMID: 17575617]
Rao RD, Currier BL, Albert TJ, et al: Degenerative cervical spondylosis: clinical syndromes, pathogenesis, and management. Instr Course Lect 2008;57:447. [PMID: 18399602]
Riew KD, Buchowski JM, Sasso R, Zdeblick T, Metcalf NH, Anderson PA: Cervical disc arthroplasty compared with arthrodesis for the treatment of myelopathy. J Bone Joint Surg Am 2008;90:2354. [PMID: 18978404]
Rihn JA, Lawrence J, Gates C, Harris E, Hilibrand AS: Adjacent segment disease after cervical spine fusion. Instr Course Lect 2009;58:747. [PMID: 19385583]
Ryken TC, Heary RF, Matz PG, et al: Cervical laminectomy for the treatment of cervical degenerative myelopathy. J Neurosurg Spine 2009;11:142. [PMID: 19769493]
Ryu JS, Chae JW, Cho WJ, Chang H, Moon MS, Kim SS: Cervical myelopathy due to single level prolapsed disc and spondylosis: a comparative study on outcome between two groups. Int Orthop 2010 Jan 29. [Epub ahead of print] [PMID: 20108087]
Suk KS, Kim KT, Lee JH, Lee SH, Lim YJ, Kim JS: Sagittal alignment of the cervical spine after the laminoplasty. Spine (Phila Pa 1976) 2007;32:E656. [PMID: 17978640]
Wang X, Chen Y, Chen D, et al: Removal of posterior longitudinal ligament in anterior decompression for cervical spondylotic myelopathy. J Spinal Disord Tech 2009;22:404. [PMID: 19652565]
OSSIFICATION OF THE POSTERIOR LONGITUDINAL LIGAMENT (ICD-9 723.7)
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Common cause of myelopathy in Asian population.
• Peak age of onset in the sixth decade of life.
• Disorder associated with other rheumatologic conditions.
• Males are more affected than females.
![]() General Considerations
General Considerations
Ossification of the posterior longitudinal ligament (OPLL) is a relatively common cause of spinal canal stenosis and myelopathy in the Asian population (Figure 4–13). Its overall incidence is 2–3% in Japan, compared with 0.6% in Hawaii and 1.7% in Italy. Males are affected more often than females, and the peak age at onset of symptoms is the sixth decade.

![]() Figure 4–13. (A) Preoperative axial T2-weighted MRI image showing severe cervical stenosis from ossification of the posterior longitudinal ligament (OPLL). (B) Postoperative axial CT image demonstrating the OPLL lesion after decompression.
Figure 4–13. (A) Preoperative axial T2-weighted MRI image showing severe cervical stenosis from ossification of the posterior longitudinal ligament (OPLL). (B) Postoperative axial CT image demonstrating the OPLL lesion after decompression.
![]() Pathogenesis
Pathogenesis
Although the cause of the disorder is unknown, it may be controlled by autosomal dominant inheritance because it is found in 26% of the parents and 29% of the siblings of affected patients. The disorder is associated with several rheumatic conditions, including diffuse idiopathic skeletal hyperostosis (ICD-9 728.89), spondylosis (ICD-9 721.0), and ankylosing spondylitis (ICD-9 720.0).
![]() Prevention
Prevention
Currently, there are no preventive measures that affect the development of OPLL. Once symptomatic, fusion of the affected area halts the growth of the ossification.
![]() Clinical Findings
Clinical Findings
Almost all patients have only mild subjective complaints at the onset, although 10–15% of them complain of clumsiness and spastic gait. Nevertheless, minor trauma can lead to acute deterioration of symptoms and can result in quadriplegia. Spastic quadriparesis is the most common neurologic presentation.
OPLL can easily be diagnosed on plain lateral radiographs. The levels most frequently involved are C4, C5, and C6. A segmental type of disorder is distinguished from the continuous, local, and mixed type based on the distribution of lesions behind the vertebral bodies. CT scanning is helpful in assessing the thickness, lateral extension, and AP diameter of the ossified ligament. More than 95% of the ossification is localized in the cervical spine, although extension into the thoracic spine is reported to be a cause of persistent myelopathy following cervical decompression.
Endochondral ossification is mainly responsible for the formation of the ossified mass, which connects to the upper and lower margins of the vertebral bodies. In many cases, the ossified material is closely adherent to the underlying dura and makes excision quite hazardous. Compression of the spinal cord results in atrophy and necrosis in the gray matter and demyelinization of the white substance.
![]() Differential Diagnosis
Differential Diagnosis
OPLL must be considered in every case of cervical spondylotic myelopathy (ICD-9 721.1). OPLL must also be distinguished from idiopathic disk space calcification (ICD-9 722.91).
![]() Complications
Complications
Complications of surgical treatment include nerve injury (especially C5 root neurapraxia), paralysis, and infection. Removal of the ossification through an anterior approach has a high incidence of durotomy and subsequent spinal fluid leakage.
![]() Treatment
Treatment
Neurologic improvement with either conservative or surgical treatment is achieved in a significant proportion of patients. Patients with severe myelopathy require neural decompression by an anterior, posterior, or combined approach. Sophisticated posterior decompression techniques, such as the open-door laminoplasty (CPT 63050, 63051), have yielded excellent long-term results for OPLL lesions that do not comprise more than 50% of the spinal canal cross-sectional area and in cases where the overall alignment of the cervical spine is neutral or lordotic.
Andres RH, Binggeli R: Ossification of the posterior longitudinal ligament. J Rheumatol 2008;35:528. [PMID: 18322975]
Chen Y, Chen D, Wang X, Guo Y, He Z: C5 palsy after laminectomy and posterior cervical fixation for ossification of posterior longitudinal ligament. J Spinal Disord Tech 2007;20:533. [PMID: 17912131]
Chen Y, Guo Y, Chen D, et al: Diagnosis and surgery of ossification of posterior longitudinal ligament associated with dural ossification in the cervical spine. Eur Spine J 2009;18:1541. [PMID: 19452175]
Dalbayrak S, Yilmaz M, Naderi S: “Skip” corpectomy in the treatment of multilevel cervical spondylotic myelopathy and ossified posterior longitudinal ligament. J Neurosurg Spine 2010;12:33. [PMID: 20043761]
Hida K, Yano S, Iwasaki Y: Considerations in the treatment of cervical ossification of the posterior longitudinal ligament. Clin Neurosurg 2008;55:126. [PMID: 19248677]
Inamasu J, Guiot BH: Factors predictive of surgical outcome for ossification of the posterior longitudinal ligament of the cervical spine. J Neurosurg Sci 2009;53:93. [PMID: 20075820]
Kim TJ, Bae KW, Uhm WS, Kim TH, Joo KB, Jun JB: Prevalence of ossification of the posterior longitudinal ligament of the cervical spine. Joint Bone Spine 2008;75:471. [PMID: 18448378]
Miyazawa N, Akiyama I: Ossification of the ligamentum flavum of the cervical spine. J Neurosurg Sci 2007;51:139. [PMID: 17641578]
Mochizuki M, Aiba A, Hashimoto M, Fujiyoshi T, Yamazaki M: Cervical myelopathy in patients with ossification of the posterior longitudinal ligament. J Neurosurg Spine 2009;10:122. [PMID: 19278325]
![]() DISEASES AND DISORDERS OF THE LUMBAR SPINE
DISEASES AND DISORDERS OF THE LUMBAR SPINE
Brett A. Freedman, MD; John M. Rhee, MD; Scott D. Boden, MD
OVERVIEW
Symptomatic degenerative conditions of the lumbar spine are among the most common reasons for referral to a spine surgeon. The differential diagnosis of back and radiating lower extremity pain is extensive (Table 4–1). Five of the most common forms of symptomatic degenerative lumbar spinal conditions, as well as spinal infections, tumors, and scoliosis, will be addressed.
Table 4–1. Differential diagnoses for degenerative lumbar spinal conditions.

DISK HERNIATION
![]() General Considerations
General Considerations
A disk herniation occurs when a piece of the nucleus pulposus (central gelatinous portion of the disk) pushes through a tear in the tougher annulus fibrosis, a dense collagenous ring encircling the nucleus pulposus. This can occur in the central (Figure 4–14A), posterolateral (Figure 4–14B), foraminal (Figure 4–14C), and extraforaminal zones (Figure 4–14D). The more central the herniation, the more likely it is to compress the traversing nerve root (ie, S1 at the L5/S1 disk), whereas the more lateral the herniation, the more likely it is to compress the exiting nerve root (ie, L5 at L5/S1 disk). Thus, a disk herniation at one spinal level can have different clinical presentations.

![]() Figure 4–14. Diagram depicting the four locations/zones for a disk herniation.
Figure 4–14. Diagram depicting the four locations/zones for a disk herniation.
Herniated nucleus pulposus (HNP) can cause nerve root symptoms by two mechanisms. First, the herniation can cause a mechanical deformation of the nerve root. Second, the release of various inflammatory mediators by the herniated disk elicits a robust inflammatory response and irritates the nerve root. It is thought that most cases of symptomatic radiculopathy from disk herniations have both a mechanical and inflammatory basis, which helps to explain in part why the size of the herniation does not necessarily correlate with the severity of symptoms.
Annually, about 5–20 per 1000 adults sustain disk herniations, resulting in a 13–40% lifetime incidence, occurring most commonly in the fifth decade. The L4/5 and L5/S1 levels are most commonly affected (80%); however, with age, proximal levels (such as L2/3 and L3/4) become increasingly involved. Recently, studies have identified genetic inheritance as the most important risk factor for developing lumbar disk herniations. Occupational and recreational exposures, obesity, and smoking are also important potentially modifiable risk factors.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
Disk herniations classically present with prodromal low back pain that acutely changes to a radiating leg pain. When this occurs, the low back pain may resolve or remain, but the leg symptoms most commonly predominate. More than 50% of the time, there is no inciting event noted. Along with pain, the patient can have numbness and/or weakness in the pattern of the compressed nerve. Diskogenic problems typically are worse in forward flexion and better in extension. Because sitting increases intradiskal pressure, many with symptomatic disk herniations feel more pain sitting than in other positions, such as lying down or even standing. Additionally, positions that put tension on the nerve root (ie, fully extending the knee) tend to reproduce or exacerbate the radiating pain.
All patients presenting with painful lumbar degenerative conditions need a thorough neurologic and physical examination (Tables 4–2 to 4–4). Representative dermatomes of the L1-S1 nerve roots should be assessed for light touch and pinprick sensation and should be compared to the contralateral side. The patient should be asked to stand and ambulate. Gait should be assessed for independence, stability, and antalgia. Frequently patients will not bear full weight on the leg ipsilateral to a large disk herniation and may list to the contralateral side. To further test motor strength and coordination, patients should be asked to walk on their heels and toes. If possible, spine range of motion should be assessed, specifically seeking out whether spinal flexion or extension is more provocative. Last, tension signs should be assessed. In the supine position, each lower extremity should be elevated between 20 and 70 degrees. Reproduction of pain down the leg below the knee constitutes a positive straight leg raise (SLR) test. A crossed SLR occurs when elevation of the contralateral extremity reproduces pain in the symptomatic extremity. Prone SLR (femoral stretch test) can be performed to test for tension in the upper lumbar roots (L2, L3, and L4), indicated by anterior thigh pain. The SLR can be repeated in the sitting position, during motor strength testing of the quadriceps as the patient fully extends the knee.
Table 4–2. Lumbar spine physical exam.

Table 4–3. Motor strength grades.

Table 4–4. Major motor groups and their innervation.

B. Imaging
MRI is the gold standard for detecting and defining lumbar disk herniations but can be overly sensitive, because high proportions of asymptomatic volunteers may have abnormal-appearing disks on MRI. Thus, it is paramount to correlate MRI findings with clinical signs and symptoms when diagnosing and treating HNPs. Disk herniations typically have three distinct morphologies on MRI—protrusion (often called a “disk bulge”), extrusion, and sequestration. A protrusion is a general bulge of the posterior disk that is seen both in conjunction with disk degeneration and the normal aging process. It is the most common form of HNP, seen in 20–30% of asymptomatic adults under the age of 40, 60% between age 40 and 60, and 80–100% over the age of 60. Protrusions have the most variable outcomes following diskectomy, with a 7–13% reherniation rate and up to 38% with persistent sciatica complaints. Protrusions are a relatively poor prognosticator for either operative or nonoperative outcomes with treatment, which makes sense because a protrusion tends to indicate global disk failure, which is less likely to respond to focal interventions like injections or microdiskectomy. Disk extrusion is an HNP in which nuclear material pushes through and beyond the annulus, yielding a mushroom-like appearance with a cap and a stalk. Extrusions represent the HNP morphology most commonly treated with diskectomy (approximately two thirds of cases) (Figure 4–15). Sequestration is a progression of extrusion, in which the herniated nuclear fragment is no longer in continuity with the disk of origin. Extrusions and sequestrations typically have better outcomes than protrusion. Unlike protrusions or bulges, extrusions and sequestrations are seen in fewer than 1–10% of asymptomatic volunteers. Thus, the clinical relevance of extrusion and sequestration is generally more certain than that of a protrusion or disk bulge.

![]() Figure 4–15. Different types of disk herniations. (A) Axial T2 MRI of a large, extruded, posterolateral disk herniation at L5-S1 compressing the S1 root (purple arrow). (B) Axial T2 MRI of an extraforaminal disk herniation at L4-L5 (purple arrow). This herniation compresses the exiting L4 root. (C) Sagittal MRI demonstrates disk herniation (extrusion type) that has migrated cephalad from the disk space of origin (L5-S1). (D) Sagittal MRI demonstrates disk herniation that has stayed at the level of the disk space (L5-S1).
Figure 4–15. Different types of disk herniations. (A) Axial T2 MRI of a large, extruded, posterolateral disk herniation at L5-S1 compressing the S1 root (purple arrow). (B) Axial T2 MRI of an extraforaminal disk herniation at L4-L5 (purple arrow). This herniation compresses the exiting L4 root. (C) Sagittal MRI demonstrates disk herniation (extrusion type) that has migrated cephalad from the disk space of origin (L5-S1). (D) Sagittal MRI demonstrates disk herniation that has stayed at the level of the disk space (L5-S1).
![]() Treatment
Treatment
The first-line treatment for most degenerative, symptomatic conditions of the lumbar spine is nonoperative. Despite the multiple nonoperative therapies advocated, overall, they have had surprisingly limited evidential support; however, researchers in the last decade have made a concerted effort to overcome this prior limitation. Nonoperative treatment is initially attempted because of the overall favorable natural history of the disorder. Symptoms in 60–90% of HNPs will resolve spontaneously in the first 6–12 weeks. Table 4–5 lists some of the more common nonoperative treatment options. Treatments are added in a stepwise fashion, beginning with medications such as nonsteroidal anti-inflammatory drugs (NSAIDs). A short course of oral steroids often provides significant symptom relief as well. Muscle relaxants may help diminish spasms and pain. Narcotics should generally be used sparingly for a short period of time due to their addictive potential. Physical therapy may be helpful in those whose symptom level allows them to participate. Epidural steroid injections (ESIs) are a common nonoperative treatment option. These are best performed under fluoroscopic guidance, with the medication placed transforaminally at the level and side of the herniation to provide the maximal dose at the needed area. The results of clinical studies on ESIs have been variable, but there is level 1 evidence indicating that they may be superior to placebo at least in the acute phase. It is unclear, however, whether ESIs change the natural history of disk herniations. Regardless, the immediate response to these injections can be diagnostic and prognostic. Patients with good to excellent initial response, regardless of whether it diminishes with time, have better outcomes following diskectomy than those with little to no response at any point following injection.
Table 4–5. Nonoperative treatment options.

Indications for operative treatment include: (1) persistent symptoms despite a reasonable course of nonoperative treatment, (2) profound or progressive motor deficit, (3) cauda equina syndrome, (4) intractable pain, and (5) patient preference. Level 1 studies have consistently shown that surgical decompression of the nerve root by removal of the offending disk fragment is significantly more efficacious than nonoperative care at time points up to 1–4+ years. These advantages exist for all outcomes commonly assessed, including pain reduction, functional improvement, and even improvement in mental health. Without question, the single most consistent finding is that the time to achieve maximal clinical outcome is substantially reduced with surgery compared with nonoperative care. The most impactful of these studies is the Spine Patient Outcomes Research Trial (SPORT), a National Institutes of Health–funded, 13-site multicenter study with observational and randomized clinical trial arms that evaluated the outcomes of surgical treatment versus conservative care for three of the most common degenerative spinal conditions—disk herniation (n = 289 randomized, n = 365 observational), spinal stenosis, and spondylolisthesis. The ample data collected from this landmark 5-year prospective study have been subanalyzed to evaluate the impact of multiple factors, including cost and complications. The more proximal the herniated level (ie, L3/4 or L4/5 and above), the more likely surgical decompression is superior to nonoperative care.
For all comers, the major advantage of surgery over non-operative care is quicker time to resolution of symptoms, especially radicular pain. This faster symptom resolution leads to a cost-benefit advantage over nonoperative care. Most patients who undergo microdiskectomy notice substantial improvement in radicular leg pain in the immediate postoperative period. Not infrequently, preoperative weakness and less commonly numbness may also improve quickly, although usually not as quickly or as completely as pain does, particularly if the numbness and weakness were long-standing and constant rather than intermittent. Diabetic patients may also have less recovery of nerve root function.
Conversely, nonoperatively treated patients have an overall slower time to improvement, with maximal outcomes not achieved until 1–2 years following symptom onset; however, at the 1- to 2-year mark, some studies have shown that the significant clinical advantage of surgery over nonoperative care may lessen or no longer exist. On the other hand, the results of the SPORT study show that patients who presented for treatment after 6 or more months of symptom duration had significantly less improvement than those who received treatment more acutely, which is consistent with prior prospective studies. Thus, there appears to be an optimal time window for maximal benefit from surgery that is somewhere between 6 weeks and 6 months. In the end, it is patient preference that should most direct treatment following symptomatic HNPs that do not resolve spontaneously or fail to respond to initial attempts at nonoperative care. Those with the most significant symptoms tend to select surgery, and those with more mild or intermittent symptoms may be best treated in a nonoperative fashion. There is little evidence that the method of surgery (eg, microdiskectomy, minimally invasive diskectomy, traditional “open” diskectomy) has a significant effect on outcomes as long as the nerve root is appropriately decompressed.
While the decision to treat an HNP with surgery or nonoperative therapy is largely one of patient and surgeon preference, there are rare exceptions in which acute operative treatment is absolutely indicated. The most agreed upon urgent surgical indication following HNP is cauda equina syndrome. Patients with cauda equina syndrome may demonstrate acute, profound bilateral lower extremity neurologic deficits, with saddle anesthesia and bowel/bladder incontinence (usually bowel incontinence and urinary retention). Cauda equina syndrome represents one of the true emergent/urgent conditions of the lumbar spine. Thus, all patients with suspected cauda equina syndrome should be immediately referred to a spine surgeon and undergo emergent MRI. Neurologic outcomes are traditionally thought to be best when decompression occurs within the first 48 hours, although recently, it has been shown that the severity (regardless of duration) of incontinence is the most important preoperative predictor of bowel/bladder functional recovery. Even when treated quickly, about 25–50% of patients will have continued bowel/bladder dysfunction. In addition to cauda equina syndrome, patients with any form of progressive neurologic deficit or those in whom pain is not well controlled in an outpatient setting are also candidates for early surgery.
Careful patient selection is mandatory when considering surgery. In addition to the disk pathology, psychosocial factors weigh heavily on outcomes. The presence of three or more of the five Waddell signs is associated with poor outcomes following surgical treatment due to underlying psychosocial issues. These signs include (1) a discrepancy between the seated and the supine SLR; (2) superficial and widespread tenderness that is nonanatomic; (3) pain with axial loading of the head or twisting the pelvis; (4) nondermatomal sensory deficits (“my whole leg is numb”); and (5) overreaction on exam.
![]() Complications
Complications
The most common complications of lumbar diskectomy include infection (1–3%), dural tears (3–10%), nerve root injury (<1%), and recurrent herniations (4–27%). Persistent low back pain is a not uncommon complaint despite excellent relief of radicular pain. It must be understood by the patient preoperatively that the primary goal of surgery is to relieve radicular nerve root pain and that midline axial low back pain may or may not improve even with an otherwise successful surgery.
Ahn UM, Ahn NU, Buchowski JM, Garrett ES, Sieber AN, Kostuik JP: Cauda equina syndrome secondary to lumbar disc herniation: a meta-analysis of surgical outcomes. Spine (Phila Pa 1976)2000;25:1515. [PMID: 10851100]
Buttermann GR: Treatment of lumbar disc herniation: epidural steroid injection compared with discectomy. A prospective, randomized study. J Bone Joint Surg Am 2004;86-A:670. [PMID: 15069129]
Carragee EJ, Han MY, Suen PW, Kim D: Clinical outcomes after lumbar discectomy for sciatica: the effects of fragment type and annular competence. J Bone Joint Surg Am 2003;85-A:102. [PMID: 12533579]
Casal-Moro R, Castro-Menéndez M, Hernández-Blanco M, et al: Long-term outcome after microendoscopic diskectomy for lumbar disk herniation: a prospective clinical study with a 5-year follow-up. Neurosurgery2011;68:1568. [PMID: 21311384]
Katayama Y, Matsuyama Y, Yoshihara H, et al: Comparison of surgical outcomes between macro discectomy and micro discectomy for lumbar disc herniation: a prospective randomized study with surgery performed by the same spine surgeon. J Spinal Disord Tech 2006;19:344. [PMID: 16826006]
Lebow R, Parker SL, Adogwa O, et al: Microdiscectomy improves pain-associated depression, somatic anxiety, and mental well-being in patients with herniated lumbar disc. Neurosurgery 2012;70:306. [PMID: 22251975]
Lurie JD, Berven SH, Gibson-Chambers J, et al: Patient preferences and expectations for care: determinants in patients with lumbar intervertebral disc herniation. Spine (Phila Pa 1976) 2008;33:2663. [PMID: 18981962]
Lurie JD, Faucett SC, Hanscom B, et al: Lumbar discectomy outcomes vary by herniation level in the Spine Patient Outcomes Research Trial. J Bone Joint Surg Am 2008;90:1811. [PMID: 18762639]
Manchikanti L, Buenaventura RM, Manchikanti KN, et al: Effectiveness of therapeutic lumbar transforaminal epidural steroid injections in managing lumbar spinal pain. Pain Physician 2012;15:E199. [PMID: 22622912]
McCarthy MJ, Aylott CE, Grevitt MP, Hegarty J: Cauda equina syndrome: factors affecting long-term functional and sphincteric outcome. Spine (Phila Pa 1976) 2007;32:207. [PMID: 17224816]
Osterman H, Seitsalo S, Karppinen J, Malmivaara A: Effectiveness of microdiscectomy for lumbar disc herniation: a randomized controlled trial with 2 years of follow-up. Spine 2006;31:2409. [PMID: 17023847]
Peul WC, van den Hout WB, Brand R, et al: Prolonged conservative care versus early surgery in patients with sciatica caused by lumbar disc herniation: two year results of a randomised controlled trial. BMJ2008;336:1355. [PMID: 18502911]
Qureshi A, Sell P: Cauda equina syndrome treated by surgical decompression: the influence of timing on surgical outcome. Eur Spine J 2007;16:2143. [PMID: 17828560]
Rhee JM, Schaufele M, Abdu WA: Radiculopathy and the herniated lumbar disk: controversies regarding pathophysiology and management. Instr Course Lect 2007;56:287. [PMID: 17472314]
Rihn JA, Hilibrand AS, Radcliff K, et al: Duration of symptoms resulting from lumbar disc herniation: effect on treatment outcomes: analysis of the Spine Patient Outcomes Research Trial (SPORT). J Bone Joint Surg Am2011;93:1906. [PMID: 22012528]
Ryang YM, Oertel MF, Mayfrank L, Gilsbach JM, Rohde V: Standard open microdiscectomy versus minimal access trocar microdiscectomy: results of a prospective randomized study. Neurosurgery2008;62:174. [PMID: 18300905]
Stafford MA, Peng P, Hill DA: Sciatica: a review of history, epidemiology, pathogenesis, and the role of epidural steroid injection in management. Br J Anaesth 2007;99:461. [PMID: 17704089]
Weinstein JN, Tosteson TD, Lurie JD, et al: Surgical versus non-operative treatment for lumbar spinal stenosis four-year results of the Spine Patient Outcomes Research Trial. Spine (Phila Pa 1976)2010;35:1329. [PMID: 20453723]
Weinstein JN, Tosteson TD, Lurie JD, et al: Surgical vs. nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT): a randomized trial. JAMA 2006;296:2441. [PMID: 17119140]
Weinstein JN, Tosteson TD, Lurie JD, et al: Surgical vs. nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT) observational cohort. JAMA 2006;296:2451. [PMID: 17119141]
SPINAL STENOSIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Often due to degenerative conditions of the spine but can also occur from congenitally narrowed spinal canal or inflammatory and traumatic conditions.
• Symptoms are exacerbated by extension of the spine and improved by flexion of the spine.
• Need to assess for vascular insufficiency and osteoarthritis of the hips and knees.
![]() General Considerations
General Considerations
Spinal stenosis refers to a narrowing of the spinal canal, most commonly due to the accumulation of space-occupying, degenerative material such as hypertrophic ligamentum flavum, osteophytes, and disk herniations or bulges (Figure 4–16). In rare instances, accumulation of epidural fat can also lead to spinal stenosis. Stenosis can occur in the central portion of the canal, the lateral recess (ie, the area underneath the facet joints), or the foramen. In most cases, stenosis occurs at the level of the facet joints. It is at that level that pathologic changes in the disk, facets, and ligamentum flavum hypertrophy converge to produce the greatest amount of narrowing. In contrast, stenosis is relatively uncommon at the level of the pedicles, and when it occurs at this level, it often indicates an underlying congenital or developmental stenosis of the bony canal.

