Alan Saunder, Ken Farrell
7.1 Introduction
History — analysis of abdominal pain
Abdominal pain is the most common presenting symptom of surgical diseases of the gastrointestinal tract. It is usually categorised by its site and mode of onset, for example, chronic epigastric pain or acute right iliac fossa pain. Analysis of abdominal pain lends itself very well to the principles of problem solving. The patient is encouraged to describe the pain in their own terms. Specific questions are interpolated to clarify and elaborate particular points and to detect the most significant symptoms. Abdominal pain is often accompanied by associated symptoms that establish a pattern and assist the clinician in making a diagnosis. Precise localisation of the pain is the most useful step in its characterisation. Although visceral pain is less precisely localised than somatic pain, in most cases the pain can be localised to an abdominal area or region: upper, lower, right, left, central or generalised. Of almost equal importance to site is the mode of onset of the pain — whether acute or chronic. For pain in each abdominal region, a limited and manageable list of common causes exists — some specific to the area and some not. With these causes in mind a careful history is taken and physical examination performed focusing on the identification of the most likely cause. A general systems review is always important in the analysis of abdominal pain, particularly in relation to the gynaecological and urinary systems. Disorders of other systems (such as diabetes) sometimes present solely with abdominal pain or in themselves need attention and may have a bearing upon prognosis.
Location and migration
Whether abdominal pain is generalised or localised to an abdominal segment is the most important piece of information to obtain first. Migration of abdominal pain also often assists in diagnosis (Fig 7.1). Central or generalised abdominal pain is a common initial symptom of many diseases (appendicitis, bowel obstruction, visceral perforation). Persisting severe generalised abdominal pain suggests generalised peritonitis. Epigastric or right upper quadrant pain extending in a band around the abdomen suggests a biliary origin (‘colic’). Migration of central pain to the right iliac fossa over the course of several hours indicates appendicitis. This is an example of visceral pain evolving into a somatic pattern as local peritonitis develops with pathological progression of appendicitis. Another example of this phenomenon is migration of pain from the left iliac fossa to the whole abdomen may be associated with initial sigmoid diverticulitis with subsequent perforation and the development of generalised peritonitis.
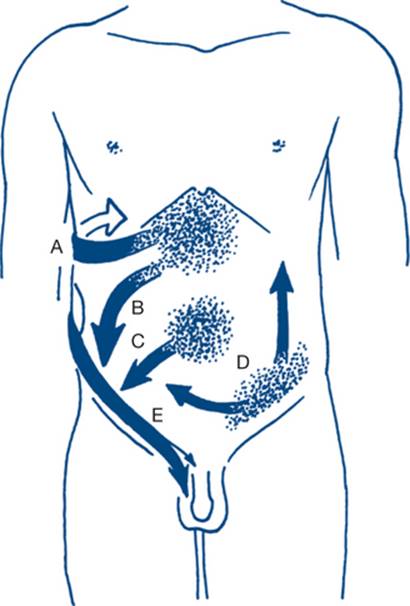
Figure 7.1 Migration of pain in common abdominal conditions
A: biliary pain — migration to below the top of the right scapula (‘wing sign’); B: perforated ulcer — down the right paracolic gutter; C: acute appendicitis — to the right iliac fossa; D: perforated diverticulitis — over the abdomen starting from the left iliac fossa; E: renal colic — from loin to groin
Onset and duration
The time, pattern and character of onset and the duration of symptoms are elicited. Acute abdominal pain that persists for six or more hours suggests the presence of a surgical abdomen; so does a history of awakening at night with the pain, especially if pain precedes other symptoms such as vomiting or diarrhoea. A sudden and dramatic onset of pain suggests rupture of a previously intact structure (e.g. duodenum, colonic diverticulum, splenic capsule, aortic aneurysm). A gradual onset, progressively worsening over hours or days, correlates with inflammation. The onset after excessive alcohol intake suggests pancreatitis. A history of abdominal injury may be of importance. A latent period can exist between the time of trauma and presentation, as is seen with delayed rupture of the spleen. A latent period can also be seen in other acute abdominal conditions such as perforated peptic ulcer, intussusception, large bowel obstruction, gallstone ileus and intestinal infarction. Sometimes such a latent period gives the illusion of resolution of the problem and can delay or obscure diagnosis.
Type and intensity
The character of the pain is subjective. Patients often relate pain to previous or imagined occurrences (‘stabbed by a knife’, ‘struck by lightning’, ‘burning’). It is important to establish whether the pain has been constant and unremitting since onset or is periodic. If periodic, is it a true colic? Colic causes a spasmodic, gripping abdominal pain that returns every few minutes with a crescendo rise and fall in intensity often described by women as similar to labour pains. Colic associated with an urge to defaecate or pass flatus and with abdominal borborygmi is pathognomonic of intestinal colic due to obstruction or irritation. The colicky pain of obstruction is usually more severe than is found with gastroenteritis. Severe colicky pain that becomes constant suggests ischaemic bowel. Constant pain often varies in severity during the course of the illness, but careful questioning will distinguish this from a true colic. The need for narcotics to control upper abdominal pain is suggestive of renal or biliary pain. Severe pain persisting without relief requires hospital admission.
Severity is also subjective and best assessed by the effect of pain on the patient, the response to the suggestion that an operation may be necessary and the necessity for and whether relief from analgesics already administered has occurred. Inability to sleep because of pain suggests a surgical problem.
Severe unremitting pain may be due to localised peritonitis because of appendicitis, cholecystitis or diverticulitis, or due to widespread peritonitis from visceral rupture or mesenteric ischemia. Biliary pain tends to plateau, after a gradual onset; it is continuous but fluctuates in severity. Acute renal pain is a very severe and constant pain. Neither is a true colic.
Radiation
Radiation or referral is extension of the pain to another site while the initial pain persists. Occasionally the site of referral may be dominant and simulate disease in another organ. Biliary pain may be felt primarily in the back or in the precordium, simulating angina. Pain from a penetrating duodenal ulcer (or from pancreatitis) and from carcinoma of the pancreas often pass directly through to the back. Renal pain radiates from the flank (loin) to groin, testis or thigh. Conversely, the pain of testicular torsion may sometimes be referred to the hypogastric region. Segmental nerve root pain is referred to the appropriate dermatome. Shoulder pain can be referred from the mediastinum or result from diaphragmatic irritation from an abdominal cause — for example, blood or intestinal contents beneath the diaphragm.
Associated symptoms
Nausea, vomiting and anorexia often precede pain of non-surgical origin: profuse vomiting as a main feature suggests upper small bowel obstruction; persistent anorexia following pain favours an organic cause; persistent vomiting from any cause can occasionally give haematemesis from a Mallory-Weiss tear in the lower oesophagus.
Nausea, diarrhoea and vomiting preceding colicky pain is a feature of gastroenteritis; diarrhoea may be due to pelvic appendicitis or abscess.
An association with menstrual disorders often indicates gynaecological disease. Abnormal menstrual loss before pain suggests abortion, or after pain suggests ectopic pregnancy. A missed period implies the possibility of a complication of pregnancy. Intermenstrual pain and bleeding may be associated with ovarian problems and vaginal discharge with salpingitis. Urinary symptoms such as frequency, scalding or haematuria suggest pyelonephritis, cystitis or stone. Chills and rigors hint at significant infective conditions such as pyelonephritis, salpingitis or viral gastroenteritis.
Offsetting factors
The factors that relieve pain may clarify the diagnosis. Relief of pain by food and alkali suggests peptic ulceration. Certain movements can relieve pain; the pain of pancreatitis may be relieved by sitting forward. A hot-water bottle is often used for biliary pain and nonsteroidal anti-inflammatory agents commonly relieve musculoskeletal pain. Relief of pain by vomiting indicates possible ulcer disease and relief of pain by defaecation or passage of flatus suggests gastroenteritis or functional bowel disease. Biliary and renal pain often require analgesic injection for relief.
Factors that worsen pain, such as movement, may indicate that the pain is musculoskeletal in origin. However, the pain of peritonitis is worsened by abdominal movement, deep breathing or coughing, as is pain from diaphragmatic irritation. If food worsens the pain, conditions such as benign or malignant gastric ulceration need consideration.
Past history
Previous episodes of pain, previous abdominal operations, a history of previous trauma, past and current drug treatment and alcohol intake are commonly relevant to the assessment of the current presentation of abdominal pain.
Physical examination
A general inspection of the patient should include a careful assessment of alertness, demeanour and hygiene. This is an ideal opportunity to identify features of chronic liver disease such as the presence of jaundice, pallor, bruising or purpura, pigmentation and loss of body hair (Fig 7.2). A record of the patient’s nutritional state should note whether body habitus is normal, obesity levels (or morbid obesity), whether there is loss of body mass and cachexia or evidence of fluid and electrolyte depletion. The preferred posture of the patient should be noted (e.g. sitting forward in acute pancreatitis, or supine and still in peritonitis.)
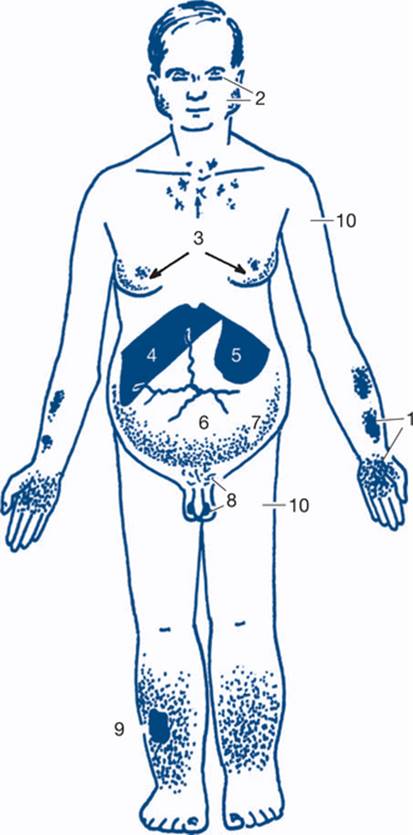
Figure 7.2 Physical signs of liver disease
1: palmar erythema and ecchymoses; 2: xanthelasma, parotid enlargement; alcoholic facies; 3: spider naevi and gynaecomastic; 4: hepatomegaly; 5: splenomegaly (the main sign of portal hypertension); 6: dilated veins on the abdominal wall; 7: ascites; 8: testicular atrophy and loss of body hair; 9:pigmentation, ulceration and oedema; 10: proximal muscle wasting
In the emergency and urgent situations, the abdomen is assessed prior to the periphery. However, in the elective ambulatory situation, the periphery is often examined initially to gain both vital information and the confidence of the patient before examining the abdomen.
Examination of the periphery
The hands are examined. Nail changes include clubbing, which may be found in a number of gastrointestinal conditions, chronic liver disease, inflammatory bowel disease and malabsorption. Other changes such as leuconychia, indicating malnutrition and hypoproteinaemia, or koilonychia, suggesting iron deficiency, may be present. Palmar erythema, spider naevi and Dupuytren’s disease (palmar nodules, bands, contractures, pits and sinuses and knuckle pads) suggest alcoholic liver disease (Fig 7.2). Asterixis is a flapping tremor best seen with the hands in extension and is found in patients with portal systemic encephalopathy.
The head and neck are then examined for abnormal pigmentation, spider naevi, xanthelasma of the eyelids, scleral jaundice or pallor and parotid enlargement. The neck is examined for cervical lymphadenopathy, especially left supraclavicular nodal enlargement (Troisier’s sign) as a sign of metastatic node involvement from carcinoma of the stomach.
The mouth is then examined for the smell of fetor hepaticus suggesting encephalopathy, mucosal thickening and ulceration, atrophy or erythema, gingivitis and angular stomatitis. A ketotic breath and coated tongue is suggestive of appendicitis or related conditions. Angular stomatitis (‘perlèche’) is seen frequently in patients with iron deficiency and malnutrition. Glossitis occurs in patients with iron, folate and vitamin B12 deficiency. Mouth ulceration may be present. The hydration status (i.e. skin turgor and moistness of mucous membranes) should be assessed at this time, especially in the acute setting.
Abdominal examination
The patient lies supine with arms by the side and with groin and hernial orifices exposed. The examination should be performed in a good light. The abdomen is inspected for abnormal contour, either distension or scaphoid appearance, for generalised or localised distension, for scars, discolouration, pigmentation or striae, for distended veins radiating from the umbilicus (caput medusae) and for visible pulsations or peristalsis. The patient is then asked to breathe deeply in and out through the mouth, to see if the contour changes or if a mass moves or becomes more obvious and to see if respiratory movement is painful. The patient is asked to cough and scars and the hernial orifices are watched to detect an expansile impulse. The patient is asked to lift the legs or shoulders from the bed and similar observations are made for an impulse. With this manoeuvre a visible mass, if subcutaneous, will become more prominent, an intra-abdominal mass less so.
Figures 7.3 and 7.4 depict anterior and posterior surface markings of the abdominal viscera.

Figure 7.3 Anterior surface markings of upper abdominal viscera

Figure 7.4 Posterior surface markings of abdominal viscera
A: lung; B: pleura; C: kidney; D: pancreas; E: spleen
After inspection, the patient is asked if there is any tenderness and, if there is, its site. Palpation is started gently with the hand flat, at a site away from the site of maximal tenderness and then in all quadrants (Fig 7.5).

Figure 7.5 Region of the abdomen
A: epigastrium; B: right hypochondrium; C: lumbar region; D: periumbilical region; E: right iliac fossa; F: hypogastrium
Then the abdomen is palpated more deeply using the flexor surface of the fingers — the use of two hands often helps to define masses (Fig 7.6).
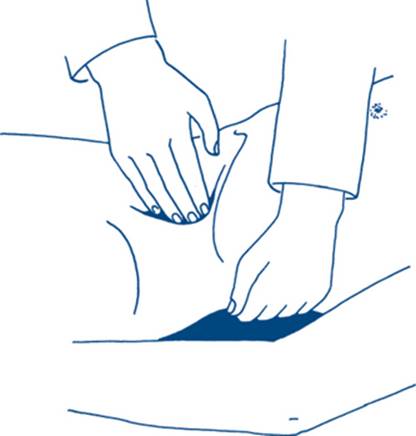
Figure 7.6 Method of palpating the spleen
The hernial orifices and genitalia are examined. It is important to distinguish normally palpable structures from abnormal masses (Fig 7.7).

Figure 7.7 Palpable normal abdominal structures
1: xiphoid process; 2: liver; 3: edge of rectus abdominis; 4: lower pole of right kidney; 5: caecum; 6: aorta; 7: ala of sacrum; 8: pregnant uterus; 9: full bladder; 10: sigmoid colon; 11: faecal loading
Routine examination for ascites is essential in the distended abdomen, by eliciting shifting dullness or a fluid thrill. Shifting dullness is shown to be present by rotating the patient about the long axis and demonstrating a change in the ‘Plimsoll line’ or transition line of dullness to percussion (Fig 7.8). Fluid thrill is detected by flicking the side of the abdomen and palpating for a transmitted impulse on the other side. Transmission of the impulse via the abdominal wall is blocked by another hand placed on the midabdomen.

Figure 7.8 Shifting dullness
Area 1 is resonant when supine and dull in lateral position. Area 2 is dull when supine and resonant in lateral position.
Based on Burkitt, 2007
The following physical signs are sought: tenderness or resistance on palpation (voluntary or involuntary guarding); rigidity (extreme guarding); and rebound tenderness (best assessed by gentle percussion). A full description of any detected mass is often most usefully recorded as a simple schematic sketch in the medical record. The edge of a mass may be best delineated by deep palpation during inspiration (liver and spleen) or by percussion (Table 7.1). The site and depth of a mass are very important diagnostic features. Other important features include shape and consistency and whether the mass is mobile or fixed. Faecal masses are indentable.
Table 7.1 Typical physical characteristics of enlarged liver, spleen and kidney

Anorectal examination
Anorectal examination is usually performed on patients in the left lateral position if there are appropriate symptoms requiring this to assess the clinical problem. It is not required as part of all abdominal examinations. The spine and hips are fully flexed; the knees are flexed to a little less than 90° to obtain better access for sigmoidoscopy. The equipment required is a good light, examination gloves, lubricating jelly and paper tissues to clean the anus after examination. Proctoscopy (anoscopy) and sigmoidoscopy (rectoscopy) should be part of a routine office or outpatient examination by appropriately trained practitioners. The proctoscope permits visualisation of the anal canal up to 10 cm; the standard rigid sigmoidoscope up to 30 cm. The flexible sigmoidoscope is a specialist instrument, visualising up to 50 cm from the anal verge. Long biopsy forceps are also necessary should sampling for histology be necessary. Neither form of sigmoidoscopy is part of a standard non-specialist examination. Bowel preparation is not usually necessary for routine anorectal examination.
Initial inspection may reveal prolapsed haemorrhoids, perianal haematomas, external openings of fistulas and the dry or moist skin changes of pruritus. Examination of the anus during straining will be necessary to reveal prolapsing haemorrhoids or rectal prolapse. Anal fissures are normally not initially obvious on inspection, as they are located above the anal verge. Most fissures are seen in the midline posteriorly. Laterally placed fissures suggest an association with inflammatory bowel disease. Gentle retraction of the anal verge will usually expose the lower edge of a posterior fissure. Rectal examination (Fig 7.10) and proctoscopy may not be possible in such instances because of painful anal spasm. Palpation commences using the pulp of the index finger introduced slowly into the anorectum. The finger will usually reach 10 cm with a little pressure on the perineum or with the assistance of bimanual compression of the lower abdomen. As the finger is withdrawn the whole circumference of the rectum is examined. The indurated, elevated and ulcerated lesion of a carcinoma is characteristic. The capacity of the rectum should be noted. Masses outside the rectum in the pouch of Douglas may be palpable anteriorly. Anteriorly in the male the normal prostate is a firm rubbery bilobed structure about 3 cm in diameter. A shallow central sulcus may be palpated and the mucosa over the prostate should be freely mobile. In the male it is difficult to define anatomical structures above the base of the prostate. The prostate feels larger on examination if performed while the patient has a full bladder. In the female the cervix of the uterus or a vaginal tampon may be palpable anteriorly and should not be mistaken for a mobile extrarectal tumour. Faecal pellets in an intrapelvic sigmoid colon can simulate abnormal extrarectal lumps but faeces are regular and indentable. The glove should be examined for blood after completion of the digital examination.

Figure 7.10 Rectal examination
Digital detection of a carcinoma of the rectum at about 10 cm from the anal verge. The characteristics on palpation are induration, elevation and ulceration. Blood may be seen on the glove after completion of the examination.