![]() Figure 4–16. (A) Axial MRI demonstrating lateral recess (subarticular) stenosis due to thickening of the ligamentum flavum (purple arrows). There is also an associated right posterolateral disk bulge (white arrow). (B) Severe central stenosis causing a pinhole-size spinal canal. Note associated facet joint synovitis and arthropathy (purple arrow).
Figure 4–16. (A) Axial MRI demonstrating lateral recess (subarticular) stenosis due to thickening of the ligamentum flavum (purple arrows). There is also an associated right posterolateral disk bulge (white arrow). (B) Severe central stenosis causing a pinhole-size spinal canal. Note associated facet joint synovitis and arthropathy (purple arrow).
Spinal stenosis is often seen in patients with a developmentally narrow spinal canal. Such patients may have had sufficient room for their nerve roots in youth, but possess little reserve capacity for the accumulation of degenerative lesions, which occur in all humans with age. Concomitant spinal instability (lateral listhesis or spondylolisthesis) and/or deformity (eg, scoliosis) may accentuate spinal stenosis: as one vertebra translates on the other, the portion of the spinal canal in between the two segments becomes narrowed in a manner similar to the closing of a cigar cutter. When foraminal stenosis occurs, it is usually due to the hypertrophy and/or proximal migration of the superior articular facet from the level below (ie, the S1 superior facet in L5-S1 foraminal stenosis), along with associated encroachment of the ligamentum flavum into the foramen. A decrease in disk height or disk herniation or bulging into the foramen can also lead to foraminal stenosis. The L4/5 level is the most commonly involved segment in spinal stenosis. Symptomatic patients can present at any adult age, with congenitally stenotic patients presenting as early as their late teen years, but most degenerative stenosis patients present in their 50s and beyond.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
Classically, patients complain of the insidious onset of radiating buttock and leg pain that is worse as the compromised spinal canal is further narrowed in extension (eg, standing upright or walking [downhill especially]) and improved as the canal is relatively enlarged in flexion (eg, sitting, lying down in a “fetal” position, or walking while bent over a shopping cart). Associated numbness, weakness, or a feeling of “heaviness” or easy fatigue in the legs with walking may be present. Patients may also complain of concomitant low back pain. Not all patients, however, present with these classic symptoms. Many will have significant radicular leg pain even at rest. Not infrequently, the leg symptoms may not radiate all the way down the leg but rather localize only to the buttock or posterior thigh area.
A thorough physical examination is necessary but is commonly nonfocal. Severe motor and sensory deficits are relatively uncommon. The hips and knee joints of all patients should be assessed, as osteoarthritis of those joints may often mimic spinal stenosis symptoms. Greater trochanteric bursitis can also masquerade as radiating leg pain. Sacroiliac joint pain, which can be very difficult to definitively diagnose, can also mimic stenotic pain localized only to the buttocks. In addition, a vascular examination should be performed in all patients to rule out vascular claudication.
B. Imaging
A noncontrast MRI scan is the most useful diagnostic tool for identifying spinal stenosis. In those who cannot have MRI scans (eg, those with pacemakers), a CT myelogram is necessary. There is little role for a plain CT scan in evaluating spinal stenosis, although it may be useful in identifying associated bony abnormalities or for surgical planning. Standing plain AP, lateral, and flexion-extension x-rays should be taken to rule out associated spinal instability or deformity.
![]() Treatment
Treatment
Nonoperative treatment modalities include the same ones used to treat lumbar disk herniations. Because stenosis rarely leads to progressive neurologic injury, nonoperative modalities are generally attempted first. However, whereas disk herniations may spontaneously resolve over time with resorption of fragments, spinal stenosis due to bony or ligamentous hypertrophy does not spontaneously regress with either time or nonoperative treatment. Similar to lumbar disk herniations, nonoperative treatment in lumbar spinal stenosis is purely symptomatic, meaning that it may help to improve pain symptoms but does not modify the underlying stenosis or make it “go away.” As such, it is debatable whether nonoperative therapies alter the natural history of spinal stenosis, and the literature shows little evidence of such. A recent meta-analysis demonstrated that the evidence in support of various nonoperative treatments is low grade at best. ESIs are a common nonoperative treatment; however, unlike for disk herniation, there does not appear to be a clinical advantage between intralaminar and transforaminal techniques. In milder cases of lumbar spinal stenosis, nonoperative treatment may lead to long-term improvement in symptoms. More commonly, however, especially in those with severe stenosis, the symptoms tend to recur and progress with time.
In those with persistent symptoms or progressive neurologic deficits, surgical treatment affords excellent outcomes. In the majority of cases of central and lateral recess stenosis, the procedure of choice is a decompressive laminectomy of the stenotic areas (Figure 4–17). A fusion is not generally indicated for spinal stenosis in the absence of instability. In those with symptomatic foraminal stenosis, a foraminotomy is additionally performed. If there is symptomatic foraminal stenosis with a tilting of the segment in the coronal plane that further narrows the foramen, then a fusion may be necessary to keep the foraminal height sufficient to relieve root compression postoperatively, even if a foraminotomy has been performed. In certain instances, particularly in the upper lumbar spine where the pars tends to be narrower, a satisfactory foraminotomy may require enough resection of the pars such that a fusion should be added because of the iatrogenic instability created in the process of decompressing the root.

![]() Figure 4–17. Anteroposterior radiographs before (A) and after (B) L4-L5 laminectomy for spinal stenosis. In most cases, central and lateral recess stenoses occur at the level of the disk space, where the facets, ligamentum flavum, and disk can converge to compress the neural elements. Thus, adequate decompression is generally achieved when the area spanning the rostral to the caudal portion of the facet joints has been relieved of neurologic compression (purple arrow).
Figure 4–17. Anteroposterior radiographs before (A) and after (B) L4-L5 laminectomy for spinal stenosis. In most cases, central and lateral recess stenoses occur at the level of the disk space, where the facets, ligamentum flavum, and disk can converge to compress the neural elements. Thus, adequate decompression is generally achieved when the area spanning the rostral to the caudal portion of the facet joints has been relieved of neurologic compression (purple arrow).
In the properly chosen patient, laminectomy provides excellent improvement in spinal stenosis symptoms. Reduction or elimination of radicular/claudicatory pain and improvement in physical function can be expected in 70–90% of cases. Again, patients should be counseled that the primary goal of treatment is relief of neurogenic rather than axial back pain, although a reduction in the latter may occur in up to 75% of cases.
Ammendolia C, Stuber K, de Bruin LK, et al: Nonoperative treatment of lumbar spinal stenosis with neurogenic claudication: a systematic review. Spine (Phila Pa 1976) 2012;37:E609. [PMID: 22158059]
Athiviraham A, Yen D: Is spinal stenosis better treated surgically or nonsurgically? Clin Orthop Relat Res 2007;458:90. [PMID: 17308483]
Atlas SJ, Keller RB, Wu YA, Deyo RA, Singer DE: Long-term outcomes of surgical and nonsurgical management of lumbar spinal stenosis: 8 to 10 year results from the Maine lumbar spine study. Spine (Phila Pa 1976)2005;30:936. [PMID: 15834339]
Koc Z, Ozcakir S, Sivrioglu K, Gurbet A, Kucukoglu S: Effectiveness of physical therapy and epidural steroid injections in lumbar spinal stenosis. Spine (Phila Pa 1976) 2009;34:985. [PMID: 19404172]
Malmivaara A: Surgical or nonoperative treatment for lumbar spinal stenosis? A randomized controlled trial. Spine (Phila Pa 1976) 2007;32:1. [PMID: 17202885]
Ruetten S, Komp M, Merk H, Godolias G: Surgical treatment for lumbar lateral recess stenosis with the full-endoscopic interlaminar approach versus conventional microsurgical technique: a prospective, randomized, controlled study. J Neurosurg Spine 2009;10:476. [PMID: 19442011]
Smith CC, Booker T, Schaufele MK, et al: Interlaminar versus transforaminal epidural steroid injections for the treatment of symptomatic lumbar spinal stenosis. Pain Med 2010;11:1511. [PMID: 20735751]
Weinstein JN, Tosteson TD, Lurie JD, et al: Surgical versus nonsurgical therapy for lumbar spinal stenosis. N Engl J Med 2008;358:794. [PMID: 18287602]
Zouboulis P: Functional outcome of surgical treatment for multilevel lumbar spinal stenosis. Acta Orthop 2006;77:670. [PMID: 16929447]
DEGENERATIVE DISK DISEASE
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Aging of the disk leads to dehydration and internal disk disruption.
• Most degenerative disks are asymptomatic.
• It is not possible to diagnose a painful disk by imaging studies alone.
![]() General Considerations
General Considerations
Aging of the intervertebral disk leads to a loss of water content, decrease in height, and an alteration in its normal mechanical properties. As a result, motion segment hypermobility, along with tears in the annulus, may occur. Although this degenerative cascade occurs ubiquitously in all spines with aging, it becomes symptomatic in only a small minority. It is currently unclear why some degenerative disks cause pain, whereas the vast majority do not, and there are no MRI or other imaging criteria that reliably differentiate symptomatic from nonsymptomatic degenerative disks. Thus, it is not possible to diagnose a painful degenerative disk on the basis of imaging studies alone.
We differentiate the term “degenerative disk,” which is a radiographic and anatomic description, from “degenerative disk disease” (DDD), which refers to the clinical syndrome of pain in the presence of a degenerative disk (Figure 4–18). Because the diagnosis of DDD is difficult to make, surgically treating diskogenic low back pain has relatively low success rates compared to other types of spinal surgery. Thus, surgical treatment of DDD is reserved for patients with severe symptoms that fail to respond to extensive nonoperative measures and in whom a clear disk pain generator has been identified (see Table 4–5). Heritability and genetics have been shown to be important risk factors for developing DDD. Potentially modifiable risk factors include smoking, obesity, and occupational exposures.

![]() Figure 4–18. (A) A 75-year-old man with symptoms of spinal stenosis and multilevel disk degeneration, but no low back pain. For reasons that are unclear, the majority of degenerative disks do not cause axial low back pain. Thus, one must not presume that degenerative changes found on imaging studies are necessarily responsible for symptoms, although they certainly can be. (B) A 37-year-old woman with single-level symptomatic degenerative disk disease at L5-S1. Note the relative loss of hydration (the central portion of the disk is darker, essentially black, compared to the more proximal disks) and disk height, along with Modic endplate changes (purple arrow).
Figure 4–18. (A) A 75-year-old man with symptoms of spinal stenosis and multilevel disk degeneration, but no low back pain. For reasons that are unclear, the majority of degenerative disks do not cause axial low back pain. Thus, one must not presume that degenerative changes found on imaging studies are necessarily responsible for symptoms, although they certainly can be. (B) A 37-year-old woman with single-level symptomatic degenerative disk disease at L5-S1. Note the relative loss of hydration (the central portion of the disk is darker, essentially black, compared to the more proximal disks) and disk height, along with Modic endplate changes (purple arrow).
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
Diskogenic low back pain classically is worse in flexion and better in extension. As with disk herniations, sitting is often the most aggravating position because it leads to the highest intradiskal pressures. Axial back pain may radiate to the buttock, thigh, or groin, but usually not past the knee in the absence of neurologic compression. Radiation past the knee in a dermatomal distribution typically indicates concomitant nerve root compression/irritation. It is important to determine the degree of relief afforded by rest. Pain that never improves, even at rest, suggests that mechanically based procedures such as spinal fusion may be unlikely to improve the pain.
B. Imaging
Radiographs typically show disk space narrowing, vertebral body osteophytes, or endplate and facet sclerosis. Radiographs should be taken upright to evaluate for potential deformities or instabilities that may not be apparent in the recumbent position. Flexion-extension films may rule out spondylolisthesis and instability. CT scans may rule out occult spondylolysis or tumors. MRI without contrast is the study of choice and demonstrates decrease in disk height and water content (dark on T2 imaging) with disk degeneration. High-intensity zones (HIZs) are focal areas of increased signal on T2-weighted images in the normally dark posterior/outer annulus indicative of annular tears or fissures. Associated endplate changes (“Modic” changes) indicative of endplate edema, fatty degeneration, or sclerosis may occur with disk degeneration as well. Patients with HIZs and Modic changes who undergo surgery for DDD tend to have better outcomes than those without such MRI findings. However, the presence of these findings is not diagnostic of symptomatic DDD.
Diskography is a useful but imperfect diagnostic modality. Because it is impossible to determine whether a disk is painful simply by its radiographic appearance, the concept behind provocative diskography is to identify the symptomatic disk by injecting it with contrast material under pressure. If the injection reproduces the patient’s typical low back pain (ie, demonstrates “concordant” pain), then that disk may be symptomatic. A CT scan can be obtained immediately following diskography to evaluate the morphology of the disk and its competence. Typically, a control level (an adjacent normal-appearing disk) is tested as well. Testing this level should produce no pain or a modest pressure sensation. If the patient reports severe pain at multiple levels, even those that are morphologically normal on MRI and postdiskography CT scan, he/she may be too sensitive to pain and less likely to benefit from a surgical procedure. The limited information to be gained from this test must be balanced against the risk of the procedure. A matched cohort study of patients who underwent diskography 7–10 years prior demonstrated significantly more disk degeneration and herniation at the side and levels of disks that had undergone diskography. This finding was consistent with those of previous animal studies.
![]() Treatment
Treatment
DDD is a challenging problem to manage, and there are no excellent treatment options. Because surgery is appropriate for only a select few, treatment always starts in a nonoperative fashion. Nonoperative options include those listed in Table 4–5. Lifestyle modifications, behavioral therapies, and management of associated psychosocial stressors may help DDD patients better cope with and manage their pain. Level 1 evidence demonstrates that patients with symptomatic DDD that is refractory to at least 6 months of nonoperative therapy may benefit from surgical treatment.
The most common surgical treatment of symptomatic DDD is spinal fusion. Surgery can be performed either anteriorly or posteriorly. Anterior surgery has the benefits of high fusion rates and avoiding the morbidity related to disruption of lumbar extensor muscles, but carries risks intrinsic to the anterior approach, such as vascular injury, retrograde ejaculation in men, and injury to the abdominal contents (Figure 4–19). Posterior fusion (either posterolateral and/or posterior interbody) generally involves pedicle screw instrumentation and provides rigid fixation, but it requires extensive posterior muscle dissection, which may exacerbate low back pain, and it also has higher infection rates and overall lower fusion rates than anterior surgery. All fusion techniques have been shown to demonstrate significantly better outcomes over nonoperative treatment in well-selected patients with single-level disk disease. Overall, about two thirds to three fourths of patients obtain significant and lasting relief from spinal fusion surgery directed at treating DDD.

![]() Figure 4–19. Anteroposterior and lateral radiographs of a 43-year-old man status post anterior lumbar inter-body fusion with cage and plate construct. Although this particular threaded, lumbar-tapered interbody cage can be used quite successfully (>95% fusion rate) as a stand-alone device, when the vascular anatomy permits and cage fixation is suboptimal, a plate can be added to further secure the construct. Excellent relief of symptoms was obtained.
Figure 4–19. Anteroposterior and lateral radiographs of a 43-year-old man status post anterior lumbar inter-body fusion with cage and plate construct. Although this particular threaded, lumbar-tapered interbody cage can be used quite successfully (>95% fusion rate) as a stand-alone device, when the vascular anatomy permits and cage fixation is suboptimal, a plate can be added to further secure the construct. Excellent relief of symptoms was obtained.
Nonfusion approaches include posterior dynamic stabilization and anterior lumbar disk arthroplasty. There is little evidence to support the use of posterior dynamic stabilization. On the other hand, recent 2-year FDA Investigational Device Exemption clinical trials comparing anterior arthroplasty to single- and two-level fusion in well-selected patients have shown significant improvements from baseline for both surgical treatments. In these studies, arthroplasty had an equivalent or superior clinical effect over fusion. However, it must be stressed that these are short-term results; the long-term functionality and survival characteristics for lumbar arthroplasties are unknown. Potential catastrophic implant failures, including potentially life-threatening expulsion of implants into the iliac vessels, have been reported, tempering enthusiasm for this approach.
![]() Complications
Complications
Commonly reported complications of instrumented posterior fusions include infection, nonunions, and implant-related issues such as malposition, loosening of screws, or backout of cages. Anterior complications include vascular injury (up to 3%), retrograde ejaculation due to hypogastric plexus injury (up to 5%), and cage migration. Fusion rates of anterior lumbar interbody fusions using rhBMP-2 and a threaded cage approach 100%. However, it has recently been reported that the rate of retrograde ejaculation may be higher when rhBMP-2 is used versus autograft. Fusion rates for posterior one- and two-level arthrodesis are 80–95%.
![]() Prognosis
Prognosis
DDD displays relatively poor outcomes compared to other lumbar spinal diagnoses regardless of either surgical or nonoperative management. About one third of patients fail to respond to all forms of treatment. Careful patient selection is the key to successful surgical outcomes. Patients with refractory low back pain and untreated underlying psychosocial conditions that exacerbate pain perception consistently have poorer surgical outcomes. Other poor prognostic factors include worker’s compensation status, pending litigation, obesity, and smoking. Proper counseling as to realistic expectations for pain reduction following surgery is paramount.
Berg S, Tullberg T, Branth B, Olerud C, Tropp H: Total disc replacement compared to lumbar fusion: a randomised controlled trial with 2-year follow-up. Eur Spine J 2009;18:1512. [PMID: 19506919]
Burkus JK: Six-year outcomes of anterior lumbar interbody arthrodesis with use of interbody fusion cages and recombinant human bone morphogenetic protein-2. J Bone Joint Surg Am 2009;91:1181. [PMID: 19411467]
Carragee EJ: Retrograde ejaculation after anterior lumbar inter-body fusion using rh-BMP-2: a cohort controlled study. Spine J 2011;11:511. [PMID: 21612985]
Carragee EJ, Don AS, Hurwitz EL, et al: 2009 ISSLS Prize Winner: does discography cause accelerated progression of degeneration changes in the lumbar disc: a ten-year matched cohort study. Spine (Phila Pa 1976)2009;34:2338. [PMID: 19755936]
Carragee EJ, Lincoln T, Parmar VS, Alamin T: A gold standard evaluation of the “discogenic pain” diagnosis as determined by provocative discography. Spine (Phila Pa 1976) 2006;31:2115. [PMID: 16915099]
Carreon LY, Glassman SD, Howard J: Fusion and nonsurgical treatment for symptomatic lumbar degenerative disease: a systematic review of Oswestry Disability Index and MOS Short Form-36 outcomes. Spine J 2008;8:747. [PMID: 18037354]
Cheh G: Adjacent segment disease following lumbar/thoracolumbar fusion with pedicle screw instrumentation: a minimum 5-year follow-up. Spine (Phila Pa 1976) 2007;32:2253. [PMID: 17873819]
Delamarter R, Zigler JE, Balderston RA, et al: Prospective, randomized, multicenter FDA IDE study of the ProDisc-L total disc replacement compared to circumferential arthrodesis for the treatment of two-level degenerative disc disease: results at twenty-four months. J Bone Joint Surg Am 2011;93:705. [PMID: 21398574]
Dimar JR: Clinical and radiographic analysis of an optimized rhBMP-2 formulation as an autograft replacement in posterolateral lumbar spine arthrodesis. J Bone Joint Surg Am 2009;91:1377. [PMID: 19487515]
Glassman SD, Polly DW, Bono CM, Burkus K, Dimar JR: Outcome of lumbar arthrodesis in patients sixty-five years of age or older. J Bone Joint Surg Am 2009;91:783. [PMID: 19339561]
Gornet MF, Burkus JK, Dryer RF, et al: Lumbar disc arthroplasty with MAVERICK disc versus stand-alone interbody fusion: a prospective randomized controlled multicenter IDE trial. Spine (Phila Pa 1976)2011;36:E1600. [PMID: 21415812]
Guyer RD: Prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: five-year follow-up. Spine J 2009;9:374. [PMID: 18805066]
Hsieh PC, Koski TR, O’Shaughnessy BA, et al: Anterior lumbar interbody fusion in comparison with transforaminal lumbar interbody fusion: implications for the restoration of foraminal height, local disc angle, lumbar lordosis, and sagittal balance. J Neurosurg Spine 2007;7:379. [PMID: 17933310]
Manchikanti L, Glaser SE, Wolfer L, Derby R, Cohen SP: Systematic review of lumbar discography as a diagnostic test for chronic low back pain. Pain Physician 2009;12:541. [PMID: 19461822]
Mirza SK, Deyo RA: Systematic review of randomized trials comparing lumbar fusion surgery to nonoperative care for treatment of chronic back pain. Spine (Phila Pa 1976) 2007;32:816. [PMID: 17414918]
Putzier M, Hoff E, Tohtz S, et al: Dynamic stabilization adjacent to single-level fusion: part II. No clinical benefit for asymptomatic, initially degenerated adjacent segments after 6 year follow-up. Eur Spine J2010;19:2181. [PMID: 20632044]
Soegaard R, Bünger CE, Christiansen T, Høy K, Eiskjaer SP, Christensen FB: Circumferential fusion is dominant over posterolateral fusion in a long-term perspective: cost-utility evaluation of a randomized controlled trial in severe, chronic low back pain. Spine (Phila Pa 1976) 2007;32:2405. [PMID: 18090078]
Videbaek TS: Circumferential fusion improves outcome in comparison with instrumented posterolateral fusion: long-term results of a randomized clinical trial. Spine (Phila Pa 1976) 2006;31:2875. [PMID: 17139217]
FACET SYNDROME
![]() General Considerations
General Considerations
Facet arthropathy and facet syndrome are relatively common but often underappreciated pain generators in patients with degenerative lumbar conditions. Unlike disk degeneration, which is ubiquitous in symptomatic and asymptomatic people, 15–90% of patients with chronic low back have obvious arthritic changes to their lumbar facet joints, whereas fewer than 15% of asymptomatic volunteers have arthritic changes on advanced imaging. Upward of 10% of patients following diskectomy may develop clinically symptomatic lumbar facet joint syndrome, which can be responsive to facet blocks. Revision or radical diskectomies significantly increase this risk.
Facet joints appear to be an independent generator of back pain. Although facet arthrosis may follow disk degeneration, facet syndrome and diskogenic back pain syndrome usually do not occur concurrently, as facet anesthetic blocks and diskogram are positive at the same level in fewer than 3–10% of cases. L5/S1 is the most commonly involved level, followed by L4/5 and L3/4. Facet degeneration is characterized by changes similar to those seen at other synovial joints—osteophytes, subchondral cysts, cartilage wear, joint space narrowing, and deformation (Figure 4–20).