Figure 7.9 Anatomy of the anorectum
The anal canal extends from the anorectal ring, which is formed by the puborectalis, down to the anal verge, a distance of about 3 cm. 1: puborectalis muscle 2: inferior haemorrhoidal plexus or cushion; 3: anal crypt; 4: dentate line; 5: interhaemorrhoidal groove; 6: anal verge; 7:external haemorrhoidal plexus; 8: external haemorrhoidal artery; 9: internal sphincter; 10: external sphincter
Proctoscopy is the best method of diagnosing haemorrhoids, as these lesions are impalpable on digital examination unless thrombosed. Proctoscopy also provides a means of close viewing of tumours in the lower third of the rectum for biopsy and for the office treatment of haemorrhoids.
7.2 ‘Acute abdomen’ (acute abdominal surgical emergency)
The ‘acute abdomen’ is difficult to define but vital to recognise. The essential task is to recognise that an acute abdominal emergency exists and that surgery, when necessary, must not be delayed. Patients present with various combinations of pain, collapse, shock and peritonitis, but not all of these clinical features are present in each patient. Severe pain is the most striking symptom and is often generalised. Less severe forms of acute abdominal pain where more time exists to make a diagnosis are characterised by their localisation and are discussed under separate headings (acute upper abdominal pain, acute right iliac fossa pain). The requirement for the patient with an acute abdomen is usually to ‘operate and see’ rather than ‘wait and see’. Some will prove on exploration to have non-surgical conditions, but this price is necessary in the interests of survival.
The timing of urgent surgery is also important; there must be adequate resuscitation but no undue delay before surgery. Early evaluation of the patient for signs of hypovolaemia and fluid depletion is therefore essential. The timing of surgery depends upon the response to resuscitation. As resuscitation begins, plans are made for any investigations required prior to surgery.
Causes
1. Acute appendicitis with perforation
2. Severe acute (haemorrhagic) pancreatitis
3. Perforated peptic ulcer
4. Diverticulitis with perforation
5. Strangulating intestinal obstruction
6. Vascular catastrophes
7. Ruptured ectopic pregnancy
8. Gastroenteritis
9. Less common causes
History and physical examination
The most vital concern is to identify those patients with a diagnosis requiring abdominal operation from those whose treatment is non-surgical while measures to resuscitate the patient are in progress. Pain usually precedes anorexia and vomiting in surgical conditions but often follows them in non-surgical conditions. Efforts should be taken to make and record a presumptive specific diagnosis in each case. A careful history and examination, and the grouping of symptoms into recognised clinical syndromes and patterns, enable a correct diagnosis to be made in a high proportion of cases.
The symptoms and signs associated with abdominal pain need careful analysis. These include nausea and vomiting, abdominal distension, change in bowel habit, peritonitis, pyrexia and prostration (shock).
Commonly, a typical association of symptoms and signs gives an early clue to the diagnosis. For example:
• Severe continuous abdominal pain with prostration (shock) suggests acute pancreatitis or vascular catastrophes such as ruptured aneurysm, dissecting aneurysm, mesenteric infarction, strangulated bowel obstruction and ruptured ectopic pregnancy.
• Abdominal pain of explosive onset with general peritonitis suggests that a visceral perforation is likely — for example, perforated peptic ulcer, perforated diverticulitis, perforated appendicitis and perforated gall bladder.
• Abdominal colic, vomiting, distension and constipation indicate the presence of the classic symptom complex of small bowel obstruction. If signs of peritonitis are also present, the bowel is probably strangulated. The hernial orifices should always be checked.
• The sequence of constipation, distension, pain and vomiting is suggestive of large bowel obstruction.
The integration of salient features of the history with examination findings will often disclose the clinical pattern or characteristics of a particular surgical condition or, at the very least, allow the formulation of a well-ordered differential diagnosis. Therefore, particular attention must be given to abdominal examination; the care with which the examination is performed often clinches the diagnosis. Systematic examination detects associated medical problems and evaluates circulatory status. The presence of fever suggests infection or aseptic inflammation and narrows down the diagnostic possibilities, as do signs of shock early in the illness.
A routine for physical examination in assessing the acute abdomen is as follows:
• Inspect for distension, scars and hernias.
• Marked pain on coughing or deep breathing suggests that a surgical condition is present. Examination of hernial orifices and external genitalia is obligatory.
• Abdominal examination checks for muscle spasm — either voluntary or involuntary guarding (true spasm). True muscle spasm indicates the presence of peritonitis. Guarding, when most severe, is called rigidity — when extreme, rigidity feels board-like.
• Test for localised tenderness by one-finger palpation; rebound tenderness is best elicited by gentle percussion. Tenderness without guarding is a feature of gastroenteritis. Percussion may also demonstrate loss of liver dullness because of overlying air from a perforated viscus.
• Carefully palpate for the presence of a mass.
• Listen for bowel sounds. A silent abdomen suggests peritonitis; exaggerated peristaltic rushes suggest bowel obstruction.
• Rectal examination is essential in all patients with acute abdomen.
1. Acute appendicitis with perforation (see also acute right iliac fossa pain)
Perforation, with presentation as an acute abdomen, is seen most frequently in the young, the old and in patients with diabetes. In many series, perforation at the time of appendicectomy has occurred in almost half those patients under 10 years and over 50 years. It is unusual for the acutely inflamed appendix to perforate within the first 12 hours. Pain in appendicitis is often initially central and diffuse, followed by a shift to the right iliac fossa within a few hours. Pain is deep-seated, continuous and gradually increases in intensity. Nausea and vomiting are common, but vomiting is rarely pronounced or persistent and is rarely the first symptom. Diarrhoea is also rarely the first symptom (Table 7.2). Its presence suggests pelvic appendicitis. The development of perforation is accompanied by more severe generalised abdominal pain and higher fever.
Table 7.2 Comparison of clinical features of perforated pelvic appendicitis and gastroenteritis
|
Perforated pelvic appendicitis |
Gastroenteritis |
|
|
Progress |
Steady deterioration |
Usually nonprogressive |
|
Pattern |
Insidious onset of pain; later development of diarrhoea and tenesmus |
Sudden onset of anorexia, nausea, vomiting and diarrhoea before pain |
|
Associated upper respiratory tract infection (URTI) |
No |
URTI common with myalgia, photophobia and headache |
|
Movement |
Exacerbates pain |
Writhing with spasms of pain |
|
Abdominal signs |
Often minimal early in the disease |
Diffuse tenderness |
|
PR examination |
Tenderness and fullness in pouch of Douglas |
Normal rectal examination |
|
WCC |
Leucocytosis |
No leucocytosis |
Signs. With general peritonitis, there is diffuse abdominal tenderness with guarding, which is often maximal in the right iliac fossa. The degree of guarding depends upon the rapidity of onset of the peritonitis. Bowel sounds often persist for some time. If untreated the abdomen becomes silent and progressively distends, with diminution of tenderness as exudate accumulates. High fever, toxicity and eventually septic shock develop. A tender local mass may be felt in the right iliac fossa suggesting abscess formation. In such a case the patient is usually toxic and febrile due to irritation of the rectum. In pelvic appendicitis, abdominal signs are often delayed or less marked but rectal examination may reveal the presence of pelvic tenderness and peritonitis.
2. Severe acute (haemorrhagic) pancreatitis
A small but important proportion of patients with severe acute pancreatitis present with an acute abdomen. The remaining patients with less severe disease present with localised acute upper abdominal pain (Ch 7.3). The most severe haemorrhagic form of the disease is associated with collapse and shock.
The onset of symptoms is occasionally explosive and can mimic a visceral perforation. Usually, however, increasingly severe pain develops over a period of several hours and spreads from the epigastrium throughout the whole abdomen and through to the back. Progressive dyspnoea and prostration (shock) are common. Persistent vomiting is often a feature of the illness (Table 7.3). Change of posture may aggravate or relieve the pain. Fever is variable.
Table 7.3 Comparison of perforated duodenal ulcer with acute pancreatitis
|
Perforated ulcer |
Acute pancreatitis |
|
|
Age and sex |
Middle-aged males |
Younger males |
|
Pain and peritonitis |
Severe pain and board-like ridigity |
Severe pain, less marked guarding, marked release tenderness |
|
Vomiting |
Repeated vomiting uncommon |
Vomiting common and persistent |
|
Dyspnoea and cyanosis |
Uncommon |
Common |
|
Abdominal distension |
Scaphoid abdomen |
Mild distension common |
|
Mass |
Uncommon |
Epigastric mass common |
Signs. Signs of generalised peritonitis are present. There is often a ‘doughy’ feel to the abdomen and the signs of peritonitis and muscle rigidity are less than one would expect from the severity of the pain and the degree of prostration (an important point of differentiation from perforated ulcer). Disparity between the signs of peritonitis and the severity of pain is sometimes a feature of bowel ischaemia.
Prostration and shock, dyspnoea, ventilatory insufficiency and cyanosis indicate a severe attack with poorer prognosis. Extraperitoneal fluid and blood extravasation may be indicated by staining in the flanks (Grey-Turner’s sign) or around the umbilicus (Cullen’s sign).These signs are typically delayed in onset and may not be present at the initial presentation. The common predisposing causes of severe pancreatitis are gallstones and alcohol. In alcoholic pancreatitis the patient may be agitated and confused, indicating imminent delirium tremens.
3. Perforated peptic ulcer
The history is usually typical, with a sudden onset of very severe abdominal pain spreading from the epigastrium to the whole abdomen; radiation to the back and shoulder is less common. The patient is anxious, grey, sweaty and still. Movement and coughing exacerbate the pain. Breathing is shallow and is inhibited by pain. Many patients have a past history of peptic ulcer.
Signs. The clinical signs are usually unmistakable. Generalised peritonitis is present, with board-like rigidity in the epigastrium and elsewhere. The term is very appropriate and, once felt, is difficult to confuse with anything else. The abdomen is silent and loss of liver dullness to percussion may be elicited. Pulse, temperature and blood pressure are all likely to be normal. Shock does not develop until a late stage. The delayed ‘stage of reaction’ or of ‘masked’ peritonitis takes several hours to develop. This should not confuse the careful observer, as rigidity and ileus persist. More difficulty is found with a localised duodenal leak when the history is less typical and the abdominal signs are restricted to local tenderness in the upper abdomen (Ch 7.3).
4. Diverticulitis with perforation
Acute abdomen from diverticulitis results usually from the rupture of a pericolic abscess. Sometimes faecal peritonitis results from sudden rupture of an uninflamed diverticulum. Thus the onset may be without warning or after a recent history of acute diverticulitis. In many cases, a long history of bowel upset and episodic lower abdominal distension and discomfort precedes the acute attack. Very severe widespread pain originating from the left iliac fossa with prostration suggests perforated diverticulitis.
Signs. General peritonitis is present with maximum tenderness and guarding in the left iliac fossa. A tender mass due to a peridiverticular phlegmon or abscess may be palpable. Fever and toxicity are common associated features: they suggest a septic cause for the acute abdomen and help to distinguish diverticulitis from perforated ulcer.
5. Strangulating intestinal obstruction
Simple intestinal obstruction is usually readily diagnosed on clinical assessment and a plain X-ray film of the abdomen and is discussed in more detail in Chapter 7.9. Strangulating obstruction is a complication of simple obstruction; it can be notoriously difficult to diagnose and is lethal without prompt surgery. In simple small bowel obstruction the abdominal pain is colicky and periumbilical. If the pain becomes continuous, strangulation should be suspected.
Obstruction with ischaemia or strangulation is also seen in the large bowel. In an important minority of patients with malignant large bowel obstruction, the ileocaecal valve is competent and prevents regurgitation into the small bowel and decompression of the large bowel so that a ‘closed loop’ obstruction develops. The main danger is then progressive distension and perforation of the caecum. Such patients may present with right-sided pain and tenderness with distension and will require urgent surgical treatment to prevent caecal perforation and the consequent faecal peritonitis. Sigmoid or caecal volvulus is of more abrupt onset with severe constant pain due to early ischaemia and rapid massive distension and imminent perforation.
In malignant large bowel obstruction, a history of altered bowel habit often precedes complete constipation for faeces and flatus. This is often due to left-sided colonic stenotic lesions. Pain is often late and is felt in the lower abdomen or back. Nausea is common, but vomiting is generally a late feature.
Signs. Although certain clinical features can create a suspicion that strangulation is present (Box 7.1), no clinical or laboratory findings exist that exclude with certainty the possibility of strangulation. In small bowel obstruction the bowel sounds are hyperactive. Signs of local peritonitis and a palpable mass strongly suggest strangulation. A strangulated external hernia (femoral, inguinal, umbilical or incisional) will be tense, tender, irreducible and will have lost the cough impulse. Always search for a hernia, even in the patient who has had previous abdominal operations. Signs of interstitial fluid and blood volume depletion will be present when bowel obstruction has persisted for one or more days. Early shock or poor response to resuscitation suggests the presence of strangulation.
Box 7.1
Clinical features suggesting bowel strangulation
Continuous severe pain
Early shock or fever
Poor response to resuscitation
Local peritonitis, with or without a tender mass
In large bowel obstruction, distension is particularly marked in the flanks and in the right iliac fossa. Signs of fluid depletion are late. The distension is predominantly gaseous and signs of shock or peritonitis suggest that perforation has occurred. Careful sigmoidoscopy may reveal the cause of the obstruction.
6. Vascular catastrophes
Ruptured aortic aneurysm usually presents in elderly men with the sudden onset of central abdominal pain radiating to the back and/or groin, associated with collapse, pallor and severe shock. There may be previous knowledge of a pulsatile abdominal swelling. The patient with ruptured aortic aneurysm is pale and sweating with collapsed veins, the signs of haemorrhagic shock. The majority of patients have ‘contained retroperitoneal haematoma’ rather than a free intraperitoneal rupture. A poorly defined pulsatile upper abdominal swelling is present in the epigastrium to the left of the midline. Femoral pulses may be weak or absent, but this is not diagnostic. Ruptured visceral artery aneurysms should be considered as well in patients with hypotension and severe sudden upper abdominal pain.
Aortic dissection presents with sudden severe tearing pain felt in the interscapular region or lower chest, radiating to the back and abdomen. Signs of distal vascular insufficiency in the limbs with absent or diminished peripheral pulses may be present, depending on the anatomy of the dissection, with possible anuria and with mild general abdominal tenderness. The patient is not always elderly in contrast to most patients with ruptured abdominal aortic aneurysms.
Acute mesenteric ischemia is typified by the presence of generalised severe, continuous abdominal pain of sudden with surprisingly few abdominal signs initially. The development of peritonism and mental confusion, associated with gross prostration and often dark rectal bleeding is usually associated with a grave outcome. Often evidence of an underlying embolic focus exists — such as atrial fibrillation or a recent myocardial infarct. The triad of pain, rectal bleeding and prostration in an elderly patient with fibrillation is suggestive of the diagnosis.
7. Ruptured ectopic pregnancy
Acute lower abdominal and shoulder-tip pain associated with pallor, prostration or fainting in a woman of child-bearing age is the characteristic presentation. Gastrointestinal symptoms are unusual. Rupture usually occurs between six and eight weeks’ gestation, so a missed period is common but not invariable. Death of the embryo causes the pain to be followed by dark, withdrawal vaginal bleeding in most but not all cases. This contrasts with the acute, bright, copious bleeding of abortion that often precedes pain. Acute abdomen from a ruptured ectopic pregnancy usually results from the rupture of isthmial implantations — in contrast to the problem of acute pelvic pain seen with a leak from an ampullary implantation.
Signs. The patient is pale and shocked, with a tender silent lower abdomen that may be mildly distended. There are usually no obvious signs of pregnancy such as breast enlargement but vaginal examination shows a soft cervix that is exquisitely tender to palpation. A tender pelvic mass may be present in one or other fornix.
8. Gastroenteritis
This non-surgical cause of an acute abdomen presents a problem in diagnosis, particularly in general practice. The main difficulty is the exclusion of appendicitis (particularly in infants), pelvic appendicitis and intestinal obstruction (in adults). In gastroenteritis, nausea, vomiting or diarrhoea precede the pain in contrast to the sequence of events in appendicitis, for example. The pain is less severe (though it may be colicky) than with obstruction. Early high fever is more suggestive of a viral illness, as are the presence of diffuse myalgias, photophobia and headache.
Signs. Abdominal tenderness tends to be poorly localised and diffuse. Abdominal guarding is not a feature of the illness and bowel sounds are hyperactive. Early high fever is a feature. As well as the common viral aetiology, enteritis may be caused by bacteria such as Yersinia, Salmonella or Campylobacter.
9. Less common causes
Primary peritonitis is now mainly found in association with alcoholic liver disease and ascites and can lead to a ‘negative’ laparotomy or laparoscopy. Foreign body perforation is not rare and may be difficult to locate at laparotomy. Periodic peritonitis (familial Mediterranean fever), gonococcal peritonitis, tuberculous peritonitis and granulomatous peritonitis, are other causes to be considered in the differential diagnosis of the acute abdomen. Complications of inflammatory bowel disease need to be considered.
The pain of renal infection can mimic an acute abdomen. Associated urinary frequency, dysuria and pyuria usually suggest the diagnosis. Rupture of an inferior epigastric vessel may be associated or follow treatment with oral anticoagulants. Rectus sheath haematoma may be spontaneous or precipitated by minor trauma and can mimic an acute abdomen or appendicitis. Similarly, ‘spontaneous’ retroperitoneal haematoma can occur in anticoagulated patients and present with lateralised abdominal or flank pain.
Medical conditions do not usually simulate an acute abdomen to the degree where surgical intervention is necessary. Basal pneumonia, however, can present difficulties. In these patients respiratory signs may be minimal, although respiratory distress is often the clue that suggests the diagnosis. Myocardial infarction is rarely a cause of an acute abdomen, but the initial pain may be in the epigastric region. Acute painful hepatic engorgement secondary to acute right ventricular failure may give rise to diagnostic confusion such as occurs in large pulmonary embolism. Acute porphyria, precipitated by barbiturates and diabetic ketosis with abdominal pain and vomiting, are occasionally problems in diagnosis, especially when associated with collapse and a confused mental state.
Diagnostic plan
Always aim to have a differential diagnosis. The list of probable to possible diagnoses dictates the investigation and management plan. Selected judiciously ordered investigations can confirm or exclude specific diagnoses, thereby allowing a logical management plan to be devised.
Urine examination
All patients should have a dipstick test on admission. Haematuria may accompany renal colic. Proteinuria, pus cells and organisms on microscopy suggest urinary infection. Urinary polymorphs may also accompany retrocaecal or pelvic appendicitis. Bile suggests acute cholecystitis or pancreatitis. Urinary glucose testing will detect the undiagnosed diabetic. The additional presence of acetone makes diabetic ketosis a possible cause of abdominal pain. Urine osmolality may be useful in assessing fluid depletion. A specific urine β-HCG should be done in all female patients of reproductive age.
Haematological examination
Haemoglobin and white cell count are often useful. Anaemia suggests that peritonitis may be due to blood in the peritoneal cavity or that there is underlying malignant or benign ulceration of the gastrointestinal tract. Leucocytosis (greater than 11 × 10∧9 per litre) suggests sepsis or inflammation. If very high (over 20) an abscess is likely. High white cell count suggests also the possibility of strangulated bowel and makes a diagnosis of perforated ulcer, pancreatitis and gastroenteritis less likely. Leucopenia may occur with severe sepsis. Blood culture is indicated for any patient with a suspected septic cause.
An elevated haemoglobin or haematocrit level is indicative of plasma volume depletion. Diminished levels indicate pre-existing anaemia or blood loss.
Biochemistry
Few tests are specifically helpful in making a diagnosis but several are useful in monitoring the treatment of metabolic and ventilatory disturbances.
Serum lipase levels over 100 iu/L are very suggestive of pancreatitis. Lipase is much more specific for pancreatitis than serum amylase where moderate elevations up to 1000 iu/L may occur with intestinal obstruction, strangulation or perforation. Serum electrolyte, urea and creatinine levels help assess fluid and electrolyte requirements. Urea elevation is commonly due to isotonic extracellular fluid (‘saline’) depletion.
Arterial blood gas analysis is important in monitoring shocked patients, those with ventilatory problems or with pancreatitis. Serum lactate levels are often disproportionately high in mesenteric vascular occlusion. Liver function tests rarely influence early operative management but are useful baselines in jaundiced patients and in alcoholics.
Imaging techniques
These include plain radiology, ultrasound, contrast and computed tomography (CT scan) and are very often valuable. Erect chest X-ray (Fig 7.11), together with erect and supine films of the abdomen, are indicated in nearly all patients. (For patients too moribund to undergo an erect chest X-ray, a lateral decubitus film may be requested.) These X-rays may show primary chest pathology (pneumonia) or basal changes secondary to a subdiaphragmaticcondition such as pancreatitis. Free gas under the diaphragm indicates a perforated viscus, usually a perforated ulcer or perforated diverticulitis. A grossly dilated stomach may be seen in patients in diabetic coma, falsely suggesting the possibility of a surgical condition.

Figure 7.11 Chest X-ray with gas under the diaphragm
Based on Burkitt, 2007
If small bowel obstruction is suspected, erect and supine views show significant distension of the small bowel with gas fluid levels and a ladder pattern (Fig 7.12). In large bowel obstruction, the colon is distended down to the site of obstruction and small bowel dilatation may coexist. If the ileocaecal valve is incompetent specific causes such as sigmoid or caecal volvulus may show localised distended large bowel loops. Gastroenteritis can be associated with small gas–fluid levels with moderate intestinal distension. Air swallowing in association with severe pain and recent injury may cause confusion. The absence of free gas does not exclude perforated viscus, nor does the absence of fluid levels in the bowel exclude strangulation. Free gas is seen in only about two-thirds of cases of perforated peptic ulcer. In appendicitis, distended bowel with fluid levels on plain X-ray often indicates a localised ileus in the right iliac fossa. Pancreatic calcification or lithiasis or a sentinel small bowel loop in the region of the pancreas or a colonic ‘cut-off’ sign may be seen in pancreatitis. More commonly, generalised ileus is present, with evidence of ascites. Distended, gas-filled, small and large bowel loops with fluid levels are present and large bowel gas extends to the rectum. Mesenteric infarction causes a diffuse small bowel ileus. In ruptured aortic aneurysm a rim of calcium may be seen in the aneurysm, particularly in the lateral decubitus films. Radio-opaque gallstones may be seen in cholecystitis (20%); urinary calculi are usually visible (80%).

Figure 7.12 Films of small bowel obstruction
A: supine abdominal X-ray with dilated small bowel loops; B: erect abdominal X-ray with multiple air fluid levels (arrowed); C: typical ’ladder’ pattern (arrowed) on supine abdominal X-ray
From Feldman, 2006
Contrast-enhanced X-rays may be required in special instances to diagnose bowel leakage. A gastrografin meal or enema will not damage the peritoneum and can be very useful, especially if combined with a CT scan at the same time. A limited contrast enema is often used to confirm the diagnosis and the site of a large bowel obstruction prior to operation.
Ultrasound is the investigation of first choice in the diagnosis of gallstones and hepatobiliary conditions.
Aortic aneurysms are readily diagnosed with ultrasound and CT angiography may be useful in diagnosis of ruptured aortic aneurysm or dissecting aneurysm if the diagnosis is not clear, the patient is stable and renal function known to be satisfactory. When a focus of infection is suspected, CT scan with oral contrast can both facilitate the diagnosis and direct interventional or surgical treatment. Ascites may be seen in pancreatitis — aspiration and amylase level on the fluid may be diagnostic.
Catheter angiography is rarely required, apart from the context of mesenteric vascular insufficiency.
Peritoneal tap and lavage
Peritoneal tap or lavage is much less commonly used to assess the acute abdomen than in assessing blunt abdominal trauma. Abdominal distension and previous incisions are contraindications. False negative results can mislead and most surgeons prefer to explore the abdomen by laparoscopy or laparotomy if a surgical cause cannot be excluded by other means.
Treatment plan
Recognition of the presence of the acute abdomen immediately raises the question of surgical treatment. Patients usually can be rapidly categorised at this stage of assessment into three groups. Concurrent resuscitation and fluid replacement is essential to the treatment of all three groups.
1. Early operation is necessary for a clear surgical diagnosis of such conditions as acute appendicitis with perforation, perforated peptic ulcer, intestinal obstruction with strangulation, mesenteric infarction, ruptured aortic aneurysm and ectopic pregnancy.
2. Early operation is necessary because surgically treatable causes cannot be excluded: an operative diagnosis is mandatory. Persisting peritonitis without a clear-cut cause requires early operative diagnosis.
Investigations in the above two groups may aid diagnosis, but they should not affect the clinical decision to operate and must not delay surgery. If the diagnosis is not clear but the patient has an acute abdomen requiring surgical intervention, the next step is often to proceed to diagnostic laparoscopy, which then allows the specific diagnosis (e.g. appendicitis) to be made and appendicectomy to be completed at the same time.
3. The diagnosis is uncertain, so early treatment should be conservative and expectant. A period of careful observation in hospital is required. Investigations are often diagnostic and may need to be repeated.
Principles of conservative treatment comprise control of concurrent illness, resuscitation and control of sepsis.
Shock and fluid depletion are treated as expeditiously as possible. An intravenous line and urinary catheter are first essentials. Monitoring of right atrial or pulmonary artery pressure may be required in severely ill patients with septic or cardiogenic shock or pancreatitis. Nasogastric suction is commenced if obstruction or perforation of the bowel is suspected. Antibiotics are given for specific problems. The abdomen should always be examined under anaesthesia by the operating surgeon prior to laparotomy/laparoscopy. This may reveal a mass and assist the choice of an incision for best exposure.
Local control of disease by correctly timed surgery is basic to survival in most patients presenting with an acute abdomen.
1. Acute appendicitis with perforation
Appropriate antibiotics are given perioperatively prior to surgery. The appendix is exposed and removed through a skin crease right iliac fossa incision if not done via laparoscopy. Thorough peritoneal lavage is performed in patients with general peritonitis. Antibiotics may be continued intravenously. (This may also be managed laparoscopically in selected cases by those with appropriate expertise, although the incidence of postoperative intra-abdominal abscess may be higher.)
If a localised appendiceal abscess is found, the appendix is removed and the abscess is drained. The wound may be left open for later delayed closure if there is contamination of the wound.
2. Acute severe (haemorrhagic) pancreatitis
Exploratory operation is occasionally unavoidable because another surgical condition (e.g. perforated ulcer, bowel obstruction, appendicitis) cannot be excluded (Box 7.2). Diagnosis is confirmed by finding an inflammatory pancreatic mass with fat necrosis and ascites (‘beef-tea’ fluid). A peritoneal dialysis catheter can be left in situ for subsequent lavage.
Box 7.2
Acute pancreatitis: diagnostic plan
Suggestive history
Serum lipase
Ultrasound to detect gallstones
Laparoscopy or laparotomy if the diagnosis remains in doubt
Treatment of pancreatitis otherwise is initially conservative:
• Monitor intravenous fluid replacement and nasogastric suction. Large volumes may be required to replace blood, plasma and electrolyte deficits. Shock is monitored by vital signs and central venous or pulmonary arterial pressure measurements. Nasogastric suction is only indicated if there is significant ileus. Otherwise there is no evidence to suggest any benefit in the management of acute pancreatitis.
• Monitor arterial blood gas values. Hypoxia is common, requiring oxygen treatment and sometimes intermittent positive pressure ventilation. Lactic acidosis may be severe.
• Monitor serum calcium. Hypocalcaemia may require supplementation and indicates a poorer prognosis.
• Monitor sepsis. Pancreatic and extrapancreatic sepsis are common and a major cause of mortality (Table 7.4). Antibiotics may be given to prevent or manage local or symptomatic sepsis.
• Monitor local complications. Pancreatic mass or phelgmon may cause temporary gastric holdup. A pancreatic pseudocyst will cause more prolonged gastric retention and may require drainage if unresolved within six weeks. Infected pancreatic necrosis and abscess are serious complications requiring surgical debridement and drainage (Fig 7.13).
Table 7.4 Indicators of severity of acute pancreatitis (Glasgow system)
|
Factor |
Level |
|
Age |
>55 years |
|
Leucocytosis |
>15 × 109/ L |
|
Blood urea concentration |
>16 mmol/L (no response to fluid administration) |
|
Blood glucose concentration |
>10 mmol/L in the non-diabetic patient |
|
Serum albumin concentration |
<32 g/L |
|
Serum calcium concentration |
20 mmol/L |
|
Lactate dehydrogenase |
600 IU/L |
|
Aspartate aminotransferase |
>100 IU/L |
|
Arterial Po2 |
<60 mmHg (8.0 kPa) |

Figure 7.13 Common complications of acute pancreatitis
A: pseudocyst of the pancreas in the lesser sac secondary to a leak from the main pancreatic duct; B: necrosis, haemorrhage and sepsis extends to the retroperitoneum. 1: inferior surface of the right lobe of the liver; 2: gall bladder; 3: stomach; 4: greater omentum; 5: transvese colon; 6: third part of the duodenum
3. Perforated peptic ulcer
Nasogastric suction is accompanied by early operation. On most occasions, and particularly in the poor risk case (Box 7.3), the ulcer is covered with an omental plug and peritoneal washout is performed (Fig 7.14). The patient is commenced on H. pylori eradication postoperatively and risk factors addressed. For the unusual combination of perforation with serious bleeding, or for very large ulcers, partial gastrectomy may be necessary. A perforated gastric ulcer may be a carcinoma and should be biopsied and preferably treated by definitive partial gastrectomy.
Box 7.3
Significant risk factors in perforated duodenal ulcer
Major concurrent medical illness
Preoperative shock
Delayed treatment until more than 24 hours from perforation

Figure 7.14 Simple closure of a perforated anterior duodenal ulcer (Roscoe-Graham method)
Sutures are passed deep to the ulcer and tied over the omentum.
4. Perforated diverticulitis
Perioperative antibiotics are given. The effected segment of colon (usually sigmoid) is resected. A restorative anastomosis is usually not performed in the presence of sepsis. The two ends of the bowel may be exteriorised as a double-barrel colostomy (Paul-Mikulicz) or, more commonly, the proximal end is brought out and the distal end is closed and left within the pelvis. This is Hartmann’s procedure (Fig 7.15). It is essential that the septic focus of diverticulitis is excised and not simply drained — drainage alone will not control infection. Later, a rectosigmoid anastomosis is performed electively when the patient is considered fit for the procedure.