![]() Figure 4–20. CT myelogram demonstrates severe spinal stenosis at L3-L4 along with bilateral facet arthropathy (black arrows).
Figure 4–20. CT myelogram demonstrates severe spinal stenosis at L3-L4 along with bilateral facet arthropathy (black arrows).
The facet capsule is richly innervated by the medial branch of the posterior ramus. Each posterior ramus divides to innervate two to three facet joints, including the joints associated with its neuroforamen and the ones above and below.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
One of the challenges with diagnosing facet syndrome (the cluster of chronic low back pain worse in extension, in the setting of facet arthritic changes that respond to facet local anesthetic blocks) is that it has no reliable signs or symptoms. Often there is referred pain that radiates into the buttock and posterior thigh (not below the knee) and sometimes into the groin as well. Lumbar extension, especially with the addition of truncal rotation, is often limited and painful. Controlled, comparative local anesthetic blocks are the diagnostic modality of choice. This requires two injections on two separate occasions of two types of local anesthetic—short acting (lidocaine) and long acting (bupivacaine). To confirm the diagnosis, the patient should have a positive response (pain relief and/or ability to perform previously painful maneuvers) after both injections and the response should be longer following the longer-acting anesthetic. Response to one and not the other is considered a false-positive result, which occurs in up to 50% of the cases. Unfortunately, multiple studies have demonstrated poor or no correlation between response to facet injections and response to nonoperative treatment or subsequent spinal fusion. Thus, while many spine surgeons believe facet degeneration is a pain generator in chronic low back pain, defining it as a unique entity or determining its contribution to a multifactorial etiology for low back pain remains unclear.
B. Imaging
It is thought in most instances that disk degeneration and height loss precede facet arthrosis. Facet orientation, which is best measured on CT scan, tends to become progressively more sagittal as arthrosis progresses. Patients with more sagittally oriented facet joints are more prone to develop spondylolisthesis. MRI and CT scans often demonstrate lateral recess and foraminal stenosis from hypertrophy of the superior articulating process in advanced facet arthropathy. Like other synovial joints, arthrosis can lead to the formation of synovial cysts. Facet cysts most commonly form on the posterior aspect of the joint, but those on the ventral or medial surface can compress nerve roots, causing radiculopathy.
![]() Treatment
Treatment
Facet syndrome is primarily treated nonoperatively with the modalities listed in Table 4–5. After clinical examination, facet local anesthetic blocks are a potential next step for the diagnosis and treatment of facet syndrome. Patients who have a positive response to facet blocks (>50–80% pain reduction) are candidates for intraarticular steroid injections or radiofrequency ablations (RFAs). Steroid injections provide long-term back pain relief in 16–63% of patients. RFA involves placing a radiofrequency probe under radiographic guidance and thermocoagulating the medial branch of the posterior ramus as it enters the facet capsule. Using strict selection criteria (positive controlled comparative blocks), it is thought that RFA provides up to 1 year of 80–90% pain relief in 60% of patients, with 87% of patients getting at least 60% pain relief. Randomized controlled trials have shown that RFA is superior to placebo. Other studies have found no significant treatment effect following facet steroid injections or RFA compared to saline placebo. RFA and intraarticular steroid injections, like many nonoperative interventions, tend to produce diminishing returns with repeated application. Last, posterior fusion may be a final treatment option for facet syndrome, with limited and contradictory evidential support. In general, facet syndrome should not be viewed as a surgical disease; further, favorable results from facet injections may not predict good outcomes from fusion surgery.
Cohen SP: Lumbar zygapophysial (facet) joint radiofrequency denervation success as a function of pain relief during diagnostic medial branch blocks: a multicenter analysis. Spine J 2008;8:498. [PMID: 17662665]
Cohen SP, Hurley RW: The ability of diagnostic spinal injections to predict surgical outcomes. Anesth Analg 2007;105:1756. [PMID: 18042881]
Cohen SP, Raja SN: Pathogenesis, diagnosis, and treatment of lumbar zygapophysial (facet) joint pain. Anesthesiology 2007;106:591. [PMID: 17325518]
Dreyfuss P, Halbrook B, Pauza K, Joshi A, McLarty J, Bogduk N: Efficacy and validity of radiofrequency neurotomy for chronic lumbar zygapophysial joint pain. Spine (Phila Pa 1976) 2000;25:1270. [PMID: 10806505]
Esses SI, Moro JK: The value of facet joint blocks in patient selection for lumbar fusion. Spine (Phila Pa 1976) 1993;18:185. [PMID: 8441932]
Jackson RP, Jacobs RR, Montesano PX: 1988 Volvo award in clinical sciences. Facet joint injection in low-back pain. A prospective statistical study. Spine (Phila Pa 1976) 1988;13:966. [PMID: 2974632]
Nath S, Nath CA, Pettersson K: Percutaneous lumbar zygapophysial (facet) joint neurotomy using radiofrequency current, in the management of chronic low back pain: a randomized double-blind trial. Spine (Phila Pa 1976)2008;33:1291. [PMID: 18496338]
Steib K, Proescholdt M, Brawanski A, et al: Predictors of facet joint syndrome after lumbar disc surgery. J Clin Neurosci 2012;19:418. [PMID: 22277562].
Stojanovic MP, Sethee J, Mohiuddin M, et al: MRI analysis of the lumbar spine: can it predict response to diagnostic and therapeutic facet procedures? Clin J Pain 2010;26:110. [PMID: 20090436]
van Wijk RM, Geurts JW, Wynne HJ, et al: Radiofrequency denervation of lumbar facet joints in the treatment of chronic low back pain: a randomized, double-blind, sham lesion-controlled trial. Clin J Pain2005;21:335. [PMID: 15951652]
Wong DA, Annesser B, Birney T, et al: Incidence of contraindications to total disc arthroplasty: a retrospective review of 100 consecutive fusion patients with a specific analysis of facet arthrosis. Spine J2007;7:5. [PMID: 17197326]
SPONDYLOLISTHESIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• There are six distinct types of spondylolisthesis.
• Signs and symptoms are dependent on type but often include a combination of low back pain and leg pain.
• Degree of slippage and slip angle and overall sagittal balance are important considerations in choosing the optimal treatment.
![]() General Considerations
General Considerations
Spondylolisthesis is derived from the Greek spondylo (meaning spine) and olisthesis (meaning slip) and refers to an abnormal slippage of one vertebra over the next. There are six types of spondylolisthesis (Table 4–6). The overall lifetime incidence of spondylolisthesis is 9–10%, with isthmic and degenerative being the most common types.
Table 4–6. Wiltse Classification of spondylolisthesis.

![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
1. Degenerative spondylolisthesis— Patients are typically older (50s and up) and complain of varying amounts of low back and/or leg pain. The radicular leg symptoms are similar to those of spinal stenosis, as central and lateral recess stenosis is exacerbated by the slip. Foraminal stenosis can also occur. Thus, the radicular symptoms may be associated with both the exiting and traversing roots at the level of stenosis. The L4/5 level is by far the most common level affected by degenerative spondylolisthesis, although it can occur at other lumbar levels. Associated axial low back pain from instability should typically be better with rest and worse with standing or walking.
2. Isthmic spondylolisthesis—Patients may present in their preteen to teen years or be relatively asymptomatic until their adulthood (30s to 50s). L5-S1 is by far the most common level involved, although isthmic defects can be seen at other levels (Figure 4–21). Central stenosis is relatively uncommon with isthmic defects, as the pars defect actually enlarges the spinal canal with slippage of the associated vertebrae. Instead, foraminal narrowing and resulting exiting root compression are typical, with the L5 root being most commonly involved with an L5-S1 isthmic slip. Low back pain when present tends to be mechanical (worse with activity) and may be acutely associated with a “clunking” sensation in flexion and extension.

![]() Figure 4–21. (A) Degenerative spondylolisthesis at L4-L5, the most common level at which they occur. (B) MRI of same patient showing fluid (white arrow) in the severely degenerative L4/5 facet. (C) Isthmic spondylolisthesis at L5-S1 (grade 1) (0–25% slippage). Although isthmic defects occur most commonly at L5-S1, they can occur elsewhere. (D) Note the pars defect (white arrow).
Figure 4–21. (A) Degenerative spondylolisthesis at L4-L5, the most common level at which they occur. (B) MRI of same patient showing fluid (white arrow) in the severely degenerative L4/5 facet. (C) Isthmic spondylolisthesis at L5-S1 (grade 1) (0–25% slippage). Although isthmic defects occur most commonly at L5-S1, they can occur elsewhere. (D) Note the pars defect (white arrow).
B. Imaging
The degree of spondylolisthesis is commonly classified according to the Meyerding grade (Table 4–7). Degenerative slips tend to be low grade, with the majority being grade I or, less frequently, grade II or III. Standing lateral x-rays are paramount to diagnosis, as the slip may not be visible on recumbent films such as supine x-rays or CT/MRI scans. Flexion-extension films may demonstrate dynamic worsening of instability (>2–4 mm motion). Lateral listhesis (slippage in the coronal plane) can also occur alone or in combination with spondylolisthesis. MRI scans are obtained to evaluate associated neural impingement. CT scans with sagittal reformatting can be helpful in identifying pars defects that may not be evident on x-ray or MRI. Bone scans are occasionally useful in identifying occult pars defects.
Table 4–7. Meyerding slippage grade.

![]() Treatment
Treatment
Slips in adults with isthmic or degenerative spondylolisthesis rarely progress. However, growing children with isthmic defects may progress. In addition, children with dysplastic slips and adults with iatrogenic slips (typically resulting from a pars insufficiency fracture after previous diskectomy/decompression procedure) are much more likely to progress and thus should be fused or at least followed carefully. The standard nonoperative treatment options (see Table 4–5) apply to the initial treatment of spondylolisthesis. Decompression alone is rarely appropriate for patients with any form of spondylolisthesis as it can further destabilize the segment or leave persistent symptoms (back and/or leg pain) related to instability. The most common treatment for either degenerative or isthmic spondylolisthesis is a posterior lumbar decompression and fusion. Recent randomized control trials have shown that surgery in the form of one- to two-level posterior decompression and fusion for degenerative spondylolisthesis with stenosis reliably and significantly outperforms nonoperative care at all times points out to 5 years. Fusion for degenerative and isthmic spondylolisthesis tends to be a very successful operation with 75% or more of patients reporting major improvement in their condition that lasts 2–4 years or longer. To put this in generic terms, the improvement in overall measures of general health and well-being (eg, Short Form-36) following fusion for spondylolisthesis is equivalent to that for hip/knee arthroplasty.
Ha KY, Na KH, Shin JH, Kim KW: Comparison of posterolateral fusion with and without additional posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis. J Spinal Disord Tech2008;21:229. [PMID: 18525481]
Hu SS, Tribus CB, Diab M, Ghanayem AJ: Spondylolisthesis and spondylolysis. J Bone Joint Surg Am 2008;90:656. [PMID: 18326106]
Kalichman L, Hunter DJ: Diagnosis and conservative management of degenerative lumbar spondylolisthesis. Eur Spine J 2008;17:327. [PMID: 18026865]
Kim JS, Kang BU, Lee SH, et al: Mini-transforaminal lumbar interbody fusion versus anterior lumbar interbody fusion augmented by percutaneous pedicle screw fixation: a comparison of surgical outcomes in adult low-grade isthmic spondylolisthesis. J Spinal Disord Tech 2009;22:114. [PMID: 19342933]
Müslüman AM, Yilmaz A, Cansever T, et al: Posterior lumbar interbody fusion versus posterolateral fusion with instrumentation in the treatment of low-grade isthmic spondylolisthesis: midterm clinical outcomes. J Neurosurg Spine 2011;14:488. [PMID: 21314280]
Rampersaud YR, Wai EK, Fisher CG, et al: Postoperative improvement in health-related quality of life: a national comparison of surgical treatment for (one- or two-level) lumbar spinal stenosis compared with total joint arthroplasty for osteoarthritis. Spine J 2011;11:1033.
Remes V, Lamberg T, Tervahartiala P, et al: Long-term outcome after posterolateral, anterior, and circumferential fusion for high-grade isthmic spondylolisthesis in children and adolescents: magnetic resonance imaging findings after average of 17-year follow-up. Spine (Phila Pa 1976) 2006;31:2491. [PMID: 17023860]
Resnick DK, Choudhri TF, Dailey AT, et al: Guidelines for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 9: fusion in patients with stenosis and spondylolisthesis. J Neurosurg Spine2005;2:679. [PMID: 16028737]
Swan J: Surgical treatment for unstable low-grade isthmic spondylolisthesis in adults: a prospective controlled study of posterior instrumented fusion compared with combined anterior-posterior fusion. Spine J2006;6:606. [PMID: 17088191]
Weinstein JN: Surgical compared with nonoperative treatment for lumbar degenerative spondylolisthesis. Four-year results in the Spine Patient Outcomes Research Trial (SPORT) randomized and observational cohorts. J Bone Joint Surg Am 2009;91:1295. [PMID: 19487505]
Weinstein JN, Lurie JD, Tosteson TD, et al: Surgical versus non-surgical treatment for lumbar degenerative spondylolisthesis. N Engl J Med 2007;356:2257. [PMID: 17538085]
SPINAL INFECTIONS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Most common forms of spinal infections include diskitis, osteomyelitis, epidural abscess, and postoperative infections.
• Lumbar spine is most commonly involved followed by the thoracic spine in bacterial infections.
• Thoracic spine most commonly involved in fungal infections.
• Pathophysiology of infection differs between children and adults.
• Back pain is the most common symptom and is severe and unremitting.
![]() General Considerations
General Considerations
Spinal infections represent fewer than 5% of orthopedic infections. The lumbar spine is the most common region involved (50%), followed by the thoracic spine (35%). The most common forms of spinal infection include (1) diskitis, (2) osteomyelitis, (3) epidural abscess, and (4) postoperative (infection complicates 0–5% of spinal surgeries). Staphylococcus aureus is the most common (40–60%) organism for all types of spinal infection, followed by coagulase-negative Staphylococcus and gram-negative enteric organisms, and a culture-specific diagnosis remains paramount to treatment. Propionibacterium acneand Staphylococcus epidermidis are common pathogens in late-onset spinal infections following instrumented spinal fusion. These low-virulence pathogens are cultured from the skin in over 80% of subjects prior to skin preparation for surgery.
The pathoanatomy of pyogenic diskitis and osteomyelitis is different in children versus adults. In children, the disk can become primarily infected via blood vessels that cross the endplate and connect the vertebrae to the nucleus. In adults, the disk is avascular and becomes secondarily infected from a primary focus of infection in the vertebral body (ie, osteomyelitis). Hematogenous osteomyelitis typically starts in the anterior-inferior corner of the vertebral body. In this portion of the vertebral body, the end-arterioles loop and create sluggish blood flow. Additionally, the venous drainage of the vertebral body is valveless (Batson plexus) and communicates with the pelvic venous system. These two anatomic features may facilitate bacterial sludging and extravasation. As the infection progresses, the endplates erode, allowing spread of infection into the adjacent avascular disk (Figure 4–22). It is the involvement of adjacent vertebral levels and the intervening disk that most reliably differentiates pyogenic diskitis from neoplasm, as tumors rarely cross the disk space. From the disk space, infection can spread to adjacent vertebral bodies and/or paravertebral structures. In some advanced cases (5–18%), infection can spread posteriorly into the spinal canal as an epidural abscess. Epidural abscess can also form hematogenously into the posterior epidural space without associated diskitis/osteomyelitis. An epidural abscess is the most urgent of spinal infections due to the potential for progressive neurologic deficit.