Figure 7.15 Hartmann’s procedure
Resection of the site of complicated diverticular disease is usually necessary if infection is to be controlled in patients with perforation. After excising the diseased segment, the risk of immediate bowel anastomosis in continuity is avoided by an end sigmoid colostomy (1) with closure of the rectal stump (2).
5. Strangulating intestinal obstruction
An operation becomes urgent when strangulation is suspected. Preoperative resuscitation aims to restore at least the blood volume prior to exploration but must not delay surgery.
At operation for small bowel obstruction the cause is relieved and necrotic or doubtful bowel is resected, with primary anastomosis.
Decompression of the proximal large bowel is indicated in large bowel obstruction because of the danger of progressive distension and perforation of the caecum. At operation, the obstructing lesion is usually removed, but it is almost always unsafe and unwise to perform primary anastomosis of obstructed large bowel.
6. Vascular catastrophes
Urgent operation is necessary for ruptured aortic aneurysm. Bleeding is controlled by proximal and distal aortic clamping. The aneurysm is replaced by an in situ prosthetic graft. Perioperative intensive care is essential.
In aortic dissection with deterioration, despite control of hypertension, an operation is required, often using endovascular techniques to limit further dissection and end-organ compromise.
For mesenteric vascular occlusion, nonviable bowel is resected at operation. If a large proportion of the small bowel is lost, it is prudent to exteriorise the ends of bowel as enterostomies. Alternatively, a planned second-look laparotomy may be performed after 24 hours to check the viability of the remaining small bowel. Anastomosis is deferred until the patient’s condition improves. If the entire small bowel is infarcted from the duodenojejunal flexure, excision and permanent intravenous feeding can be considered in younger patients without gross associated pathology.
7. Ruptured ectopic pregnancy
Immediate surgery to control bleeding may be necessary in the shocked patient. The affected fallopian tube may be excised. The patient is at risk of a further ectopic implantation in future pregnancies, but sometimes repair of the tube is possible after removal of the ectopic tissue.
8. Gastroenteritis
Appropriate medical treatment is required. A major difficulty is differentiating non-surgical causes of abdominal pain (e.g. nonspecific abdominal pain, mesenteric adenitis, gastroenteritis, urinary infection) from appendicitis. When this differentiation cannot be made with certainty and pain and tenderness persist, a diagnostic laparoscopy may be performed. Most surgeons accept the necessity of removing a small number of normal appendices (∼10%) to avoid missing early appendicitis. Faecal culture may reveal a specific organism and thus specific treatment may be instituted. Organisms, such as Salmonella, Yersiniaand Campylobacter, may require appropriate antibiotic therapy and consultation with infectious diseases physicians.
The remaining miscellany of causes of abdominal pain will require appropriate treatment.
7.3 Acute upper abdominal pain
The patients in this group are not as seriously ill as those with an ‘acute abdomen’ for which indications for early laparotomy are usually present. Various diseases are common to both forms of presentation. These diseases present in a less severe form in patients presenting with acute upper abdominal pain. More time exists for a diagnosis to be made without the urgent need for early surgery. Most causes can therefore be managed by confirmatory investigations during initially conservative non-surgical treatment.
Causes
1. Acute gastritis and nonulcer dyspepsia
2. Acute exacerbation of duodenal ulcer
3. Biliary ‘colic’ and acute cholecystitis
4. Acute (oedematous) pancreatitis
5. Less common causes
History
1. Acute gastritis and nonulcer dyspepsia
Acute gastritis may be due to bacterial or viral infection or to duodenal reflux gastritis. These patients may be admitted to hospital with burning epigastric pain of sudden onset, often with a provisional diagnosis of perforated ulcer. In the past ‘abdominal pain of unknown cause’ was often the eventual diagnosis in patients who recovered without surgery. Nowadays, subsequent endoscopy reveals that many of those patients have gastritis secondary to bacterial infection. Viral and bacterial gastroenteritis may have acute epigastric pain and vomiting as the main symptoms; diarrhoea is less prominent. Pain may also be triggered by substances toxic to the gastric mucosa, such as alcohol and analgesic agents. On occasions, after cholecystectomy or partial gastrectomy, acute epigastric pain is secondary to bile reflux gastritis.
2. Acute exacerbation of duodenal ulcer
Complicated duodenal ulcer can present with acute upper abdominal pain due to an acute exacerbation or to a localised perforation. In many instances gastric risk factors, such as alcohol, nonsteroidal anti-inflammatory drugs (NSAIDS) and bacterial or bile gastritis, have triggered the relapse. About three-quarters of the patients have a past history of ulcer. Sealing of a small perforation is more common in patients who have had past abdominal surgery, but a scar should also suggest that other causes for the pain are possible, such as adhesive small-bowel obstruction or postgastrectomy bile gastritis. Details of any previous operative procedure performed should be sought from previous records as the patient’s account is frequently inaccurate or incomplete.
In about a third of cases with an acute abdomen from perforated ulcer, a prodromal period exists during which a small leak initially localises before free perforation occurs. This period can last for several hours and should be borne in mind when seeing a patient in the emergency department with acute epigastric pain that has partially resolved.
The presence of H. pylori should be considered as a causative agent for gastroduodenal ulcer disease, the diagnosis confirmed and eradication therapy instituted.
3. Biliary ‘colic’ and acute cholecystitis
Most episodes of biliary ‘colic’ last for no more than a few hours but recur intermittently. Many of these patients see a doctor electively with the problem of chronic episodic epigastric pain. Biliary colic is distinct from an episode of acute cholecystitis where the patient has severe persisting acute pain, will call a doctor or present at hospital. Narcotic analgesics are usually necessary for pain relief once the diagnosis of acute biliary pain has been made. Biliary pain has an abrupt onset, is felt in the epigastrium or the right hypochondrium and can fluctuate in severity. The pain is often referred to the back. Most patients have a past history of attacks of biliary colic; an attack of persisting acute cholecystitis as the first clinical evidence of gallstones is less common.
When rapid resolution of symptoms occurs, the patient can be investigated electively with ultrasound for gallstones. Continued pain for more than 12 hours suggests acute cholecystitis. Bacteria can be cultured from the bile in only 70% of patients with established cholecystitis. Admission to hospital is necessary for persistent pain and for associated systemic effects. In 95% of patients, acute cholecystitis results from persistent obstruction of the cystic duct by a gallstone impacted in Hartmann’s pouch. The natural history of acute cholecystitis depends upon whether the obstruction is relieved, whether there is secondary bacterial infection, the age of the patient and the presence of concurrent medical illness (particularly diabetes mellitus). Most attacks will resolve spontaneously in hospital; some progress to abscess formation and occasionally to free perforation with generalised peritonitis (Fig 7.16). Jaundice occurs in only 10% of patients and suggests stone in the bile duct, acalculous cholecystitis or gangrenous cholecystitis. Jaundice with high fever suggests ascending cholangitis.

Figure 7.16 Pathology and natural history of gallstones
A: asymptomatic gallstones; B: biliary pain (chronic cholecysitis); B1:obstructive jaundice; B2: pancreatitis secondary to small stones; B3: ascending cholangitis. C: acute cholecystitis; C1: mucocele of gall bladder; C2: perforated empyema — with pericholecystic abscess; C3: free perforation. D: chronic cholecystitis; D1: cancer of gall bladder; D2: cholecysto-duodenal fistula and gallstone ileus
Acute acalculous cholecystitis usually occurs in the postoperative, posttraumatic or severely ill hypotensive patient. Gangrene may proceed to perforation, which may be fatal. Physical signs may be minimal. The diagnosis is notoriously difficult to make (or to think of) in the seriously ill patient with multiple coexisting problems.
4. Acute (oedematous) pancreatitis
In about 80% of cases acute pancreatitis is of modest severity and presents as localised acute upper abdominal pain, without systemic effects. The prognosis in these cases is good and a fatal outcome unusual. The remaining 20% of patients present with the more severe acute haemorrhagic pancreatitis with necrosis. Collapse and shock occur and the presentation is that of an acute abdomen (Ch 7.2) — a potentially lethal situation with a mortality rate of 30%.
Attacks of pancreatitis frequently follow an alcoholic binge or large meal. The pain is abrupt in onset, severe and persistent and often radiates through to the back. Persistent vomiting is a feature. The majority of cases of mild or moderate oedematous pancreatitis settle down rapidly in hospital.
Acute oedematous pancreatitis is often secondary to gallstones. A past history of biliary pain may be present. With pancreatitis, the pain tends to be central rather than right-sided and the illness more severe and prostrating. Most attacks resolve, as small stones pass through the sphincter of Oddi and are passed unnoticed in the faeces. Stones in the bile duct are found in only about 10% of patients when investigations are delayed for more than a week after the attack of pancreatitis. The earlier the duct is investigated (by surgery or endoscopic retrograde cholangiopancreatography: ERCP), the more patients are found who have stones in the bile duct. The pathology of gallstone pancreatitis is characterised by the presence of small stones in a functioning gall bladder with a patent, large cystic duct and stones can be found in the faeces, if diligently sought.
Examination
Gastritis and duodenal ulcer
In acute gastritis moderate epigastric tenderness is present. In patients with sealed perforation, muscle tenderness and guarding are present to a greater extent than in patients with acute oedematous pancreatitis. Unless a periduodenal abscess has developed, patients with an acute exacerbation of duodenal ulcer have no systemic signs of toxicity.
Cholecystitis
Right upper quadrant tenderness and guarding are present. Tenderness beneath the right costal margin on inspiration is characteristic (Murphy’s sign). In about a third of patients the inflamed gall bladder is palpable. A palpable mass is found more frequently after the first 24 hours; before this, the abdominal tenderness often masks the presence of the gall bladder mass (Fig 7.17). Mild fever is common, as are moderate tachycardia and leucocytosis. In contrast, patients with biliary colic have no significant findings on examination.

Figure 7.17 Acute cholecystitis
An acutely tender, palpable gall bladder is virtually diagnostic of acute cholecystitis. The gall bladder enlarges laterally rather than towards the umbilicus.
High fever and chills are uncommon and suggest either the presence of empyema of the gall bladder, in which case the diagnosis is incorrect, or that common duct stone with cholangitis is a complication.
Acute pancreatitis
The signs found depend upon the severity of the disease. Despite severe pain, examination of the abdomen usually reveals that guarding is not as marked as would be expected in a patient with comparable pain due to perforated ulcer. An epigastric abdominal mass may be found. A mass is due to inflammatory oedema of the pancreas, rather than to the necrotic mass with slough that occurs with more severe forms of acute haemorrhagic pancreatitis. Peripheral circulatory and respiratory failures are uncommon — these, also, are features of the more severe form of acute pancreatitis (Ch 7.2). The temperature is usually normal or slightly elevated; leucocytosis, if present, is of moderate degree. Clinical evidence of secondary basal atelectasis and pleural effusion on the left side of the chest are common. In most cases with mild disease the patient’s clinical condition rapidly settles with treatment, with complete resolution of physical signs.
Diagnostic plan
Early endoscopy is contraindicated in the investigation of acute upper abdominal pain because of the danger of converting a localised perforation from duodenal ulcer into a general peritonitis.
Haematological and biochemical examination
In acute pancreatitis the serum lipase usually rises to above 100 iu/L within six hours of the onset of an acute episode and remains elevated for about two days. The serum lipase is usually a reliable test for diagnosing acute pancreatitis. A mild elevation can occur in other diseases causing acute abdominal pain, particularly those associated with ischaemia or perforation of bowel. The serum lipase may be normal in the hyperlipidaemic patient or because the level has returned to normal before the estimation was carried out.
A transitory rise in serum bilirubin is common in patients with gallstone pancreatitis. Urinary bilirubin is often present in patients with acute cholecystitis or pancreatitis and is a helpful diagnostic clue. Daily review of electrolytes, calcium, glucose and renal function is essential.
The full blood count is normal in most patients presenting with acute upper abdominal pain. Marked leucocytosis suggests the presence of sepsis — as is found with empyema of the gall bladder, paraduodenal abscess or a high-retrocaecal appendicitis with abscess. Mild or moderate leucocytosis can occur in uncomplicated acute pancreatitis or acute cholecystitis. Hypochromic anaemia suggests blood loss from peptic ulcer or a colonic malignancy.
Radiology: plain erect film of chest and abdomen
In cases of perforated ulcer, where the leak of gastric contents is confined to the upper abdomen, free gas under the diaphragm on an erect chest X-ray may not be found. In pancreatitis, X-ray changes of left basal pulmonary atelectasis and effusion are often seen. Plain film of the abdomen may also reveal opaque gallstones or pancreatic calcification. About 20% of gallstones are radio-opaque. An ileus pattern on the plain film, localised to the right upper quadrant, suggests cholecystitis or pancreatitis. A localised small bowel loop (‘sentinel loop’) or large bowel ileus (‘colonic cutoff’ sign) over the pancreas may occur.
Ultrasound and CT scanning
Ultrasound is the best method of detecting gallstones in patients with acute upper abdominal pain. It is sometimes difficult to detect gallstones by ultrasound in patients with pancreatitis because the distended bowel gas of ileus makes ultrasound examination more difficult. Ultrasound is also a valuable method of detecting and following a pancreatic mass and the evolution of pseudocysts, especially in the thin patient. Ultrasound can show the increased thickness and oedema of the gall bladder wall that is a feature of acute cholecystitis and thus can be of considerable diagnostic value in patients with acalculous cholecystitis. Ultrasound is less effective in the obese patient with excess gastrointestinal gas. Occasionally in such patients, a CT scan is a better examination — CT imaging of the gall bladder is not as satisfactory as ultrasound, but CT imaging with contrast of the pancreas can be highly discriminatory in patients with acute pancreatitis.
Radionuclide excretion scan
When a policy of early surgery for acute cholecystitis is being followed, a firm diagnosis of the presence of gallstones, and preferably of lack of function of the gall bladder, is very valuable. The diagnosis on clinical grounds will be wrong in 10–20% of cases. Ultrasound and radionuclide (99Tcm CHIDA) scan make accurate diagnosis possible in most cases. HIDA scan of the gall bladder, if available, is a useful method of diagnosing the presence of acute cholecystitis, by demonstrating nonfunction of the gall bladder.
Gastrografin swallow and meal
An early, water-soluble contrast study of the upper gastrointestinal tract is indicated in the patient suspected of having a localised perforation of an ulcer. Indications include a past history of ulcer in a patient with persistent pain and moderate tenderness with a negative plain X-ray. A localised pool of contrast connected to and lying outside the duodenum confirms the diagnosis of a sealed perforation.
Late endoscopy and ERCP
Endoscopy should be delayed in the undiagnosed case until resolution of symptoms occurs. In patients with acute gastritis or nonulcer dyspepsia, biopsy and silver stain of antral mucosa may reveal the presence of H. pylori. ERCP may be indicated in the patient with postcholecystectomy pain if a stone in the bile duct is suspected, particularly in patients with dilated extrahepatic ducts and altered liver function.
Treatment plan
1. Acute gastritis and non-ulcer dyspepsia
These patients are observed in hospital until they settle down, at which time gastroscopy is performed to exclude ulcer. The later treatment of non-ulcer dyspepsia includes measures used in the treatment of functional bowel disease and a course of eradication treatment of helicobacter if appropriate.
2. Acute exacerbation of duodenal ulcer
For exacerbation of ulcer, conservative treatment is indicated and the symptoms resolve in a few days. Local perforation is an indication for elective surgery, especially in patients with a long history, previous complications or associated stenosis. If free perforation occurs in hospital (and a diagnosis is made promptly), laparoscopy and/or laparotomy is performed. The ulcer can be oversewn with an omental plug.
3. Biliary ‘colic’ and acute cholecystitis
Intravenous fluids and antibiotics are commenced.
With acute cholecystitis, pain and tenderness often resolve within 48 hours in about two-thirds of cases. In the past it was recommended that these cases have an interval cholecystectomy after two to three months. Clinical trials have shown that early operation before discharge from hospital can be performed safely, providing the surgeon is experienced and satisfactory control of any concurrent disease can be achieved. Most patients are therefore best operated upon at the first convenient opportunity.
Ideally, operative cholangiography should be performed routinely. The incidence of a clinically undetected stone in the bile duct is about 5%. Bile duct stones may be removed during surgery using specialised retrieval devices via the cystic duct or directly from the common bile duct. Sometimes it is safer to leave duct stone removal for a postoperative ERCP/sphincterotomy.
4. Acute (oedematous) pancreatitis
Most patients with oedematous pancreatitis settle down rapidly in hospital with conservative treatment. The principles of treatment are:
• control of pain, using pethidine rather than morphine, the latter causing spasm of the sphincter of Oddi
• prevention of renal and respiratory insufficiency by careful replacement of fluids, chest physiotherapy and intranasal oxygen
• nasogastric suction may be used if there is symptomatic ileus (i.e. protracted vomiting)
• antibiotics may be used mainly to treat respiratory infection, which these patients are susceptible to. More intensive management, such as physiotherapy and ventilatory support, is necessary in severely ill patients.
In gallstone pancreatitis, current opinion favours performing a cholecystectomy during the initial hospital stay so that the risk of another attack is avoided. In aged patients, endoscopic sphincterotomy may be a safer alternative initial form of treatment.
5. Less common causes
These may be gastrointestinal or nongastrointestinal. Pain from preicteric hepatitis can be confused with acute cholecystitis. Prodromal symptoms of nausea and anorexia and signs of tender hepatomegaly with disordered liver function tests help make the diagnosis. Pain from hydronephrosis is often felt in the epigatrium. Occasionally, silent myocardial infarction, with acute right ventricular failure causing painful hepatic engorgement, can present with acute upper abdominal pain.
Basal pneumonia may present, especially in the young, with upper abdominal pain and guarding. An important sign of abdominal pain due to respiratory infection is associated respiratory distress. Chest signs may be minimal, consisting only of occasional basal crepitations.
Nerve root pain (T6–10) can cause acute upper abdominal pain. In most cases the clue to the diagnosis is radiation of the pain from the back. Osteoarthritis with spur formation and shingles are the most common causes of root pain.
Splenic infarction may present with acute left hypochondrial pain. Splenic infarcts occur in association with bacterial endocarditis, lymphoma and in patients with splenomegaly secondary to alcoholic cirrhosis of the liver. Occasionally, blood from the pelvis may produce signs primarily in the upper abdomen with left shoulder tip pain, as may delayed rupture of the spleen occurring some days after injury.
Sometimes aortic aneurysm presents with acute epigastric or left hypochondrial pain when rupture is imminent. More commonly the problem presents as an acute abdomen (Ch 7.2). An interval of several hours may exist between the first episode of self-limited bleeding and later retroperitoneal rupture.
Appendicitis in a high retrocaecal position beneath the liver can closely mimic acute cholecystitis, as may obstructing carcinoma of the right transverse colon with a mass. Obstructing carcinomas of the transverse colon can present with colicky upper abdominal pain or lesser discomfort that is made worse or triggered by the ingestion of food. These patients often have an iron deficiency anaemia — an important clue that can suggest the diagnosis.
Occasionally, patients with alcoholic liver disease develop acute epigastric pain, probably because of low-grade infected ascites or primary peritonitis.
7.4 Acute right iliac fossa pain
Localisation of acute pain to the right iliac fossa is a very common clinical problem. The age and sex of the patient determine the possible causes considerably. Most young men with acute right iliac fossa pain will prove to have appendicitis. In young women gynaecological disorders often also present with acute pain in the right iliac fossa.
The first question is whether the patient has acute appendicitis. The classic clinical presentation of appendicitis can be expected in about half the patients. Atypical cases of appendicitis often mimic other abdominal disorders; too long a delay before surgery can lead to serious consequences. Appendicectomy is therefore performed unless acute appendicitis can be excluded beyond reasonable doubt. The proportion of cases in which a normal appendix is removed should not, in general, exceed 10%.
Characteristically, appendicitis has a progressive course, described by J B Murphy as the ‘march of events’:
1. pain (usually starting as umbilical or epigastric and radiating to the right iliac fossa)
2. nausea or vomiting
3. localised right iliac fossa tenderness
4. fever (mild)
5. leucocytosis.
Most patients with acute appendicitis present at stage 3 and acute right iliac fossa tenderness and guarding is the key to the diagnosis. Lack of localised tenderness and variations in the march of events suggest alternative diagnoses for acute right iliac fossa pain.
Causes
1. Acute appendicitis
2. Acute mesenteric adenitis and acute Meckel’s diverticulitis
3. Nonspecific right iliac fossa pain
4. Gynaecological disorders
5. Acute urinary tract stone, infection or hydronephrosis
6. Less common causes
History and physical examination
1. Acute appendicitis
Acute appendicitis can occur in infants and the old but most cases are found during adolescence and early adult life. Appendicitis at the two extremes of age is more difficult to diagnose. The symptoms usually begin with pain that is central, symmetrical and often colicky. As with many other viscera, the early pain of appendicitis is often referred to the somatic dermatome of the midgut — around the umbilicus. The patient is often awakened in the early morning by vague abdominal discomfort progressing to central or epigastric cramping pain, followed some time later by increasing nausea, anorexia and indigestion. Neither vomiting nor diarrhoea is an early symptom. An illness starting with nausea or diarrhoea, progressing to abdominal pain, suggests that the diagnosis is gastroenteritis, not appendicitis (Table 7.2). Early rigor and high fever are also uncommon and favour another diagnosis, such as urinary tract infection. Absence of fever must not preclude the diagnosis of appendicitis. Fever is absent throughout the course of the disease in a significant number of patients with appendicitis. Within several hours the pain shifts and becomes localised in the right iliac fossa; the patient can often point to the site of tenderness with one finger. Movement and coughing increase the abdominal discomfort.
Examination, when pain is localised to the right iliac fossa, usually reveals sharply localised tenderness, accompanied by mild or moderate involuntary guarding. Coughing often elicits local pain (‘cough tenderness’) and is a helpful sign. Tenderness is usually localised over the appendix: the site may be at McBurney’s point but is often at some other site in the right iliac fossa. Rebound tenderness on percussion may be found in the same area. The patient often looks flushed and unwell, with a rapid pulse, furred tongue and abdominal breath. From this stage the inexorable progression of untreated appendicitis usually continues, although resolution can occur at any stage with milder attacks. Many patients are severely ill and toxic (because of peritonitis or abscess) by 72 hours from the onset of the illness.
Tenderness on rectal examination is very significant in the diagnosis of pelvic appendicitis. Abdominal tenderness is often not prominent with pelvic appendicitis.
Most atypical presentations of appendicitis are due to appendicitis in an anomalously positioned appendix (Fig 7.18).