![]() Figure 4–22. (A) Lateral radiograph of a morbidly obese brittle diabetic man with osteomyelitis-diskitis involving L2-L3 and L4-L5. (B) CT reconstruction demonstrates erosion of the endplates at the bottom of L2, top of L3, bottom of L4, and top of L5 due to infection. (C, D, and E) Sagittal, axial, and coronal MRIs show extensive infection at L2-L3 and L4-L5 with bilateral psoas abscesses (black arrows).
Figure 4–22. (A) Lateral radiograph of a morbidly obese brittle diabetic man with osteomyelitis-diskitis involving L2-L3 and L4-L5. (B) CT reconstruction demonstrates erosion of the endplates at the bottom of L2, top of L3, bottom of L4, and top of L5 due to infection. (C, D, and E) Sagittal, axial, and coronal MRIs show extensive infection at L2-L3 and L4-L5 with bilateral psoas abscesses (black arrows).
Nonbacterial infections are less common. Fungal infections are often indolent but can lead to multilevel involvement and significant deformity. Tuberculous spinal infection (Pott disease) is also an indolent infection, which has a predilection for the thoracic spine, but only affects about 1–5% of patients with tuberculosis (TB). Like fungal infections, kyphotic deformity is often the most clinically significant adverse effect. TB may mimic tumor because it tends to spare the disk space, presumably because TB is an obligate aerobic organism and the oxygen tension in the disk space is low. A purified protein derivative test will be positive in >95% of immune-competent patients, but may be anergic in immune-compromised patients. Traditional TB cultures took 21 days or longer for results, but recently polymerase chain reaction has become the test of choice to confirm TB infection.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
Symptoms of spinal infection are widely variable. Back pain localized to the area of infection is the most common symptom. Back pain associated with infections is classically more severe and unremitting (often worse at night) compared to that seen with degenerative disorders, although many patients do have mechanical symptoms that modulate with level of activity. Constitutional symptoms such as fever are not sensitive and may be present in only about 33–50% of spinal infections. Furthermore, fever is a common (>40%, >75% following major fusion surgery) phenomenon in the immediate postoperative period for uncomplicated spinal surgeries. Neurologic symptoms, such as radiating pain, numbness, or weakness, may occur and are associated with epidural extension of infection or pathologic fracture of infected vertebrae causing kyphosis and compression of the neural elements. Those at risk for infection include the elderly, diabetics, and those on chronic steroids.
Hemodynamically unstable septic shock is rare but can occur with spinal infections. The physical exam may be normal or demonstrate limited range of motion of the affected spine. Neurologic deficits are relatively uncommon at presentation (10–17%).
B. Laboratory Studies
Baseline laboratories for patients presenting with suspected spinal infection include complete blood count (CBC) with differential, C-reactive protein (CRP), ESR, and blood cultures from two separate sites. Leukocytosis and a left shift will be present in less than 50%, but CRP and ESR will be elevated in more than 90% at presentation. The CRP starts to elevate within 24–48 hours of infection onset, whereas ESR can take up to 1 week to elevate. In addition to being first to elevate, CRP is the first to normalize following initiation of successful treatment. As such, it is an important tool in monitoring treatment response. Blood cultures are positive in only 30–50% of patients. Antibiotics should be withheld until blood cultures and preferably tissue cultures can be obtained. CT-guided needle biopsy of the most accessible site of presumed infection is the culture method of choice and yields positive results in 50–80% of patients, but it is imperative to obtain large enough samples to culture. If an organism cannot be identified or in patients with obvious infections requiring surgical debridement and reconstruction, open biopsy is performed with 80–90% positive yield in patients without recent antibiotic exposure.
C. Imaging
Plain radiographs in osteomyelitis may demonstrate radio-lucency from osteolysis or sclerosis in chronic infections. However, plain films may be negative early in the disease process, as radiolucency is not seen until 30–50% of the trabeculae have been lost, which may take up to 3–4 weeks to develop. Disk space narrowing is an early radiographic finding of diskitis. As the infection progresses, spinal deformities such as focal kyphosis may be seen.
MRI with gadolinium contrast is the study of choice for diagnosing all forms of spinal infections. Its sensitivity and specificity are more than 95%. MRI changes occur within 24–48 hours of infection onset. MRI additionally allows for evaluation of neurologic compression. However, the true extent of bony destruction is better evaluated on CT scans. Typically, both an MRI and CT scan are needed to fully evaluate spinal infections. Nuclear medicine studies (technetium-99 or gallium studies) reliably (90% accuracy) show uptake in areas of infection within 3–7 days of onset but are less commonly used now that MRI is widely available and provides better anatomic detail.
![]() Treatment
Treatment
The goals of treating pyogenic infections are fourfold: (1) to eradicate the infection; (2) relieve pain; (3) preserve or improve neurologic function; and (4) maintain or restore spinal alignment and stability. Indications for surgical treatment include the following: (1) to obtain a microbiologic diagnosis; (2) presence of abscess (intradiskal, bony, soft tissue, or epidural); (3) presence of neurologic deficit; (4) spinal instability; (5) progressive or severe spinal deformity; and (6) failure of nonoperative treatment, including refractory pain. The major goal of surgery is thorough debridement of all infected, nonviable tissue. Spinal reconstruction with instrumentation is often used to stabilize the spine after debridement. Although somewhat counterintuitive, the use of spinal implants, particularly those made of titanium, to fuse and stabilize the infected spine helps to successfully eradicate the infection, provided there has been adequate debridement and institution of culture-specific antibiotics. In the absence of the above indications, a term of nonoperative treatment may be considered. Failure of nonoperative treatment is indicated by lack of improvement in pain, fever, and/or laboratory parameters despite 2–3 weeks of appropriate antibiotic coverage.
In most cases, an anterior approach is necessary to radically debride the infection. Supplemental posterior instrumentation and fusion are usually needed to provide additional stability in the majority of cases, as the spine is rendered unstable either by the infection or the debridement needed to remove the infection. Anterior bone quality is generally poor in patients with infection and should not routinely be relied upon alone for fixation. Epidural abscesses are most commonly drained through a dorsal approach and are most common in the thoracolumbar spine. Classically, autograft has been used to graft anterior column deficits. More recently, allograft or titanium mesh cages combined with rhBMP-2 have been used with success to avoid the morbidity of autograft harvest. A prolonged course of postoperative antibiotics, typically parenteral for a minimum of 6 weeks, is needed. Nutritional supplementation plays a crucial role in healing of these patients, who are or become malnourished by the chronic infection.
![]() Prognosis
Prognosis
Spinal infection is a serious disease, which historically was fatal in 50–70% of cases and even today has an overall mortality rate of 5–20%. Debridement and instrumented fusion can be successfully performed in a single stage with 90–100% initial response and less than 10% recurrence. When postoperative infections are identified and adequately treated in the acute phase (<4–6 weeks after surgery), originally placed implants can generally be left.
Allen RT, Lee YP, Stimson E, Garfin SR: Bone morphogenetic protein-2 (BMP-2) in the treatment of pyogenic vertebral osteomyelitis. Spine (Phila Pa 1976) 2007;32:2996. [PMID: 18091493]
Grane P, Josephsson A, Seferlis A, Tullberg T: Septic and aseptic post-operative discitis in the lumbar spine—evaluation by MR imaging. Acta Radiol 1998;39:108. [PMID: 9529438]
Hahn F, Zbinden R, Min K: Late implant infections caused by Propionibacterium acnes in scoliosis surgery. Eur Spine J 2005;14:783. [PMID: 15841406]
Kuklo TR, Potter BK, Bell RS, Moquin RR, Rosner MK: Single-stage treatment of pyogenic spinal infection with titanium mesh cages. J Spinal Disord Tech 2006;19:376. [PMID: 16826013]
Mok JM, Pekmezci M, Piper SL, et al: Use of C-reactive protein after spinal surgery: comparison with erythrocyte sedimentation rate as predictor of early postoperative infectious complications. Spine (Phila Pa 1976)2008;33:415. [PMID: 18277874]
Ogden AT, Kaiser MG: Single-stage debridement and instrumentation for pyogenic spinal infections. Neurosurg Focus 2004;17:E5. [PMID: 15636575]
O’Shaughnessy BA, Kuklo TR, Ondra SL: Surgical treatment of vertebral osteomyelitis with recombinant human bone morphogenetic protein-2. Spine (Phila Pa 1976) 2008;33:E132. [PMID: 18317180]
Petignat C, Francioli P, Harbarth S, et al: Cefuroxime prophylaxis is effective in noninstrumented spine surgery: a double-blind, placebo-controlled study. Spine (Phila Pa 1976) 2008;33:1919. [PMID: 18708923]
Savage JW, Weatherford BM, Sugrue PA, et al: Efficacy of surgical preparation solutions in lumbar spine surgery. J Bone Joint Surg Am 2012;94:490. [PMID: 22437997]
Schimmel JJ, Horsting PP, de Kleuver M, et al: Risk factors for deep surgical site infections after spinal fusion. Eur Spine J 2010;19:1711. [PMID: 20445999]
SPINAL TUMORS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Metastatic tumors are the most common tumors of the spine.
• Eighty percent of metastases to the spine come from cancer of the prostate, breast, or lung.
• Most common benign primary tumor of the spine is hemangioma.
• Back pain is the most common presenting symptom.
![]() General Considerations
General Considerations
Metastases represent the most common tumors of the spine. The spine is the most common location for skeletal metastasis, with the thoracic spine being the most commonly involved region. Typical spinal metastases include tumors of the prostate, breast, lung, thyroid, and kidney. Not unexpectedly, since they are the most prevalent cancers, 80% of spinal metastases originate from prostate, breast, or lung primary malignancies. In contrast, primary malignant bone tumors are comparatively rare and include chondrosarcoma, osteosarcoma, and chordoma. Lymphomas, solitary plasma-cytomas, and multiple myelomas are also seen in the spine. Most (70%) malignant tumors arise in patients older than 21 years and typically occur in the anterior spine (vertebral body and pedicles). Chondrosarcoma is a notable exception that typically arises from the posterior elements.
The most common benign tumor of the spine is hemangioma, present in 10–12% of people. Most patients present with asymptomatic, incidental findings on MRI scans for other reasons (eg, back pain or sciatica), but in rare cases, hemangiomas can become large and symptomatic. Other than hemangiomas, which are frequently seen in older patients, benign bone tumors are much more common in younger patients. Osteoid osteomas and osteoblastomas are benign primary bone tumors that tend to involve the posterior elements. They are classically associated with non-mechanical back pain that tends to be worse at night and that improves with NSAIDs. Other benign bone tumors of the spine include aneurysmal bone cyst, fibrous dysplasia, and giant cell tumors. Giant cell tumors, although histologically benign, may be locally aggressive and even metastasize.
Another tumorlike finding that is a relatively common incidental finding on routine MRI for evaluation of back pain or sciatica is syrinx (a fluid-filled space within the spinal cord) or persistence of the central canal of the spinal cord. When seen, pathologic reasons, such as intramedullary tumor, Chiari malformation, spinal canal anomaly, tethered cord, or severe spinal stenosis, can be excluded with a screening MRI of the whole spine, preferably with contrast. However, about 90% of syringes are stable incidental findings not correlated with the clinical symptoms that elicited the MRI. In such cases, in the absence of progressive neurologic deficits or planned surgical intervention, expectant observation is a reasonable option as well.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
The presentation of spinal tumors is often similar to that of spinal infections. Axial back pain is the most common complaint, present in about 90% of cases. Associated neurologic symptoms may be present if the tumor impinges neural elements or is associated with pathologic fracture and neural compression (ie, metastatic spinal cord compression). The presence of constitutional symptoms such as fevers, night sweats, or weight loss should be elicited. Back pain is often constant and unremitting, classically worse at night time, but may also have a component made worse by mechanical loading. Patients with new-onset back pain and a history of cancer need to be ruled out for metastatic disease to the spine. Physical findings include local tenderness over the tumor, limited range of motion, and, in advanced cases, deformity from vertebral body collapse and kyphosis.
B. Imaging and Laboratory Data
The radiologic workup of spinal tumors is similar to that of spinal infections. MRI remains the mainstay of diagnosis, with CT scans also obtained to assess the degree of bony destruction, the pattern of bone response to the lesion, and the presence of matrix, and weight-bearing plain films to assess overall alignment and deformity. Plain films are poorly sensitive for tumors: approximately 40% trabecular bone loss must be present before being detectable on plain film as a lucent area. Thus advanced imaging is critical to making a prompt diagnosis. Because of its ability to provide fine bony detail, CT scans are helpful in determining the aggressiveness of lytic lesions. Slow-growing benign tumors usually display a sharp transition zone with reactive, sclerotic bone surrounding the tumor as the bone has had time to respond. This is called a geographic 1A lesion. Aggressive, fast-growing tumors usually demonstrate a moth-eaten appearance with little reactive bone and a wide transition zone. Biopsy should be considered for spinal tumors that cannot be diagnosed based on imaging alone. Most commonly, needle biopsy under CT guidance is sufficient, but several large-bore specimens should be obtained to ensure a tissue diagnosis. In the case of widely metastatic disease, biopsy of a more superficial tumor may be considered if it is easier to access. If a needle biopsy is inconclusive, then open biopsy may be needed. In cases in which a curative resection is contemplated, the biopsy must be planned appropriately to allow for excision of the biopsy tract during the definitive operation.
A metastatic workup should be considered in all patients with spinal tumors, which includes a CT scan of the chest, abdomen, and pelvis. Whole-body bone scan can also be helpful in detecting malignant or metastatic foci. Unfortunately, only about 50% of multiple myelomas display increased uptake on bone scans. Bone scans detect tumors based on increased regional blood flow to the tumor and by radiolabeling (with technetium-99) phosphates, which are deposited in areas of active bone formation. Routine laboratory data include CBC with differential, calcium, Mg, Po4, and liver function tests. Serum and urine electrophoresis can be helpful in diagnosing multiple myeloma.
Preoperative embolization of tumors may be helpful in decreasing intraoperative bleeding, making surgery safer, easier, and more effective. Likewise, selective embolization is a treatment option for large, symptomatic hemangiomas. In addition, with tumors involving the lower thoracic spine, angiography may be helpful in identifying the major feeding vessel to the spinal cord (the artery of Adamkiewicz) so that it can be spared surgically and avoid potential vascular infarction of the spinal cord.
![]() Treatment
Treatment
Treatment is dictated by a number of factors, including age (<65 or >65 years), tumor type and stage, symptom severity, presence of neurologic compression or neurologic symptoms (most importantly, ambulatory status), expected life span (<3 or >3 months), and mechanical stability. Most benign asymptomatic lesions require no treatment, but depending on the certainty of the diagnosis, periodic follow-up with CT scans is prudent. Timely repeat imaging is also indicated if symptoms worsen during clinical observation. Indications for surgery include absence of viable nonsurgical treatment modalities, intractable pain despite nonsurgical treatment, mechanical instability, neurologic preservation or need for decompression, and, rarely, in certain primary tumors of the spine, curative resection. Percutaneous cement augmentation is a minimally invasive intervention with an expanding role in the treatment of painful spinal neoplastic lesions. The treatment of hemangiomas was the initial indication for this technique, which is now most commonly used to treat osteoporotic vertebral compression fractures.
Steroids are often used in the setting of tumors causing myelopathy from spinal cord compression. Bisphosphonates are also commonly used by oncologists to manage bone loss associated with neoplastic disease of the spine. They have been shown to significantly reduce the incidence of skeletally related (adverse) events (pathologic fracture, need for radiation or surgery to bone, or spinal cord compression) in patients with multiple myeloma and osteolytic metastases.
A. Metastatic Tumors
Most metastatic tumors without neurologic compression are treated nonsurgically with radiation and/or chemotherapy, depending on the tumor type and its responsiveness to those modalities. However, in the presence of symptomatic spinal cord compression, recent level 1 evidence, which has since been corroborated in other studies, concludes that surgery prior to radiation therapy is superior to radiation therapy alone or radiation prior to surgery in terms of neurologic outcomes. An exception arises if neurologic compression is due solely to a highly radiosensitive tumor, with no significant spinal instability or bony compressive component, in which case radiation may be considered first. Unless the tumor is highly radiosensitive or the patient is a poor surgical candidate, we generally advocate performing surgery first in patients with symptomatic neurologic compression in order to avoid progressive neurologic deterioration and complications associated with surgery through a previously irradiated field.
Surgery may also be considered in those without actual but impending neurologic compromise due to spinal instability. In general, those with more than 50% vertebral body involvement or destruction of the facet joints, pars, and pedicles are at risk for instability and resultant neurologic compromise. Unfortunately, in clinical practice, the assessment of spinal stability is often not straightforward, typically requiring a nuanced interpretation of biomechanical criteria based on the “personality” or pattern of tumor involvement seen on a combination of plain film, CT, and MRI.
The choice of surgical approach depends on a number of factors. In the vast majority of metastatic cases, the goal is palliation of pain and prevention or reversal of neurologic impairment. Thus the operation should be designed to achieve those goals while minimizing morbidity. Depending on the location and pattern of compression, the decompression and reconstruction can be performed anteriorly, posteriorly, or combined.
Postoperative radiation may be helpful in removing residual tumor left behind in order to decrease recurrence. However, acute postoperative radiation therapy may increase the rate of nonunions and wound infection. Thus, it is generally preferable to wait anywhere from 3 to 6 weeks after surgery to institute radiation. The choice of spinal reconstruction will depend on the patient’s overall prognosis. Those with a reasonable life expectancy beyond 1–2 years may benefit from achieving solid bony union, but this must be balanced against the potentially higher bleeding and infection risks associated with bone grafting and fusion. Those with poor prognoses for survival may be best managed with decompression and instrumentation only. In these circumstances, cement can be used to augment vertebral corpectomies and provide immediate mechanical stability of vertebral body defects. Pain related to impending or actual pathologic fractures may be managed in certain instances with percutaneous cement procedures, such as vertebroplasty or kyphoplasty, which have the advantage of being less invasive.
B. Benign Primary Bone Tumors
Osteoid osteomas are typically seen in the posterior elements of teenagers (10–20 years old) and may be associated with acute scoliosis. CT scans demonstrate a central (lucent) nidus of tumor (sometimes with a small focus of bone at its center) surrounded by a sclerotic rim of reactive bone. Spontaneous regression can occur. If the symptoms are severe or persist despite time and NSAIDs, thorough curettage of the lesion yields good outcomes. If the nidus is removed, recurrence is relatively unlikely. Good results have also been reported with RFA. Most cases of osteoid osteoma or osteoblastoma-related scoliosis are not structural and spontaneously improve after tumor resection.
Osteoblastomas represent osteoid osteomas that are larger than 2 cm. They typically occur in patients younger than 30 years and can also be associated with scoliosis. When they occur in the spine, they tend to be in the posterior arch. Marginal excision provides excellent outcomes with relatively low recurrence rates. Depending on the extent of the excision, a fusion may be necessary.
Aneurysmal bone cysts cause hyperemic lytic lesions, most commonly in the posterior elements but not infrequently extending into the anterior column of the lumbar spine as well (Figure 4–23). As with most benign primary bone tumors, patients tend to be younger, typically younger than 20 years. Expansion may cause neural compression and radiculopathy or myelopathy. MRI demonstrates characteristic fluid-fluid levels due to the layering of blood within the lesion. Curettage and resection of aneurysmal bone cysts with bone grafting is the treatment of choice. Again, fusion may be needed depending on the extent of the lesion. Preoperative embolization should strongly be considered to limit intraoperative bleeding.

![]() Figure 4–23. Aneurysmal bone cyst of the spine in a 27-year-old woman with severe low back pain and leg weakness. (A) Anteroposterior radiograph demonstrates destruction of the L2 pedicle along with lytic changes in the L2 vertebra (black arrow). (B) Axial MRI shows an expansile, cystic lesion with fluid-fluid level causing compression of the spinal canal. (C) CT scan shows expansile cortical margin with lytic erosion.
Figure 4–23. Aneurysmal bone cyst of the spine in a 27-year-old woman with severe low back pain and leg weakness. (A) Anteroposterior radiograph demonstrates destruction of the L2 pedicle along with lytic changes in the L2 vertebra (black arrow). (B) Axial MRI shows an expansile, cystic lesion with fluid-fluid level causing compression of the spinal canal. (C) CT scan shows expansile cortical margin with lytic erosion.
Giant cell tumors are seen in slightly older individuals (20s to 40s) and arise most commonly in the anterior spine. Curettage and bone grafting may be performed but have a high recurrence rate of up to 45%. Thus, en bloc resection should be considered when feasible to prevent long-term recurrences. Radiation treatment may be associated with sarcomatous transformation in up to 15% of patients and thus is not routinely recommended.
C. Malignant Primary Bone Tumors
Multiple myeloma is the most common primary malignant bone tumor of the spine and is usually seen in patients in their 50s to 60s. It and its unifocal counterpart, solitary plasmacytoma, are B-cell lymphoproliferative cancers. In the absence of instability, myeloma can be treated with radiation, chemotherapy, and stem-cell transplants. However, surgery may be the option of choice in the face of neurologic compression associated with spinal instability, pathologic fractures, or impending fractures. Typically, patients will receive an antiresorptive medicine (like bisphosphonates) to reduce the incidence of skeletally related adverse events.
Chordomas are tumors arising from notochordal cells within the vertebral body, typically during the fifth and sixth decades and beyond. The sacrum accounts for over half of cases involving the spine, with the occipitocervical region accounting for another third. Because the local recurrence rate of chordoma is extremely high, care must be taken to obtain wide margins and avoid tumor contamination into the surgical field. Adjuvant radiation therapy may be helpful, particularly if complete resection is not possible or tumor contamination occurs during resection. Most patients with chordoma eventually die from complications related to local recurrence.
Chondrosarcomas are cartilaginous tumors typically arising in the posterior elements of the spine in patients in their 40s to 60s. Radiographs and CT scans demonstrate calcification within the tumor. Because they are radio-resistant, wide surgical excision is the mainstay of treatment. It is important to try to obtain clean surgical margins in order to avoid local recurrences, because local recurrence and progression are the most common causes of death from chondrosarcoma.
![]() Prognosis
Prognosis
The prognosis will vary widely depending on the tumor. In the case of acute myelopathy associated with metastatic disease, it has been shown in a randomized controlled trial that immediate surgical decompression followed by radiation therapy is superior to radiation therapy alone. In general, surgical counseling must include specific discussion of quality of remaining life weighed against the degree of pain and/or actual versus impending neurologic deficit. Patients with less than 3 months of expected survival are typically provided nonoperative care. Surgery becomes a more reasonable option in those with better prognoses, and it should at least be considered in all patients with actual or impending neurologic deficits, especially spinal cord level compression. Even in those with relatively poor prognoses, surgery may provide substantial benefit in terms of neurologic preservation and pain relief during a patient’s remaining life.
Acosta FL Jr, Dowd CF, Chin C, Tihan T, Ames CP, Weinstein PR: Current treatment strategies and outcomes in the management of symptomatic vertebral hemangiomas. Neurosurgery 2006;58:287. [PMID: 16462482]
Barr JD, Barr MS, Lemley TJ, McCann RM: Percutaneous vertebroplasty for pain relief and spinal stabilization. Spine (Phila Pa 1976) 2000;25:923. [PMID: 10767803]
Berenson J, Pflugmacher R, Jarzem P, et al: Balloon kyphoplasty versus non-surgical fracture management for treatment of painful vertebral body compression fractures in patients with cancer: a multicentre, randomised controlled trial. Lancet Oncol 2011;12:225. [PMID: 21333519]
Chaichana KL, Woodworth GF, Sciubba DM, et al: Predictors of ambulatory function after decompressive surgery for metastatic epidural spinal cord compression. Neurosurgery 2008;62:683. [PMID: 18425015]
Chi JH, Gokaslan Z, McCormick P, Tibbs PA, Kryscio RJ, Patchell RA: Selecting treatment for patients with malignant epidural spinal cord compression-does age matter? Results from a randomized clinical trial. Spine (Phila Pa 1976) 2009;34:431. [PMID: 19212272]
Gerszten PC, Monaco EA 3rd: Complete percutaneous treatment of vertebral body tumors causing spinal canal compromise using a transpedicular cavitation, cement augmentation, and radiosurgical technique. Neurosurg Focus2009;27:E9. [PMID: 19951062]
Henry DH, Costa L, Goldwasser F, et al: Randomized, double-blind study of denosumab versus zoledronic acid in the treatment of bone metastases in patients with advanced cancer (excluding breast and prostate cancer) or multiple myeloma. J Clin Oncol 2011;29:1125. [PMID: 21343556]
Kondo T, Hozumi T, Goto T, Seichi A, Nakamura K: Intraoperative radiotherapy combined with posterior decompression and stabilization for non-ambulant paralytic patients due to spinal metastasis. Spine (Phila Pa 1976)2008;33:1898. [PMID: 18670344]
Magge SN, Smyth MD, Governale LS, et al: Idiopathic syrinx in the pediatric population: a combined center experience. J Neurosurg Pediatr 2011;7:30. [PMID: 21194284]
Patchell RA, Tibbs PA, Regine WF, et al: Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet 2005;366:643. [PMID: 16112300]
Singh K, Samartzis D, Vaccaro AR, Andersson GB, An HS, Heller JG: Current concepts in the management of metastatic spinal disease. The role of minimally-invasive approaches. J Bone Joint Surg Br2006;88:434. [PMID: 16567775]
![]() DEFORMITIES OF THE SPINE
DEFORMITIES OF THE SPINE
Bobby K.B. Tay, MD; Harry B. Skinner, MD, PhD
SCOLIOSIS
![]() Essentials of Diagnosis
Essentials of Diagnosis
• Scoliosis is deviation of the spine from the vertical axis greater than 10 degrees.
• Ninety percent of cases do not progress.
• Curve magnitude greater than 45 degrees is likely to progress into adulthood.
• Most cases in young people are asymptomatic; adults are more likely to have symptoms of back pain and sciatica.
![]() General Considerations
General Considerations
Scoliosis as defined by the Scoliosis Research Society is any lateral deviation of the spine from the normal vertical line of 10 degrees or greater. Minor “wiggles” less than 10 degrees are termed spinal asymmetry and do not require follow-up. Using the 10-degree standard, about 2–4% of the U.S. population has scoliosis with an equal distribution between the sexes. At this low threshold for diagnosis, over 90% of cases will not progress to the point of warranting further intervention. Thus, referral to a spine surgeon is typically reserved for curves that are more than 15–20 degrees, for curves that demonstrate progression on serial assessment by the primary care provider, or for patients with worrisome history or physical exam findings. When coronal curvature is greater than 20–30 degrees, a strong female predominance (8–10:1) is noted. Idiopathic scoliosis is a disease that is driven by skeletal growth until a significant magnitude of curvature is obtained (>30–50 degrees). As such, there are essentially three stages of treatment for scoliosis—observation alone (the most common), bracing (only appropriate in the skeletally immature), and surgical correction.
If by skeletal maturity the coronal curve has not exceeded 30 degrees, then this mild spinal deformity is not likely to progress in adulthood, and it is unlikely to have future clinical impact on the patient. On the other hand, when the magnitude of curvature exceeds 45–50 degrees, progression in adulthood is predicted, typically at a pace of 1 degree per year. With continued progression, clinical manifestations are likely, which include back pain and radiculopathy/sciatica (especially when the major curve involves the lumbar spine), and in the most severe cases (>75–90 degrees), pulmonary and gastrointestinal (GI) compromise can occur. As such, surgery, in the form of instrumented spinal fusion, is typically indicated when coronal curvature exceeds 45–50 degrees. The goals of surgical correction are to arrest curve progression and provide sufficient curve correction to produce a stable, well-balanced spine centered over the pelvis by fusing as few segments as possible. In addition to this primary structural goal, additional benefits may include improved cosmesis and reduced back pain and neurologic dysfunction. Traditionally, scoliosis clinical research has focused on the primary goal, which has been deformity correction. Recent clinical studies have focused on the overall well-being of scoliosis patients as assessed by validated outcome tools.
Scoliosis is most commonly divided into subsets, based on the age at diagnosis. When scoliosis is present at birth, it is termed congenital scoliosis, which occurs by one of two structural anomalies. Either there is a failure of formation of part of the spine (for instance, a hemi-vertebra, which is the most common anomaly producing congenital scoliosis), or there is failure of segmentation (ie, a block vertebra). The crucial period for formation and segmentation of the spine occurs in the fourth to sixth week of gestation, which is also a critical time for spinal cord, renal, and cardiac development. As a result, when congenital scoliosis is diagnosed, the health of these additional structures should be screened as well. When scoliosis presents within the first 3 years of life, it is termed infantile scoliosis. Infantile scoliosis is a rare (<1% of all cases) form of scoliosis, which has strong genetic associations, and it too requires further screening (ie, screening MRI of the spine). It commonly produces a left thoracic curve, and the vast majority (~90%) resolves spontaneously. When it presents between 3 and 10 years of age, it is called juvenile scoliosis. This type behaves and is treated like adolescent scoliosis, although the greater growth potential predisposes to a greater risk of progression to the point of surgical indication. Adolescent idiopathic scoliosis, the most common type of scoliosis, is diagnosed when scoliosis presents after 10 years of age and prior to skeletal maturity (typically 2 years after menarche in females). Adult scoliosis refers to scoliosis detected after skeletal maturity. It typically has two primary distinctions—de novo (meaning it developed in adulthood, often as an advanced form of spinal degeneration, typified by low-degree lumbar curves with significant rotatory subluxation[s]) and progressive (meaning it was the progression of a curve that started earlier in life). Aside from congenital and infantile forms of scoliosis, which have unique anatomic considerations, the distinctions between juvenile, adolescent, and adult are not as significant.
Last, the aforementioned classifications describe idiopathic scoliosis, which is the etiology of 85% of cases; however, scoliosis can be associated with skeletal syndromes (eg, Marfan syndrome or neurofibromatosis), termed syndromic scoliosis, or neuromuscular conditions (cerebral palsy being most common). These two rarer causes of scoliosis tend to produce more significant degrees of curvature and more commonly require surgical intervention. Neuromuscular scoliosis is often treated with fusion of the entire thoracic and lumbar spine to the pelvis. An important consideration for these two rare forms of scoliosis is that they are part of a syndrome. When diagnosed, comprehensive evaluation for other known associated manifestations (eg, aortic dilation in Marfan syndrome) of the syndrome should be undertaken.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
Scoliosis is most commonly detected incidentally on screening examinations or during routine pediatric appointments in the preteen years. Uneven ribs, shoulders, or pelvis may be noted by the patient or parents and should be assessed on examination. While the vast majority of patients are asymptomatic at presentation, about one third of patients will complain of back pain, which is often in the interscapular area. When it is in the low lumbar area, radiographs should be inspected for evidence of spondylolisthesis, which can coexist in scoliotic patients. Since reports of back pain can be elicited in a similar percentage of young adults with straight spines, the clinical significance of this complaint and the direct association with the underlying spinal deformity may not be clear. That being said, reports of severe back pain may prompt further evaluation, to include screening whole-spine MRI to ensure that no intraspinal anomalies (eg, syrinx, tumor, or diastematomyelia) are present. The yield from these additional studies is exceedingly low and likely does not offset the cost and difficulty (eg, some level of sedation is often needed to obtain the MRI). Since indications for MRI in the setting of scoliosis are not universally accepted, this determination is probably best left to the consultant. When patients present with positive findings on neurologic examination (eg, weakness, sensory deficits, altered gait, bowel or bladder problems), then screening MRI should be ordered.
The Adams forward bend test is the classic screening assessment for scoliosis. It is carried out in most states on an annual basis in school for adolescent females (and commonly males as well). This test requires the examiner to stand posterior to the patient, while the child bends over at the waist until their trunk is level with the ground. Scoliosis is not simply a lateral deviation, but a rotation of the spine. The rotation at the thoracic level causes the ribs to rise up on the side of the curve apex, which is most commonly the right side (90% of cases). It is so common for the main thoracic curve to be apexed to the right that many experts feel screening MRI is indicated whenever the main thoracic curve points to the left. This truncal rotation can simply be viewed or palpated, but a scoliometer is a device, which looks like a carpenter’s level, used to directly measure the degree of rotation in the chest cavity. Screening thresholds for radiograph and/or spine surgical consultation vary between 5- and 7-degree tilt, as measured by the scoliometer. The lower threshold is more sensitive (minimal to no false negatives), whereas the upper limit is more specific (less false positives).
B. Imaging
The gold standard for diagnosing scoliosis is an upright posterior-anterior long cassette (36”) frontal and lateral radiograph of the entire thoracic and lumbar spine. This is often called a scoliosis survey. This should be obtained at a radiology suite that is versed in the proper protocol for obtaining high-quality scoliosis films. The radiologist or spine surgeon then views the spine and looks for the three common curves that occur in scoliosis—proximal thoracic, main thoracic, and lumbar. The magnitude of the curvature is measured using the Cobb method, which subtends the angle between lines drawn at the upper endplate of the upper vertebra in the measured curve (termed the upper end vertebrae) to the lower endplate of the lower vertebrae (lower end vertebra). The apex is the direction that the convex side of the curve points (often reported using the Latin prefixes dextro, meaning right, or levo, meaning left); specifically, it is the lateral-most deviated point in the curve, which can be a vertebral body or disk space. The largest of the curves is called the major curve. Any additional curves are called minor curves. The major curve, in addition to having the greatest degree of coronal curvature, also has the greatest degree of rotation. Rotation is graded based on the degree to which the apical vertebral pedicles are rotated from their normal owl’s eyes appearance. Minor curves are frequently compensatory. Compensatory curves typically do not have a rotatory component, as they are simple lateral deviations in the spine that naturally occur to compensate for the major curve, thereby keeping the head centered over the pelvis.
The most common method in current use for classifying adolescent idiopathic scoliosis curves is the Lenke classification system. It replaces the King classification system, because it is more reliable, better predicts surgical plan, and takes into account the three-dimensional nature of scoliosis. In addition to standing scoliosis films, supine bending films are also needed to assess the flexibility and structural nature of each curve. The Lenke classification system has three components. First, there are six curve types. Then there is a lumbar modifier, which demonstrates the degree to which the lumbar curve deviates from the vertical midline. Last, there is a sagittal modifier, which assesses the degree of thoracic (T5-T12) kyphosis.
Many patients with main thoracic scoliosis (Lenke type 1, most common type) have flat (hypokyphotic) thoracic spines, as the normal thoracic kyphosis has rotated into the coronal plane. Kyphosis (abnormal forward curvature of the spine; normal = 10–50 degrees) can occur in the setting of scoliosis or as an independent deformity. When global thoracic kyphosis of more than 50 degrees occurs in the setting of three or more consecutive thoracic vertebral bodies having a wedging of 5 degrees or more, typically with Schmorl nodes present, it is termed Scheuermann kyphosis. Most cases of Scheuermann kyphosis require no specific treatment, although bracing can be attempted during growth. Corrective surgical fusion may be considered in severe cases (>75–90 degrees).
![]() Treatment
Treatment
As stated earlier, there are three stages of treatment for scoliosis. The first and commonly the only stage needed is serial observation. Because scoliosis is a disease of progression, it takes at least two points in time to fully assess the significance of the deformity, unless the deformity is severe at presentation. Three factors most affect the likelihood of progression: (1) female gender, (2) growth remaining, and (3) current curve magnitude. To minimize repeated radiation exposure, serial follow-up typically occurs at 4-month intervals during periods of peak growth and 6-month intervals during periods of less expected growth, especially in lower magnitude curves (<20 degrees). Curves that are less than 30 degrees in adulthood do not require routine follow-up. While there is some variation, a consensus of the reported methods for predicting progression to more than 40–45 degrees shows that major curves that are less than 20–30 degrees at or in the year prior to menarche or the start of the skeletal growth spurt in males have less than 5–20% risk of progression to the point of requiring surgical intervention. Asymptomatic curves of less than 30 degrees detected as incidental findings in females more than 2 years out from menarche and skeletally mature males do not require spine surgical consultation solely for evaluation of scoliosis. Since curve progression in adulthood occurs at a much slower pace than in adolescence, serial images, when indicated, should be separated by 1–3+ years in adults. The measurement error for coronal curve measurement on scoliosis films is 3–5 degrees. Thus, with an expected progression of 1 degree per year in adulthood, it would take at least 3 years to demonstrate a real progression exceeding measurement error. Indications for scoliosis surgery in adults are largely based on clinical complaints, as opposed to curve magnitude and deformity.
If the major curve progresses to 20–25 degrees (up to 35 degrees) in a skeletally immature patient, then bracing is the next stage of treatment offered. There are several types of braces, with two major subsets—thoracolumbar orthoses and nighttime braces. Thoracolumbar orthoses (eg, Boston brace) are the most common type of brace and are intended to be worn 20–22 hours a day. They are typically made of thin plastic, so they can be worn under clothes to reduce the stigma of wearing a brace. Compliance significantly affects the ability of braces to prevent progression, which is the goal of bracing. Bracing is not intended to permanently correct scoliosis, but rather to arrest curve progression at a magnitude below which it is unlikely to produce lifelong impact (ie, <30 degrees). With compliant wear, bracing can arrest/slow scoliosis progression and prevent need for surgical intervention in up to 75% of those treated. Bracing is a challenging treatment for image-conscious adolescents to tolerate, because when it is started, it must be maintained through the remainder of skeletal growth and then some time after (often 2–3 years of total wear time). To avoid brace wear during school, nighttime braces were introduced, which hold the spine in an overcorrected position during nighttime only. This technique is more painful and may be less effective. Physical therapy and activity modification do not influence the natural history of scoliosis. There is no reason to restrict activity in patients diagnosed with scoliosis. That being said, reducing the weight of book bags, encouraging the wear of both shoulder straps (versus using just one shoulder), and best yet, rolling book-bags with two sets of books (one for school and one for home) can help reduce back pain complaints and do demonstrate short-term measureable difference in curvature.
Last, when the degree of curvature exceeds 45–50 degrees, surgical intervention in the form of instrumented spinal fusion is indicated. This can occur via posterior, anterior, or combined posterior-anterior spinal fusion. Clear clinical superiority between the approaches is uncertain. Each has its own specific risk-benefit profile. Multiple techniques exist for correcting the curvature seen in scoliosis. All methods include rigid fixation of the spine, some degree of destabilization of the instrumented spinal segment (ie, facetectomies and ligament releases), correction of the deformity via forces applied through the implants, and securing the correction by locking the screws (or fixation devices) to rods and then placement of bone graft to encourage solid fusion across the instrumented segment. The most common means for instrumenting the spine is a pedicle screw and rod construct. Pedicle screws provide strong three-column fixation capable of delivering three-dimensional corrective forces to the spine.
Outside the scope of this chapter is the complex decision process that goes into level selection and whether to fuse all curves or only the major curve, expecting the minor/compensatory curves to spontaneously correct. Current surgical techniques, especially in young healthy patients, produce solid fusions with excellent correction in the vast majority of patients with very low rates of complications. The most dreaded complication is spinal cord injury (<1%). This can occur through implant malposition, but more commonly is related to the acute correction of the deformity, especially in the setting of significant kyphosis. Intraoperative neuro-monitoring allows for real-time or near real-time assessment of spinal cord function during scoliosis surgery to reduce the risk of this severe complication. Comorbidities, such as osteoporosis, obesity, cardiac disease, and diabetes, increase the rate of complication associated with adult scoliosis surgery. Further, younger patients have significantly more flexible curves, which can be more effectively corrected with simpler techniques, than the rigid curves seen in adult scoliosis. When the primary presenting symptom is pain or neurologic deficit, deformity correction in the setting of low-magnitude curves (30–50 degrees) in patients with neutral sagittal balance (ie, the spine is maintained in a rather vertical stature, without fixed forward tipping of the spine) is not always a goal of surgery in adult scoliosis. When sagittal imbalance is present through scoliosis or kyphoscoliosis, long-segment thoracolumbar fusions are often indicated to secure the spine and restore the sagittal balance in adults. This is crucial, because sagittal balance is the structural parameter most predictive of successful clinical outcome following adult scoliosis surgery. More recently, it has been shown that in addition to achieving these technical goals, scoliosis surgery significantly improves cosmesis, pain, and overall well-being.
Belmont PJ Jr, Kuklo TR, Taylor KF, Freedman BA, Prahinski JR, Kruse RW: Intraspinal anomalies associated with isolated congenital hemivertebra: the role of routine magnetic resonance imaging. J Bone Joint Surg Am 2004 Aug;86-A(8):1704-1710. [PMID: 15292418]
Bridwell KH, Baldus C, Berven S, et al: Changes in radiographic and clinical outcomes with primary treatment adult spinal deformity surgeries from two years to three- to five-years follow-up. Spine (Phila Pa 1976)2010;35:1849. [PMID: 20802383]
Carreon LY, Sanders JO, Diab M, Sturm PF, Sucato DJ; Spinal Deformity Study Group: Patient satisfaction after surgical correction of adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 2011;36:965. [PMID: 21224771]
Charles YP, Daures JP, de Rosa V, Diméglio A: Progression risk of idiopathic juvenile scoliosis during pubertal growth. Spine (Phila Pa 1976) 2006 Aug 1;31(17):1933-1942. [PMID: 16924210]
Clements DH, Marks M, Newton PO, Betz RR, Lenke L, Shufflebarger H; Harms Study Group: Did the Lenke classification change scoliosis treatment? Spine (Phila Pa 1976) 2011 Jun 15;36(14):1142-1145. [PMID: 21358471]
Diab M, Landman Z, Lubicky J, et al: Use and outcome of MRI in the surgical treatment of adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 2011;36:667. [PMID: 21178850]
Fu KM, Smith JS, Polly DW Jr, et al: Correlation of higher preoperative American Society of Anesthesiology grade and increased morbidity and mortality rates in patients undergoing spine surgery. J Neurosurg Spine2011;14:470. [PMID: 21294615]
Hamilton DK, Smith JS, Sansur CA, et al: Rates of new neurological deficit associated with spine surgery based on 108,419 procedures: a report of the scoliosis research society morbidity and mortality committee. Spine (Phila Pa 1976) 2011;36:1218. [PMID: 21217448]
Howard A, Wright JG, Hedden D: A comparative study of TLSO, Charleston, and Milwaukee braces for idiopathic scoliosis. Spine (Phila Pa 1976) 1998 Nov 15;23(22):2404-2411. [PMID: 9836354]
Isaacs RE, Hyde J, Goodrich JA, Rodgers WB, Phillips FM: A prospective, nonrandomized, multicenter evaluation of extreme lateral interbody fusion for the treatment of adult degenerative scoliosis: perioperative outcomes and complications. Spine (Phila Pa 1976) 2010;35(26 Suppl):S322. [PMID: 21160396]
Lange JE, Steen H, Gunderson R, Brox JI: Long-term results after Boston brace treatment in late-onset juvenile and adolescent idiopathic scoliosis. Scoliosis 2011;6:18. [PMID: 21880123]
Lehman RA Jr, Lenke LG, Keeler KA, et al: Operative treatment of adolescent idiopathic scoliosis with posterior pedicle screw-only constructs: minimum three-year follow-up of one hundred fourteen cases. Spine (Phila Pa 1976) 2008;33:1598. [PMID: 18552676]
Lenke LG: Lenke classification system of adolescent idiopathic scoliosis: treatment recommendations. Instr Course Lect 2005;54:537. [PMID: 15948478]
Little DG, Song KM, Katz D, Herring JA: Relationship of peak height velocity to other maturity indicators in idiopathic scoliosis in girls. J Bone Joint Surg Am 2000;82:685. [PMID: 10819279]
Lowe TG, Line BG: Evidence based medicine: analysis of Scheuermann kyphosis. Spine (Phila Pa 1976) 2007;32(19 Suppl): S115. [PMID: 17728677]
Newton PO, Faro FD, Lenke LG, et al: Factors involved in the decision to perform a selective versus nonselective fusion of Lenke 1B and 1C (King-Moe II) curves in adolescent idiopathic scoliosis. Spine (Phila Pa 1976)2003;28:S217. [PMID: 14560195]
Potter BK, Kuklo TR, Lenke LG: Radiographic outcomes of anterior spinal fusion versus posterior spinal fusion with thoracic pedicle screws for treatment of Lenke Type I adolescent idiopathic scoliosis curves. Spine (Phila Pa 1976) 2005;30:1859. [PMID: 16103856]
Ramirez N, Johnston CE, Browne RH: The prevalence of back pain in children who have idiopathic scoliosis. J Bone Joint Surg Am 1997;79:364. [PMID: 9070524]
Reames DL, Smith JS, Fu KM, et al: Complications in the surgical treatment of 19,360 cases of pediatric scoliosis: a review of the Scoliosis Research Society Morbidity and Mortality database. Spine (Phila Pa 1976)2011;36:1484. [PMID: 21037528]
Smucny M, Lubicky JP, Sanders JO, Carreon LY, Diab M: Patient self-assessment of appearance is improved more by all pedicle screw than by hybrid constructs in surgical treatment of adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 2011;36:248. [PMID: 21248593]
Thuet ED, Winscher JC, Padberg AM, et al: Validity and reliability of intraoperative monitoring in pediatric spinal deformity surgery: a 23-year experience of 3436 surgical cases. Spine (Phila Pa 1976)2010;35:1880. [PMID: 20802388]
NEUROFIBROMATOSIS
Spinal deformity associated with neurofibromatosis poses some special considerations. Curvatures seen in affected patients may be of the idiopathic type or the dysplastic type. Curvatures of the first type exhibit the same curve patterns as seen in patients with idiopathic scoliosis and are most commonly right thoracic curves and may be managed in a similar manner. Curvatures of the second type can be much more malignant in behavior.
Dysplastic curves can be identified by evidence of dys-plastic bone: penciling of the ribs or transverse processes, enlargement of the foramina, erosion of the vertebrae, and evidence of a shorter, more abrupt curve than that seen in idiopathic scoliosis. Dysplastic curves usually are associated with kyphosis, which also exists through a fairly short sharp segment. They may occur in the thoracic, thoracolumbar, or lumbar spine.
Dysplastic curves in patients with neurofibromatosis can progress rapidly and lead to severe deformity. Bony erosion can occur secondary to neurofibromas or dural ectasia (expansions of the dural sac, which can account for enlargement of the foramina or erosion of the vertebrae). The short kyphotic curves and erosion of bone can, in severe cases, result in neurologic impairment, including paraplegia.
Surgery in patients with dysplastic curves is associated with a high incidence of pseudarthrosis. If surgery is indicated, it is usually recommended to perform both an anterior and a posterior fusion. This combined approach results in a satisfactory fusion rate of up to 80%. Because of the dysplastic bone stock, it may be necessary to use a combination of sublaminar wires, hooks, and screws. Preoperative MRI may be useful in assessing the extent of dural ectasia. Fusion levels are selected according to the end vertebra of the curvature. The end fusion level must lie centered over the middle of the sacrum (the Harrington stable zone), much like the selection for idiopathic scoliosis. Clearly, however, the fusion should not end above or below a dysplastic vertebra, although it would be rare for such a level not to be within the curve.
Funasaki H, Winter RB, Lonstein JB, et al: Pathophysiology of spinal deformities in neurofibromatosis: an analysis of 71 patients who had curves associated with dystrophic changes. J Bone Joint Surg Am1994;76:692. [PMID: 8175817]
Greggi T, Martikos K: Surgical treatment of early onset scoliosis in neurofibromatosis. Stud Health Technol Inform 2012;176:330. [PMID 22744522]
Vitale MG, Guha A, Skaggs DL: Orthopaedic manifestations of neurofibromatosis in children: an update. Clin Orthop Relat Res 2002;401:107. [PMID: 12151887]
CONGENITAL SCOLIOSIS
Congenital scoliosis is caused by one of the two types of structural bony abnormality (Figure 4–24). Type I is a failure of formation, such as that seen with hemivertebrae. Type II is a failure of segmentation, such as that seen with block vertebrae and that seen with unsegmented bars, where there is a tether to growth on one side of the spine. Mixed abnormalities are also found in patients with congenital scoliosis. Unilateral unsegmented bars with contralateral hemiverte-brae have the greatest tendency for rapid progression and should be surgically fused as soon as the bony abnormality is evident. Unilateral unsegmented bars also tend to progress.