Figure 7.18 Anatomy of the appendix
The caecum may be high (A) normal (B) or low (C) in position. The appendix may be free lying, paracaecal or retrocaecal, pre-ileal or postileal, or pelvic in position.
In rectrocaecal or paracaecal appendicitis the pain can be confined to the epigastrium or right upper abdomen without moving to the right lower quadrant or pain may shift to a more lateral point in the flank than is usually the case. The appendix may lie against the ureter and the psoas muscle, causing urinary frequency with red and white cells in the urine and pain that radiates to unusual sites such as the right testis and front of the thigh. Retrocaecal appendicitis can closely resemble acute cholecystitis or acute pyelonephritis. The patient often presents with a limp due to painful hip flexion, even suggesting hip disease.
Pelvic appendicitis is probably the most sinister form of the disease. Pain and tenderness are often suprapubic and abdominal tenderness less marked. Abdominal tenderness may be absent during the first 48 hours of the illness or until general peritonitis ensues. The early diagnosis often made in these cases is gastroenteritis, cystitis or salpingitis. The diarrhoea of pelvic appendicitis, however, is usually late. The diagnosis of pelvic appendicitis depends on performing a rectal examination.
Retro-ileal appendicitis may produce a classic picture of small bowel obstruction. This unusual form of appendicitis can be associated with early vomiting and diarrhoea. Meckel’s diverticulitis can produce a similar clinical picture.
Young children and the aged tend to have a more rapid and severe course with early peritonitis or early abscess formation and poor localisation of infection. A similar natural history is often found in obese patients. Appendicitis is a particularly dangerous condition in psychiatrically disturbed patients and in infancy. One must always be aware of the possibility of appendicitis in an infant who is lethargic, irritable and difficult to examine, especially when vomiting and abdominal distension are present. The younger the patient, the more appendicitis appears to resemble gastroenteritis.
Appendicitis in pregnancy can also be extremely difficult to diagnose because the appendix is in a higher position than usual and the infective process tends to localise poorly. Appendicitis mainly occurs during the second trimester and must be distinguished from other common causes of abdominal pain at that time — including degeneration of fibromyoma, acute pyelitis, cholecystitis, nonulcer dyspepsia and reflux oesophagitis.
2. Acute mesenteric adenitis and acute Meckel’s diverticulitis
These conditions are considered together as they can closely mimic appendicitis and are usually best treated by surgical exploration, often with laparoscopy.
Acute mesenteric adenitis. This condition is more common in children and adolescents and often follows or accompanies an upper respiratory viral illness. The clinical course of abdominal pain and tenderness is very similar to appendicitis. Pain tends to be more generalised and less severe. Tenderness is less well localised and less constant in position. Fever can be higher and constitutional signs less. There may be evidence of a coexisting viral illness. The disease is self-limiting and nonprogressive. Some patients who are seen at the stage of resolution with minimal abdominal and general signs are obviously getting better; conservative treatment and observation are indicated. In other cases the safest treatment is early laparoscopy and possible appendicectomy. The diagnosis is established at operation by finding a normal appendix and enlarged succulent nodes in the ileal mesentery with a serous exudate.
Acute Meckel’s diverticulitis. Meckel’s diverticulitis can also simulate acute appendicitis very closely. Other forms of presentation are intestinal obstruction, gastrointestinal haemorrhage or an umbilical fistula. Presentation in childhood is more common than in adults and the diagnosis will usually be made only at operation. Occasionally the clinical features of appendicitis, together with rectal haemorrhage, will suggest the diagnosis, as will associated low small bowel obstruction.
3. Nonspecific acute right iliac fossa pain
Although called nonspecific, a more precise presumptive diagnosis can be made in many cases. Conditions causing nonspecific right iliac fossa pain separate into two main groups: those due to viral or bacterial gastroenteritis and those due to functional bowel disease (irritable colon).
Gastroenteritis secondary to infection is probably a more common cause of acute right iliac fossa pain than functional bowel disease. Severe vomiting and diarrhoea start early in gastroenteritis in contrast to appendicitis, which usually has a more insidious onset with pain as the first symptom. An early high fever is common and tenderness is more diffuse than is found in acute appendicitis. Signs of upper respiratory tract infection are frequently found. This condition is perhaps the most common reason for a negative appendicectomy.
Functional bowel disease. Patients with functional bowel disease usually do not appear ill. Symptoms and signs are less than is found with gastroenteritis or appendicitis. A recent history of anxiety and depression is common, as is a past history suggestive of functional bowel disease — particularly constipation. On examination there is mild diffuse tenderness and voluntary guarding. A scar, through which a normal appendix was removed in the past, is common.
4. Gynaecological disorders
Salpingitis. Women with salpingitis generally present with poorly localised pain, often starting in the lower right abdomen and then becoming more diffuse. Most patients have associated urinary frequency. Early high fever is common, with diffuse lower abdominal tenderness and guarding, tenderness in both lateral fornices (especially on rocking the cervix) and vaginal discharge.
Complicated ovarian cyst. An acute torsion of an ovarian cyst may be difficult to distinguish from acute pelvic appendicitis. The pain is severe and usually more diffuse. Pain may radiate to the flank or thigh. It may not be possible to examine the patient adequately because of exquisite pelvic tenderness. The temperature is usually normal but the patient looks ill. A ruptured ovarian follicle (mittelschmerz) can mimic appendicitis. The history usually reveals that the onset of the pain is mid-cycle. The pain rapidly settles and, as with most gynaecological causes of acute right iliac fossa pain, gastrointestinal symptoms are insignificant. In addition these patients do not look as ill as patients with appendicitis and the condition is often resolving when seen.
Ectopic pregnancy. Ectopic pregnancy is an important condition that can resemble acute appendicitis, particularly the subacute presentation that follows ampullary implantation. In these patients there may not be a history of missed period. On examination, as well as poorly localised tenderness in the right lower abdomen, the cervix is tender and soft and the uterus is enlarged. An elevated beta HCG is often helpful in the diagnosis.
5. Urinary tract stone, infection or hydronephrosis
Urinary stone. In most patients with pain due to urinary tract stone the diagnosis is obvious. Typical renal colic is associated with severe loin pain often radiating to the ipsilateral groin. There may be haematuria and frequency. Difficulty may arise in cases without classic pain or a stone later impacted in the lower right ureter may cause an acute periureteral inflammatory reaction with local tenderness and guarding.
Renal tract infection. Renal tract infection and perinephric abscess are more frequent causes of pain that resembles high retrocaecal appendicitis. Classically the patient with urinary tract infection has an early high fever, rigors and frequency, and dysuria from the time of onset of the pain. Pyuria is found on urine examination. The diagnosis may be especially difficult in children with a ‘closed’ infected hydronephrosis and thus no pyuria.
Hydronephrosis. Hydronephrosis is an important cause of intermittent attacks of abdominal pain that can mimic appendicitis. Renal tenderness is usually present and the disease is not usually progressive. Occasionally, abdominal signs are so marked that appendicitis is difficult to exclude.
Diagnostic plan
In patients with a typical clinical presentation of appendicitis no further investigations are necessary. Appendicectomy should proceed forthwith. The surgeon will decide whether to remove the appendix by laparoscopy or open operation.
When doubt about the diagnosis exists, the following investigations can be performed.
Full blood count
This may reveal a moderate leucocytosis typical of appendicitis. Anaemia suggests the possibility of an unusual cause of right iliac fossa pain, such as carcinoma of the right colon. In a patient with equivocal symptoms and signs, and in whom pain has been present for 72 hours or more, a normal white cell count is helpful and supports the decision to treat the patient by observation and review. It cannot be too strongly stressed that a normal white cell count in the first two to three days of the clinical history does not exclude appendicitis. If a white cell count is done and found to be normal in a patient with a clinical diagnosis of acute appendicitis, operation should proceed without delay on the basis of the clinical findings.
Urine examination
This is essential and may detect the presence of sugar and diagnose diabetes. Leucocytes and red cells may be present in the urine in renal tract infection and in some cases of retrocaecal appendicitis. Again, positive urinary findings, when the diagnosis is clinically suggestive of appendicitis, should not delay operation.
Cervical swab and culture
This is indicated when a vaginal discharge suggests the possibility of salpingitis. This is especially so if a sexually transmitted disease (e.g. gonorrhoea) is suspected.
Plain X-ray
This may be indicated when renal stone is suspected or diagnostic doubt exists for other reasons. Acute appendicitis is diagnosed without radiographic studies in most young patients. Plain films of the abdomen may be valuable in atypical cases, especially in the very young and very old. In about 50% of patients with appendicitis, evidence of localised ileus in the right iliac fossa is seen on X-ray. The visualisation of a faecolith on plain abdominal X-ray in a patient with acute right iliac fossa pain is typically diagnostic of appendicitis.
Ultrasound
This technique is more helpful in the diagnosis of gallstones or of hydronephrosis — it can be useful confirming these diagnoses in equivocal presentations. Pelvic and/or transvaginal ultrasound can be very helpful in confirming gynaecological abnormalities such as ovarian cyst, salpingitis or ectopic pregnancy.
CT scan
CT scan may be of value in the neglected case referred with gastrointestinal disease for diagnosis. It should be noted also that, in many centres, it is the investigation of choice for appendicitis where clinical features are not typical.
Laparoscopy
This is a useful procedure in cases of doubt and is a particularly important diagnostic procedure in the patient suspected of having a gynaecological cause of acute right iliac fossa pain, especially in those who have missed a period and ectopic pregnancy is suspected.
In many cases a safe appendicectomy can be performed by laparoscopy. If the appendix is normal many of the other possible surgical conditions can be seen and treated laparoscopically.
If a mass is palpable under general anaesthesia, the procedure should be converted to an ‘open’ appendicectomy performed through a transverse, muscle-splitting, right lower quadrant abdominal incision.
HCG pregnancy test
Urinary hormonal (β-HCG) testing for pregnancy usually becomes positive by 14 days from conception. It is prudent to perform the test in women of child-bearing age with acute right iliac fossa pain when the pattern is not typical of appendicitis. The test should be routine if complications of pregnancy are suspected.
Treatment plan
1. Acute appendicitis
Most cases of typical acute appendicitis occur in young fit patients; appendicectomy can be performed promptly with safety. Perioperative antibiotics are given (usually in conjunction with preoperative pain relief/sedative). Preoperative resuscitation is usually not required, although special care must be taken with infants. Resuscitation is necessary in patients with advanced disease, prolonged vomiting and peritonitis. The extent of resuscitation depends upon the patient’s response to intravenous infusion and the urgency of surgery. Under anaesthesia the abdomen is palpated to detect the presence of a mass.
In most cases the appendix can be removed laparoscopically. The open approach is performed through a muscle-splitting right lower abdominal incision. The incision is centred on a mass if one is palpable. If the appendix is found to be normal, further laparoscopic examination (or, in the open case, a limited laparotomy) is performed to exclude other pathology. Whether inflamed or not, the appendix is removed if an incision has been made in the right lower quadrant. Peritoneal drainage is unnecessary unless there is local abscess formation. Intravenous antibiotics are continued postoperatively only in patients with abscess or peritonitis.
2. Acute mesenteric adenitis and Meckel’s diverticulitis
The diverticulum is removed and bowel continuity restored. The appendix is also removed.
3. Nonspecific acute right iliac fossa pain
These patients may be admitted and kept under careful observation in hospital until their symptoms settle down. Surgery is indicated with deterioration or when appendicitis cannot be excluded in the patient with persistent pain and tenderness. Laparoscopy can be a helpful diagnostic procedure that allows therapeutic interventions if appropriate pathology is encountered.
4. Gynaecological disorders
When an acute gynaecological disorder is suspected, laparoscopic confirmation is indicated in most cases. Patients with anaemia and a diagnosis of ectopic pregnancy may need blood to be cross-matched and available for transfusion. Open laparotomy may be required.
5. Acute urinary tract stone, infection or hydronephrosis
Renal tract infections are treated conservatively with antibiotics. Subsequent investigation of the urinary tract may be necessary to discover the cause of hydronephrosis.
6. Less common causes
These are numerous. Perforated ulcer may present with acute pain in the right iliac fossa due to gastric fluid tracking down the right paracolic gutter. Carcinoma of the right colon or caecum may present acutely, like appendicitis, with a complication such as obstruction or localised perforation. A mass in the right iliac fossa with accompanying anaemia suggests the diagnosis. Crohn’s disease may present with acute right iliac fossa pain. There is usually a history of altered bowel habit and general deterioration in health with weight loss. Occasionally acute regional ileitis (which may be the earliest phase of Crohn’s disease) presents de novo with acute right iliac fossa pain. At operation in these cases, a copious straw-coloured intraperitoneal fluid discharges on opening the peritoneum. The appendix is normal and an acutely inflamed distal ileum is found. Appendicectomy is usually necessary in these cases and, if performed with care, will not lead to a faecal fistula. If the appendix is not removed, the presence of a right iliac fossa incision may delay necessary surgery at a later date. Diverticulitis in a redundant sigmoid colon in the right iliac fossa; an isolated caecal diverticulitis or benign caecal ulcer are occasional causes of acute right iliac fossa pain. Right basal pneumonia may also present with predominantly right-sided abdominal pain and guarding. The diagnosis may be difficult, but the clue to the diagnosis is the presence of respiratory distress even though clinical signs on chest examination may be minimal. Herpes zoster (shingles) can mimic appendicitis but the pain follows the nerve root distribution. Often a heraldic vesicle will be found on careful examination, in which case non-surgical observation and review confirm the diagnosis.
7.5 Acute lower abdominal (pelvic) pain
Acute lower abdominal pain may be suprapubic or felt in one or other iliac fossa or associated with deeper pelvic pain. It is often difficult for the patient to localise or the doctor to interpret whether the site of pain is confined to the lower abdomen or extends into the pelvis. Most patients can separate pelvic and associated lower abdominal pain from anorectal or perineal pain: the latter is considered as a separate problem in Chapter 7.22. Acute right iliac fossa pain usually presents as a separate problem (Ch 7.4)
Pain secondary to spinal disease and referred along the T12 and L1 dermatome and pain from local soft tissue injury or groin hernias may occasionally give somewhat similar pain. Musculoskeletal pain rarely requires hospital admission and most musculoskeletal conditions of the lumbosacral spine and pelvis are easy to distinguish (because of the relation to posture and movement) from visceral conditions causing lower abdominal or pelvic pain. Acute renal colic (Ch 9.2) with its characteristic pattern is usually easily distinguishable. In cystitis, frequency and burning of urination dominate the clinical picture (Ch 9.3).
Conditions causing acute lower abdominal and pelvic pain separate into two main groups:
• patients over 60 years in whom diverticulitis and carcinoma of the colon are likely causes of acute pain
• patients under 50 years, with gynaecological disorders or appendicitis.
Causes
1. Diverticulitis
2. Carcinoma of the colon
3. Pelvic appendicitis
4. Gynaecological disorders
5. Less common causes
History and physical examination
1. Diverticulitis
Acute colonic peridiverticulitis results from obstruction, inflammation and sometimes localised rupture of a diverticulum. The pain is of gradual onset, its site depending upon the position of the sigmoid colon (which often lies in the pelvis) and how well localised the inflammatory process is. A history of previous bowel irregularity and lower abdominal discomfort and distension, partially relieved by defaecation, is common. Fresh rectal bleeding is not a feature of diverticular disease and when present indicates that another condition such as haemorrhoids or carcinoma is likely to be present. However, profuse rectal bleeding can occur with diverticular disease — but rarely, if ever, with acute diverticulitis (Fig 7.19). Diverticulitis adjacent to the bladder may produce dysuria. In younger patients a very similar acute clinical picture may be due to spastic colon or functional bowel disease but, more commonly, this condition causes chronic pain in the lower abdomen.

Figure 7.19 Complicated diverticulitis
Acute abdomen due to: A: perforation of a pericolic abscess (purulent peritonitis); or B: perforation of an uninflamed diverticulum (faecal peritonitis).
Large bowel obstruction due to diverticular disease (i.e. diverticular stricture) is uncommon. Associated small bowel obstruction, however, may occur due to adhesion of a loop of small bowel to the area of sigmoid diverticulitis.
Four main forms of presentation of diverticulitis can be defined. They are associated with acute lower abdominal pain of increasing severity:
• stage 1 — colonic wall and peridiverticular inflammation
• stage 2 — localised abscess
• stage 3 — generalised purulent peritonitis caused by rupture of a pericolic abscess
• stage 4 — generalised faecal peritonitis due to major acute rupture of a diverticulum.
The localised forms of disease (stage 1 or 2) present as acute lower abdominal or pelvic pain often localised to the left iliac fossa. Patients with stage 3 or 4 disease present with an acute abdomen (Ch 7.2).
With stage 1 or 2 diverticulitis, left lower abdominal tenderness and local peritonitis are present. A tender mass can often be felt abdominally or as an extrarectal mass on rectal examination. When spastic colon causes acute pain, no mass can be felt. Sigmoidoscopy is usually normal in diverticular disease, although the instrument can rarely be passed beyond the rectosigmoid junction. Occasionally, pus may be seen in the lumen at the limits of sigmoidoscopy. A low-grade fever is often present with a leucocytosis.
2. Carcinoma of the colon
Carcinoma may sometimes mimic diverticular disease. Partial obstruction of the left colon usually develops insidiously and the presenting symptom is usually altered bowel habit rather than pain (Ch 7.20). Sometimes, however, acute deep-seated lower abdominal cramping pain is the predominant initial symptom. The pain of partial obstruction is usually felt in the hypogastrium, often more so on the left side and sometimes in the back. Complete large bowel obstruction may develop soon after the onset of colicky pain. These cases usually reveal a short history of constipation and distension, partially relieved by defaecation. Rectal bleeding may have been noted by the patient.
Sometimes a locally advanced carcinoma of the distal colon may perforate at or near the site of the tumour, thus presenting with left iliac fossa peritonitis and a mass that very closely resembles the clinical pattern of a perforated diverticular abscess or advanced pelvic appendicitis.
Abdominal examination may demonstrate some abdominal distension. The colonic carcinoma is rarely palpable on abdominal examination. Liver enlargement may indicate the presence of metastatic disease. Rectal examination is usually normal but a mass may be felt extrarectally. Flexible sigmoidoscopy or colonoscopy may show an obstructing carcinoma. Blood in the lumen of the bowel suggests a carcinoma above the limit of the sigmoidoscope.
3. Pelvic appendicitis
When the appendix becomes inflamed in a pelvic site, the usual presentation is acute lower abdominal or pelvic pain, progressing later to iliac fossa peritonitis. The diagnosis of appendicitis can be difficult because abdominal tenderness may be absent or minimal until late in the illness. Diarrhoea follows well after the onset of pain, in contrast to gastroenteritis where diarrhoea is a presenting symptom. The diarrhoea of pelvic appendicitis with a pelvic abscess comprises the frequent passage of small volumes of mucus and liquid faeces associated with severe tenesmus.
Signs of lower abdominal peritonitis are variable. The key to the diagnosis of pelvic appendicitis is tenderness on rectal examination. A tender pelvic mass may also be palpable. In late cases with an established pelvic abscess, oedematous mucosa can be felt over a boggy swelling in the pouch of Douglas.
4. Gynaecological disorders
Acute lower abdominal and pelvic pain in a young woman immediately suggests the possibility of gynaecological disease, especially when the pain is associated with a menstrual disorder. The pain is mainly felt deep in the pelvis or suprapubically. Acute abdominal pain in pregnancy forms a special group. The causes are shown in Table 7.5.
Table 7.5 Acute abdominal pain in pregnancy
|
First trimester |
Second trimester |
Third trimester |
|
Ectopic pregnancy |
Red degeneration in fibromyoma |
Premature labour |
|
Abortion |
Pyelonephritis and cystitis |
Abruptio placentae |
|
Ruptured corpus luteum |
Cholecystitis |
Uterine rupture |
|
Liver rupture and haematoma |
||
|
Appendicitis |
HELP* |
|
|
Nonulcer dyspepsia and reflux |
* Haemolysis. Elevated liver enzymes. Low platelets. A pre-eclamptic condition presenting with acute epigastric pain.
Salpingitis. This is characterised by high fever, chills and a mucopurulent vaginal discharge. The inflammation generally affects both tubes. There is diffuse, lower abdominal suprapubic tenderness and rebound tenderness. Extreme pelvic tenderness may prevent the palpation of uterine adenexa, but a purulent discharge can be seen issuing from the external cervical os.
Acute rupture of an ectopic pregnancy. This presents with acute pain, which may be referred to the shoulder from diaphragmatic irritation, progressing to pallor and shock. The cervix is soft and extremely tender to palpation and dark vaginal bleeding of modest amount occurs, usually after two or three months of amenorrhoea. Ectopic pregnancy may be difficult to distinguish from appendicitis. Ectopic pregnancy with ampullary implantation has a more benign course than acute tubular rupture associated with implantation in the isthmus. A more gradual onset of lower abdominal and pelvic pain precedes dark vaginal bleeding after variable amenorrhoea (Fig 7.20). Blood also passes retrogradely into the peritoneum to collect in the pouch of Douglas forming a pelvic haematocele. The temperature may be normal or elevated. On vaginal examination dark blood may be seen escaping from the external os. Extreme tenderness elicited by movement of the soft cervix makes palpation of any tubal swelling difficult — signs that are not found with mittelschmerz.

Figure 7.20 Sites of ectopic pregnancy implantation
1: interstitial 4%; 2: isthmial 18%; 3: ampullary 55%; 4: abdominal cavity 1%; 5: fimbrial 20%; 6: ovarian 2%
Bleeding corpus luteum cyst or ovulatory bleeding (mittelschmerz). These are commoner causes of haemoperitoneum than ectopic pregnancy. The clinical features are less dramatic and signs of local peritoneal irritation less intense. A history of mid-cycle pain may be obtained.
Torsion of an ovarian cyst results in severe sustained pain of sudden onset, usually over a wide area from loin to groin and is usually accompanied by vomiting. Pelvic tenderness is extreme and a very tender mass may be palpable. There is usually no fever or leucocytosis.
Diagnostic plan
Full blood count, blood grouping and pregnancy test
Anaemia suggests blood loss or severe sepsis. Leucocytosis suggests the presence of inflammation, particularly salpingitis, appendicitis and diverticulitis.
The pregnancy test for human chorionic gonadotrophic in urine or serum is highly sensitive and becomes positive within 10 to 12 days of conception. The test may therefore be positive before the expected next period.
Urine microscopy and analysis
This checks for renal tract infection and haematuria. Swab, microscopy and culture of any vaginal discharge must also be performed.
Plain X-ray of the abdomen
This may show evidence of large bowel dilatation. Sometimes the short ‘shouldered’ stricture of a carcinoma of the left colon is outlined with gas. Gas may be seen to have collected within a peridiverticular abscess with a characteristic ‘soap bubble’ appearance outside the bowel gas limits. Calcification within the aorta or iliac arteries revealing aneursmal changes may also be evident. Plain X-ray should be avoided if pregnancy is a possibility.
Pelvic ultrasound
Pelvic ultrasound and expertly performed transvaginal studies can be particularly valuable in women when the pregnancy test is positive but no pregnancy is detectable in the uterus although fluid is detected in the pouch of Douglas. In these patients ectopic pregnancy is highly likely. Ovarian cysts show up easily on ultrasound, but ectopic pregnancies are less easy to visualise. The presence of cysts up to 30 mm in diameter within the ovary is normal. Excision of simple ovarian cysts of less than 50 mm diameter is inadvisable. Drainage of cysts at laparoscopy is a treatment that may relieve pelvic pain.
Laparoscopy
This is indicated in patients where a gynaecological disorder is suspected and will help to exclude appendicitis and diverticulitis in difficult cases (Fig 7.21).

Figure 7.21 Laparoscopy
The uterus is stabilised per vaginum. The pelvis is inspected after insufflation of CO2 into the peritoneal cavity. An ectopic pregnancy is diagnosed here.
Laparotomy is necessary for diagnosis and treatment in patients with clinical deterioration, with peritonitis or with a pelvic mass.
CT contrast study
The assessment of diverticular disease and its complications is often best pursued with a CT scan with rectal gastrograffin contrast. This may reveal complicated diverticular disease such as:
• an abscess cavity or sinus tract outside the colonic wall
• an intraluminal abscess producing indentation
• extrinsic compression by a paracolic mass
• intramural sinus or fistula
• extramural fistula formation to the bladder or uterus.
There may also be evidence of additional diverticula, segmental spasm and narrowing of the lumen with a sawtooth appearance of the colonic mucosa.
Colonoscopy
Colonoscopy may be performed at a later time. It should not be encouraged in the acute setting due to the risk of perforation
Treatment plan
1. Diverticulitis
Patients with acute diverticulitis (stage 1) are treated by fasting, intravenous fluids and broad-spectrum antibiotics covering anaerobes and both Gram-positive and -negative bacteria. Many episodes settle down within 48 hours. After the acute attack has settled, the basis of treatment is a lifelong high-fibre diet to reduce the incidence of further acute attacks. Elective investigations, such as colonoscopy performed at least a month later, are essential to confirm the diagnosis and exclude carcinoma. Contrast CT studies can complement colonoscopy to exclude malignancy with certainty.
Elective surgery. The indications for elective resection are:
• recurring attacks of left iliac fossa pain, fever and leucocytosis with radiological or endoscopic evidence of diverticulitis despite adequate treatment with a high-fibre diet
• CT evidence of contained perforation
• X-ray or clinical evidence of obstruction
• associated urinary tract symptoms or fistula
• inability to rule out carcinoma, despite colonoscopy and barium enema.
Urgent surgery is indicated when there is failure to improve within 48 hours from admission or if the patient deteriorates prior to this. Primary resection and anastomosis with a defunctioning loop ileostomy can be considered for stages 1 and 2 disease — especially in fit patients and in those in whom systemic sepsis is controlled.
Patients with stage 3 or 4 diverticular disease present with an acute abdomen. They require exteriorisation–resection without anastomosis and are discussed in Chapter 7.2.
2. Carcinoma of the colon
The basis of treatment for impending obstruction due to carcinoma is to resect the lesion after adequate preoperative preparation. Efforts are made to relieve the obstruction and to prepare the patient for semi-elective surgery, rather than being forced to operate as an emergency for complete obstruction. Preoperative measures include: the control of concurrent illness; enemas and bowel washouts to relieve obstruction and produce a clean colon; prophylactic perioperative antibiotics to prevent infection; CT scan with IV contrast to determine whether ureteric obstruction is present; and correction of anaemia by transfusion of packed cells — preferably given at least 48 hours before surgery. Some patients benefit from urgent distal colonic stenting to alleviate obstructive symptoms while their general condition is assessed and optimised.
Where operation is performed in the presence of partial obstruction, it is often prudent to form a loop ileostomy to protect the anastomosis after resection of the tumour, especially if there is inadequate bowel washout and technical difficulty because of obesity.
3. Pelvic appendicitis
Early appendicectomy should be performed laparoscopically or through an iliac fossa or midline incision. The peritonium is washed out and perioperative antibiotics are given. An associated pelvic abscess will require formal drainage as well. The drainage tube is preferably brought out through a separate stab incision.
4. Gynaecological disorders
If the diagnosis is equivocal it is helpful to perform laparoscopy, proceeding immediately to laparotomy if indicated.
Salpingitis is treated with antibiotics; mittelschmerz is treated conservatively; ectopic pregnancy and torsion of an ovarian cyst by early surgery.
Persisting haemorrhage from a ruptured ovarian cyst is treated by laparoscopic aspiration and diathermy or by local oversewing of the cyst. Meticulous pelvic toilet at operation and a gentle operative technique using fine atraumatic synthetic sutures help minimise the risk of tubal adhesions and later infertility.
5. Less common causes
Unusual causes of acute lower abdominal pain include acute pelvic congestion syndrome, perinephric abscess, pancreatic abscess or necrosis extending down the left paracolic gutter, small bowel obstructionsecondary to diverticulitis, Crohn’s colitis of the left colon and spontaneous rupture of the inferior epigastric artery in patients on oral anticoagulants. Contained rupture of an iliac artery aneurysm may also present with sudden severe lower abdominal pain and requires emergency surgical intervention. Laparoscopy may be indicated because of persistent signs of peritonitis in the absence of a firm diagnosis in some of these conditions.
7.6 Chronic epigastric pain
Patients with chronic or recurrent epigastric pain generally present electively to their general practitioner; they may subsequently be referred for specialist consultation. Causes of chronic and of acute epigastric pain thus differ somewhat in emphasis, but overlap. Chronic epigastric pain may be described by the patient as indigestion or dyspepsia (food-related discomfort) if discomfort is less severe. Various more descriptive (but subjective) terms such as fullness, bloating, pressure and hunger pain may also be used by the patient to describe the upper abdominal discomfort. Chronic pain associated with weight loss always warrants thorough assessment and investigation.
Chronic epigastric pain is attended by a limited and manageable list of common causes (Table 7.6). A wide spectrum of less common causes exists.
Table 7.6 Features of the common causes of chronic epigastric pain
|
Cause |
Feature |
|
Nonulcer dyspepsia |
Atypical patterns |
|
Gallstones |
Episodic pain |
|
Duodenal ulcer |
Periodic or cyclical pain |
|
Carcinoma of stomach |
Onset of dyspepsia in a patient over 40 years |
Causes
1. Nonulcer dyspepsia
2. Gallstones and chronic cholecystitis
3. Duodenal ulcer
4. Gastric ulcer
5. Carcinoma of the stomach
6. Less common causes
History
1. Nonulcer dyspepsia
Nonulcer dyspepsia is a diagnosis of exclusion applied to a heterogeneous group of patients with chronic epigastric pain without proven ulcer or other extragastric pathology. Other terms used include functional dyspepsia, pylorospasm, nervous dyspepsia and gastritis. When the pain is situated in the right hypochondrium and resembles biliary pain, the condition is often labelled biliary dyskinesia. The symptoms can be identical to duodenal ulcer but usually the illness lacks a clear descriptive focus and the pain fits no definite pattern. Bloating is a characteristic symptom that many patients find quite disabling. Many patients have a long history of poor response to antacids, prolonged medical treatment and negative investigations. Alcoholism and depressive illness are common associated problems. Other features of functional bowel disease can coexist. Patients may have a history of intermittent change in bowel habit, lower abdominal distension, flatulence and straining at stool. Symptomatic gastritis secondary to bile reflux may also develop in both the postcholecystectomy and postgastrectomy patient. Such patients often have a history of early morning pain, anorexia, flatulence with bile vomiting and diarrhoea with urgency.
2. Gallstones and chronic cholecystitis
Patients with gallstones frequently present with episodic upper abdominal pain. Biliary pain (‘colic’) begins abruptly and subsides gradually over a period of hours and is usually felt in the epigastrium or right hypochondrium. It can be postprandial or nocturnal and commonly radiates around the costal margin to the back with a sometimes severe and usually constant nature. Patients have often had a number of similar attacks requiring narcotic analgesics or admission to hospital. The pain may occasionally be felt in the left hypochondrium or precordially, making it difficult to distinguish from oesophageal pain or angina. The acute upper abdominal pain of acute cholecystitis usually leads to hospital admission (Ch 7.3) and early cholecystectomy and is related to persistent rather than transitory obstruction of the cystic duct by stone.
Chronic or recurrent dyspeptic symptoms are also common. These may have been present for many years and are associated with flatulence, abdominal distension, nausea and fatty food intolerance.
3. Duodenal ulcer
Characteristically, patients with duodenal ulcer have a chronic, fluctuating, remitting and relapsing natural history. The pain is felt in the epigastrium and is described as dull, boring, aching, burning, gnawing or hunger-like. The pain is generally relieved by antacids, food and milk. A high intake of milk may produce weight gain, but occasionally food or alcohol can make the pain worse, with associated weight loss. Occasionally the patient localises the pain precisely in the epigastrium just to the right of the midline. Radiation directly to the back in the interscapular region suggests posterior penetration of the ulcer. This complication generally causes loss of the cyclical character of the pain, loss of response to antacids and vomiting.
Periodicity and relapse of symptoms is classic. Relapse is often triggered by smoking, stress or nonsteroidal anti-inflammatory agents. During relapse, attacks of pain come on each day, from half an hour to three hours after meals. Relapses and remissions follow each other in cycles. Relapses last for days or weeks and often occur during spring and autumn. Remissions last from weeks to months. In about half the patients, the daily cycle of pain during a relapse may occur so close to the next meal that it is described as ‘hunger’ pain. Nocturnal pain is a common complaint, usually wakening the patient in the early morning hours and also relieved by food and alkali.
The basis of over 90% of duodenal ulcers has been proven to be due to H. pylori and the basis of modern diagnosis and treatment is to confirm the presence of H. pylori and then institute eradication therapy.
The Zollinger-Ellison syndrome (gastrinoma of the pancreas) should be considered when a severe and resistant ulcer diathesis is present. Features suggesting the diagnosis include ulcers situated more distally, multiple and recurrent ulcers, severe diarrhoea (found in 30% of cases) and the combination of severe upper and lower abdominal symptoms (Box 7.4).
Box 7.4
Causes of combined upper and lower gastrointestinal symptoms
![]() Functional bowel disease (dyspepsia and altered bowel habit)
Functional bowel disease (dyspepsia and altered bowel habit)
![]() Zollinger-Ellison syndrome (vomiting, epigastric pain and diarrhoea)
Zollinger-Ellison syndrome (vomiting, epigastric pain and diarrhoea)
![]() Carcinoma of the caecum (indigestion and diarrhoea)
Carcinoma of the caecum (indigestion and diarrhoea)
![]() Crohn’s disease (vomiting and diarrhoea)
Crohn’s disease (vomiting and diarrhoea)
![]() Pyloric stenosis (constipation and vomiting)
Pyloric stenosis (constipation and vomiting)
![]() Thyrotoxicosis (vomiting and diarrhoea)
Thyrotoxicosis (vomiting and diarrhoea)
![]() Chronic pancreatitis (epigastric pain and diarrhoea with steatorrhoea)
Chronic pancreatitis (epigastric pain and diarrhoea with steatorrhoea)
4. Gastric ulcer
The history of patients with gastric ulcer is very similar to that of duodenal ulcer (Table 7.7). Pain, however, tends to be more severe, more loss of work occurs and relapse is more frequently triggered by analgesic agents. Relapses last longer, generally from one to two months. Food and alcohol appear to worsen the pain, which generally occurs within 30 minutes of a meal. Anorexia and weight loss are also more common. Associated gastritis is a feature of gastric ulcer and contributes to weight loss. Vomiting is more common and more likely to relieve the pain.
Table 7.7 Comparison of the clinical features of duodenal and gastric ulcers
|
Features |
Gastric ulcer |
Duodenal ulcer |
|
Onset of pain |
Occurs within 30 minutes of a meal |
Occurs 2–3 hours after meals — ‘hunger pain’ |
|
Wakes patient at night |
||
|
Relieving factors |
Vomiting |
Food and milk, antacids |
|
Precipitating factors |
Eating (may lose weight) |
Smoking and other risk factors |
|
Analgesics |
||
|
Nonsteroidal anti-inflammatory drugs |
||
|
Periodicity |
2–3-month cycle |
4–6-month cycle |
5. Carcinoma of the stomach
Stomach cancer should be suspected when dyspepsia starts for the first time in a patient aged over 40 years. Early gastric cancer, with minimal mucosal changes, needs to be diagnosed if prognosis is to be improved. Unfortunately many patients present (or are diagnosed) late in Western countries.
Symptoms usually begin with lethargy and vague postprandial heaviness, discomfort and fullness: a symptom complex usually described as indigestion or dyspepsia. Anorexia is an early symptom (especially to meat) leading to early weight loss. Early satiety is a common feature, with distortion of the senses of smell and taste. Most patients have lost more than 5% of their normal body weight on presentation. Eventually continuous intractable epigastric pain is present.
Complications may cause additional symptoms, depending on the site of the tumour. Nausea and vomiting can result from pyloric obstruction (10–20% of patients) or there may be dysphagia due to oesophageal obstruction (10% of patients). Haematemesis and melaena is unusual, but iron deficiency anaemia from chronic blood loss is found on investigation in almost half the patients. Anaemia may be the sole form of presentation (Ch 7.18) in a small proportion of patients.
Examination
Nonulcer dyspepsia, gallstones, duodenal ulcer, reflux oesophagitis, chronic pancreatitis and gastric ulcer often have no specific physical signs to confirm the diagnosis. Chronic cholecystitis associated with gallstones and chronic epigastric pain is rarely associated with a palpable gall bladder.
Deep tenderness in the epigastrium or right upper abdomen are the most common findings in carcinoma of the stomach. An epigastric mass or hepatomegaly due to metastases are also distressingly frequent findings. Other signs of metastatic disease should be sought. Pyloric obstruction may cause a succussion splash. Significant weight loss is often present. Chronic pancreatitis may cause diffuse tenderness in the left upper abdomen; the pancreas is not palpable unless complicated by pseudocyst.
Diagnostic plan
Diagnosis usually requires additional investigations, particularly ultrasound of the upper abdomen and endoscopy.
Blood examination
Iron deficiency anaemia suggests carcinoma of the stomach. Carcinoma of the right colon should also be considered in the anaemic patient when dyspeptic symptoms are vague.
Ultrasound
This is the preferred examination for gallstones. In 80% of cases gallstones are radiolucent and will not be detected on plain X-ray examination. If ultrasound is negative but clinical suspicion of biliary disease is strong a, HIDA study of the gall bladder may be helpful in assessing function.
Endoscopy
This is the preferred investigation for the diagnosis of duodenal or gastric ulcer. Biopsy and brush cytology are necessary additions for the differentiation of benign gastric ulceration from carcinoma of the stomach. Early gastric cancer may require multiple biopsies of the gastric mucosa, as field malignant change is common. Endoscopic and histological features of reflux oesophagitis may be difficult to interpret; oesophageal motility studies may be helpful. No abnormality or antral gastritis is found with nonulcer dyspepsia. Helicobacter pylori may be found on biopsy. Biopsy should be routinely performed if the endoscopy is being performed for the investigation of epigastric pain.
Contrast radiology
Distinguishing benign from malignant gastric ulcers is often difficult; endoscopy is usually necessary to obtain a definitive diagnosis. Barium swallow is the preferred initial investigation for gastrointestinal symptoms only if dysphagia is the major presenting problem and an oesophageal or pharyngeal lesion is suspected (Ch 7.12). Endoscopy will usually be required subsequently to confirm the radiological findings. Contrast study of the small bowel (small bowel enema) is indicated in the occasional patient where chronic small bowel obstruction is suspected to be the cause of epigastric pain. Abdominal CT examination with contrast is most useful when a pancreatic lesion is suspected and for the diagnosis and the definition of focal lesions in the liver. For the latter lesions, a triple phase CT angiography study can delineate lesions very clearly
MRCP, CT cholangiography and ERCP
MRCP and CT cholangiography are newer, noninvasive modalities of imaging the hepatopancreaticobiliary system with great clarity. These investigations allow precise planning of treatment for various ductal conditions.
ERCP is indicated in the occasional patient with persistent pain where the diagnosis remains in doubt after the above investigations or treatment has failed. ERCP has a special place is diagnosing pancreatic carcinoma or inflammation. In suspected biliary disease this investigation is generally reserved for those patients with unexplained abnormalities in liver function tests or intractable postcholecystectomy pain, especially those with bile duct dilatation on ultrasound.
Where diagnosis is obscure or the clinical pattern is unusual, additional tests may be performed. These include serum amylase (or lipase), serum gastrin, hydatid serology, colonoscopy or barium enema.
Treatment plan
1. Nonulcer dyspepsia
There are two main avenues of treatment for nonulcer dyspepsia. Attention should first be given to control of gastric risk factors, particularly alcohol intake, smoking and dietary irregularity. Written instructions can be given to the patient as set down in Box 7.5. Drug treatment may also be required. Proton-pump inhibitors are superior to H2-receptor antagonists in relieving symptoms. Helicobacter pylori eradication should occur if biopsies are positive.
Box 7.5
General instructions for relief of symptoms from gastritis or dyspepsia
![]() Specific medicines or tablets may be prescribed by your doctor. In addition to medical treatment, changes in lifestyle can help reduce the severity of symptoms and the chances of recurrence.
Specific medicines or tablets may be prescribed by your doctor. In addition to medical treatment, changes in lifestyle can help reduce the severity of symptoms and the chances of recurrence.
![]() Avoid the use of aspirin or aspirin-like drugs.
Avoid the use of aspirin or aspirin-like drugs.
![]() Stop smoking.
Stop smoking.
![]() Reduce alcohol intake.
Reduce alcohol intake.
![]() Eat regular meals. Breakfast is particularly important.
Eat regular meals. Breakfast is particularly important.
![]() Regular exercise such as walking can improve your fitness and general resistance.
Regular exercise such as walking can improve your fitness and general resistance.
2. Gallstones and chronic cholecystitis
There is no satisfactory medical treatment for gallstones. Bile salts have limited usefulness and limited ability to dissolve gallstones in the long term. Shock wave lithotripsy is contraindicated. The treatment is by elective cholecystectomy after control of any concurrent disease. In about 5% of cases unsuspected stones are found in the bile duct, so operative cholangiography should be a routine (Fig 7.22). Percutaneous laparoscopic cholecystectomy is the operation of choice but open operation may be necessary if the gall bladder is very thickened or contracted or not properly visible because of chronic adhesions.

Figure 7.22 Cholecystectomy — normal operative cholangiogram
No filling defects, normal duct anatomy and diameter, free flow of contrast into the duodenum, lower and upper reaches of duct well visualised.
The small group of patients with biliary dyskinesia is treated symptomatically — similarly to patients with functional bowel disease. The results of surgical treatment tend to be disappointing.
3. Duodenal ulcer
Most duodenal ulcers have a chronic, fluctuating, periodic natural history. The key decision in patients with significant and disabling symptoms is between long-term drug therapy and surgery. Medical management consists of attention to risk factors — particularly smoking, dietary irregularity, stress, nonsteroidal anti-inflammatory agents, coffee ingestion and alcohol. The treatment of H. pylori involves the use of a proton pump inhibitor and two antibiotics (e.g. clarithromycin plus metronidazole). Modern medical treatments heal most ulcers.
Surgical procedures are rarely required and aim to reduce acid secretion. Previously a satisfactory elective surgical procedure in most instances was highly selective vagotomy (HSV) but this is seldom required in the era of proton-pump inhibitors and H. pylori eradication. For rarer cases where duodenal ulcers are intractable due to Zollinger-Ellison syndrome, gastrinoma should be searched for, usually in the pancreas, and resected.
4. Gastric ulcer
The main issue after diagnosis of gastric ulcer is to exclude malignancy. The complications of gastric ulcer are also more serious than for duodenal ulcer. The surgical approach to this problem is therefore more active. Medical treatment is the same as for duodenal ulcer and is directed at H. pylori eradication. Repeat endoscopy after six to eight weeks of medical management must confirm that healing has occurred. Biopsy and/or brush cytology and lack of recurrence at a further endoscopy after a second similar follow-up period is also essential. Partial or complete healing, with relief of symptoms and negative biopsy, may occur initially with medical management, even though the gastric ulcer is malignant. Surgery is indicated if the ulcer has not completely healed within six to eight weeks and for any recurrent ulcers.
5. Carcinoma of the stomach
Local control of the disease is best obtained by surgery (Box 7.6). Surgery should, if possible, completely excise the gastric lesion so that recurrence does not occur at this site during the life of the patient (Fig 7.23). Symptoms due to anastomotic recurrence represent a surgical failure.
Box 7.6
Management of carcinoma of the stomach
The aim is to achieve local control of disease for the life of the patient. The best method is by surgical resection.
Laparotomy is required in about 85% of patients. Resectability is determined at operation — a CT scan is unreliable in determining resectability. Resection is possible in about 50% of patients.
Excision of all macroscopic disease (with potential for cure) is possible in about 25% of patients.

Figure 7.23 Surgical management of carcinoma of the stomach
A: antral tumours often present with vomiting and are managed by partial gastrectomy; B: body tumours present with chronic epigastic pain; total gastrectomy is usually required; C: tumours of the cardia present with dysphagia; oesophagogastrectomy required
The decision as to whether the tumour is resectable usually requires laparotomy. The major reason for inability to resect the lesion is posterior fixation. Laparotomy is superior to CT scanning in deciding whether resection is feasible, especially in wasted patients where loss of fat planes makes the extent of the tumour difficult to determine on scan. Total gastrectomy (Fig 7.24) is preferred in patients who have satisfactory general health. Total gastrectomy is also indicated with diffuse or desmoplastic tumours at any site and in tumours with associated atrophic gastritis. Ivor Lewis oesophagogastrectomy is indicated in resectable carcinoma of the cardia. About half those coming to operation can have the tumour resected. Half the resections can be considered potentially curable, there being no apparent macroscopic spread beyond resected tissue. Adjuvant chemotherapy may further improve survival after resection. The total five-year survival rate in Western countries remains poor and is about 15%.

Figure 7.24 Total gastrectomy
Total gastrectomy achieves best local control of disease. Reconstruction is by Roux-en-Y oesophagojejunostomy. The letters indicate the arrangement of structures before and after the gastrectomy and reconstruction.
6. Less common causes
Non-gastrointestinal causes
Causes of chronic epigastric pain outside the gastrointestinal tract should be remembered. These include epigastric hernia, aortic aneurysm, visceral artery aneurysm (e.g. splenic or hepatic) and nerve root pain (T6–10). Persistent pain due to an aortic aneurysm is an important harbinger of rupture. The pain may be confused with pain of pancreatic origin. Examination reveals a pulsatile deep epigastric mass. Ultrasound is the most useful means of confirming the diagnosis of aortic aneurysm and CT angiography the best investigation to define the extent of aneuysmal disease and plan appropriate surgery.
Gastrointestinal causes
Reflux oesophagitis. These patients are usually identified by the dominant presenting symptom of postural heartburn and regurgitation. Retrosternal oesophageal pain is characteristically related to swallowing. The pain may be precordial and difficult to distinguish from angina (Ch 7.11). When felt lower down — behind the xiphoid process or in the epigastrium — reflux oesophagitis can be difficult to differentiate from other causes of chronic epigastric pain. Associated symptoms of fluid regurgitation suggest that reflux oesophagitis is the primary cause of the problem. Surgery is indicated for failed medical management, especially when there are complications such as aspiration pneumonitis.
Chronic pancreatitis. Epigastric pain is often vague, deep-seated and nauseating, and may radiate through to the back. The patient characteristically assumes a sitting position with spinal flexion in an attempt to relieve pain. Weight loss is likely to be more profound if carcinoma is present. Chronic pancreatitis affects a different set of patients from acute pancreatitis, the latter presenting with acute upper abdominal pain (Ch 7.3). There is considerable overlap between acute and chronic pancreatitis, particular where the aetiological factor is alcohol. Both conditions are associated with heavy alcohol consumption. In about half the patients with chronic pancreatitis the disease presents with recurrent episodes of epigastric pain and the complications of malabsorption and diabetes should be considered.
The principles of medical treatment of chronic pancreatitis include abstinence from alcohol (which is rarely effective for late disease with continuous pain), narcotic analgesics, splanchnic nerve block, nutritional support, pancreatic enzymes plus antacids and the control of diabetes (which is found in about a third of cases). Surgical treatment is indicated for intractable pain. Alcoholism should not necessarily influence the decision but indicates the need for careful preoperative preparation. Most surgeons would in fact stipulate abstinence as a prerequisite for surgery. Duodenal ulcer is often associated with chronic pancreatitis and should be excluded by endoscopy. Duct dilatation is found on ERCP in 30–40% of cases. In these patients a Roux-en-Y pancreaticojejunostomy (often combined with resection of the pancreatic head) is the surgical treatment of choice (Fig 7.25).

Figure 7.25 Puestow’s operation
Puestow’s operation (side-to-side Roux-en-Y pancreaticojejunostomy): for painful chronic pancreatitis with duct dilatation.
Pseudocyst may be a factor in clinical deterioration and pain and is an indication for internal surgical drainage of the cyst into the stomach or small bowel. Pseudocysts in chronic pancreatitis do not have the same significance as those seen following an attack of acute pancreatitis. Surgical drainage of small pseudocysts is not advocated, as they are unlikely to be the cause of pain.
Carcinoma of the body of the pancreas. Management is unsatisfactory. The disease usually presents late with locally advanced disease or metastases. Careful preoperative planning, with percutaneous fine needle aspiration biopsy to establish the diagnosis and CT scanning to determine the extent of disease, is desirable. Resection is restricted to the occasional case with potentially curable disease.
Hepatic diseases — including hydatid disease, primary and secondary carcinoma and congestive hepatomegaly secondary to heart failure — can present with chronic epigastric pain.
Post-cholecystectomy syndrome. This term is applied to patients who complain of biliary pain, dyspepsia, flatulence and fatty food intolerance after cholecystectomy (Box 7.7). Retained common duct stone should be excluded by CT cholangiogram or MRCP. If stones are found, they are extracted with a basket after ERCP sphincterotomy. Other causes of pain, such as peptic ulcer and bile reflux gastritis, may be seen on endoscopy. Occasionally post-cholecystectomy pain may be due to stones in a cystic duct remnant. In patients who have had a cholecystectomy for biliary dyskinesia — that is, no stones were found at the original operation — the diagnosis usually proves to be functional bowel disease. Spasm of the sphincter of Oddi can produce a dilated bile duct and episodes of pain, but the results of sphincterotomy are unpredictable in these cases.
Box 7.7
Causes of post-cholecystectomy syndrome
Incorrect diagnosis
![]() Functional bowel disease (biliary dyskinesia, stenosis of the sphincter of Oddi)
Functional bowel disease (biliary dyskinesia, stenosis of the sphincter of Oddi)
![]() Peptic ulcer
Peptic ulcer
![]() Chronic pancreatitis
Chronic pancreatitis
Retained stone in the bile duct
Bile duct injury
Liver pathology (e.g. hydatid disease)
Carcinoma of the transverse colon. Partially obstructing colonic cancers may be associated with postprandial pain. Patients with right colonic tumours may complain of dyspepsia in the absence of obstruction. Anaemia, with a normal gastroscopy, suggests the diagnosis of carcinoma of the colon. Chronic upper small bowel obstruction, especially the partial form seen with small-bowel tumours (mainly lymphomas) or Crohn’s disease, can occasionally cause chronic upper abdominal pain.
Post-gastrectomy syndromes can in most cases be treated conservatively (Table 7.8).
Table 7.8 Postgastrectomy syndromes: management
|
Syndrome |
Medical treatment |
Surgical treatment |
|
Dumping |
Beta-blocker |
Roux-en-Y bile diversion |
|
Early |
Small, frequent dry meals, low sugar intake, rest after meals |
|
|
Late |
Sugar at two hours after meals |
|
|
Bile reflux and vomiting, with epigastric pain |
Exclude recurrent ulcer |
Roux-en-Y diversion plus vagotomy or stomal reconstruction |
|
H2 receptor antagonist |
||
|
Bile salt binding agents |
||
|
High-fibre diet |
||
|
Avoid risk factors |
||
|
Diarrhoea |
As for irritable colon |
Reversal of pyloroplasty or reversed ileal loop |
7.7 Chronic lower abdominal (pelvic) pain
Many diseases of the lower abdomen and pelvis are associated with pain and discomfort; relatively few patients present solely because of chronic pain. Patients with chronic cervicitis commonly present with vaginal discharge, less commonly with pelvic discomfort and dyspareunia alone. Common conditions, such as ovarian cysts and uterine fibroids, can present as chronic pain, but are more commonly discovered on routine vaginal examination, present with abnormal uterine bleeding or cause acute lower abdominal pain (Ch 7.5).
The periodic premenstrual deep pelvic pain associated with dysmenorrhoea and mid-cycle ovulatory pain (mittelschmerz) is usually easily recognisable. Progressive dysmenorrhoea may suggest the presence of endometriosis, one of the common causes of chronic pelvic pain.
Hip, sacroiliac and lower back pain is usually readily distinguished from visceral pain. Pain of musculoskeletal origin is closely influenced by posture and movement. Hip disease is suggested by localisation of pain to the front of the hip, radiation to the knee and exacerbation of the pain by walking. Spasm of pelvic muscles (such as the iliopsoas and obturator internus) induced by pelvic inflammatory disease is more likely to simulate hip pain, rather than the reverse. Lower back pain due to musculoskeletal disease is also relatively easy to distinguish from pelvic causes of pain. Buttock claudication due to aortoiliac arterial obstruction is usually readily recognised. Distinction of ischaemic limb pain from sciatica due to spinal stenosis can be more difficult.
As with acute abdomino-pelvic pain, common causes of chronic pain separate into two main groups:
• younger women with associated menstrual upsets and gynaecological disorders
• older patients with alteration in bowel habit, in whom bowel disease is the likely diagnosis.
Causes
1. Chronic pelvic inflammatory disease
2. Pelvic congestion syndrome
3. Endometriosis
4. Spastic colon and diverticular disease
5. Less common causes
History and physical examination
Careful bimanual vaginal and rectal examination is essential to determine the cause of deep pelvic pain. The state of the hip joints and spine and blood supply to the legs, must always be assessed.
1. Chronic pelvic inflammatory disease
Pelvic inflammatory disease is an imprecise (and often overused) term. Pelvic inflammatory disease should be suspected in a patient presenting with chronic lower abdominal and pelvic pain, with associated pelvic tenderness and vaginal discharge. Fever is variable and inconstant. A firm diagnosis of endometritis and tubal inflammation requires laparoscopic confirmation. Most patients with endometritis or tubal infection have sexually transmitted disease of gonococcal or chlamydial origin or are infected by other organisms following spontaneous or induced abortion. Intra-uterine devices are associated with infections with organisms such as Staphylococcus aureus, Escherichia coli, chlamydia, Actinomyces or Bacteroides fragilis. Sexually transmitted diseases often cannot be definitely excluded as the cause of such bacterial infection. Patients with sexually transmitted pelvic inflammatory disease commonly also exhibit urethritis, cystitis and Bartholin’s gland infection.
2. Pelvic congestion syndrome
The pelvic congestion syndrome is a common cause of chronic dull pelvic ache in premenopausal women. Local tenderness on pelvic examination is variable. Pressure over the ovarian point may reproduce the pain. No pathological cause is found on investigation; pelvic varicosities are consistently found on laparoscopy or laparotomy.
3. Endometriosis
In endometriosis, functioning endometrial tissue is found in ectopic sites. The areas most often involved are the ovaries, the pouch of Douglas, uterosacral ligaments, rectovaginal septum, sigmoid colon, small intestine and pelvic peritoneum. The severity of symptoms in endometriosis imperfectly correlates with the extent of implantation. Characteristically the disease first presents as progressive dysmenorrhoea developing in early adult life. Pelvic pain eventually becomes severe and continuous or a vague sense of lower abdominal and pelvic discomfort persists throughout the menstrual cycle and is exaggerated during menstruation. Associated symptoms include menorrhagia and dyspareunia. Endometriosis is often associated with infertility. Miscarriage is common. Defaecation may be painful during the menstrual period, this symptom being characteristic of deposits in the pouch of Douglas and rectovaginal septum.
The picture may be further complicated by bladder symptoms, ureteric obstruction with flank and lower abdominal pain, partial obstruction of the large bowel during menstruation and occasionally rupture of endometrial deposits causing acute lower abdominal pain.
Rectovaginal bimanual examination is an essential part of diagnosis. Examination just before a period may reveal shotty nodules in the pouch of Douglas.
4. Spastic colon and diverticular disease
Functional bowel disease is a very common cause of chronic lower abdominal and pelvic pain. In most patients a careful history reveals a long history of intermittent change in bowel habit, not usually found in patients with a gynaecological cause of chronic pelvic pain. Episodic diarrhoea, bloating and pain partially relieved by defaecation and mild tenesmus, suggest that functional bowel disease may be the cause of pelvic pain.
It needs to be noted that some patients give a history of obstructed defaecation (anismus), probably due to spasm of the puborectalis muscle. Patients present because of the sensation of a painful obstructing lump in the rectum (Ch 7.24). Painful defaecation associated with obstructed defaecation can thus be due to functional bowel disease, as well as endometriosis. Differential diagnosis may require laparoscopy.
A variant of the descending perineum syndrome, occurring more commonly in men, is known as proctalgia fugax (Ch 7.22). Patients have episodes of fleeting, severe, rectal pain, often waking them at night. This condition should not be confused with coccydynia — where pain is felt over the coccyx and the coccyx is tender to palpate. Patients with proctalgia fugax usually show no abnormality on clinical examination but may have a long history of straining on defaecation and poor defaecatory habits.
Diagnostic plan
Blood examination
This is normal in most patients with chronic pelvic pain, with the notable exception of patients with pelvic abscess in whom leucocytosis is found.
Radiological examination
Pelvic X-ray is usually required to exclude bony disease. Contrast abdominal and pelvic CT scan will often reveal the cause of chronic pelvic pain and direct subsequent management. It may show abscess or sinus tracks communicating with the colon, indentation of the colon by intramural abscess or extrinsic compression by a paracolic mass.
Microbiological examination
On vaginal examination, the external os of the cervix is swabbed and cultures taken. The urethra and rectum should also be swabbed for organisms, which may be the cause of pelvic inflammatory disease. In practice a chlamydiazyme test is an easier method than culture to detect chlamydial infection. Prostatic massage and culture of urethral discharge may assist in the diagnosis and treatment of chronic prostatitis. Urine examination and culture, especially the first pass of urine if prostatitis is suspected, is an essential part of the investigation of a patient with pelvic pain.
Laparoscopy
This is necessary when a gynaecological disorder, such as endometriosis, is suspected. Diagnostic laparoscopy in endometriosis reveals reddish blue peritoneal implants with pale contracted points of scar tissue, which look like coarse grains of pepper and are described as ‘powder burns’.
Treatment plan
1. Pelvic inflammatory disease
In most cases the infection is either gonococcal or chlamydial and responds to an appropriate antibiotic. Clinically significant infection in association with an intra-uterine device indicates the need for its removal. Tubo-ovarian abscess requires incision and drainage. Infertility is a common sequel of pelvic inflammatory disease. Vaginal thrush can complicate broad-spectrum antibiotic treatment.
2. Pelvic congestion syndrome
Most patients respond to conservative measures: these include reassurance and suppression of ovarian activity. Surgery is not required.
3. Endometriosis
The treatment of endometriosis depends upon the severity of symptoms, the age of the patient, the extent of the disease and the desire for children. Conservative treatment is indicated in young patients with mild symptoms and minimal findings on pelvic examination. If the symptoms become more severe, if pelvic masses develop or if infertility becomes a problem, then measures to suppress ovulation are added to simple treatment with analgesics. Hormone therapy to suppress ovulation may be contraindicated in patients with fibroids or when there is a history of venous thrombosis or breast cancer. Conservative surgery by laparoscopic cauterisation of implants may improve fertility. Occasionally, definitive excisional surgery, with removal of uterus, tubes and ovaries, must be performed in order to cure the condition. Endometrial bowel implants can mimic cancers, so histologic confirmation of diagnosis is always required.
4. Spastic colon and diverticular disease
Most patients with functional bowel disease will respond to the gradual introduction of bulking agents and fibre to the diet, after colonoscopic exclusion of other causes. Treatment may be unsuccessful if introduced too precipitously in patients with obstructed defaecation and the descending perineum syndrome, where the concomitant correction of poor defaecatory habits is of primary importance. Reassurance that cancer is not present is also very important. Chronic diverticulitis may require surgical treatment, but most patients with localised diverticulitis in the pelvis will settle with conservative management. This includes high-fibre diet, anti-spasmodic agents and dietitian review. Prolonged antibiotic treatment should be avoided. Surgery for complicated diverticular disease is preferably by resection and primary anastomosis. A covering loop ileostomy may be indicated to protect the anastomosis.
5. Less common causes
Pelvic abscess secondary to diverticulitis or appendicitis. Such patients occasionally present with lower abdominal discomfort and mucous diarrhoea, extending over several weeks. Symptoms are associated with fever and varying degrees of toxicity, anorexia and weight loss.
With pelvic diverticulitis, a previous history of altered bowel habit is common. Bimanual palpation reveals a large firm, tender, tethered mass in the pouch of Douglas. Patients with pelvic appendiceal abscesses are usually much younger, but otherwise have similar clinical findings to those of pelvic diverticulitis. A recent episode of pain suggestive of appendicitis, with apparent resolution, is common. This episode is often assessed to have been a bout of gastroenteritis. Pelvic examination reveals a firm mass in the pouch of Douglas, which in time may soften and point into the rectum or vagina. For pelvic appendiceal abscess laparotomy and appendicectomy with drainage of the pelvic abscess is indicated. Diverticular abscesses also require abdominal surgery and drainage. Pelvic abscess resulting from other causes, such as a postoperative sequel of perforated ulcer, may progress to a large tense fluctuant mass protruding into the anterior rectal wall or vagina. These abscesses may be drained through the rectum or vagina, after aspiration to establish the presence of pus (Fig 7.26).