![]() Figure 4–24. The major types of congenital scoliosis are failure of formation, as shown in diagrams A through E, and failure of segmentation, as shown in diagrams F and G. (Reproduced, with permission, from Hall JE: Congenital scoliosis. In Bradford DS, Hensinger RN, eds: The Pediatric Spine. New York: Thieme; 1985.)
Figure 4–24. The major types of congenital scoliosis are failure of formation, as shown in diagrams A through E, and failure of segmentation, as shown in diagrams F and G. (Reproduced, with permission, from Hall JE: Congenital scoliosis. In Bradford DS, Hensinger RN, eds: The Pediatric Spine. New York: Thieme; 1985.)
With respect to progression, hemivertebrae have a variable prognosis, depending on whether a contralateral hemivertebra is present that results in overall balance of the spine, whether multiple hemivertebrae are on one side of the spine, and how much growth potential is predicted for each endplate of the hemivertebra. Hemivertebrae at the cervicothoracic junction and the lumbosacral junction have a relatively poor prognosis because the spine above or below the abnormality cannot compensate. Hemivertebrae should be observed so as to delineate their growth potential and progression.
Bracing is ineffective in treating congenital scoliosis because the curves are inflexible. Bracing is sometimes used to prevent progression of the compensatory curve, however.
In patients with congenital scoliosis, the incidence of cardiac abnormalities is increased, as is the incidence of renal abnormalities (20–30%) and intracanal abnormalities (10–50%). Abdominal ultrasound or other imaging tests should be used to rule out absent or abnormal kidneys. Intracanal abnormalities may include a syrinx (cyst within the cord), diastematomyelia or diplomyelia (division or reduplication of the cord, respectively), and tethered cord (presence of a tight filum terminale that does not permit the conus medullaris to migrate upward normally with growth).
If surgical intervention in patients with congenital scoliosis is indicated, several options are available. “Growth-friendly” surgical management is preferred in early-onset scoliosis so that the curve can be controlled while the spine and thorax continue to grow. Various distraction/compression systems are available for such surgery without fusion. Fusion in situ is the simplest procedure. For very young (<10 years) patients, however, a posterior fusion alone results in tethering of the posterior elements while the anterior elements continue to grow. This situation may lead to the crankshaft phenomenon, whereby the anterior growth in the spine results in a twisting deformity around the fused posterior elements. For this reason, combined anterior and posterior fusion is usually recommended for very young patients, halting growth circumferentially about the spine. (The crankshaft phenomenon can also occur in very young patients with non-congenital forms of scoliosis who were treated by posterior fusion. Age younger than 10 years, Risser stage 0 or 1, and the presence of an open triradiate cartilage are indicators of skeletal maturity at risk for development of crankshaft.)
In some cases of hemivertebra, hemiepiphysiodesis may be performed, arresting growth on the curve convexity but permitting continued growth on the curve concavity, with resultant gradual curve correction. This procedure has good results in selected patients but can be unpredictable with respect to the amount of actual correction that can be achieved.
In cases in which a hemivertebra is accompanied by significant coronal decompensation and compensatory growth would not be adequate to result in spinal balance, consideration can be given to hemivertebra excision via a combined anterior and posterior approach. Although this procedure is technically more demanding and has greater potential risks, it allows for better overall curve correction and improvement of coronal balance. Newer surgical techniques may allow for a single-stage posterior hemivertebral decancellation and excision. This approach may obviate the need for a separate anterior approach to the spine. Hemivertebra excision may be the preferred option in the lumbar spine or lumbosacral junction, where the neurologic risk is to the cauda equina rather than the spinal cord and where oblique takeoff of the vertebra above the hemivertebra can result in significant truncal decompensation.
Bradford DS: Partial epiphyseal arrest and supplemental fixation for progressive correction of congenital spinal deformity. J Bone Joint Surg Am 1982;64:610. [PMID: 7068703]
Gomez JA, Lee JK, Kim PD, et al: “Growth friendly” spine surgery: management options for the young child with scoliosis. J Am Acad Orthop Surg 2011;19:722. [PMID: 22134204]
Lenke LG, Newton PO, Sucato DJ, et al: Complications following 147 consecutive vertebral column resections for severe pediatric spinal deformity: a multicenter analysis. Spine (Phila Pa 1976)2013;38:119. [PMID: 22825478]
Thompson AG, Marks DS, Sayampanathan SR, et al: Long term results of combined anterior and posterior convex epiphysiodesis for congenital scoliosis due to hemivertebrae. Spine (Phila Pa 1976)1995;20:1380. [PMID: 7676336]
Wang S, Zhang J, Qiu G, et al: Dual growing rods technique for congenital scoliosis: More than 2 years outcomes: the preliminary results of a single centre. Spine (Phila Pa 1976) 2012;37:E1639. [PMID: 22990366]
Yaszay B, O’Brien M, Shufflebarger HL, et al: Efficacy of hemivertebra resection for congenital scoliosis: a multicenter retrospective comparison of three surgical techniques. Spine (Phila Pa 1976)2011;36:2052. [PMID: 22048650]
KYPHOSIS
The normal sagittal contour of the spine includes cervical lordosis, thoracic kyphosis, and lumbar lordosis (Figure 4–25). Increases or decreases in any of these can be seen. If they are severe enough, they can cause disability, as discussed later in the cases of congenital kyphosis and Scheuermann kyphosis.

![]() Figure 4–25. The normal sagittal contour of the spine. (Reproduced, with permission, from Bullough PG, Boachie-Adjei O: Atlas of Spinal Diseases. London: Gower; 1988.)
Figure 4–25. The normal sagittal contour of the spine. (Reproduced, with permission, from Bullough PG, Boachie-Adjei O: Atlas of Spinal Diseases. London: Gower; 1988.)
1. Congenital Kyphosis
As in congenital scoliosis (see previous discussion), congenital kyphosis can result from a failure of formation or a failure of segmentation. In congenital kyphosis, however, failures of formation have a much more dangerous clinical prognosis. These can lead to congenital or progressive “dislocation” of the spinal column (Figure 4–26) and paralysis if not treated appropriately. If performed early enough, posterior fusion may be sufficient to prevent neurologic problems. Severe deficiencies, however, may require anterior and posterior fusion to achieve stability.