Figure 7.26 Drainage of pelvic abscess
A: An artery forcep is guided over the index finger through the point of maximum softening in the anterior rectal wall.
Chronic prostatitis. Pain is usually felt more in the perineum and may be associated with low backache. There may be mild symptoms of bladder irritation and penile pain on micturition. The prostate is enlarged, firm, irregular and tender. Calcific deposits may be palpable. The lesion can be difficult to distinguish from prostatic carcinoma on examination. Urethral massage produces a urethral discharge, which should be cultured and examined cytologically. The organisms responsible for prostatitis are best detected in a specimen of early morning urine. Chronic prostatitis is a difficult condition to treat. Antibiotic treatment is usually unsuccessful. Regular prostatic massage may be helpful.
Chronic anal fissure. This common disorder usually presents as acute anal pain (Ch 7.22). Bright red bleeding at the end of defaecation is also common. Fissures are characterised by their chronicity and may be neglected for years, especially in patients where the condition developed during pregnancy or childbirth. The complaint is often of a vague continuous pelvic and perineal ache, which radiates down the legs and may not be localised by the patient to the anus. Unless a careful examination is performed, these lesions can be missed because they are located above the anal verge. Anal spasm will also inhibit the performance of an adequate examination.
Chronic cervicitis. Chronic cervicitis often dates from childbirth. Pain is usually associated with profuse vaginal discharge. The key to diagnosis is that the cervix itself is tender and pressure on it will reproduce the pain. There is no uterine or adnexal tenderness. Most of the other less common causes of chronic pelvic pain are diagnosed on pelvic examination. Diagnosis may be aided by ultrasound of the pelvis, laparoscopy or occasionally angiography.
Retroperitoneal tumours. Most retroperitoneal tumours are of connective tissue origin. The clinical presentation is characterised by lower abdominal discomfort or pain, fever and, after some time, the appearance of a lower abdominal mass. Most will need to be explored with a view to excision. Careful delineation of the lesion before surgery by organ imaging helps in the planning of resection.
7.8 Bowel obstruction
Bowel obstruction usually presents as a classic syndrome complex comprising symptoms and signs of pain (colic), vomiting, distension, constipation (for flatus and faeces) and features of body water and electrolyte depletion. Most patients with small bowel obstruction present acutely, with pain an early symptom. This contrasts to malignant large bowel obstruction where the onset of symptoms is gradual, pain is late and altered bowel habit is the usual first presentation. A significant clinical problem in general practice is that severe gastroenteritis can resemble acute intestinal obstruction.
Initial assessment: small or large bowel obstruction?
The diagnosis of bowel obstruction is usually made soon after the patient arrives at hospital with a diagnosis of acute abdomen (Ch 7.2) — from the clinical features, aided by a plain X-ray of the abdomen. The important questions to answer then are whether the obstruction is of small or large bowel, whether due to functional or mechanical causes and whether strangulation is present.
In most cases small bowel and large bowel obstruction can be distinguished from each other, firstly by their different clinical patterns. For small bowel obstruction the usual sequence is: abdominal colic — vomiting — distension — constipation. For large bowel obstruction the sequence is: constipation — distension — pain — vomiting.
In small bowel obstruction, supine and erect plain abdominal films reveal a central ladder-like pattern of dilated loops of small bowel with air-fluid levels (Fig 7.12). The colon is devoid of gas, unless right colonic carcinoma is the cause of small bowel dilatation when a small portion of dilated colon may be seen on X-ray.
In large bowel obstruction, plain abdominal films reveal gas-filled and distended colon, with maximal dilatation of the caecum, especially if the ileocaecal valve is competent.
The major management problems in small bowel obstruction are restoring extracellular fluid depletion, detecting strangulation and the indications for and timing of surgery (Box 7.8).
Box 7.8
Management plans in small bowel obstruction
Correct extracellular fluid loss
Detect strangulation
Consider indications for and timing of surgery
A major management problem in large bowel obstruction is to detect and relieve closed-loop obstruction of the colon between the tumour and the ileocaecal valve, with impending perforation of the caecum.
Small bowel obstruction
Common causes
1. Adhesive obstruction
2. Hernias
3. Malignancies
4. Sigmoid diverticulitis and intraperitoneal abscess
5. Gallstone ileus and enteroliths
History
In simple small bowel obstruction the character of the pain is colicky (with a crescendo–decrescendo pattern). The pain is felt symmetrically in the periumbilical region. Vomiting is early and profuse and eventually bile-stained then faeculent, if the obstruction is complete. The degree of abdominal distension depends on the site of obstruction and whether it is complete. Distension tends to be central. The patient may continue to have one or two small bowel actions but constipation is later complete for faeces and (more importantly) for flatus. Clinical manifestations of small bowel obstruction vary according to the level of blockage (Fig 7.27). High small bowel obstruction may present with profuse vomiting that seldom becomes faeculent — even when prolonged — and little distension. Pain may be less severe, rather than the classic cramping, colicky pain. Most episodes of small bowel obstruction are sudden in onset and complete, although spontaneous relief may follow after a period. Chronic small bowel obstruction is less common and is mainly seen in Crohn’s disease and small bowel malignancy.

Figure 7.27 Clinical presentations of small bowel obstruction depend upon the level and cause of obstruction
A: high obstruction (adhesive): vomiting early and profuse, pain atypical or absent, distention minimal, X-ray may be negative; B: distal small bowel, strangulated hernia: colicky pain, distension, vomiting; local symptoms not always prominent; C: obstructing carcinoma, right colon, distal small bowel obstruction
1. Adhesive obstruction
Adhesive obstruction is a late postoperative complication. Prevention of adhesions is achieved by a surgical technique that avoids ischaemia of tissue and preserves omentum, but the disease process itself (e.g. ruptured appendicitis, ruptured spleen) is often a major cause of postoperative adhesions. The operations most commonly followed by adhesive obstruction are lower abdominal — appendicectomy, hysterectomy and colorectal resection — but any intraperitoneal operation can be followed by adhesive bowel obstruction.
2. Hernias
Indirect inguinal, femoral and umbilical hernias are the hernias most commonly complicated by small bowel obstruction. Femoral hernia is more prone to this complication than either of the others. The onset is usually sudden, with the hernia becoming tense, tender and irreducible and loss of the cough impulse on examination. Femoral hernias can, however, be difficult to diagnose as the cause of obstruction. The femoral hernia is often small, with little local pain, the main initial complaint being vomiting and central colic. A femoral hernia often has been present for many years unnoticed, so a history of hernia may not be forthcoming. Richter’s hernia is the term used to describe strangulation of part of the bowel circumference within a hernial sac (usually femoral). Obstructive symptoms and signs are often delayed in these hernias. If the omentum alone is obstructed and strangulated within a hernia, abdominal pain is vague, unless general peritonitis ensues.
3. Malignancies
Obstructing carcinoma of the caecum or proximal carcinoma of the right colon can present as small bowel obstruction. This diagnosis must be kept high on the list in older patients with small bowel obstruction, even if previous abdominal surgery has been performed, especially if the surgery was performed many years before. Suspicion of a colonic cause is also suggested by the presence of right colonic distension on X-ray. Barium enema or colonoscopy is indicated in these patients even if the obstruction resolves. Surgery must not be delayed if the clinical signs do not rapidly remit.
Secondary malignancy may cause obstruction, especially with peritoneal seedings. Obstruction may be acute in presentation but is often slower in onset and initially incomplete. The common primary sites of such secondary malignant tumours are the ovary, melanoma and colorectum. Melanoma, and also renal cell carcinoma, may truly metastasise to the small bowel. In these cases, intussusception may occur as the mechanism of small bowel obstruction. Intussusception due to enlarged Peyer’s patch is a common cause of bowel obstruction and possibly acute abdomen in infants. The mechanism, imaging and pathology of this are shown in Figure 7.28.

Figure 7.28 Intussusception
1: possible foci for intussusception and diagrammatic representation of mechanism; 2: barium enema of the small bowel ‘into’ colon intussusception; 3: pathological specimen illustrating C — colon, I — intestine and A — appendix
Credit: Based on Burkitt, 2007
4. Sigmoid diverticulitis and intraperitoneal abscess
Prodromal symptoms of diverticular disease are accompanied by developing small bowel obstruction due to adhesion of a small-bowel loop to the diverticular mass or abscess. Adherence to an intraperitoneal inflammatory focus or abscess is an important cause of small bowel obstruction in the elderly (Fig 7.29) without previous scars or hernias. Usually, systemic manifestations of the abscess will coexist.

Figure 7.29 Complicated diverticular disease
Small bowel obstruction secondary to diverticulitis with small bowel adherence to sigmoid colon affected by diverticulitis.
5. Gallstone ileus and enteroliths
Gallstone ileus is due to acute cholecystitis causing a fistula between Hartmann’s pouch of the gall bladder and the second part of the duodenum. The stone, if too large, will impact in the lower ileum. The condition should always be suspected in elderly patients in whom intermittent small bowel obstruction progresses to complete obstruction with no hernia or previous operation indicating a likely cause. A previous history of gall bladder pain is helpful, but often not present. Radiological signs of gas in the biliary tree confirm that a fistula exists between the gut and biliary system. The calculus is not usually radio-opaque.
Enterolith. Previous gastric resection or diversion makes obstruction by an enterolith more common. Focal concretions in the lumen of the gut are called bezoars and can be trichobezoars (hairballs, seen in patients with psychiatric disorders) or more commonly phytobezoars (pith of fruits or vegetables). It is important for patients after gastric resection to avoid fruits such as oranges because of the indigestible pith. Adhesions due to previous peritonitis or abdominal surgery may also enhance the obstructive potential of food boluses consisting of indigestible material.
Examination
Local signs of mechanical obstruction include distension, which is predominantly central, visible peristalsis and hyperactive bowel sounds. Signs of peritonitis suggest strangulation, as does a tender mass. A mass may also be palpable with carcinoma of the caecum or intraperitoneal abscess. General signs may be minimal in the early stages of obstruction, but signs of extracellular fluid loss increasingly develop. The fluid lost is isotonic, so changes in serum electrolyte levels are minimal, as body fluid osmolality remains constant. Thirst is therefore not a feature, unless there is significant blood loss into a loop of strangulated bowel or prolonged water depletion.
The general clinical effects depend upon the amount of extracellular fluid lost, as follows.
• Reduced tissue turgor, with a dry firm tongue and loss of skin elasticity, is detectable with loss of 3 L of fluid (or 5% of the body weight in an average adult).
• Tachycardia and postural hypotension develop with 4–5 L of fluid loss (or a deficiency of from 7–10% of body weight).
• Shock and oliguria appear with more than 6 L loss (or more than 10% loss of body weight).
Shock is more likely to be due to strangulation or some other cause unless it is clearly related to a prolonged history of vomiting from unrelieved bowel obstruction.
Diagnostic plan
Haematological and biochemical examination
Haemoconcentration with a raised haemoglobin is a possible early finding in patients with small bowel obstruction, due to extracellular fluid loss. Anaemia suggests that blood loss has occurred into a long strangulated loop or chronic blood loss from a colonic malignancy. A hypochromic microcytic anaemia strongly suggests malignant obstruction. Leucocytosis suggests sepsis or strangulation. The serum ureaand creatinine levels are increased pari passuwith the degree of prerenal injury due to extracellular fluid loss.
Radiology
Plain X-ray film of the chest and vertical and supine abdominal films will show small bowel dilatation, with the ladder pattern of jejunal dilatation or the ‘sausage’-like appearance of ileal dilatation (Fig 7.30). The combination of gas and fluid-filled loops of small bowel on the erect film with an empty colon, particularly if the gas-fluid levels are arranged in a ‘string of beads’ pattern, is virtually diagnostic of small bowel obstruction. Intraperitoneal fluid and gas (‘soap bubble’ sign) suggest perforation with abscess formation. An area of ‘white out’ or ‘pseudo-tumour’ suggests the presence of a fluid-filled, distended, strangulated small bowel loop. The strangulated loop may be air-filled giving a ‘coffee bean’ sign on X-ray. Opaque gallstones and gas in the biliary tree should be looked for routinely. This finding suggests the possibility of gallstone ileus, but may follow previous papillotomy or sphincterotomy.

Figure 7.30 Diagrammatic depiction of radiological features of small bowel obstruction
A: erect film; B: supine film
In closed loop obstruction, strangulation, ischaemic necrosis, peritonitis, perforation and septicaemic shock occur much earlier in the course of the disease. Continuous pain is present, with early formation of a tender mass and signs of peritonitis. This type of strangulation is the usual one seen in complicated internal or external hernias.
Gastrograffin follow-through. This can be performed by the delivery of contrast material via a nasal tube passed into the upper small bowel. This can be helpful in the less acute case when no hernia or abdominal scar exists on physical examination but a small bowel lesion is suspected or when high small bowel obstruction is suspected even though the plain X-ray is unremarkable or when it is not possible to distinguish either the cause of obstruction or whether it is of small or large bowel. Such a study, due to the osmotic effect of the contrast agent, may also be therapeutic for small bowel obstructions precipitated by food bolus.
Abdominal CT scan is increasingly used to confirm the diagnosis of small bowel obstruction (where a transition zone between dilated and collapsed bowel can be seen) and also the possible cause (such as a tumour).
Treatment plan
In small bowel obstruction the main pathophysiological processes producing clinical features and influencing treatment are as follows.
Loss of alimentary secretions and depletion of extracellular fluid. With high small bowel obstruction, early and profuse vomiting causes rapid loss of extracellular fluid (‘saline’). With more distal small bowel obstruction, vomiting is later and loss from the body occurs at a somewhat slower rate. Isotonic fluid accumulates within distended small bowel above the obstruction.
Distension of the small bowel. Obstruction causes distension of the small bowel because of accumulated swallowed gas, continuing secretion, failure of onward passage and decreased reabsorption of fluid.
Stasis. Stasis of fluid above the blockage results in bacterial multiplication within the lumen. Developing bowel ischaemia leads to rapid entry of organisms into the circulation, with septicaemia.
Strangulation. Strangulation comprises developing ischaemia and threatened necrosis of the obstructed segment. Strangulation can occur as a result of bowel volvulus where there is mesenteric vascular compromise. Ischaemic necrosis develops more rapidly in cases of internal or external hernias; the narrow neck to the hernial defect constricts the blood supply of the contents.
The clinical effects of strangulation depend upon the length of the entrapped or twisted loop. With strangulation of a large loop of small bowel, haemorrhage and fluid loss can be sufficient to produce hypovolaemic shock at an early stage. Signs of peritonitis may be relatively late.
Management of small bowel obstruction thus comprises nasogastric suction and intravenous resuscitation (in all cases) and surgical intervention to relieve persisting obstruction or strangulation.
Nasogastric suction
This is indicated to decompress the bowel, to control vomiting and to empty the stomach so that the danger of pulmonary aspiration is minimised, especially at the time of induction of anaesthesia. The volume of aspirate lost is replaced by intravenous saline. Prolonged decompression by nasogastric suction is used in patients when conservative treatment is indicated, such as for recurrent adhesive obstruction.
Resuscitation
The major principle is to correct ECF (‘saline’) depletion (Box 7.9).
Box 7.9
Extracellular fluid (ECF) replacement in small bowel obstruction*
Volume of replacement fluid
Isotonic fluid and electrolyte intravenously to replace:
![]() established fluid deficit
established fluid deficit
![]() continuing losses (aspirate, fistula loss, vomitus)
continuing losses (aspirate, fistula loss, vomitus)
![]() basal daily requirements.
basal daily requirements.
Composition of replacement fluid
Isotonic saline or Hartmann’s solution, plus potassium requirements when urine output is adequate.
Rate of replacement
Depends upon clinical response, urine output and central venous pressure.
* Failure of adequate resuscitation suggests strangulation
Volume of replacement is calculated by adding:
• the estimated fluid deficit on admission
• the continuing sensible loss of aspirate, vomitus or fistula loss over each 24-hour period
• basal daily requirements of fluid and electrolyte.
The fluid deficit is estimated from clinical and biochemical features. The normal haemocrit is 40%. The normal blood volume is 7% (male) or 6.5% (female) of the body weight.
Composition of the fluid given is isotonic saline or Hartmann’s solution, plus potassium additives according to the estimated potassium deficiency and daily requirement (1 mmol/kg/day). Potassium infusion is not commenced until urine output is satisfactory.
The rate of intravenous fluid replacement is judged according to urine output and clinical response, aided by central venous pressure estimations in the severely septic or shocked patient. Shock is corrected by transfusion of colloid. Intranasal oxygen is given in all shocked patients. If anaemia is present, packed cells are used. The rate of transfusion in shocked patients is monitored by clinical response and central venous pressure. Blood culture is taken if the patient is febrile and antibiotics are started early to assist in resuscitation, reduce the toxic effects of septicaemia and to prepare the patient for definitive surgical treatment.
In high-risk patients with chronic pulmonary disease or chronic renal impairment, volume loading should be done cautiously; the rate is best controlled by monitoring the left atrial pressure. Endotracheal intubation and positive pressure ventilation may be necessary in critically ill patients.
Surgical intervention
Surgery is indicated in nonstrangulated obstruction when: the cause of obstruction is not known; adhesive obstruction has not responded to conservative treatment within 48 hours of presentation; and there is any hint of developing strangulation. The timing of surgery depends upon the cause of obstruction, the severity of extracellular fluid loss, the response to resuscitation and whether strangulation is present. If possible, surgery is optimally carried out after rehydration has been achieved and tissue perfusion has been restored.
Surgical intervention should, however, always be prompt if strangulation is suspected. If the pain becomes continuous, if shock and fever appear early in the course of obstruction, if there is a poor response to resuscitative measures and if signs of local peritonitis are present, then a strangulated obstruction that requires urgent surgery must be assumed. These features may also be associated with a septic focus (such as peridiverticular or appendiceal abscess) causing bowel obstruction. Both a strangulated loop obstruction and an abscess with obstruction are often associated with a tender mass on physical examination. Early surgery in patients with strangulated obstruction is first-line treatment. The obstruction is usually released by dividing the relevant adhesions or the hernial neck. The dilated small bowel is decompressed into the stomach and then through a nasogastric tube. Nonviable bowel is resected; the creation of a temporary stoma is considered when there is obstruction distal to the primary site of obstruction, such as is found in Crohn’s disease, or if the blood supply to the resection margins in doubt.
Management of other causes
Initially conservative treatment is given for nonstrangulated postoperative obstruction, presumed secondary malignancy (this may not be easily diagnosed preoperatively), radiation enteritis and in cases of numerous previous episodes of spontaneously resolving obstruction. A long intestinal tube may be of value in these patients and is inserted at laparotomy.
Large bowel obstruction
Most cases of large bowel obstruction are due to carcinoma of the distal large bowel. The history is usually more insidious, but a relatively short history of altered bowel habit can be followed by sudden complete constipation for faeces and flatus, with early and marked distension, followed by pain.
Extracellular fluid loss is not a feature of large bowel obstruction unless a significant degree of secondary small bowel dilatation occurs (Fig 7.31). Careful preoperative preparation and resuscitation is, however, necessary in most cases. The patients are often elderly, with concurrent medical illness, have iron deficiency anaemia from chronic blood loss and may be severely ill with sepsis secondary to impending or established perforation.

Figure 7.31 Large bowel obstruction
A: often the ileocaecal valve prevents retrograde decompression into the small bowel, creating a closed loop obstruction; B: alternatively, with spontaneous decompression into the small bowel the presentation is less acute and the danger of caecal perforation is less
Common causes
1. Colonic carcinoma
2. Sigmoid or caecal volvulus
3. Diverticular disease
4. Colonic pseudo-obstruction
5. Faecal impaction
History and physical examination
1. Colonic carcinoma
In most patients with obstructing carcinoma of the distal colon a history of increasing constipation and increasing use of laxatives and progressive distension with discomfort precedes the onset of acute symptoms. Pain is often late in onset. Pain may be colicky or continuous and is felt in the lower abdomen, pelvis, left iliac fossa or back. Nausea is common, but vomiting is generally a late feature. On examination there may be fullness and tenderness in the flanks and right iliac fossa or a uniform fullness due to associated small bowel distension. A mass may be palpable with caecal or sigmoid tumours. Irregular hepatomegaly may be found due to secondaries in the liver. On rectal examination and sigmoidoscopy, an extrarectal carcinoma of the sigmoid may be felt or a rectosigmoid carcinoma seen. Blood may also be seen in the rectum or sigmoid.
2. Sigmoid or caecal volvulus
Volvulus of the colon accounts for about 10% of cases of large bowel obstruction. Most are due to sigmoid volvulus; about 2–3% are due to caecal volvulus. Sigmoid volvulus can occur at any age but is most common in the aged, immobilised and constipated patient and in intellectually impaired patients. Some patients with sigmoid volvulus have a history of recurrent episodic lower abdominal pain and distension, relieved by explosive diarrhoea, until the eventual acute onset of continuous, severe, ischaemic pain and massive distension of unresolved sigmoid volvulus. A spiral fold at the site of obstruction may be seen on sigmoidoscopy at about 15–20 cm above the anus (Fig 7.32).