![]() Figure 4–26. Congenital kyphosis and congenital “dislocation” of the spinal column. (Reproduced, with permission, from Dubousset J: Congenital kyphosis. In Bradford DS, Hensinger RN, eds: The Pediatric Spine. New York: Thieme; 1985.)
Figure 4–26. Congenital kyphosis and congenital “dislocation” of the spinal column. (Reproduced, with permission, from Dubousset J: Congenital kyphosis. In Bradford DS, Hensinger RN, eds: The Pediatric Spine. New York: Thieme; 1985.)
2. Scheuermann Kyphosis
Normal thoracic kyphosis ranges from 25 to 45 degrees. Postural kyphosis can increase this curvature, but if no abnormalities are present, the curve is flexible and the posture can be easily corrected by the child. If endplate abnormalities are present and three or more vertebral bodies are wedged as seen on the lateral radiograph, the diagnosis of Scheuermann kyphosis can be made. Schmorl nodules, characterized by herniation of the disk material at the vertebral endplates, and increased thoracic kyphosis are also seen. Clinically, patients with this type of kyphosis have a curvature that is more abrupt than that observed in people with postural roundback, and this type is only partly correctable by forced extension. It can be demonstrated either by having the patient hyperextend or by taking a lateral radiograph with the patient lying over a bolster at the apex of the kyphosis so the Cobb angle can be measured. Thoracic curves may cause pain and discomfort, although some report that pain is more commonly seen in thoracolumbar curves.
Bracing can be instituted if the kyphosis measures more than 45 or 55 degrees in a skeletally immature patient, particularly if the curvature is progressive or accompanied by pain. If lesser degrees of deformity are symptomatic, they can be treated with physical therapy exercises and observed for progression. Brace treatment requires the use of the Milwaukee brace, with two paraspinal pads placed over the apical ribs posteriorly. Radiographs should be taken with the patient in the brace to confirm that adequate correction is being effected. The brace can be removed for sports and bathing but should otherwise be worn 23 hours a day. Repeat lateral radiographs should be taken at intervals of 4–6 months. If bracing is successful at controlling the curve, it should be continued until the patient nears skeletal maturity. Weaning should be performed slowly, so as to maintain correction. Although some correction may be lost, proper use of the Milwaukee brace can result in long-lasting improvement in many patients with kyphosis (which is not the case with brace treatment of adolescent idiopathic scoliosis).
Surgical treatment of kyphosis may be indicated if the curve magnitude increases despite bracing, if the patient has significant associated symptoms, or if the patient who is nearing skeletal maturity has a severe curvature. Posterior spinal fusion with multilevel posterior Smith-Peterson osteotomies is the treatment of choice in these cases. Segmental instrumentation using pedicle screws with a compression-type construct is used to correct the deformity and hold the correction until fusion occurs. If the curve flexibility does not permit adequate correction as demonstrated on a hyperextension lateral radiograph, an anterior release and fusion prior to the posterior spinal fusion is indicated. Care should be taken to extend the fusion to the upper thoracic spine, sometimes to T1, to minimize the risk of developing junctional problems at the cervicothoracic junction.
Reports describe the natural history of Scheuermann kyphosis, suggesting some functional limitations but little actual interference with lifestyle. The deformity can worsen over time. It appears clear, however, that many patients have their symptoms of back pain and deformity improved by surgery. Proper patient education and selection are essential for appropriate treatment of these patients.
Arlet V, Schlenzka D: Scheuermann’s kyphosis: surgical management. Eur Spine J 2005;14:817. [PMID: 15830215]
Noordeen MH, Garrido E, Tucker SK, et al: The surgical treatment of congenital kyphosis. Spine (Phila Pa 1976) 2009;34:1808. [PMID: 19644332]
Tsirikos AI, McMaster MJ: Infantile developmental thoracolumbar kyphosis with segmental subluxation of the spine. J Bone Joint Surg Br 2010;92:430. [PMID: 20190317]
MYELODYSPLASIA
Neural tube defects can result in complex spinal deformities secondary both to the neuromuscular collapsing nature of the spine and to the vertebral anomalies that can give rise to congenital kyphosis or congenital scoliosis. Myelomeningocele or meningocele is present at birth in a patient whose neural tube failed to close in utero. Sac closure is usually performed shortly after birth. In many cases, the affected infant also requires placement of a ventriculoperitoneal shunt because of hydrocephalus. The level of neurologic function usually corresponds to the level of the defect. For example, a low thoracic myelomeningocele patient has no lumbar nerve roots functioning and therefore no lower extremity function. An L4 myelomeningocele patient has a functioning tibialis anterior but no extensor hallucis and no gastrocnemius and usually no voluntary bowel and bladder control.
Neurologic function in patients with myelodysplasia is static and should not deteriorate with growth. Neurologic changes, especially during growth spurts, require evaluation for tethered cord, a common occurrence in affected children, which results in traction on the spinal cord.
Orthopedic management includes maximizing the function of patients through the use of braces, ambulatory aids, wheelchairs, or surgery. The degree of spinal deformity is related to the neurologic level, with spinal collapse more likely in those with a higher neurologic level of involvement than in those with a lower level. The presence of bony abnormalities can affect this prognosis, of course.
As with many neuromuscular spinal deformities, curvatures may present early in life. If the clinician elects to treat a patient with bracing, it is important to remember that bracing in the presence of insensate skin can result in pressure sores if the brace is not adequately padded and the parents are not instructed regarding skin care.
In many cases, the curvature eventually requires surgical stabilization. Because of the magnitude and stiffness of the curvature as well as the absence of posterior elements, the preferred treatment is anterior and posterior fusion. Anterior instrumentation may improve rigidity of the surgical construct. In patients with myelodysplasia, fusion to the sacrum is invariably required because of pelvic obliquity or lack of sitting balance. Instrumentation from the pelvis (iliac fixation) to the proximal thoracic spine is preferred, as with many neuromuscular deformities.
The lack of posterior elements in the myelodysplastic spine can lead to congenital kyphosis. Although kyphosis in these patients does not compromise neurologic function, it can lead to pressure sores over the prominent area. The treatment of choice for this problem is posterior kyphectomy and fusion. The neural elements can be cut and tied off at the site of the kyphotic deformity since the patient usually has no neurologic function below the kyphus.
Banit DM, Iwinski HJ Jr, Talwalkar V, et al: Posterior spinal fusion in paralytic scoliosis and myelomeningocele. J Pediatr Orthop 2001;21:117. [PMID: 11176365]
Hwang SW, Thomas JG, Blumberg TJ, et al: Kyphectomy in patients with myelomeningocele treated with pedicle screw-only constructs: case reports and review. J Neurosurg Pediatr 2011;8:63. [PMID: 21721891]
THORACIC DISK DISEASE
Disk herniation is found much less commonly in the thoracic spine than in the cervical and lumbar spine, presumably because of the decreased mobility seen in this region with the rib cage and sternum. Herniated thoracic disks account for 1–2% of operative disks, although the reported incidence in autopsy series is 7–15%.
Patients with thoracic disk disease may present with radicular symptoms at the level of involvement and complain of back or lower extremity pain, extremity weakness, numbness corresponding to the level of the disk herniation or below, and bowel or bladder dysfunction. They may demonstrate a spastic gait, with long-tract signs, if the disk is more central. Diagnosis is made by myelography, sometimes in conjunction with CT scanning or MRI.
In the absence of long-tract signs and paraparesis, conservative measures may include rest, anti-inflammatory medications, and physical therapy, with a 70–80% success rate.
Surgical treatment is recommended for patients with signs of myelopathy, including paraparesis or hyperreflexia. Decompression is most safely performed via an anterior approach. The anterior extrapleural approach is advocated and yields good results.
When an anterior approach is used, 58–86% of patients show neurologic improvement and 72–87% experience pain relief. Neurologic deterioration is reported in up to 7% of patients who undergo surgery via an anterior or anterolateral approach and in 28–100% of patients who undergo posterior decompression. A major complication rate of 6.7% and a reoperation rate of 5% were reported for a minimally invasive lateral approach. Posterior laminectomies are associated with a high rate of complications, including worsening neurologic function from manipulation of the cord and incomplete decompression of an inadequately visualized disk.
Brown CW, Deffer PA Jr, Akmakjian J, et al: The natural history of thoracic disc herniation. Spine (Phila Pa 1976) 1992;17:97. [PMID: 1631725]
Russo A, Balamurali G, Nowicki R, et al: Anterior thoracic foraminotomy through mini-thoracotomy for the treatment of giant thoracic disc herniations. Eur Spine J 2012;21(Suppl 2):S212. [PMID: 22430542]
Uribe JS, Smith WD, Pimenta L, et al: Minimally invasive lateral approach for symptomatic thoracic disc herniation: initial multicenter clinical experience. J Neurosurg Spine 2012;16:264. [PMID: 22176427]
Vanichkachorn JS, Vaccaro AR: Thoracic disk disease: diagnosis and treatment. J Am Acad Orthop Surg 2000;8:159. [PMID: 10874223]
OSTEOPOROSIS AND VERTEBRAL COMPRESSION FRACTURES
Osteoporosis is characterized by a decline in overall bone mass in the axial and appendicular skeleton. The disease affects between 15 and 20 million people in the United States. Peak bone mass, attained between 16 and 25 years of age, slowly declines with age as the rate of bone resorption exceeds that of bone formation. This phenomenon occurs in both men and women and is known as senile osteoporosis. Women are also susceptible to postmenopausal osteoporosis that occurs during the 15–20 years after the onset of menopause and is directly linked to estrogen deficiency. Environmental factors also play a role in accelerating the rate of skeletal bone loss. These include chronic calcium deficiency, smoking, excessive alcohol intake, hyperparathyroidism, and inactivity. Genetic influences may also play a role.
Vertebral compression fractures are one of the most frequent manifestations of osteoporosis in the elderly (>60 years). Over 700,000 vertebral compression fractures occur each year. Fortunately, the overwhelming majority of patients are asymptomatic or become asymptomatic after a period of conservative care.
![]() Clinical Findings
Clinical Findings
Patients with symptomatic vertebral compression fractures typically complain of axial pain localized to the fractured level. Occasionally, the patient’s family notices that the patient’s back is becoming increasingly rounded, and significant loss of height has occurred. This spinal deformity is known as the “dowager’s hump.” In general, there is no neurologic dysfunction and no radiation of the pain in any dermatomal distribution. There is often no history of significant trauma or an inciting event.
![]() Imaging
Imaging
Plain radiographs and densitometric scans are the major imaging modalities in the assessment of osteoporotic bone and their pathologic counterparts (insufficiency fractures). Dual-energy x-ray absorptiometry (DXA or DEXA) is the most useful of the densitometric imaging techniques because it carries a high degree of precision (0.5–2%) and subjects the patient to minimal amounts of radiation. It is also quite accurate for assessment of osteoporosis in both the axial and appendicular skeleton. It is important to note, however, that hypertrophic osteophytes in the spine that are radiodense can cause an inaccurate reading on the DXA and should not be used as the sole site of assessment for osteoporosis. Other imaging modalities include single-energy x-ray absorptiometry (SXA), quantitative computed tomography (QCT), and radiographic absorptiometry.
Posterior/anterior and lateral radiographs of the affected area of the spine are likely to reveal the location and severity of the osteoporotic fracture(s). In the thoracic spine, wedge compression fractures are most commonly encountered. In the lumbar spine, both compression and burst fractures can occur. Other imaging modalities include technetium bone scans and MRI scans. These studies should be reserved for the evaluation of fractures that remain symptomatic or progress after a course of conservative treatment. MRI is extremely useful in differentiating nonunited fractures from those that have healed and in differentiating osteoporotic fractures from those caused by malignancy.
Bone biopsy is indicated if a metabolic bone disease or a malignancy is suspected as the cause of the osteoporosis. The sample, typically retrieved from the anterior iliac crest or from the vertebral body at the time of vertebral augmentation, is examined using bone histomorphometry.
![]() Treatment
Treatment
Prevention still remains the best treatment for osteoporosis. Maximizing bone mineral density prior to the onset of bone loss and minimizing the bone loss that occurs is the optimal regimen to prevent the painful sequelae of the disease. In women, estrogen replacement therapy can be initiated if there is no history of breast cancer, thromboembolic disease, or endometrial disease. Routine gynecologic examination is necessary once therapy is initiated. Calcitonin therapy can be used if estrogen therapy is contraindicated. Parathyroid hormone is currently under clinical trials for the treatment of osteoporosis. Early evidence suggests that it may help to increase skeletal bone mass significantly and may be useful as a first-line treatment for severe osteoporosis.
The bisphosphonates, etidronate and alendronate, prevent osteoclastic resorption of bone. They are the only FDA-approved compounds in widespread use that increase bone mineral density. However, the increase is relatively small. The use of chronic continuous bisphosphonate therapy has been linked to insufficiency fractures of the proximal femur.
The initial treatment of symptomatic vertebral compression fractures involves a trial of analgesic therapy and bracing for comfort. Evaluation and treatment for osteoporosis can be initiated if not done already. Conservative therapy should be attempted for at least 6–12 weeks or longer if the patient is improving.
![]() Surgical Treatment
Surgical Treatment
Patients who have fractures that cause neurologic deficits or significant spinal cord compression should be treated with anterior decompression and fusion followed by posterior segmental instrumentation and fusion. The poor bone quality makes correction of deformity and maintenance of posterior only constructs a challenging task (Figure 4-27).

![]() Figure 4–27. Complex revision osteotomy surgery. A 65-year-old woman with incapacitating back and leg pain after two failed prior operations done at an outside hospital for an osteoporotic burst fracture. (A) Preoperative lateral x-ray demonstrates pullout of the screw construct, multiple nonunions, sagittal imbalance, and inability to walk due to pain. She underwent third-time revision decompression, complete removal of the L2 vertebral body (vertebral column resection [VCR]), and revision instrumentation and fusion, all done through a posterior approach. This procedure allows for shortening of the spinal column and correction of kyphotic deformity. (B) Postoperative lateral x-ray demonstrates markedly improved alignment. Patient was able to walk with minimal pain.
Figure 4–27. Complex revision osteotomy surgery. A 65-year-old woman with incapacitating back and leg pain after two failed prior operations done at an outside hospital for an osteoporotic burst fracture. (A) Preoperative lateral x-ray demonstrates pullout of the screw construct, multiple nonunions, sagittal imbalance, and inability to walk due to pain. She underwent third-time revision decompression, complete removal of the L2 vertebral body (vertebral column resection [VCR]), and revision instrumentation and fusion, all done through a posterior approach. This procedure allows for shortening of the spinal column and correction of kyphotic deformity. (B) Postoperative lateral x-ray demonstrates markedly improved alignment. Patient was able to walk with minimal pain.
Patients who have recalcitrant back pain from a non-united vertebral compression fracture and have failed a course of conservative management can obtain excellent symptomatic relief from fracture stabilization through injection of PMMA bone cement into the fracture through a percutaneous technique. The two most popular procedures, vertebroplasty and kyphoplasty, are both safe and efficacious. In both techniques, a cannula is inserted intrapedicularly or extrapedicularly (lateral to the pedicle) into the anterior portion of the affected vertebral body, and acrylic cement is instilled into the fractured bone under fluoroscopic control. Once the cement cures, the fracture is immediately stabilized. In the kyphoplasty technique, a balloon is inflated in the vertebral body in an attempt to compress the existing bone, create a void for instillation of more viscous cement under lower pressure, and correct the wedge deformity. This technique has the theoretical advantage of allowing some deformity correction and preventing high-pressure-related extrusion of PMMA into the spinal canal.
The mechanism of pain relief achieved through vertebroplasty and kyphoplasty is unclear. Multiple mechanisms may play a role, including fracture stabilization, denervation of pain fibers by the heat generated during the cement curing process, and neurotoxicity of the PMMA monomer. In addition, longer follow-up has raised concerns over pre-disposing the adjacent segment to fracture by overstiffening the affected level. These concerns are currently under active investigation.
Asenjo JF, Rossel F: Vertebroplasty and kyphoplasty: new evidence adds heat to the debate. Curr Opin Anaesthesiol 2012;25:577. [PMID: 22914353]
Coumans JV, Reinhardt MK, Lieberman IH: Kyphoplasty for vertebral compression fractures: 1-year clinical outcomes from a prospective study. J Neurosurg 2003;99(Suppl 1):44. [PMID: 12859058]
Do HM, Kim BS, Marcellus ML, et al: Prospective analysis of clinical outcomes after percutaneous vertebroplasty for painful osteoporotic vertebral body fractures. AJNR Am J Neuroradiol 2005;26:1623. [PMID: 16091504]
Garnier L, Tonetti J, Bodin A, et al: Kyphoplasty versus vertebroplasty in osteoporotic thoracolumbar spine fractures. Short-term retrospective review of a multicentre cohort of 127 consecutive patients. Orthop Traumatol Surg Res 2012;98:S112. [PMID: 22939104]
Grohs JG, Matzner M, Trieb K, et al: Minimal invasive stabilization of osteoporotic vertebral fractures: a prospective nonrandomized comparison of vertebroplasty and balloon kyphoplasty. J Spinal Disord Tech 2005;18:238. [PMID: 15905767]
Steinmann J, Tingey CT, Cruz G, et al: Biomechanical comparison of unipedicular versus bipedicular kyphoplasty. Spine (Phila Pa 1976) 2005;30:201. [PMID: 15644756]
Svedbom A, Alvares L, Cooper C, et al: Balloon kyphoplasty compared to vertebroplasty and nonsurgical management in patients hospitalised with acute osteoporotic vertebral compression fracture: a UK cost-effectiveness analysis. Osteoporos Int 2013;24:355. [PMID: 22890362]
![]() INJURIES OF THE CERVICAL SPINE
INJURIES OF THE CERVICAL SPINE
The cervical spine is the most mobile area of the spine, and as such, it is prone to the greatest number of injuries. Injuries to the cervical spine and spinal cord are also potentially the most devastating and life altering of all injuries compatible with life. In the United States, approximately 10,000 spinal cord injuries occur each year (about 39 per million). An estimated 80% of the victims are younger than 40 years, with the highest proportion of injuries reported in those between 15 and 35 years of age. Approximately 80% of all people who suffer from spinal column injuries are male. Falls account for 60% of injuries to the vertebral column in patients older than 75 years. In younger patients, 45% of injuries result from motor vehicle accidents, 20% from falls, 15% from sports injuries, 15% from acts of violence, and the remainder from other causes.
With the use of seat belts and air bags in motor vehicles and the advent of trauma centers and improved emergency service awareness of potential cervical injuries, fewer patients with cervical spine injuries are dying secondary to respiratory complications. The approach in treating these patients is early recognition of cervical spine injuries with rapid immobilization to prevent neurologic deterioration while the evaluation and treatment of associated injuries are carried out. After the patient is stabilized, the goals are restoration and maintenance of spinal alignment to provide stable weight bearing and facilitate rehabilitation.
![]() Identification and Stabilization of Life-Threatening Injuries
Identification and Stabilization of Life-Threatening Injuries
Eighty-five percent of all neck injuries requiring medical evaluation are a result of a motor vehicle accident. Many of the affected patients are multiple-trauma victims and therefore may have more urgent life-threatening conditions. The ABCs of trauma are followed in order of priority, with airway, b reathing (ventilation), and c irculation secured before further evaluation proceeds. Throughout the evaluation of other body systems, the cervical spine should be presumed injured and thus immobilized. Approximately 20% of patients with cervical trauma are hypotensive upon presentation. The hypotension is neurogenic in origin in approximately 70% of cases and related to hypovolemia in 30%. Concomitant bradycardia is suggestive of a neurogenic component. Another finding suggestive of cervical spine injury is an altered sensorium secondary to head trauma or lacerations and facial fractures. Appropriate diagnosis and fluid management are critical in the early hours of postinjury management. After all life-threatening injuries are identified and stabilized, the secondary evaluation, including an extremity examination and neurologic examination, can be safely carried out.
![]() History and General Physical Examination
History and General Physical Examination
Details of the history of the injury should be obtained. If the patient is conscious, much of the information can be obtained directly. If not, family members or witnesses of the injury should be questioned. In the case of a motor vehicle accident, for example, pertinent questions include the following: Which part of the patient’s body was the point of impact? Was the patient thrown from the car? Was there head trauma or a loss of consciousness? Were there any transient signs of paresis? Was the patient able to move any of his or her extremities at any time following the accident and before loss of function? What were the speeds of the involved motor vehicles? Was the patient restrained with a seat belt? Did an air bag deploy?
The history taken from the patient or family members should also include information about preexisting conditions such as epilepsy or seizures and about preexisting injuries. If the patient had any previous radiographic examinations, the radiographs might be useful for comparison.
It is helpful to question patients about what they are experiencing at the time of the examination. Are there areas of numbness, paresthesia, or pain? Can they move their extremities? The examiner should then proceed with the physical evaluation, beginning by observing the face and head of the patient for any areas of potential injury and attempting to determine the potential mechanism of injury. For instance, any lacerations or contusions to the forehead might indicate a hyperextension-type injury because as the head hits the windshield and stops, the body continues to move forward from the momentum of the impact. Observation should next include watching the extremities for any signs of motion. A genital examination should be performed because a sustained penile erection may be indicative of severe spinal cord injury. Then without moving the patient, palpation can be performed. Although palpation can be helpful in identifying potential levels of injury of the spine, it should not be used as the sole screening examination because false-negative results are possible.
![]() Neurologic Evaluation
Neurologic Evaluation
A meticulous neurologic examination should be performed following the history and general physical examination.
A. Neurologic Tests
The neurologic evaluation should start with documentation of the function of the cranial nerves, working proximally to distally. Observation is particularly important in the unconscious patient. Spontaneous motion in an extremity may be a sole source of information regarding spinal cord function. Respiratory efforts made with intrathoracic musculature versus abdominal musculature are also significant. In the conscious patient who is able to follow commands, a motor examination should be fairly straightforward. Rectal and perianal sensations should be documented because these may be the sole signs of intact distal spinal cord function.
An extensive sensory examination should also be performed with careful attention to dermatomal innervation. In the acute setting, it is useful to document sharp and dull sensations as well as proprioception. Sharp and dull sensations are carried via the lateral spinothalamic tract, whereas proprioception is carried through the posterior columns. Sharp and dull sensations are effectively tested with the sharp and blunt ends of a pen, and proprioception is tested by having the patient verify the position of the large toe and other joints as the examiner places them in dorsiflexion and plantarflexion. It proves helpful to make ink markings directly on the patient’s skin to show the level of the dermatomal deficit, which decreases the chance for intraobserver or interobserver error over sequential examinations.
Reflexes should be checked bilaterally. In the upper extremity, the biceps reflex at the flexor side of the elbow evaluates the C5 nerve root, and the brachioradialis stretch reflex at the radial aspect of the forearm just proximal to the wrist checks the C6 nerve root. The triceps reflex is innervated by C7. In the lower extremity, the knee jerk reflex is innervated by L4, and the ankle jerk is innervated by S1.
The presence or absence of the four reflexes listed in Table 4–8 should be checked. The Babinski reflex (plantar reflex) is evaluated by firmly stroking the lateral plantar aspect of the foot distally and then medially over the metatarsal heads and then observing the toes. If the toes flex, the response is considered negative (normal). If the toes extend and spread, the response is considered positive (abnormal) and indicative of an upper motor neuron lesion. The bulbocavernosus reflex has its root in the S3 and S4 nerves and is evaluated by squeezing on the glans in a male patient or applying pressure to the clitoris in a female patient. This action should elicit a contraction of the anal sphincter. If a Foley catheter is in place, simply pulling on the Foley catheter can stimulate the anal sphincter contraction. The cremasteric reflex is evaluated by stroking the inner thigh and observing the scrotal sac, which should retract upward secondary to contraction of the cremasteric muscle. This function is innervated by T12 and L1. Finally, the anal wink, innervated by S2, S3, and S4, is elicited by stimulating the skin about the anal sphincter and eliciting a contraction.
Table 4–8. Evaluation of reflexes in patients with injuries of the cervical spine.

The presence of spinal shock causes the absence of all reflexes below the level of injury and typically lasts up to 24 hours after the injury. The bulbocavernosus reflex is the reflex that returns first (see Table 4–8), thus marking the end of spinal shock. This point has prognostic importance because recovery from a complete neurologic deficit that is still present at the end of spinal shock is extremely unlikely. A complete neurologic examination should be repeated over time as the patient is manipulated and treated.
B. Anatomic Considerations
The ability to interpret the results of a patient’s neurologic examination appropriately depends on a thorough knowledge of the anatomy of the spinal cord and peripheral nerves.
Peripheral nerves are a combination of afferent fibers, which carry information from the periphery to the central nervous system, and efferent fibers, which carry information away from the central nervous system. As the peripheral nerve approaches the spinal cord, it becomes known as the spinal nerve. Prior to entering the spinal cord, the fiber splits, with the afferent fibers becoming the dorsal root or sensory root and the efferent fibers becoming the ventral root. The afferent fibers are often regrouped in various plexuses that are located between the spinal cord and the periphery. This regrouping takes place before the fibers enter the dorsal root, therefore leading to significant overlap between the dorsal root and the respective dermatomes. The implications of this anatomic fact should be kept in mind by the clinician when performing a sensory examination. For example, a sectioned peripheral nerve is demonstrated by a highly specific sensory loss in that particular area served by that nerve, whereas the clinical findings are more variable for a sectioned dorsal root.
The spinal cord is a caudal continuation of the brain, extending in an organized fashion from the foramen magnum at the base of the skull down to the proximal lumbar spine. The spinal cord has three primary functions: It provides a relay point for sensory information; it serves as a conduit for ascending sensory information and descending motor information; and it mediates body and limb movements because it contains both interneurons and motor neurons. Headed from caudal to rostral, the spinal cord is highly organized with a central butterfly-shaped area of gray matter and surrounding white matter.
The overall diameter of the spinal cord varies as a relative percentage of the spinal canal. The cord fills approximately 35% of the canal at the level of the atlas but increases to approximately 50% of the canal in the lower cervical spine. This variation results from the relative increasing and decreasing size of the spinal gray matter and spinal white matter. As the spinal roots become larger, as occurs at the base of the cervical spine, the size of the gray matter increases relative to the white matter, whereas the size of the white matter decreases linearly from cephalad to caudal.
The gray matter, so called because it appears gray on unstained cross sections, is divided into three zones: the dorsal horn, the intermediate zone, and the ventral horn. Made up predominantly of lower motor neurons, it is prominent in the cervical swellings and lumbar swellings, where axons concentrate before exiting to innervate the upper extremities and lower extremities, respectively.
The white matter derives its name from the fact that the axons in this area are myelinated, casting a white hue on unstained sections. White matter is functionally and anatomically divided into three bilaterally paired columns: the ventral columns, the lateral columns, and the dorsal columns.
The two major ascending systems that relay somatic sensory information are the dorsal columns and the anterolateral system. The ascending axon has its cell body located in the dorsal root ganglion before proceeding without synapsing through the dorsal horn at that level and then ascending along the dorsal column before synapsing at the approximate level of the medulla and crossing over to the contra-lateral side before proceeding to the cerebral cortex. The topography of the dorsal column is such that the sacrum and lower extremities are medial, with the trunk and cervical region being lateral. The anterolateral system carries pain and temperature sensorium. The afferent fibers have a cell body in the dorsal root ganglion and then synapse at that given level in the dorsal horn before crossing directly to the contralateral side and traveling up the spinothalamic tract.
Motor pathways originate in the cerebral cortex and travel distally to the contralateral side approximately at the level of the medulla and travel down the lateral corticospinal tract before synapsing with the lower motor neuron in the ventral horn of the gray matter. The topography of the corticospinal tract is such that the sacrum and legs lie lateral to the trunk and cervical axons. Thus, at the level of the cervical spine, the spinal cord contains both lower motor neurons traversing to the upper extremities and upper motor neurons being transmitted to the lower extremities. Therefore, injury in this area can give both upper and lower motor findings.
The anatomy of the reflex arc and especially its relationship to spinal shock should be kept in mind. The basic reflex circuitry is an afferent nerve coming from a stretch receptor through the dorsal horn of gray matter before synapsing with the lower motor neuron in the ventral horn of the gray matter, which sends a positive signal to the same muscle via an alpha motor neuron. This simple arc, however, is modulated by input from higher centers. If all descending influence is interrupted, such as would occur in a traumatic transection of the spinal cord, all reflexes are lost. This is also seen during spinal shock. If the local circuitry of the reflex arc is not disturbed, reflexes return at the end of spinal shock. The earliest reflex to return is the bulbocavernosus reflex, which typically returns within 24 hours of injury. Peripheral reflexes may take several months before they return.
C. Risk of Neurologic Damage
As mentioned earlier, the spinal cord varies in its diameter from cephalad to caudad. In the upper cervical spine, it occupies approximately a third of the spinal canal. In the lower cervical spine, it occupies approximately half of the canal. As inferred from this anatomy, the risk of neurologic damage from injury is greater in the lower cervical spine.
Cord compromise extends from two causes: mechanical destruction resulting directly from the trauma and vascular insufficiency. With vascular insufficiency, hypoxia and edema follow and result in further tissue damage. By approximately 6 hours after the trauma, axonal transport ends, and by 24 hours, cord necrosis begins.
D. Classification of Neurologic Status
1. Intact—Approximately 60% of injuries to the cervical spine result in no neurologic sequelae. In most of these cases, the injuries are in the upper cervical spine, where the ratio of the spinal cord to the spinal canal is smaller. It is obviously critical to identify unstable injuries of the cervical spine in the intact patient because the evolution of neurologic deficits is both potentially catastrophic and preventable.
2. Nerve root injuries—Eight cervical nerve roots correspond to the seven cervical vertebral bodies. Each of the first seven nerve roots exits above its respective body (the C1 nerve exiting above the C1 vertebral body, the C2 nerve exiting above the C2 body, and so forth), whereas the C8 nerve root exits through the foramen between the C7 and T1 vertebral bodies. Nerve root injuries can happen either in isolation or in conjunction with more severe spinal cord injuries. Injury to the nerve root alone may result from a compression or fracture of the lateral bone mass and thus impingement on the neural foramen. The clinical findings of a root injury would be those of a lower motor neuron lesion. If the nerve root is still intact and the ongoing pressure to the root is removed, the prognosis for recovery of nerve root function is good.
3. Incomplete versus complete neurologic injury—In the acute setting, any evidence of neurologic function distal to the level of injury is significant and defines the lesion as being incomplete rather than complete. As Lucas and Ducker reported in a prospective study published in 1979, “The less the injury, the greater the recovery,” and “partial lesions partially recover, whereas complete lesions do not.”
The motor and sensory examination outlined by the American Spinal Injury Association (ASIA) is the most widely accepted and utilized system to assess the impact of spinal cord injury on the patient. It involves the use of a grading system to evaluate the remaining sensory and motor function. The system allows the patients to be assessed through scales of impairment and functional independence.
The sensory level is determined by the patient’s ability to perceive pinprick (using a disposable needle or safety pin) and light touch (using a cotton ball). Testing of a key point in each of the 28 dermatomes on the right and the left sides of the body and evaluation of perianal sensation are required. The variability in sensation for each individual stimulus is graded on a three-point scale:
0 = absent
1 = impaired
2 = normal
NT = not testable
In the cervical spine, the C3 and C4 nerve roots supply sensation to the entire upper neck and chest in a cape-like distribution from the tip of the acromion to just above the nipple line. The next adjacent sensory level is the T2 dermatome. The brachial plexus, C5-T1, supplies the upper extremities.
ASIA also recommends testing of pain and deep pressure sensation in the same dermatomes as well as an evaluation of proprioception by testing the position sense of the index fingers and great toes on each side.
The motor level is determined by manual testing of a key muscle in the 10 paired myotomes from rostral to caudal. The strength of each muscle is graded on a six-point scale:
0 = total paralysis
1 = palpable or visible contraction
2 = full range of motion of the joint powered by the muscle with gravity eliminated
3 = full range of motion of the joint powered by the muscle against gravity
4 = active movement with full range of motion against moderate resistance
5 = normal strength
NT = not testable
For myotomes that are not clinically testable by manual muscle evaluation, the motor level is presumed to be the same as the sensory level (C1-C4, T2-L1, S2-S5).
ASIA also recommends evaluation of diaphragmatic function (via fluoroscopy, C4 level) and the abdominal musculature (via the Beevor sign, which is the upward migration of the umbilicus from upper abdominal contraction in the absence of lower abdominal contraction due to paralysis at the T10 level). Evaluation of medial hamstring and hip adductor strength is also recommended but not required.
E. Clinical Features of Spinal Cord Syndromes
Combining the findings on examination with knowledge of the cross-sectional anatomy of the spinal cord allows the examiner to identify specific injury patterns (Figure 4–28).

![]() Figure 4–28. Diagrams illustrating cross-sectional views of the normal and injured spinal cord. The diagram of the normal spinal column shows the segmental arrangement (C, cervical; L, lumbar; S, sacral; T, thoracic) and the area of flexors and extensors (FLEX and EXT). Central cord syndrome, anterior cord syndrome, Brown-Séquard syndrome, and posterior cord syndrome are incomplete injuries, with affected areas shaded. In complete spinal cord injury, all areas are affected.
Figure 4–28. Diagrams illustrating cross-sectional views of the normal and injured spinal cord. The diagram of the normal spinal column shows the segmental arrangement (C, cervical; L, lumbar; S, sacral; T, thoracic) and the area of flexors and extensors (FLEX and EXT). Central cord syndrome, anterior cord syndrome, Brown-Séquard syndrome, and posterior cord syndrome are incomplete injuries, with affected areas shaded. In complete spinal cord injury, all areas are affected.
1. Central cord syndrome—The most common of the incomplete cord syndromes is the central cord syndrome, which occurs most frequently in elderly (>65 years) people with underlying degenerative spondylosis but can also be found in younger people who have had a severe hyperextension injury with or without evidence of a fracture, known as spinal cord injury without radiographic abnormality (SCIWORA). Central cord syndrome is defined by ASIA as a clinical presentation characterized by “dissociation in degree of motor weakness with lower limbs stronger than upper limbs and sacral sensory sparing.” The syndrome typically occurs following a hyperextension injury and is thought to be caused by an expanding hematoma or edema forming in the central aspect of the spinal cord. Central cord syndrome can be quite variable in presentation and in recovery. A mild presentation may consist of a slight burning sensation in the upper extremities, whereas a severe central cord syndrome includes motor impairment in both the upper and lower extremities, bladder dysfunction, and a variable sensory deficit below the level of injury. The pattern of clinical presentation is directly related to the cross-sectional anatomy of the spinal cord. Because the lower extremity and sacral tracts of the spinothalamic and corticospinal tracts are lateral, these areas are often spared in central cord syndrome. In cases in which they are involved, they are the areas whose function returns first. The upper extremity deficit is caused by a lesion in the gray matter, and the damage here is largely irreversible.
From 50 to 75% of patients with central cord lesions show some neurologic improvement, but the amount of improvement varies considerably among patients. The usual order in which motor function recovery occurs is as follows: return of lower extremity strength, return of bladder function, return of upper extremity strength, and return of intrinsic function of the hand.
2. Anterior cord syndrome—The patient with an anterior cord syndrome typically presents with immediate paralysis and loss of pain and temperature sensation. Both the spinothalamic and corticospinal tracts are located in the anterior aspect of the spinal cord and are therefore involved. With the dorsal columns preserved, the patient still has intact proprioception and vibratory sense as well as intact sensation to deep pressure. This clinical presentation is the most common in the younger (<35 years) trauma victim. The mechanism of injury is typically a flexion injury to the cervical spine. It is usually associated with an identifiable lesion of the cervical spine, most commonly a vertebral body burst fracture or a herniated disk. Return of useful motor function is reported in only 10–16% of patients with anterior cord syndrome. The prognosis is slightly improved, however, if evidence of spinothalamic tract function is present early.
3. Brown-Séquard syndrome—Patients with this syndrome have a motor weakness on the ipsilateral side of the lesion and a sensory deficit on the contralateral side caused by a functional hemisection of the spinal cord. For example, a cervical lesion on the right side of the spinal cord disrupts the ipsilateral corticospinal tract, which is the tract that carries motor function to the right side of the body distal to the level of the lesion. The right spinothalamic tract is also disrupted. This tract carries pain and temperature fibers from the contralateral side of the body distal to the level of injury. Position sense and vibratory sense, which are carried in the posterior column, have not yet crossed the midline; therefore, these sensory functions are disrupted on the ipsilateral side of the injury.
Brown-Séquard syndrome may result from a closed rotational injury such as a fracture-dislocation or may result from a penetrating trauma such as a stab wound or from iatrogenic injury while placing surgical instruments within the spinal canal. The prognosis in cases resulting from a closed injury is quite favorable, with 90% of patients regaining function of the bowel and bladder as well as the ability to walk.
4. Posterior cord syndrome—The posterior cord syndrome is the least common of the incomplete syndromes and typically a result of an extension-type injury. Its clinical presentation is one of loss of position and vibratory sense below the level of injury secondary to disruption of the dorsal columns. With these deficits as isolated findings, the prognosis for recovery of ambulation and function of the bowel and bladder is excellent.
5. Complete spinal cord injury—A complete neurologic deficit is characterized by a total absence of sensation and voluntary motor function caudal to the level of spinal cord injury in the absence of spinal shock. Initial evaluation must rule out any evidence of sacral sparing and the presence of a bulbocavernosus reflex. In the absence of sacral sparing and with the return of the bulbocavernosus reflex, which typically occurs within 24 hours, the spinal cord injury is termed complete and there is virtually no likelihood of functional spinal cord recovery. Affected patients may gain some root function about the level of the injury—a phenomenon called root escape because this damage to nerve roots is a peripheral nerve injury. Although the presence of root escape should not be taken as a potential return of spinal cord function, it can significantly improve the patient’s rehabilitation efforts because vital function of the upper extremities may be regained.
![]() Imaging Studies
Imaging Studies
A. Radiography
1. Screening radiograph—A lateral radiograph of the cervical spine may be the only screening tool obtained upon initial radiographic evaluation of the multiple-trauma patient. This radiograph must be carefully reviewed. Should a patient present with a complete neurologic injury or a densely affected incomplete neurologic injury indicating a traumatically malaligned cervical spine, closed reduction of the cervical spine should be urgently attempted with axial traction through Gardner-Wells tongs. Once the patient is fully evaluated and life-threatening injuries are stabilized, secondary diagnostic studies can then be undertaken. If the patient is fully alert, has full pain-free rotational range of motion, no palpable tenderness, and no other injuries, the cervical spine can be cleared on clinical grounds.
2. Subsequent plain radiographs—Full radiographic evaluation of the cervical spine with plain radiographs includes lateral, AP, open-mouth (odontoid), right oblique, and left oblique views. The lateral radiograph, if adequate, visualizes approximately 85% of significant cervical spine injuries. It must display the base of the skull with all seven cervical vertebrae, as well as the proximal half of the T1 vertebral body. If the C7-T1 junction is not visualized, a repeat radiograph should be done with axial traction on the upper extremities caudally to attempt to visualize the C7-T1 junction. If this is unsuccessful, a swimmer’s view, which is a transthoracic lateral with the patient’s arm fully abducted, should be taken. If this plain radiograph is not satisfactory and if suspicion of injury is still high, a CT scan must be obtained.
When evaluating a lateral cervical spine radiograph, the clinician should first evaluate the bony anatomy. Four lines or curves should be kept in mind (Figure 4–29). The anterior spinal line and the posterior spinal line are imaginary lines drawn from the anterior cortex and posterior cortex, respectively, of the cervical vertebral body from C2 all the way down to T1. The spinal laminar curve is an imaginary line drawn from the posterior aspect of the foramen magnum connecting the anterior cortex of each successive spinous process. These three lines (labeled A, B, and C in Figure 4–29) should have a gentle, continuous lordotic curve with no areas of acute angulation. The fourth line (labeled D in Figure 4–29) is known as the basilar line of Wackenheim, and it is drawn along the posterior surface of the clivus and should thus be tangent to the posterior cortex of the tip of the odontoid process. After the clinician examines the radiograph in terms of these four lines or curves, he or she should look at the individual vertebral bodies to see if there is loss of height of any of them or if a rotational deformity is present with alterations in the alignment of the facets.

![]() Figure 4–29. Diagram illustrating normal lines and curves in the bony anatomy of the cervical spine. The anterior spinal line (line A), the posterior spinal line (line B), and the spinal laminar curve (line C) should have a gentle, continuous lordotic curve. The basilar line of Wackenheim (line D) is drawn along the posterior surface of the clivus and should thus be tangent to the posterior cortex of the tip of the odontoid process. (Reproduced, with permission, from El-Khoury GY, Kathol MH: Radiographic evaluation of cervical spine trauma. Semin Spine Surg 1991;3:3.)
Figure 4–29. Diagram illustrating normal lines and curves in the bony anatomy of the cervical spine. The anterior spinal line (line A), the posterior spinal line (line B), and the spinal laminar curve (line C) should have a gentle, continuous lordotic curve. The basilar line of Wackenheim (line D) is drawn along the posterior surface of the clivus and should thus be tangent to the posterior cortex of the tip of the odontoid process. (Reproduced, with permission, from El-Khoury GY, Kathol MH: Radiographic evaluation of cervical spine trauma. Semin Spine Surg 1991;3:3.)
The evaluation of soft tissues can also prove valuable diagnostically. Prevertebral soft tissues have an upper limit of normal width beyond which a prevertebral hematoma indicative of vertebral injury can be suspected. The upper limits of normal are 11 mm at C1, 6 mm at C2, 7 mm at C3, and 8 mm at C4. The measurements below C4 become more variable and therefore less reliable clinically.
The AP view of the cervical spine is at first a confusing projection to those who are unfamiliar with cervical anatomy, yet careful attention to bony detail in the AP view can be of significant diagnostic aid in picking up subtle injuries. The bony and soft-tissue anatomy seen on the AP projection should be symmetric. The spinous processes should be equally spaced because a single level of increased intraspinous process distance suggests posterior instability. Abrupt malalignment of the spinous processes suggests a rotatory injury such as a unilateral facet dislocation. After checking for these problems, the clinician should inspect the lateral masses. The facet joints are typically angled away from the vertical and therefore not clearly seen on the AP projection. If, however, the facet joint can be seen at a particular level, this is indicative of a fracture through the lateral masses and a rotational malalignment of the facet.
The open-mouth (odontoid) view is the projection most useful for looking at C1-C2 anatomy. It permits visualization of both the dens in the AP plane and the lateral masses of C1 on C2.
The right and left oblique views can be taken of the cervical spine with the patient in the supine position. These views are useful as confirmatory studies in ruling in or out lateral mass injuries.
3. Stress radiographs—Two techniques are used in obtaining cervical stress radiographs. The first is to apply axial distraction to the cervical spine through a halo or traction device and obtain a lateral radiograph. This technique should be carefully performed in the presence of a physician and only after gross instabilities of the cervical spine are ruled out. Serial lateral radiographs are taken as weight is sequentially added, reaching an amount equivalent to approximately a third of body weight or 30 kg, depending on the level of suspected injury. Occult instability can be inferred by noting an interspace angulation of at least 11 degrees or an interspace separation of at least 1.7 mm (Figure 4–30).

![]() Figure 4–30. (A) Diagram illustrating an increase of the C2-C3 interdisk space in a patient with type IIA traumatic spondylolisthesis. (B) Radiograph demonstrating an increased space. (Reproduced, with permission, from Levine AM, Rhyne AL: Traumatic spondylolisthesis of the axis. Semin Spine Surg 1991;3:47.)
Figure 4–30. (A) Diagram illustrating an increase of the C2-C3 interdisk space in a patient with type IIA traumatic spondylolisthesis. (B) Radiograph demonstrating an increased space. (Reproduced, with permission, from Levine AM, Rhyne AL: Traumatic spondylolisthesis of the axis. Semin Spine Surg 1991;3:47.)
The second technique, which should only be performed in a fully alert and cooperative patient, is used to obtain flexion-extension lateral radiographs that are helpful in the diagnosis of late instability. The technique is to have the patient flex the head forward as far as possible while a lateral radiograph is taken and then to have the patient put the head in full extension while another radiograph is taken. Findings presumptive of instability are facet subluxation, forward subluxation of 3.5 cm of one vertebral body on the next, and interbody angulation of greater than 11 degrees.
B. Computed Tomography
CT scanning is the most useful means for definitive delineation of bony fracture anatomy. Its advantages are its ready availability and its ability to be performed with a minimal amount of patient manipulation. CT scans provide excellent axial detail, and if thin enough sections are taken, the computer can reconstruct images in sagittal, coronal, or oblique planes. CT scans can now even be reformatted into a three-dimensional construct for excellent visualization of the bony anatomy.
C. Magnetic Resonance Imaging
MRI is the most effective way to evaluate the soft-tissue component of cervical trauma. The major advantage of MRI is that it can visualize occult disk herniation, hematoma, or edema about the spinal cord, as well as ligamentous injury. Current disadvantages are that MRI is disrupted by metallic objects, so these should be removed from the area of examination, and it also requires a prolonged amount of time to perform, therefore making close monitoring of the acutely ill patient difficult.
![]() Diagnostic Checklist of Spinal Instability
Diagnostic Checklist of Spinal Instability
The concept of spinal stability is central to the understanding and treatment of cervical spine injuries. In a broad sense, patients with injuries that are deemed unstable require surgical intervention, whereas those deemed to have stable injury patterns can be treated nonoperatively. Spinal injuries, however, are not readily divided into unstable and stable injuries, and in actuality, they fall along a spectrum of spinal instability.
White and Panjabi’s diagnostic checklist of spinal instability (Table 4–9) has nine categories, each of which is assigned a point value. If a total of 5 points is present in a given patient, the injury is deemed unstable.
Table 4–9. White and Panjabi’s diagnostic checklist of spinal instability.

Holdsworth’s two-column theory of spine stability, as well as Denis’s three-column theory, proposed for application to the thoracolumbar spine, are also applied to the cervical spine in an attempt to better predict stability in the neck.
![]() General Principles of Managing Acute Injuries of the Cervical Spine
General Principles of Managing Acute Injuries of the Cervical Spine
Management of acute cervical spine injury is predicated on two principles: protection of the uninjured spinal cord and prevention of further damage to the injured spinal cord. This is accomplished by following spine precaution principles from the very onset of medical care, starting at the accident scene. The cervical spine should be considered injured until proven otherwise and securely immobilized before the patient is transported to a medical center. The equipment for initial immobilization should not be removed until the definitive means of immobilization can be put in place or the cervical spine is cleared of injury. Use of a spinal board, with the patient’s head taped to the board and held between two sandbags, is the most secure form of immobilization readily available in the field. This technique can be supplemented by a Philadelphia collar. When the medical center is reached, if a definitive cervical spine injury is identified and deemed unstable, skeletal traction for immobilization, reduction, or both may be applied. Gardner-Wells traction is easily applied and adequate for axial traction. Halo traction affords the added advantage of four-point fixation and thus controlled traction in three planes. Prior to application of traction, it is important to make sure that the patient does not have an occipitocervical dislocation. In these cases, application of traction can lead to worsening of the dislocation and neurologic injury. These specific cases should be treated with immediate application of a halo vest. Halo traction can also be easily converted at a later time to halo-vest immobilization.
Among the various agents that show potential benefits in laboratory studies of models of spinal cord injury are corticosteroids, opiate receptor antagonists (such as naloxone or thyrotropin-releasing hormone), and diuretics (such as mannitol). The National Acute Spinal Cord Injury Studies (NASCIS) II and III reported neurologic improvement with steroid treatment given within 8 hours of injury. Those treated within 3 hours did best; those treated between hours 3 and 8 only did better by extending to 48 hours of treatment. Criticism of the NASCIS studies called to question the validity of the conclusions, and many professional organizations downgraded their enthusiasm for the use of methylprednisolone in the patient with the acutely injured spinal cord. However, many hospitals still use the protocol in blunt trauma cord injuries if the medicine can be administered within 3 hours of the injury. The recommended dosage of methylprednisolone in an acute setting is 30 mg/kg given as a bolus and followed by 5.4 mg/kg/h for 24 hours. However, some thought should be given to its use because, for example, the Congress of Neurological Surgeons stated that steroid therapy “should only be undertaken with the knowledge that the evidence suggesting harmful side effects is more consistent than any suggestion of clinical benefit.”
Cripps RA, Lee BB, Wing P, et al: A global map for traumatic spinal cord injury epidemiology: towards a living data repository for injury prevention. Spinal Cord 2011;49:493. [PMID: 21102572]
Denis F: The three-column spine and its significance in the classification of acute thoracolumbar spinal injuries. Spine (Phila Pa 1976) 1983;8:817. [PMID: 6670016]
Ito Y, Sugimoto Y, Tomioka M, et al: Does high dose methylprednisolone sodium succinate really improve neurological status in patient with acute cervical cord injury? A prospective study about neurological recovery and early complications. Spine (Phila Pa 1976) 2009;34:2121. [PMID: 19713878]
White AA III, Panjabi MM: Update on the evaluation of instability of the lower cervical spine. Instr Course Lect 1987;36:513. [PMID: 3437146]
INJURIES OF THE UPPER CERVICAL SPINE
With the exception of occipitoatlantal dissociation, traumatic injuries to the upper cervical spine are less frequently associated with significant neurologic injury than are traumatic injuries to the lower cervical spine. This is secondary to the fact that the spinal cord occupies only a third of the upper spinal canal versus a half of the lower spinal canal.
![]() Occipitoatlantal Dissociation
Occipitoatlantal Dissociation
Occipitoatlantal dissociation is a disruption of the cranial vertebral junction, and it implies a subluxation or complete dislocation of the occipitoatlantal facets. This injury is typically fatal, yet the clinician must be aware of it because unrecognized occipitoatlantal dissociation may have catastrophic results. The mechanism of dissociation is poorly understood, but it most likely results from either a severe flexion or distraction type of injury. Anterior translation of the skull on the vertebral column is a common presentation and most likely a hyperflexion injury. Bucholz, however, presented the pathologic anatomic findings of fatal occipitoatlantal dissociation and proposed a mechanism of hyperextension with resultant distractive force applied across the craniovertebral junction.
When the dissociation is a frank dislocation, the findings are clear on a lateral radiograph. When the dissociation is a subluxation, however, findings may be more subtle. In normal individuals, the distance between the tip of the dens and the basion (the anterior aspect of the foramen magnum) should be no greater than 1.0 cm, and the previously described Wackenheim line should run from the base of the basion tangentially to the tip of the dens. If the dens penetrates this line, anterior translation of the cranium is implied. Calculation of the Powers ratio can also be helpful in securing the diagnosis. Powers and his colleagues described a ratio of two lines (Figure 4–31), the first of which runs from the tip of the basion to the midpoint of the posterior lamina of the atlas (line BC) and the second of which runs from the anterior arch of C1 to the opisthion (line AO). When the ratio of BC to AO is greater than 1:1, anterior occipitoatlantal dissociation is present. Other radiographic signs include marked soft-tissue swelling and the presence of avulsion fractures at the occipitovertebral junction.

![]() Figure 4–31. Diagram showing lines used in the calculation of the Powers ratio, which is helpful in diagnosing occipitoatlantal dissociation. The distance between the basion (point B) and the posterior arch (point C) is divided by the distance between the anterior arch of C1 (point A) and the opisthion (point O). The normal ratio of BC to AO is 1:1. A ratio of greater than 1 suggests the head is dislocated anteriorly on the spine.
Figure 4–31. Diagram showing lines used in the calculation of the Powers ratio, which is helpful in diagnosing occipitoatlantal dissociation. The distance between the basion (point B) and the posterior arch (point C) is divided by the distance between the anterior arch of C1 (point A) and the opisthion (point O). The normal ratio of BC to AO is 1:1. A ratio of greater than 1 suggests the head is dislocated anteriorly on the spine.
Early recognition and surgical stabilization are the mainstays of treatment in cases of occipitoatlantal dissociation.
![]() Fractures of Vertebra C1 (Atlas Fractures)
Fractures of Vertebra C1 (Atlas Fractures)
The mechanism of injury in the fracture of the atlas is most typically axial compression with or without extension force, and the anatomic findings of the fracture are indicative of the specifics of the force and the position of the head at the time of impact. In 1920, Jefferson presented his classic description of the four-part fracture of the atlas following an axial injury. This fracture is a burst type that occurs secondary to the occipital condyles being driven into the interior portions of the ring of the atlas and driving the lateral masses outward, resulting in a two-part fracture of the anterior ring of the atlas as well as a two-part fracture of the posterior ring. More common than the classic four-part atlas fracture, however, are the two-part and three-part fractures. Isolated anterior arch fractures are the least common, and they are typically associated with fractures of the dens, whereas the more common posterior arch fracture is typically the result of a hyperextension injury.
A fracture of the atlas is typically diagnosed on plain radiographs. Findings may be subtle on the lateral cervical spine radiograph. The open-mouth (odontoid) view may show asymmetry of the lateral masses of C1 on C2 with overhang (Figure 4–32). A bilateral overhang totaling more than 6.9 mm is presumptive evidence of a disruption to the transverse ligament and suggests potential late instability. Presumptive evidence for transverse ligament disruption can also be seen on the lateral radiograph if the ADI is greater than 4 mm.