Figure 7.32 A: Diagram of sigmoid volvulus: direction of twist is usually anticlockwise B: Plain abdominal X-ray of sigmoid volvulus showing a distended loop arising from the pelvis; on sigmoidoscopy the site of obstruction can be seen at about 15–20 cm from the anal verge.
Image B from Duthie, 2007
3. Diverticular disease
Diverticular disease is an uncommon cause of sudden complete obstruction. Acute obstruction may follow fibre-bolus obstruction, occasionally peridiverticular abscess and rarely, chronic inflammatory fibrosis with stricture formation. An extrarectal mass may be felt on rectal examination. Usually the sigmoidoscope cannot be negotiated above the rectosigmoid junction.
4. Colonic pseudo-obstruction
Acute functional colonic ileus (colonic pseudo-obstruction) is the cause of large bowel obstruction in about 5–10% of cases. The condition is seen most commonly in bedridden patients with systemic illnesses such as renal or heart failure, diabetes, spinal disease or septicaemia. It is also associated with pelvic trauma or surgery, such as hip fractures and gynaecological surgery. Colonic motility may be directly impaired by drugs or indirectly by hypokalaemia induced by drugs or by diarrhoea. The danger of perforation is less than with mechanical obstruction, although massive distension of the caecum can occur. Colonic pseudo-obstruction can be very difficult to distinguish from a mechanical cause. Caecal volvulus can also occur subsequently, adding mechanical obstruction to the pathology.
5. Faecal impaction
Faecal impaction is also seen in the aged, bedridden, debilitated patient. It may closely resemble malignant obstruction in its clinical presentation. A feature of the history is ‘faecal incontinence’, which is in fact spurious diarrhoea secondary to partial obstruction and stercoral irritation of the colon.
Diagnostic plan
Plain X-ray. On the plain film a closed loop obstruction of the large bowel gives the appearance of a picture frame outlining the abdominal cavity. Variable degrees of decompression often occur into the small bowel. The colon can be distinguished from the small bowel by its haustral markings, which do not cross the entire lumen of the distended bowel, whereas the valvulae conniventes in the jejunum do. Gas under the diaphragm secondary to perforation of a carcinoma or diverticular mass or sigmoid volvulus is found occasionally with large bowel obstruction. The ‘soap bubble’ sign with loss of the psoas shadow suggests local perforation of a colonic carcinoma with abscess. Gas in the rectum on a supine film is strongly suggestive of functional large bowel obstruction, provided the gas has not already been introduced into the rectum by sigmoidoscopy or enema. The Frimann-Dahl or ‘M’ sign is a feature of sigmoid volvulus: three lines of apposed bowel are seen radiating from the pelvis and outlining gas-filled, grossly distended sigmoid colon (Fig 7.32).
A CT scan of the abdomen and pelvis (with or without rectal contrast) may define a mechanical colonic obstruction and a causative mass lesion. In the case of carcinoma, it may also be useful to demonstrate the presence of liver metastases.
Barium enema. A limited barium enema is occasionally required to establish the diagnosis of malignant obstruction and to exclude nonmechanical (functional) large bowel obstruction. A ‘parrot’s beak’ sign of cut-off at the site of sigmoid volvulus may be seen or an apple-core carcinomatous stricture.
Colonoscopy is not usually indicated with small or large bowel obstruction because of the danger of perforation secondary to air insufflations, because the bowel is imperfectly prepared and because the diagnosis usually can be easily established by other methods of investigation. If the obstruction settles, then elective colonoscopy is often necessary to diagnose carcinomatous or inflammatory strictures. Colonoscopy has been used to successfully decompress the colon in functional large bowel obstruction and sometimes to treat advanced rectal tumours with laser photocoagulation or stenting to relieve obstruction.
Treatment plan
1. Colonic carcinoma
With incomplete obstruction, an enema may relieve the obstruction sufficiently to allow more time for preoperative preparation. Concurrent illness is controlled as much as is possible in the time available. Anaemia is corrected by transfusion of packed red cells well prior to surgery. Extracellular fluid deficiency, if present, is corrected and prophylactic parenteral antibiotics are given with premedication. In some patients, consideration is given to colonic stenting as the initial decompression procedure for distal colonic stenotic lesions.
The choice of surgical procedure depends upon the site of obstruction, the tumour stage, the general health and age of the patient and the technical difficulties encountered at operation. The major technical difficulties encountered are obesity and faecal loading of the proximal colon. For lesions in the right colon or right transverse colon an extended right hemicolectomy with primary anastomosis is the procedure of choice. For the more common obstructing lesions in the left colon in poor-risk cases a three-stage operative approach may be indicated if stenting is not possible. The first stage is a laparotomy and decompression of the colon by loop transverse colostomy or loop ileostomy; the second is resection of the tumour; and the third is closure of the colostomy or ileostomy. In younger patients a subtotal colectomy with ileocolonic anastomosis or intra-operative irrigation and primary anastomosis may be possible alternatives. A loop ileostomy would be made to protect the anastomosis. In patients with advanced lower left colonic carcinomas or obstructions due to a diverticular and peridiverticular mass, end colostomy with oversewing of the distal rectal stump (Hartmann’s procedure) may be indicated. Bypass of metastatic malignancies is indicated for multiple obstructing lesions or with a fixed tumour. Perforated tumours require exteriorisation–resection by a Paul-Mikulicz or Hartmann’s technique.
2. Sigmoid or caecal volvulus
In early cases the twisted sigmoid colon can be deflated without surgery by gently passing a soft rectal tube through a rigid sigmoidoscope into the twisted loop. Colonoscopy should be attempted only with extreme caution. Laparotomy is indicated when signs of strangulation or perforation are present or when sigmoidoscopic deflation is unsuccessful. Later elective resection is indicated in relatively healthy patients, as recurrence can be expected to occur in at least half the patients. Emergency resection and anastomosis may occasionally be practical in patients in good general condition, but it is usually safer to bring the two ends out by exteriorisation–resection as a double-barrelled (Paul-Mikulicz) colostomy in the left iliac fossa as a first stage or to perform a Hartmann’s procedure.
3. Diverticular obstruction
Diverticular disease with obstruction can usually be treated by delayed semi-elective primary resection and anastomosis.
4. Colonic pseudo-obstruction
In functional large bowel obstruction, operative decompression of the caecum by caecostomy or colostomy may occasionally be required if the distension does not subside with enemas or colonoscopic deflation, as well as attending to possible drug and electrolyte causes. Some cases may also respond to medical treatment with neostigmine, provided mechanical obstruction has been definitively excluded by appropriate imaging.
5. Faecal impaction
Faecal impaction requires a combination of digital disimpaction, enemas and purgation. Subsequent investigations should establish that no mechanical cause of obstruction is present.
7.9 Abdominal mass
An abdominal mass may be discovered on routine physical examination or during assessment of a patient for deterioration in general health. These forms of presentation are surprisingly common and many common diseases can present in this way. For example, patients with mucocele of the gall bladder, pseudocyst of the pancreas, carcinoma of the colon and carcinoma of the stomach often seek medical help because of vague deterioration in general health. A mass is then found on examination, without a proffered history of abdominal pain or of altered bowel habit, which might indicate the cause of the abdominal swelling. A careful further history and systems review will, however, often reveal subtle symptoms. These, together with a precise delineation of the mass, usually suggest the clinical diagnosis.
General assessment
1. Confirm a mass is present
The first step is to be sure that a mass is present and is abnormal. In the normal abdomen, particularly in patients with emphysema and asthenic habitus, the lower poles of the left and right kidneys may be palpable. A normal liver is rarely palpable, except in infants. Palpating the liver edge with certainty requires enlargement or an abnormally firm consistency or both. Percussion helps establish the level of the liver’s lower and upper borders. The normal spleen is not palpable. In the lower abdomen, faeces may normally be palpable in the colon as indentable masses. The normal sigmoid colon is sometimes felt as an indistinct tube in the left iliac fossa; this finding is also a feature of spastic colon. Abdominal wall swellings can be excluded by contracting the abdominal musculature; subcutaneous swellings will remain clearly palpable. A tendinous intersection of the rectus abdominis is often mistaken for the liver edge in the epigastrium. Abdominal wall hernias also produce localised masses but are usually readily identified by site, by cough impulse and by reducibility through a palpable defect (Ch 8.3). A prominent xiphisternum may be noted as an upper epigastric mass of concern.
2. Categorise the mass
As with abdominal pain, intra-abdominal masses are best classified according to their site within the abdomen (Figs 7.33 and 7.34). Other features on physical examination of particular importance in establishing the diagnosis are the mobility and consistency of the mass. Most abdominal swellings are localised into one of the abdominal quadrants. Occasionally, multiple abdominal masses occur; these may be due to malignant peritoneal involvement, faecal impaction and sometimes multiple skip lesions of Crohn’s disease.

Figure 7.33 Upper abdominal masses
1: enlarged liver; 2: Riedel’s lobe; 3: carcinoma of the right colon; 4, 5: gall bladder; 6: rounder lower poles of both kidneys; 7: abdominal aortic aneurysm; 8: splenomegaly; 9: pseudocyst of the pancreas

Figure 7.34 Lower abdominal masses
1: full urinary bladder; 2: ovarian cyst; 3: fibromyomas of uterus; 4: tender mass of appendiceal abscess; 5: pregnant uterus; 6: tender mass of diverticulitis
Some solitary masses (of liver, spleen, kidney, uterus, ovary or the retroperitoneum) may become so large that they present as abdominal distension (Ch 7.10). Most of these lesions, when smaller, can present as solitary abdominal masses.
An important part of the assessment of lower abdominal masses is to ensure that the bladder is empty — by catheterisation if necessary — before physical examination.
Liver and splenic swellings usually give characteristic features on clinical examination. Hepatomegaly is considered on page 240 and that of the spleen on page 241 where an approach to generalised enlargement of both liver and spleen are considered as a separate problem.
In regard to subsequent diagnostic and treatment plans, diagnostic laparotomy is now practised less than it used to be; improved diagnostic power is given by ultrasound, CT scanning and CT-guided percutaneous biopsy, laproscopy and perhaps MRI scanning. These techniques are very helpful in localising lesions anatomically. Percutaneous or endoscopic biopsy often clinch the diagnosis and also assist preoperative planning. Laparotomy often remains the only sure method of determining resectability of tumours.
Right upper quadrant mass
Common causes
1. Single mass in the liver
2. Mucocele of the gall bladder
3. Right renal mass
4. Carcinoma of the right colon
History and examination
The patient may recall on direct questioning a past episode of upper abdominal pain suggestive of cholecystitis, haematuria suggesting a renal carcinoma or change in bowel habit or epigastric discomfort related to carcinoma of the right colon. A history of residence where hydatid disease is endemic may be important.
1. Single mass in the liver
This is a common diagnostic problem. Common causes include Riedel’s lobe, which is a variant of normal liver anatomy and may be mistaken for an enlarged gall bladder. Riedel’s lobe is an extension of the right lobe of the liver below the costal margin in the anterior axillary line (Fig 7.33). The colorectum, breast and lung are the most common primary tumours to present with a single palpable metastatic nodulein the liver. Hydatid disease is an important cause of an asymptomatic solitary liver mass (Fig 7.35). Other causes include simple cyst, polycystic disease, cystadenoma, liver abscess and primary hepatic carcinoma. About half the patients with liver abscess have a previous history of liver trauma. Patients with amoebic abscess may give a past history of dysentery contracted in a foreign country.

Figure 7.35 The life cycle of the hydatid parasite
A: Primary host — dog. After eating sheep offal (liver, lung) containing hydatid cysts, the scolices pass into the small intestine and grow into the adult tapeworm Echinococcus granulosus. The eggs produced by the terminal segments are shed and passed in canine faeces. B:Secondary hosts — sheep and humans. Ova are ingested, the chitinous enveloped is dissolved by acid and pepsin in the stomach. After absorption, ova pass via the portal blood to the liver and grow into adult hydatid cysts containing scolices.
2. Mucocele of the gall bladder
This can present as an asymptomatic mass beneath the tip of the right ninth costal cartilage. The mass is smooth and rounded, moves on respiration, cannot be separated by palpation from the liver and is dull to percussion.
3. Right renal swelling
This lies more laterally in the flank and extends posteriorly in the paracolic gutter. A renal swelling is also suggested by palpation of its rounded lower pole, moderate movement on respiration and resonance to percussion due to overlying (right) colon. A renal mass is bimanually palpable and ballotable.
4. Carcinoma of the right colon
A hepatic flexure mass may be difficult to distinguish on physical examination from a mass in the gall bladder or liver. A colonic mass tends to be hard and irregular, with less movement on respiration. It is often possible to get between a colonic mass and the liver, but the tumour may sometimes directly invade the liver and move with it. Chronic hydatid cyst rarely is extruded from the liver to produce an extrahepatic smooth lesion, similar to a colonic mass.
Diagnostic plan
A provisional diagnosis should always be attempted from the physical findings and clinical history.
Ultrasound, CT scanning and percutaneous guided biopsy are valuable means of further clarifying the diagnosis. The CT scan displays the anatomy of the liver to greater advantage than ultrasonography or a radionuclide scan. With fatty infiltration the liver is of lower density and the veins stand out. In cirrhosis the liver may be contracted and nodular, ascites may be detectable and the spleen is often enlarged. The CT scan can show whether a lesion is within the liver or is extrahepatic and can show attenuation characteristics of the lesion itself, distinguishing between haemangioma, hydatid disease, abscess, carcinoma and simple cyst. CT scanning is usually performed before and after the injection of intravenous contrast medium. Except when vascular, metastatic disease is usually of lower attenuation than normal liver and will stand out to greater degree after injection of contrast medium has rendered the liver parenchyma more dense. Primary (benign and malignant) tumours are also usually of lower density than normal liver parenchyma unless the parenchyma is infiltrated by fat. Cysts of the liver are sharply defined and contain fluid of low density similar to water. Hydatid cysts may have a calcified wall, contain denser fluid, are often multilocular and daughter cysts may be identified within the main cyst. Hydatid serology (ELISA, latex agglutination) also assists in the diagnosis of hydatid disease before surgery, although CT scanning has a high degree of diagnostic specificity. Abscesses have a lower density than normal liver parenchyma and can contain gas, readily detected by CT scan. A CT-guided percutaneous biopsy is often the next step, except in the case of hydatid disease. Percutaneous needling or core biopsy must not be performed if there is any chance of a liver cyst being a hydatid.
Gallstones are best detected by ultrasound examination. Intravenous pyelography and arteriography may be necessary to supplement the findings of CT scanning, although CT angiography will allow most renal masses to be interrogated sufficiently. Colonoscopy is necessary when the diagnosis of carcinoma of the colon is suspected.
Treatment plan
Solitary liver tumour. Both secondary and primary carcinoma may be localised and thus potentially resectable. Angiography, as well as CT scanning, is necessary in patients being considered for surgery, to assist in surgical planning. Many malignant tumours are inoperable, because of either direct spread to involve body wall outside the capsule of the liver or involvement of the inferior vena cava, hilum of the liver or caudate lobe or metastatic disease. Up to 80% of the normal liver can be removed; the residual liver regenerates rapidly. Cirrhosis is a relative contraindication to resection because of technical difficulty, insufficient liver reserve, poor general health and nutrition and limited regenerative capacity. Hepatic resections are classified as segmental or nonsegmental. Wedge resection is an example of nonsegmental resection. Segmental resections are planned according to the vascular anatomy of the liver (Fig 7.36). Careful monitoring and metabolic support is necessary after surgery if more than 50% the liver has been removed. Surgical resection is the only form of treatment for primary or secondary tumour that offers a chance of cure.

Figure 7.36 The segmental anatomy of the liver displayed ex vivo
Right hepatectomy: segments v, vi, vii, viii; left hepatectomy: segments ii, iii, iv; right lobectomy: segments iv, v, vi, vii, viii; left lobectomy: segments ii, iii; segments v, vi, vii, viii are supplied by the right hepatic artery, segments i, ii, iii, iv are supplied by the left hepatic artery.
Liver abscesses are treated by metronidazole if amoebiasis is suspected and by percutaneous CT guided aspiration. In most cases these techniques are curative. Open surgical drainage is only occasionally necessary.
Hydatid disease of the liver is treated by surgery because of the risks of rupture and secondary infection and of other complications. The principles of surgical management are as follows.
• Prevention of spillage of cyst contents into the peritoneal cavity.
• Removal of all cyst elements (the living germinal epithelium and the chitinous laminated membrane, collectively called the endocyst). This leaves behind the adventitious layer (Fig 7.37), which is merely compressed host tissue (ectocyst). The junction between the laminated membrane and host tissue is quite avascular. Attempted resection of the adventitia can cause excessive bleeding.
• Closure of biliary communications to the cyst cavity.
• Sterilisation of the cavity with a suitable scolicide (hypertonic saline, silver nitrate, formalin).
• Closure of the cavity.

Figure 7.37 Liver hydatid
A: adventitia derived from the host; B: laminated chitinous layer; C: germinal layer — which gives rise to brood capsules containing scolices. Daughter cysts float free within the main cyst, together with detached brood capsules and scolices (hydatid sand).
Most other lesions causing right upper quadrant masses require surgical treatment, which is described in more detail in sections describing their more common form of presentation: renal carcinoma — haematuria (Ch 9.2); gallstones — acute and chronic epigastric pain (Ch 7.3, 7.6); right colonic carcinoma — iron deficiency anaemia (Ch 7.18); and altered bowel habit (Ch 7.20).
Less common causes of a right hypochondrial mass include high retrocaecal appendiceal abscess, ileocaecal Crohn’s disease and pyloric masses. Gall bladder carcinoma rarely presents as a palpable mass but is more often diagnosed on pathological examination of the gall bladder after cholecystectomy.
Hepatomegaly
Liver enlargement as a cause of an abdominal mass is usually obvious on clinical examination. Apparent hepatomegaly can occur with depression of a normal liver below the costal margin in patients with emphysema, but such displacements of the normal liver are more usually diagnosed by percussion than palpation. The normal liver can span up to 14 cm in its vertical extent in the midclavicular line. Liver dullness extends from just below the costal margin up to the fifth rib in the midclavicular line.
A palpable liver is usually both enlarged and firmer than normal. A liver swelling has an edge extending across the right upper abdomen and epigastrium, moves freely on respiration and is dull to percussion. Sometimes only the left lobe of the liver is palpable to the left of the midline in cirrhosis. Riedel’s lobe is a normal variant with extension of normal liver tissue below the right costal margin in the anterior axillary line. The next question is whether the liver is smoothly or irregularly enlarged.
Common causes
Smooth regular liver enlargement
1. Alcoholic liver disease: diffuse fatty infiltration and micronodular cirrhosis
2. Chronic right heart failure, particularly when secondary to constrictive pericarditis
3. Haematological diseases — chronic leukaemia, lymphoma
4. Myeloproliferative or infiltrative disorders — myelofibrosis, polycythaemias, haemochromatosis or amyloidosis
5. Rarer causes of cirrhosis or infiltration
Irregular or nodular liver enlargement
1. Secondary carcinoma
2. Alcoholic liver disease: macronodular cirrhosis
3. Primary liver cell carcinoma (hepatoma)
4. Polycystic disease of the liver
Symptoms and signs
Direct questioning may reveal symptoms of alcoholism. Hepatomegaly can usually be distinguished easily from other masses in the right hypochondrium. A tender liver suggests inflammation or congestion; hardness and nodularity suggest malignancy; however, the cirrhotic liver is also very firm in consistency.
Very large livers are found with: alcoholic liver disease with fatty infiltration; congestive hepatomegaly from heart failure; metastases or hepatoma; myelofibrosis; and chronic myeloid leukaemia (Box 7.10).
Box 7.10
Causes of massive hepatomegaly and splenomegaly
Hepatomegaly
Metastatic cancer
Alcoholic hepatomegaly with fatty infiltration
Hepatoma
Chronic myeloid leukaemia
Myelofibrosis
Congestive hepatomegaly — CCF
Splenomegaly
Chronic myeloid leukaemia
Myelofibrosis
Lymphomas
Chronic malaria
If cirrhosis is suspected, additional signs are checked. An enlarged spleen or dilated abdominal wall veins indicates coexisting portal hypertension. Ascites may be present. Jaundice, spider naevi, palmar erythema, Dupuytren’s disease, leuconychia, skin bruising, parotid enlargement, loss of body hair, gynaecomastia and testicular atrophy are noted.
Cirrhosis is occasionally due to specific causes other than alcoholism: biliary cirrhosis — greenish jaundice and xanthelasma may be present; haemochromatosis — skin pigmentation and glycosuria (‘bronze diabetes’) or Wilson’s disease — Kayser–Fleischer rings in the iris may be present.
Diagnostic and treatment plans
Haematological tests
Blood examination may reveal leukaemia, anaemia or polycythaemia. Serum iron, total iron binding capacity and serum ferritin may be required to exclude haemochromatosis.
Liver function tests
Gamma GT level may be raised early in alcoholic liver disease. Significant elevation of serum alkaline phosphatase suggests infiltrative conditions such as secondary carcinoma or primary biliary cirrhosis.
Scanning
CT scan is a sensitive method of detecting density changes in the liver parenchyma because of hepatocellular disease. CT also gives precise anatomical localisation of masses within the liver and will show whether a mass is inside or outside the liver.
Biopsy
CT-guided needle liver biopsy is useful for localised lesions and to obtain histology in diffuse hepatic disease.
Treatment depends upon the cause of enlargement. Localised lesions in the liver may be amenable to surgical excision, depending on the pathophysiological state of the remaining liver, the anatomy of the liver and assessment by a multidisciplinary team of the resection possibilities.
Left upper quadrant mass
Common causes
1. Splenomegaly
2. Enlarged left lobe of the liver
3. Carcinoma of stomach or left colon
4. Left renal tumour, other retroperitoneal masses
History and examination
The history may reveal a likely source. An enlarged spleen is usually characteristic and its features are considered at the end of this section.
A left-sided or epigastric mass due to an enlargement predominantly affecting the left lobe of the liver is a common finding in patients of short squat build with micronodular cirrhosis and may be confused with a splenic swelling.
Splenomegaly may also be difficult to distinguish from left renal tumours, carcinomas of the stomach or left half of the transverse colon, pseudocyst of the tail of the pancreas and adrenal tumours. In contrast to an enlarged spleen, each of these lesions tends to have an irregular surface and an edge that is not rounded and notched like the spleen. Most importantly, the swellings are less mobile — movement on respiration is minimal, whereas splenic movement on respiration is usually free. Because of overlying bowel these masses tend to be resonant to percussion. It is frequently possible to get above them, again in contrast to an enlarged spleen.
An aortic aneurysm presents more as an asymmetric epigastric lump and is considered with epigastric masses.
Diagnostic plan
The differential diagnosis of the mass dictates which investigations are a priority and once again highlights the importance of arriving at a clinical diagnosis.
The physical signs of the lump may be less reliable diagnostically than is the case for masses in the right hypochondrium. In cases of doubt, splenic enlargement usually can be confirmed or excluded by ultrasound. The lesion can be further defined by CT scanning. Delineation of the lesion is often assisted by contrast studies of the stomach to locate the anatomical plane of the abdominal mass more accurately. Gastroscopy and colonoscopy exclude mucosal gastroduodenal and colonic pathology. Ultrasound is the best screening test but provides inferior anatomical definition to CT scanning. Percutaneous-guided fine needle aspiration is sometimes used to obtain cytological diagnosis: CT scan is the method of choice for the demonstration of pancreatic anatomy. Radionuclide scan is of little value, apart from definition of splenic size.
Treatment plan
Gastric and colonic tumours need endoscopic diagnosis with tissue biopsies and possible surgical resection to be planned. Most retroperitoneal tumours are lymphomas that can be histologically diagnosed by CT-guided percutaneous biopsy. Laparotomy or laparoscopy with biopsy is not usually necessary to stage the tumour and to obtain more histological detail.
Symptomatic and progressive pseudocysts of the pancreas greater than 5 cm in diameter are drained through the stomach by cystogastrostomy because of the danger of secondary infection and haemorrhage. This procedure may be done endoscopically as well but, either way, the wall of the pseudocyst needs to be mature enough to permit this internal drainage procedure to occur. Diagnosis is established by CT scanning with contrast studies of the stomach and preoperative endoscopic retrograde pancreatogram. ERCP is only done as an immediate preoperative investigation and under antibiotic cover, for there is a danger of infecting a cyst by this test.
Splenomegaly
Common causes
1. Infections: acute — viral hepatitis, glandular fever, leptospirosis, septicaemia; subacute — subacute bacterial endocarditis, brucellosis, malaria
2. Portal hypertension: secondary to liver cirrhosis or portal vein thrombosis
3. Haematological, myeloproliferative and lymphoproliferative disorders: leukaemias, lymphomas, myelofibrosis, congenital and acquired haemolytic anaemias, polycythaemia
4. Collagen disorders: systemic lupus erythematosis, polyarteritis nodosa, rheumatoid arthritis
5. Infiltrations: sarcoid, amyloid, lipid storage diseases
Transitory splenomegaly of mild degree occurs in a great many infectious diseases.
Huge spleens (Box 7.10) are seen in only a few conditions: chronic myeloid leukaemia, myelofibrosis, lymphoma, malaria.
History and examination
Factors in the past history may indicate a possible cause such as overseas travel that may imply the possibility of malaria or schistosomiasis, or occupational hazards that suggest brucellosis or Q fever. Valvular heart disease raises the possibility of endocarditis; infectious contact the possibility of leptospirosis and glandular fever. Ethnic origin may cause investigation for haemolytic anaemia and recent ingestion of certain drugs the possibility of thrombocytopenia. A history of liver disease or umbilical sepsis at birth raises the possibility of congestive splenomegaly secondary to cirrhosis or portal vein thrombosis. Evidence of alcohol abuse suggests portal hypertension and cirrhosis.
The features distinguishing the spleen are its rounded, notched lower border and edge, superficial anterior position with movement on respiration and the fact that one cannot get between it and the costal margin on deep palpation or percussion. Additional physical signs should be sought, including those of liver disease, sepsis and valvular heart disease. Associated lymphadenopathy suggests lymphoma or leukaemia, but lymphadenopathy is also common with nonspecific febrile illnesses. Bleeding disorders associated with purpura and petechiae suggest the possibility of leukaemia, auto-immune disease, drug-induced splenomegaly, polycythaemia and myelofibrosis. Skin rashes or mucous membrane lesions are often associated with acute conditions such as glandular fever, varicella, typhoid and septicaemia. Gastrointestinal haemorrhage may occur from oesophageal varices in portal hypertension and granulocytopenia may be associated with persistent leg ulceration. Nail bed, skin, conjunctival and retinal haemorrhages suggest endocarditis.
Diagnostic and treatment plans
Renal tumours, gastric leiomyomas and carcinoma of the splenic flexure of the colon may produce somewhat similar physical findings. If there is any doubt, radionuclide (technetium 99) scan can be performed to confirm that a mass in the left hypochondrium is the spleen. Patients with a palpable spleen and no other obvious symptoms or signs should be investigated by a complete blood examination and abdominal CT scan. The scan, as well as ruling out another left hypochondrial mass, may show the presence of defects in the spleen due to infarcts, tumour or other infiltrates. The blood count may be diagnostic of chronic myeloid or lymphatic leukaemia in asymptomatic patients. Haemolytic anaemia is diagnosed by tests for anaemia and haemolysis. Splenomegaly in lymphoma is almost always associated with lymphadenopathy and the diagnosis can often be made by lymph node biopsy.
There may be red cell changes consistent with liver disease (macrocytosis) or haemolysis (pigmented cells, spherocytes and Heinz bodies) or polycythaemia.
Hypersplenism is characterised by a moderately large spleen, pancytopenia and an active marrow. Platelets and white cells are particularly likely to be depressed. In hereditary spherocytosis there are spherocytes on the film, platelets and white cells are normal. In myelofibrosis, marrow biopsy shows proliferation of stem cells.
Splenectomy is indicated for hypersplenism when there is major haemolysis with a need for transfusion, marked neutropenia with recurrent infections or thrombocytopenia causing purpura or spontaneous haemorrhage, and for massive splenomegaly causing severe local symptoms. Splenectomy is also indicated in hereditary spherocytosis and chronic idiopathic thrombocytopenic purpura. Splenectomy can be performed laparoscopically in many of these conditions because the spleen is not enlarged in the latter conditions.
Combined liver and spleen enlargement
Combined enlargement of liver and spleen comprises an important clinical syndrome of haematological and myeloproliferative disorders, but many other causes also exist.
Common causes
1. Acute and chronic hepatitis
2. Alcoholic liver disease with portal hypertension and hypersplenism
3. Haematological and myeloproliferative disorders: leukaemias, lymphomas, myelofibrosis, haemolytic anaemias
4. Collagen disorders: systemic lupus erythematosus
5. Infiltrations: sarcoid, amyloid, lipid storage diseases
History and examination
Enlargement of the liver and spleen individually have been considered above. In the patient with hepatosplenomegaly, first assess whether an acute systemic illness is likely. General malaise and fever may indicate an acute infective cause.
If the clinical history suggests liver disease or a high alcohol intake, look for associated clinical features of cirrhosis, including jaundice.
Look also for associated lymph node enlargements, bleeding tendency, pallor or plethora suggesting a haematological or myeloproliferative disorder. Associated generalised lymph node swellings suggest lymphoma or leukaemia. If the clinical situation seems chronic and stable, likely diagnoses are chronic lymphatic leukaemia (suggested by moderate hepatosplenomegaly and large nodes) or chronic myeloid leukaemia (suggested by only a few nodes, but with large or massive spleen and liver). Acute leukaemia may present with an acute anaemia and bleeding tendency, lymph node swelling, mouth ulceration and sternal tenderness. Liver and spleen enlargement can be massive in myelofibrosis, whereas lymph node enlargements are uncommon.
Diagnostic and treatment plans
Full blood examination (FBE) and coagulation profile, tests of liver function, lymph node or marrow biopsy will usually be diagnostic of the common conditions causing hepatosplenomegaly. Treatment will depend upon the primary cause.
Epigastric mass
Common causes
1. Gastric outlet obstruction with gastric dilatation
2. Carcinoma of the stomach or colon
3. Abdominal aortic aneurysm
4. Retroperitoneal swellings — pancreas or lymphoma
5. Small bowel Crohn’s disease
6. Mesenteric cyst
History and examination
Gastrointestinal masses
Gastric carcinomas are often difficult to palpate because most tumours are concealed by the rib cage within the upper abdomen. The dilated thick walled stomach of gastric outlet obstruction may be apparent as a soft resonant epigastric fullness. A succussion splash is audible. Persistaltic waves running from left to right may be seen. Gastric and colonic masses may be mobile but, unlike liver and splenic swellings, do not move with respiration. Small bowel Crohn’s disease may cause multiple masses. Mesenteric cysts are soft, fluctuant abdominal swellings that can be moved most readily across the plane of the small bowel mesentery.
Retroperitoneal mass
Retroperitoneal masses are relatively immobile, indistinct on examination and often not felt.
Abdominal aortic aneurysm
This presents as a mass in the epigastrium down to the umbilicus just to the left of the midline and may be discovered on routine physical examination, sometimes because of vague epigastric discomfort. The patient may be aware of a painless throbbing mass. The fusiform, pulsatile mass just above and to the left of the umbilicus is diagnostic. The aneurysm may be slightly tender, suggesting impending rupture or rarely an inflammatory aneurysm. Sometimes transmitted pulsation associated with a large pancreatic swelling can be difficult to distinguish from an aortic aneurysm.
Diagnostic plan
Diagnosis of gastrointestinal epigastric masses is often based on gastroscopy or colonoscopy or CT studies with oral contrast. Ultrasound and CT scanning are very helpful for extragastric masses and provide the opportunity for percutaneous needle biopsy of masses. Subsequent laparoscopy may provide further information and biopsy material prior to exploration with a view to resection.
Palpable gastric carcinomas are often not resectable, but CT scanning is not a reliable discriminant of resectability. Even when CT scanning suggests invasion of adjacent organs, laparoscopy or laparotomy is usually indicated. Some will prove to be resectable, which usually provides the best palliation.
Incidental imaging findings (‘incidentaloma’)
Increasing use of highly sensitive but not necessarily specific imaging modalities may result in the identification of otherwise unsuspected abnormalities. These lesions present a clinical dilemma as the patient may not have any related symptoms but this incidental finding or ‘incidentaloma’ has been discovered.
The classic example of an ‘incidentaloma’ occurs when a patient is referred for an abdominal CT scan and an otherwise unsuspected adrenal mass is identified. Review of the patient’s history may reveal hypertension that can be related to phaechromocytoma, possible in the context of multiple endocrine neoplasia or an aldosterone-producing adenoma. The diagnoses are confirmed with serum and urine testing. Adrenal masses larger than 4 cm have an increased risk of being malignant and should be removed. Small non-functioning lesions can be monitored with serial CT scanning. The adrenal gland is also a possible site of metastatic cancer, particularly metastatic lung cancer, which may result in destructive replacement of the glands and adrenal insufficiency.
Incidental lesions are identified with other imaging modalities, including ultrasound and MRI, and in other regions of the body. In some cases, a clear diagnosis can be made through imaging, such as simple liver cysts on ultrasound. Conversely, other situations demand multiple investigations to reassure the patient and practitioner that the lesion is benign and non-functioning and will not have adverse health consequences.
Plain X-rays of the abdomen will reveal calcification in the outer layers of over 90% of atherosclerotic abdominal aneurysms. CT angiography is indicated whenever suprarenal involvement of the aorta or visceral arterial occlusion is suspected. Ultrasound is very useful for the measurement of the size and extent of an aneurysm and also as part of a screening or aortic surveillance program.
Treatment plan
The diagnosis and extent of disease dictates the role of surgery in managing most of these masses. Unfortunately, by the time of diagnosis, the disease has progressed to involve ‘vital structures’ such as major blood vessels that render curative resection an impossibility. Nevertheless a range of palliative treatments are available including surgical procedures.
With respect to abdominal aortic aneurysms (AAA), the principal indication for elective operation for aneurysm is to prevent rupture. Symptomatic or tender aneurysms require prompt surgical treatment. Rupture rarely occurs until the diameter of the aneurysm exceeds 6 cm on ultrasound examination. Repair of AAA greater than 5.5 cm is recommended in younger patients with a longer life expectancy. The aneurysm is replaced with a synthetic graft. The graft may be inserted using an endoluminal approach via the femoral artery or via laparotomy where it is placed within the lumen of the incised aneurysm. The outer layers are sutured over the graft after completion of the anastomosis: above to the end of the transected aneurysm below the renal arteries and distally to the transected ends of the common iliac arteries (Fig 7.38). The decision as to which technique is employed rests with the treating vascular surgeon in consultation with the patient.