![]() Figure 4–32. Open-mouth (odontoid) radiographic view demonstrating asymmetry of the lateral masses of C1 on C2 with overhang in a patient with a Jefferson fracture. (Reproduced, with permission, from El-Khoury GY, Kathol MH: Radiographic evaluation of cervical spine trauma. Semin Spine Surg 1991:3:3.)
Figure 4–32. Open-mouth (odontoid) radiographic view demonstrating asymmetry of the lateral masses of C1 on C2 with overhang in a patient with a Jefferson fracture. (Reproduced, with permission, from El-Khoury GY, Kathol MH: Radiographic evaluation of cervical spine trauma. Semin Spine Surg 1991:3:3.)
The treatment for fractures of the atlas as isolated injuries is typically nonoperative (Figure 4–33). If there are signs of transverse ligament disruption, halo traction is indicated with later transfer to halo-vest immobilization for a total of 3–4 months. Immediate halo-vest application is indicated in cases involving a moderately displaced fracture with lateral mass overhang up to 5 mm, although collar immobilization is preferred in cases involving a minimally displaced fracture of the atlas. At completion of bony union, flexion-extension views should be obtained to rule out any evidence of late instability. If late instability is present and the bony elements were allowed to heal, a limited C1-C2 fusion can address the instability. If a nonunion is present or if the posterior arch remains disrupted, an occiput to C2 fusion is necessary to control the late instability.

![]() Figure 4–33. Imaging studies in a patient who was in a motor vehicle accident and sustained a distractive extension injury to his cervical spine and a three-part fracture of his atlas (a Jefferson fracture). (A) Lateral radiographic view showing a fracture of the posterior arch. (B) Axial section of a CT scan further delineating the fracture anatomy. This injury was deemed stable and treated nonoperatively in a halo vest.
Figure 4–33. Imaging studies in a patient who was in a motor vehicle accident and sustained a distractive extension injury to his cervical spine and a three-part fracture of his atlas (a Jefferson fracture). (A) Lateral radiographic view showing a fracture of the posterior arch. (B) Axial section of a CT scan further delineating the fracture anatomy. This injury was deemed stable and treated nonoperatively in a halo vest.
![]() Dislocations and Subluxations of Vertebrae C1 and C2
Dislocations and Subluxations of Vertebrae C1 and C2
A. Atlantoaxial Rotatory Subluxation
Atlantoaxial rotatory subluxation is most common in children and may be associated with minimal trauma or even occur spontaneously. Although some patients are asymptomatic, others present with neck pain or torticollis (a position in which the head is tilted toward one side and rotated toward the other). Inasmuch as the mechanism of injury is often unclear, the propensity for the C1-C2 location is based on anatomic factors. In approximately 50% of cases, cervical spine rotation occurs at the C1-C2 junction, where the facet joints are more horizontal and less inherently stable in rotation.
The diagnosis of atlantoaxial rotatory subluxation is typically suspected on the basis of radiographs taken in several views. The odontoid view may show displacement of the lateral masses with respect to the dens; a lateral view may show an increased ADI; and the AP view may show a lateral shift of the spinous process of C1 on C2. CT scanning can be used to confirm the diagnosis, and a dynamic CT scan with full attempted right and left rotation can demonstrate a fixed deformity.
There are four types of atlantoaxial rotatory subluxations. In type I, the ADI is less than 3 mm, which suggests the transverse ligament is still intact. In type II, the interval is 3–5 mm, which suggests the transverse ligament is not structurally intact. In type III, the interval exceeds 5 mm, which is indicative of disruption of the transverse ligament as well as secondary stabilization of the alar ligament. In type IV, there is a complete posterior dislocation of the atlas on the axis, a finding typically associated with a hypoplastic odontoid process such as that seen in several forms of mucopolysaccharidosis (eg, the Morquio syndrome).
Treatment of atlantoaxial subluxation is typically conservative, consisting of traction followed by immobilization. Approximately 90% of patients respond to this treatment regimen. There is a high incidence of recurrence, however. For patients who do not respond to conservative measures and for patients with recurrent problems, C1-C2 arthrodesis may be required to control the deformity.
B. Disruption of the Transverse Ligament
The transverse ligament and secondarily the alar ligament are the main constraints to anterior displacement of C1 on C2. It was previously presumed that because anterior subluxation of C1 on C2 typically involves a fracture through the dens, the transverse ligament is in fact stronger than the bony elements of the dens. Fielding and his colleagues, however, showed that experimentally this was not the case, yet clinically the higher association of anterior dislocation of dens fractures still holds true.
The mechanism of disruption is typically a flexion injury, and the diagnosis is made on lateral radiographs. The ADI should not exceed 3 mm in the adult. If the interval is 4 mm or larger and the dens is intact, a rupture of the transverse ligament is presumed.
High-resolution CT scan can be used to categorize this injury into two types. Type 1 is a disruption in the substance of the transverse ligament, whereas type 2 involves an avulsion fracture of the insertion of the transverse ligament on the lateral mass of C1. Type 1 injuries predictably fail conservative treatment and should be managed with a C1-C2 arthrodesis. A trial of nonoperative care in type 2 injuries using a rigid cervical orthosis may be a reasonable alternative. A 74% success rate can be anticipated, with surgery reserved for patients who fail nonoperative care, showing persistent instability after 12 weeks in mobilization.
C. Fracture of the Odontoid Process
Fracture of the odontoid process is typically associated with high-velocity trauma, and the mechanism of injury is flexion in most cases. Depending on the fracture pattern, extension may be the predominant force in a smaller subset of cases. Associated injuries, particularly fractures of the ring of the atlas, should be ruled out. Neurologic involvement is relatively rare with odontoid fractures. In a study of 60 patients with acute fractures of the odontoid process, Anderson and D’Alonzo reported that 15 had some neurologic deficit on presentation, but only five of the 15 had major neurologic involvement, and only two of this group of five remained quadriparetic at follow-up.
Odontoid fractures may be suspected on the basis of clinical presentation and confirmed on plain radiographs, although spasm and overlying shadows can obscure the diagnosis. CT scan with sagittal and coronal reconstruction is the most sensitive study to diagnose these injuries. CT scan with axial sectioning alone may miss the horizontal fracture line typical of these injuries; thus, the reconstructions are necessary.
Both the risk of nonunion with delayed instability and the method of treating odontoid fracture depend on the classification of the fracture. Reported rates of nonunion range from 20 to 63%. According to the classification system proposed in 1974 by Anderson and D’Alonzo, there are three types of fracture of the odontoid process (Figure 4–34).

![]() Figure 4–34. Diagram showing the three types of fractures of the odontoid process.
Figure 4–34. Diagram showing the three types of fractures of the odontoid process.
Type I is a fracture through the tip of the odontoid process. In this configuration, the blood supply is maintained through the base of the odontoid process and through the attachment of the alar transverse ligaments. The mechanical stability of this fracture pattern is left intact. Symptomatic care and immobilization are the treatment of choice.
Type II, the most common type, is a fracture through the base of the odontoid process at its junction with the body of the axis. In this configuration, soft-tissue attachments to the fracture fragment cause distraction at the fracture site. Because the amount of cancellous bone available for opposition is limited, a high nonunion rate is expected, particularly if displacement is significant or the patient is older (>60 years). In this case, primary surgical treatment may be indicated. Anterior screw fixation of the odontoid process is now the treatment of choice for most type II odontoid fractures. Although it is technically demanding, it does allow for the maintenance of motion at C1-C2 (Figure 4–35).

![]() Figure 4–35. Imaging studies in a patient with a type II odontoid fracture nonunion. (A) Open-mouth radiographic view showing the fracture line at the base of the odontoid process. (B) Sagittal reconstruction using CT scanning to better delineate the fracture anatomy. (C) Radiograph taken after the patient underwent anterior placement of two odontoid screws under fluoroscopic control using a cannulated screw system.
Figure 4–35. Imaging studies in a patient with a type II odontoid fracture nonunion. (A) Open-mouth radiographic view showing the fracture line at the base of the odontoid process. (B) Sagittal reconstruction using CT scanning to better delineate the fracture anatomy. (C) Radiograph taken after the patient underwent anterior placement of two odontoid screws under fluoroscopic control using a cannulated screw system.
Type III is a fracture through the body of the axis. The blood supply is maintained through soft-tissue attachments, and abundant cancellous bone opposition at the fracture site facilitates a high rate of union. The treatment, therefore, is conservative, consisting of halo traction or halo-vest immobilization until bony union occurs. Although the rate of union is acceptable, there is a relatively high rate of mal-union that may limit the patient’s cervical rotation.
D. Hangman’s Fracture (Traumatic Spondylolisthesis of Vertebra C2)
Hangman’s fracture occurs when a fracture line passes through the neural arch of the axis. The anatomy of the axis is such that the superior facets are anterior and the inferior facets are posterior, thus concentrating stress through the neural arch. Because of the high ratio of spinal canal size to spinal cord size at this level, neurologic damage associated with hangman’s fracture should be unusual. However, in his postmortem studies, Bucholz reported that traumatic spondylolisthesis was second only to occipitoatlantal dislocations in cervical injuries leading to fatalities.
According to the scheme proposed by Levine and Rhyne, hangman’s fractures can be classified on the basis of anatomic factors and the presumed mechanism of injury. Treatment depends on the type of fracture. Imaging studies in a patient with hangman’s fracture are shown in Figure 4–36.

![]() Figure 4–36. Imaging studies in a patient who was in a motor vehicle accident and sustained a hangman’s fracture, or traumatic spondylolisthesis of C2. (A) Lateral radiographic view, which is largely unremarkable. (B) Sagittal reconstruction using CT scanning to better delineate the fracture site at the base of the posterior elements. The patient was treated nonoperatively.
Figure 4–36. Imaging studies in a patient who was in a motor vehicle accident and sustained a hangman’s fracture, or traumatic spondylolisthesis of C2. (A) Lateral radiographic view, which is largely unremarkable. (B) Sagittal reconstruction using CT scanning to better delineate the fracture site at the base of the posterior elements. The patient was treated nonoperatively.
Type I is typically caused by hyperextension with or without additional axial load. There is no angulation of the deformity, and the fracture fragments are separated by less than 3 mm. Treatment should consist of immobilization in a cervical collar or halo vest until union occurs, which is typically 12 weeks.
Type II is thought to be caused by hyperextension and axial load with a secondary flexion component leading to displacement of the fracture. Reduction of the anterior angulation in this type of fracture is necessary and typically obtained by traction therapy and then followed by placement of a halo vest until union occurs. An atypical type II hangman’s fracture is described. This fracture occurs through the posterior aspect of the vertebral body, potentially resulting in cord compromise as the anterior aspect of the vertebral body flexes forward. A higher likelihood of neurologic injury with this atypical pattern is seen, and halo-vest immobilization is recommended.
Type IIA has the same fracture pattern as type II but with a component of distraction that also occurred at the time of injury and led to disruption of the C2-C3 disk space, rendering this injury inherently unstable. Traction should be avoided in cases of type IIA fracture because it exacerbates the injury. Treatment should consist of immediate halo-vest application, with the patient’s head positioned in slight extension to afford a reduction.
Type III includes a fracture through the neural arch, a facet dislocation, and a disruption of the C2-C3 disk space that renders the injury highly unstable. Treatment generally consists of early closed reduction of the facet dislocation and application of a halo vest to maintain the reduction. If the reduction cannot be obtained in a closed fashion or cannot be maintained conservatively, treatment with open reduction of the dislocation and anterior or posterior fusion is indicated.
Anderson LD, D’Alonzo RT: Fractures of the odontoid process of the axis. J Bone Joint Surg Am 1974;56:1663. [PMID: 4434035]
Hsu WK, Anderson PA: Odontoid fractures: update on management. J Am Acad Orthop Surg 2010;18:383. [PMID: 20595131]
Huybregts JG, Jacobs WC, Vleggeert-Lankamp CL: The optimal treatment of type II and III odontoid fractures in the elderly: a systematic review. Eur Spine J 2013;22:1. [PMID: 22941218]
Ramieri A, Domenicucci M, Landi A, et al: Conservative treatment of neural arch fractures of the axis: computed tomography scan and x-ray study on consolidation time. World Neurosurg 2011;75:314. [PMID: 21492736]
INJURIES OF THE LOWER CERVICAL SPINE
As stated earlier, fractures and dislocations of the lower cervical spine have a greater frequency of catastrophic neurologic involvement because of the decreased ratio of spinal canal to spinal cord in the lower levels. Treatment of affected patients again relies on early recognition of the injury, recognition of inherent stability or instability of the injury pattern, and institution of appropriate definitive care.
In 1982, Allen and colleagues developed a classification system for closed indirect fractures and dislocations of the lower cervical spine. After reviewing numerous cases previously described by other authors as well as 165 of their own cases, they grouped the injuries into six categories, based on the position of the cervical spine at the time of impact and on the dominant mode of failure. The six categories were compressive flexion, vertical compression, distractive flexion, compressive extension, distractive extension, and lateral flexion. Of these, the distractive flexion injuries were the most common, followed by the compressive extension injuries and the compressive flexion injuries. Some of the categories were further divided into stages, as described next.
![]() Compressive Flexion Injury
Compressive Flexion Injury
There are five stages of compressive flexion injuries, which are labeled compression flexion stage (CFS) I through V (Figure 4–37). CFS I shows a slight blunting and rounding to the anterior superior vertebral margin, without any evidence of posterior ligamentous damage. CFS II shows some additional loss of height of the anterior vertebral body, again sparing the posterior elements. CFS III has an additional fracture line passing from the anterior surface of the vertebral body through to the inferior subchondral plate, with minimal displacement. CFS IV has less than 3 mm of displacement of the inferior posterior vertebral fragment into the neural canal. CFS V has severe displacement of the inferior posterior fragment into the canal, with widening of the spinous processes posteriorly, indicative of three-column disruption.

![]() Figure 4–37. Radiographs showing the five stages of compressive flexion injury. A shows compression flexion stage (CFS) I. B shows CFS II. C shows CFS III. D shows CFS IV. E shows CFS V. (Reproduced, with permission, from Allen BL, Ferguson RL, Lehmann TR, et al: A mechanistic classification of closed, indirect fractures and dislocations of the lower cervical spine. Spine (Phila Pa 1976)1982;7:1.)
Figure 4–37. Radiographs showing the five stages of compressive flexion injury. A shows compression flexion stage (CFS) I. B shows CFS II. C shows CFS III. D shows CFS IV. E shows CFS V. (Reproduced, with permission, from Allen BL, Ferguson RL, Lehmann TR, et al: A mechanistic classification of closed, indirect fractures and dislocations of the lower cervical spine. Spine (Phila Pa 1976)1982;7:1.)
Within the compressive flexion category are two types of fractures, more commonly referred to as the compression fracture and the teardrop fracture. Most compression fractures without disruption of the posterior elements are thought to be stable, so no surgical intervention is required. The more severe compression fracture injuries, however, can result in displacement of bone into the spinal canal, and if a neurologic injury is present, these require anterior decompression and stabilization. All patients should be carefully checked with flexion-extension views at the completion of their treatment to rule out any evidence of late instability.
![]() Vertical Compression Injury
Vertical Compression Injury
Vertical compression spinal (VCS) injuries occur secondary to axial loading and are divided into three stages. VCS I consists of an endplate central fracture with no evidence of ligamentous failure. VCS II is a fracture of both vertebral endplates, again with only minimal displacement. VCS III is the more commonly termed burst fracture with a spectrum of fragmentation of the vertebral body, with or without posterior element disruption.
The treatment for VCS injuries is typically nonoperative. Traction is applied to obtain and maintain alignment, and bony union is generally complete after 3 months of halo-vest immobilization. Flexion-extension views should be obtained at the completion of healing because a posterior ligamentous injury can result in late instability.
![]() Distractive Flexion Injury
Distractive Flexion Injury
The category of distractive flexion spinal (DFS) injury was the most common injury category reported by Allen and colleagues, and it includes both unilateral and bilateral facet subluxation and dislocation. There are four stages of DFS injury. DFS I, termed a flexion sprain, is characterized by subluxation of the facet joint, with possible interspinous process widening. This injury has subtle radiographic findings and may easily be missed during initial evaluation and therefore result in late symptomatic instability (Figure 4–38). DFS II is a unilateral facet dislocation, the diagnosis of which can be confirmed on plain radiographs. The lateral radiograph would reveal an anterior subluxation of one vertebra of approximately 25% of vertebral body width at the affected level. The facet itself may be perched or fully dislocated. DFS III is a bilateral facet dislocation with approximately 50% anterior dislocation at the affected level. DFS IV, which is also termed a floating vertebra, is a bilateral facet dislocation with displacement of a full vertebral width.

![]() Figure 4–38. Imaging studies in a patient with a distractive flexion injury of the cervical spine. (A) This lateral radiographic view demonstrates anterior subluxation of C5 on C6. (B) The follow-up radiograph shows progression of the subluxation. The patient was treated with a posterior spinal fusion of C5-C6.
Figure 4–38. Imaging studies in a patient with a distractive flexion injury of the cervical spine. (A) This lateral radiographic view demonstrates anterior subluxation of C5 on C6. (B) The follow-up radiograph shows progression of the subluxation. The patient was treated with a posterior spinal fusion of C5-C6.
Treatment of DFS injuries depends on the severity of the injury. Achievement of anatomic alignment and spinal stability yields the best results. Patients with unilateral facet dislocation should be treated with closed reduction in the acute phase, followed by immobilization. If closed reduction is not possible, open reduction and fusion are indicated (Figure 4–39). Bilateral facet dislocations are associated with a higher incidence of both neurologic injury and instability. Treatment consisting of closed reduction and immobilization is feasible, but because it results in a high percentage of late instability, which eventually requires posterior fusion, the use of early posterior fusion is indicated.

![]() Figure 4–39. Imaging studies in a man who fell from a height and suffered a C6-C7 fracture-dislocation with a perched facet but remained neurologically intact. (A) Lateral radiographic view demonstrating the fracture-dislocation at C6-C7. (B) MRI demonstrating the anterior subluxation of C6 on C7, with the intervertebral disk retropulsed behind the C6 vertebral body. The patient was treated with an anterior diskectomy, reduction, and fusion.
Figure 4–39. Imaging studies in a man who fell from a height and suffered a C6-C7 fracture-dislocation with a perched facet but remained neurologically intact. (A) Lateral radiographic view demonstrating the fracture-dislocation at C6-C7. (B) MRI demonstrating the anterior subluxation of C6 on C7, with the intervertebral disk retropulsed behind the C6 vertebral body. The patient was treated with an anterior diskectomy, reduction, and fusion.
Another fracture pattern that should be included in the discussion on flexion injuries is the clay shoveler’s fracture, which is a fracture of the spinous process, typically at level C6, C7, or T1. This is an avulsion injury that generally occurs in flexion by the counteractive forces of the muscular attachments. As an isolated injury, it is considered stable and usually treated nonoperatively.
![]() Compressive Extension Injury
Compressive Extension Injury
The category of compressive extension (CES) injury was the second most common injury category reported by Allen and colleagues. It is divided into five stages. CES I is a fracture of the vertebral arch unilaterally, with or without displacement, and CES II is a bilateral fracture. CES III and CES IV were not encountered in the series reported by Allen and colleagues but are theoretic interpolations between CES II and CES V. CES III is a bilateral fracture of the vertebral arch articular processes, lamina, or pedicle without vertebral displacement, whereas CES IV is the same fracture pattern but with moderate vertebral body displacement. Three patients in the Allen series had CES V injuries, which were bilateral vertebral arch fractures with 100% anterior displacement.
Treatment of CES injuries is related to the three-column theory. Stabilization with a posterior, anterior, or combined approach is indicated if there is significant disruption of the middle column or of two of the three columns.
![]() Distractive Extension Injury
Distractive Extension Injury
Distractive extension (DES) injuries are typically soft-tissue lesions and divided into two stages. DES I is a disruption of the anterior ligamentous complex or, rarely, a nondisplaced fracture of the vertebral body. Radiographs may appear entirely normal. One clue to the diagnosis is widening of the disk space, which is sometimes present. DES II is a disruption of the posterior soft-tissue complex, which can allow posterior displacement of the upper vertebral body into the spinal canal. This lesion is often reduced at the time of lateral radiographs and may show only subtle or no changes on routine radiographs. When neurologic involvement is present, it is most commonly a central cord syndrome, and provided that no coexisting compression lesions are present, some neurologic recovery is expected.
The DES injury is usually stable and does not require surgical intervention. Late flexion-extension views, however, are indicated to rule out any evidence of late instability.
![]() Lateral Flexion Injury
Lateral Flexion Injury
Allen and colleagues included the injuries of five patients in the category of lateral flexion (LFS) injury. This category is further divided into two stages. LFS I is an asymmetric compression fracture of the vertebral body and ipsilateral posterior arch, with no displacement in the coronal plane. LFS II has a similar fracture pattern but with displacement in the coronal plane, which suggests ligamentous disruption on the tension side of the injury. This mechanism can lead to brachial plexus injuries of varying degrees on the distracted side.
Because of the rarity of LFS injuries, treatment protocols are not well established. Surgical stabilization should be considered if late instability is expected or if there is a neurologic deficit.
![]() Treatment Decisions
Treatment Decisions
Ultimately the treating physician must decide on a treatment plan. The Allen classification, although quite useful to describe an injury, is a mechanistic system that is challenging to apply to the individual patient to assess operative indications. The decision whether to operate is based on a spectrum of spinal stability and neurologic compromise. A patient with a three-column injury, continued neurologic compression, and neurologic symptoms has a clear operative indication either through an anterior, posterior, or combined approach. A fully neurologically intact patient with a one-column injury generally does fine in a brace. Patients with injury patterns in between must be treated on a case-by-case basis.
CERVICAL STRAINS AND SPRAINS (WHIPLASH INJURY)
Cervical strains and sprains, which are commonly referred to as a whiplash injury when associated with motor vehicle accidents, can produce a protracted and confusing clinical picture. Pain is typically the one unifying feature, yet there may be numerous other complaints, including local tenderness, decreased range of motion, headaches that are typically occipital, blurred or double vision, dysphagia, hoarseness, jaw pain, difficulty with balance, and even vertigo. It is often difficult for the physician to correlate radiographic findings, diagnostic test results, and other objective findings with the subjective complaints of the patient. The constellation of symptoms is fairly uniform, however, and should certainly not be discounted, and many investigators propose an anatomic basis for the clinical complaints. McNabb proposed that paresthesias in the ulnar distribution may be secondary to spasm of the scalenus muscle, and certainly symptoms such as hoarseness and dysphagia can be related to retropharyngeal hematoma. The cervical zygapophysial joint and facet capsule are implicated as a source for chronic pain after whiplash injury.
Figure 4–40 presents an algorithm for management of cervical strain. Radiographs should be taken because the amount of neck trauma that the patient has sustained may be significant. Radiographic findings, however, may be subtle or entirely negative. Cervical lordosis may be reversed, indicating spasm. Subtle signs of instability may also be present, and these can be further delineated on flexion-extension views if symptoms persist. The prevertebral soft-tissue window should be within normal limits to rule out any prevertebral hematoma. MRI is not helpful in the acute setting to definitively diagnose whiplash injury.

![]() Figure 4–40. Algorithm for management of patients with cervical strain.
Figure 4–40. Algorithm for management of patients with cervical strain.
Once the stability of the spine is ensured, the care of the cervical sprain or whiplash injury should be symptomatic. Initial rest, bed rest if necessary, and soft collar immobilization are indicated, along with the use of anti-inflammatory medications. Early mobilization with progressive range of motion and weaning from external supports should be encouraged, however. Frequent reassurance is often necessary because the symptoms may be long lasting. Some patients with chronic unrelenting symptoms despite maximal conservative management may benefit from facet injections and rhizotomies.
Approximately 42% of patients have persistent symptoms beyond 1 year, with approximately a third having persistent symptoms beyond 2 years. Most patients who do improve do so within the first 2 months. Factors associated with a poor prognosis include the presence of occipital headaches, inter-scapular pain, or reversal of cervical lordosis. Women have a worse prognosis than men, and hyperextension injuries are thought to have a worse prognosis than hyperflexion injuries.
Anderson SE, Boesch C, Zimmermann H, et al: Are there cervical spine findings at MR imaging that are specific to acute symptomatic whiplash injury? A prospective controlled study with four experienced blinded readers. Radiology 2012;262:567. [PMID: 22187629]
McNabb I: The “whiplash syndrome.” Orthop Clin North Am 1971;2:389. [PMID: 5150390]