Figure 7.38 Excision and replacement of abdominal aortic aneurysm with a synthetic graft
The adventitia of the aneurysmal wall is closed over the graft.
Right iliac fossa mass
Causes
1. Carcinoma of the caecum
2. Chronic appendiceal abscess
3. Ileocaecal Crohn’s disease
4. Less common causes
History and examination
Appendicitis, carcinoma of the caecum and Crohn’s disease often present as a right iliac fossa mass. Careful analysis of the history will usually uncover symptoms suggesting the diagnosis. In appendicitis, direct questioning often reveals a recent previous episode of ‘gastroenteritis’. Carcinoma of the caecum is notorious for late diagnosis with a mass, because of a paucity of early symptoms. A right iliac fossa mass associated with nonspecific malaise is the next most common form of presentation after iron deficiency anaemia. Vague epigastric discomfort is a common early, but neglected, feature of caecal carcinoma.
Ileocaecal Crohn’s disease may also present for the first time as a mass, discovered in a patient presenting because of weakness and malaise. Usually a history of intermittent diarrhoea, abdominal pain and failing health is also present.
Appendiceal abscess presents as a tender, poorly outlined, fixed mass close to the anterior superior iliac spine (Fig 7.34). Some degree of systemic toxicity is usually evident. Carcinoma of the caecum presents as a firmer, more distinct, nontender mass that can either be mobile or fixed in the right iliac fossa. Iron deficiency anaemia is a common associated finding. In contrast to the inflammatory swellings, little or no systemic toxicity is present. Ileocaecal Crohn’s disease forms a firm, fixed and slightly tender mass. The history is longer, with evidence of obstruction and weight loss is usually more than that found in carcinoma and appendicitis. The degree of systemic toxicity is variable.
Diagnostic plan
Blood examination may reveal iron deficiency anaemia, a feature of colonic carcinoma. Leucocytosis is suggestive of appendicitis. The most valuable investigation is often colonoscopy. Colonoscopy reveals the state of the caecal mucosa and allows a biopsy to be performed; CT scanning reveals whether a lesion is cystic or solid and whether the disease extends into surrounding tissues.
Treatment plan
Treatment of these conditions is surgical. Appendiceal abscess is treated by drainage, with or without appendicectomy. Caecal carcinoma is treated by right hemicolectomy. For ileocaecal Crohn’s disease, unless there is significant obstruction, a period of medical management using a 5-aminosalicylate such as mesalazine with or without oral corticosteroids (prednisolone) may lead to remission. However, in most patients with a mass as the form of presentation, laparotomy and right hemicolectomy is likely to be required ultimately. Most patients with Crohn’s disease will require surgical treatment at some stage of their disease course.
Less common causes
These include chronic inflammatory disorders of the caecum (yersinia, tuberculosis, actinomycosis and right colonic diverticulitis). Iliac artery aneurysm, iliac lymph nodes and chondrosarcoma of the ilium are occasional causes.
Left iliac fossa mass
Causes
1. Diverticulitis
2. Carcinoma of the sigmoid colon
3. Faecal masses in the sigmoid colon
4. Less common causes
History and examination
A careful history will usually reveal evidence of altered bowel habit, which is the predominant symptom of both carcinoma and diverticulitis.
Faecal masses are often multiple and indentable.
The mass of diverticulitis comprises a lump of peridiverticular cellular and oedematous tissue (phlegmon), often progressing to abscess formation. The degree of toxicity is variable. The mass is tender, indistinct, fixed and dull to percussion, with its long axis parallel to the inguinal ligament.
Carcinoma of the colon is usually a firmer, more distinct and nontender mass, which may be mobile or fixed; faeces may be palpable in the colon above the lesion. The patient is not toxic. Liver secondaries may be present. Pelvic examination reveals that the masses of colonic origin do not arise from the pelvis, though they can often be felt extrarectally on rectal examination.
In younger males, a tumour in an undescended testis may present as a mass in either the right or the left iliac fossa, as an intraperitoneal swelling. The cardinal physical sign is an empty scrotum on the same side.
Diagnostic plan
Colonoscopy and CT scanning will establish the diagnosis in most instances. CT scanning with rectal contrast may also reveal the extent of sinus and fistula formation in patients with diverticular disease. Colonoscopy or flexible sigmoidoscopy with biopsy of suspicious mucosal lesions is often necessary to distinguish finally between carcinoma and diverticular disease.
Treatment plan
The treatment of sigmoid colonic lesions presenting as a mass is nearly always a surgical resection with the aim of achieving local control of the underlying disease. Often such patients are malnourished and a period of medical optimisation is required prior to resection.
Less common causes
These include iliac artery aneurysm, psoas abscess, iliac lymphadenopathy and tumours of the bony pelvis.
Abdominal swellings arising from the pelvis
Common causes
1. Distended bladder
2. The pregnant uterus
3. Uterine fibromyomas
4. Ovarian tumours and cysts, tubo-ovarian masses
History and examination
Symptoms may be absent or found only on careful questioning. The possibility of pregnancy may be concealed by the patient. There may be little or no history of obstructed urinary flow in association with bladder retention and distension, especially in the aged, addled or delirious patient. Relatives may describe overflow urinary incontinence. Uterine fibromyomas are often asymptomatic. Abnormal uterine bleeding and vague pelvic discomfort may be revealed by direct questioning.
Swellings arising from out of the pelvic cavity are characterised by inability to get below the swelling. When large they may present as abdominal distension. Vaginal and rectal examination and bimanual abdominopelvic palpation must not be omitted.
A distended bladder is a smooth dull midline swelling arising from the pelvis. Pressure over the lump usually produces the desire to micturate. The enlarged bladder is not distinctly felt on bimanual examination. Urinary catheterisation settles the question when doubt persists about the aetiology of such a mass.
The pregnant uterus is a firm, smooth, dull, symmetrical, pear-shaped swelling arising from the pelvis. On bimanual examination it cannot be moved independently of the cervix, which is soft. Other signs of pregnancy are present.
Uterine fibromyomas may grow to a large size forming a firm or hard, dull, bosselated swelling arising from the pelvis. On bimanual palpation the mass moves with the uterus.
An ovarian cyst forms a smooth, dull, rounded swelling arising from the pelvis that is usually palpable in the lateral fornix as an adnexal mass on bimanual examination.
Ovarian tumours are often bilateral. Ovarian enlargement must be distinguished from uterine fibroids, pyosalpynx, tubal tuberculosis, carcinoma of the rectosigmoid, diverticulitis, endometrial or metastatic peritoneal deposits in the pouch of Douglas, retroperitoneal tumours and pelvic kidney.
A pelvic kidney can present as a fixed pelvic mass and is occasionally the sole functioning kidney; it must be identified and the patient informed of the anomaly.
Diagnostic plan
Ultrasonography is very helpful in identifying the organ involved with pelvic swellings and the nature of the swelling whether it be solid, fluid or fetal. X-rays may reveal calcification within fibroids but pregnancy should be excluded before any radiation is applied to the pelvic region. Laparoscopy may be valuable in difficult cases. A pregnancy test is a prudent investigation when any possibility of pregnancy exists. Pelvic CT scanning helps identify retroperitoneal masses and infiltration.
Treatment plan
With chronic urinary retention, renal failure may be present and this needs to be assessed and managed before planning definitive treatment of the cause. The most common cause is prostatic enlargement (Ch 9.6). Catheter drainage, as well as establishing the diagnosis, may be necessary for a period of time to improve renal function before transurethral prostatic resection is performed.
A pregnant patient needs to be referred to obstetrics for appropriate antenatal care while both ovarian and uterine masses require formal gynaecological assessment and management.
7.10 Abdominal distension
Abdominal distension as a presenting problem can develop insidiously in the absence of clear symptoms that might suggest a cause. Patients with functional bowel disease may present complaining of distension or bloating but such pseudo-distension is not demonstrable on physical examination. Causes of abdominal distension are often listed descriptively under the mnemonic of the five F’s: fetus, flatus, faeces, fat and fluid. Intra-abdominal tumours that can produce localised (and sometimes generalised) abdominal enlargement are considered in Chapter 7.9. In such patients, examination usually delineates a mass and the problem becomes that of an abdominal mass. Potentially massive swellings in this group include hepatomegaly, splenomegaly, fibroids, retroperitoneal tumours, carcinoma of the stomach, pancreas and colon, polycystic kidneys, urinary retention, ovarian cysts, ganglioneuroma and nephroblastoma.
Common causes
1. Fat
2. Pregnant uterus (fetus)
3. Accumulation of gastrointestinal gas (flatus)
4. Faecal impaction (faeces)
5. Ascites or encysted liquid (fluid)
History and examination
1. Fat
Fat is a very common cause of abdominal distension (‘beer gut’, ‘pot belly’). Fat is deposited both subcutaneously and intraperitoneally. Intra-abdominal fat deposition in omentum may be extreme in beer drinkers.
2. Pregnant uterus (fetus)
Pregnancy is the most common cause of abdominal distension. The patient may not be aware of or fail to acknowledge the cause of distension. The pear-shaped uterine swelling arises from the pelvis and is usually consistent with the period of amenorrhoea. The diagnosis can be difficult during the first 20 weeks. Fetal heart sounds may be heard at a later stage. Bimanual examination reveals a soft cervix and confirms the pelvic origin of the swelling; often signs of pregnancy are present.
3. Accumulation of gastrointestinal gas (flatus)
Distended gas-filled bowel has no palpable surface or edge, the only diagnostic feature being hyperresonance, sometimes with visible peristalsis. Significant accumulation of gas can occur in various abdominal and systemic disorders, with minimal associated abdominal symptoms. Gas may distend the stomach and the small and large bowel uniformly. Gaseous distension localised to the stomach is seen in organic and functional gastric outlet obstruction. Gaseous distension of the large bowel is often maximal in the caecum.
Chronic gastric dilatation occurs with pyloric obstruction. Often epigastric pain is absent; there may be a past history of peptic ulceration. Dilatation is commonly associated with a succussion splash.
Acute gastric dilatation can cause insidious abdominal distension culminating in fatal regurgitation. The fully blown picture usually represents the end-point of inappropriate oral fluid administration in a patient with gastric ileus after operation or due to severe systemic illness. Functional large bowel obstruction (colonic ileus) is also a cause of insidious distension due to gas. This condition is commonly associated with diabetes mellitus and hypokalaemia and some of the drugs that impair motility.
Occasionally, chronic constipation leads to colonic dilatation — secondary to damage from prolonged cathartic abuse, with or without faecal impaction. This may cause massive gaseous distension.
Air swallowing (aerophagy) is perhaps the most common cause of tympanites. Occasionally, gaseous distension from air swallowing is induced to produce pseudocyesis (false pregnancy).
Chronic adhesive small bowel obstruction may produce gaseous distension without significant abdominal pain.
4. Faecal impaction (faeces)
Faecal impaction may present as abdominal distension or as abdominal masses. Faecal impaction may be associated with Hirschsprung’s disease, acquired megacolon, chronic intestinal obstruction or with chronic constipation from any cause. The patient may complain of spurious diarrhoea due to mucus and watery material leaking around the main mass of faeces. On physical examination faeces are felt as indentable masses along the line of the colon. In advanced cases faecal masses tend to coalesce to form one mass, which can be mistaken for a bowel tumour.
5. Ascites or encysted liquid (fluid)
Liquid fluid in the peritoneal cavity may be free (ascites) or encysted (where fluid is trapped in a cavity or between adhesions and is of sufficient size to cause generalised distension). Ascites has two major diagnostic physical features: fluid thrill and shifting dullness. A fluid thrill is elicited by flicking the side of the abdomen with the index finger and feeling the vibrations on the other side of the abdomen with the other hand. A third hand is placed by the patient or assistant over the mid abdomen, to prevent the percussion wave being transmitted in the abdominal wall. A fluid thrill is present across any fluid-filled cavity, so the difference between free and encysted fluid depends upon the demonstration of shifting dullness. With free ascites, dullness is found mainly in the flanks and across the lower abdomen — in contrast to encysted fluid, where dullness is localised and more central. The level of dullness in the flanks in free ascites shifts under the influence of gravity when the patient moves position (Fig 7.8).
Causes of ascites (free intraperitoneal fluid)
These include hepatic disease, cardiac and renal diseases, starvation, malignant ascites, tuberculous peritonitis and less common causes.
Ascites in hepatic disease results from four main factors:
• increased formation of hepatic lymph because of sinusoidal hypertension
• increased formation of splanchnic lymph
• hypoalbuminaemia
• salt and water retention by the kidneys.
The most common cause of hepatic ascites is alcoholic liver disease. Associated abdominal findings may assist in the diagnosis. The position of the umbilicus, nearer the pubis than the xiphoid process, helps to distinguish ascites from lower abdominal encysted fluid, where the reverse is more commonly found. Abdominal wall venous collaterals may be present (Fig 7.39). Umbilical and inguinal hernias, together with hydrocele, scrotal oedema and oedema of the lower limbs, are frequent associated findings. Pleural effusion, usually on the right side, and dyspnoea from diaphragmatic elevation are also common. Most patients will have the peripheral signs of chronic liver disease. Occasionally a sudden increase in the amount of ascites indicates deterioration of liver function after an alcoholic binge, development of a hepatoma or bacterial infection of ascitic fluid.

Figure 7.39 Collateral venous bloodflow on the anterior abdominal wall
A: portal venous obstruction with ascites; B: inferior vena caval obstruction
The separation of cardiac and renal diseases from other causes of ascites is usually obvious. In constrictive pericarditis the diagnosis is suggested by a raised jugular venous pressure, paradoxical pulse, the radiological demonstration of a calcified pericardium and the characteristic electrocardiogram. Renal failure, particularly with protein depletion, may produce marked ascites. Generalised oedema in association with ascites, together with gross proteinuria, is diagnostic.
Hypoproteinaemia secondary to starvation (kwashiorkor) will produce significant ascites, which is exaggerated by limb muscle wasting.
Tuberculous peritonitis may present in the early stages as a primary abdominal infection without pulmonary, intestinal or uterine tube involvement. Ascites may be the predominant manifestation.
Malignant ascites, including that due to mucus-forming tumours (pseudomyxoma peritonei), may be a diagnostic problem when no clear evidence exists of the primary tumour site. Common primary sites are the stomach, colon, breast and ovary. At times the primary site is small and peritoneal seeding is disseminated widely as small implants. Prognosis is poor, except in the case of certain ovarian tumours.
Pseudomyxoma peritonei is an unusual condition characterised by accumulation in the abdomen of large amounts of mucus, secreted by a neoplasm of the ovary or appendix. The incidence of primary malignancy of the peritoneum — mesothelioma — is increasing with increased contact with asbestos. Ascites, distension and weight loss with a palpable mass is the usual form of presentation.
Chylous ascites is caused by the rupture of a retroperitoneal lymph vessel, usually a result of either trauma or lymphoma.
Ascites is gross in the Budd-Chiari syndrome, which is a consequence of hepatic vein thrombosis. Pancreatic ascites is an uncommon chronic sequel of pancreatitis, often thought to be secondary to liver disease, as most of these patients are alcoholics. The amylase in the fluid is high and diagnosis is confirmed by a leak from the pancreas shown on ERCP. An uncommon finding in benign fibromas of the ovary is the development of ascites and hydrothorax — Meigs’ syndrome.
Causes of encysted fluid
Fluid trapped in a cyst or in the renal pelvis or between adhesions will demonstrate a fluid thrill, be dull to percussion and be fluctuant; however, the fluid will not shift on movement. The features of encysted fluid depend upon its anatomical origin. Focal fluid collections large enough to present as abdominal distension include ovarian cysts, hydronephrosis, polycystic kidneys, distended urinary bladder, pancreatic cysts and mesenteric cysts.
Diagnostic plan
The diagnosis of the cause of abdominal distension can usually be made on physical examination. A plain X-ray often confirms the cause of distension as ascites, gaseous distension or faecal impaction. Passage of a nasogastric or rectal tube can be helpful in the diagnosis and management of gaseous distension. Eliciting the cause of ascites may pose some difficulties. If the cause of ascites is not apparent from clinical examination, the following tests should be performed: a full blood count and film, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), serum protein estimation, liver function tests, abdominal ultrasound, and abdominal and chest X-rays. Liver biopsy, providing the clotting profile is normal, to gain a histological diagnosis may be necessary if chronic liver disease is likely as the cause of ascites.
Diagnostic paracentesis is indicated if the cause of ascites is not shown from the above investigations. Bloodstained fluid obtained by uncomplicated aspiration suggests malignancy. The detection of malignant cells is difficult on cytological examination, but anaplastic or mesothelial cells may be seen. The protein content of ascitic fluid has little specificity, but the lower the protein content, the more likely the diagnosis of liver cirrhosis. The amylase content should be routinely determined to exclude pancreatic ascites. Cultures (both aerobic and anaerobic) should be performed, together with routine culture for mycobacteria.
Gastrografin contrast studies may help elicit the cause of gaseous distension. Ultrasound and CT scanning of the abdomen are often of great value in the detection of lymphoma and pancreatic disease.
Treatment plan
Ascites due to liver disease can be controlled by salt and water restriction and careful use of a combination of a sodium-losing diuretic (e.g. frusemide, thiazide) and a potassium-conserving agent (e.g. spironolactone). Before instituting therapy, hepatoma and spontaneous bacterial peritonitis should be excluded. Refractory ascites may be treated by permanent implantation of a silastic tube to shunt fluid from the peritoneal cavity to the jugular vein. This method may also be of palliative value for malignant ascites. In cases of intractable ascites, Budd-Chiari syndrome and pancreatic ascites need to be excluded. Gaseous distension of bowel requires correction of the underlying systemic or local cause. Faecal impaction requires a combination of enemas, digital disimpaction and aperients. Exclusion of a mechanical cause of obstruction is important.
7.11 Retrosternal pain and heartburn
When patients present with a history of episodic retrosternal pain, the distinction between cardiac pain and the heartburn of reflux oesophagitis is, in most instances, clear from the history. However, gastrointestinal reflux can precipitate attacks of angina in patients with established coronary artery disease and angina-like pain can also be produced by skeletal and biliary disorders. In patients with oesophageal disease, pain identical to angina pectoris can also occur without any evidence of coronary artery disease on angiography. The history remains the most important part of the diagnostic process. Modern methods of investigation often, but not always, can distinguish between cardiac and oesophageal pain in the difficult case. Ischaemic retrosternal chest pain and pain secondary to oesophageal disease have a similar pattern of referred pain, in the area of distribution of the C3, 4, 5 and T1, 2, 3 sensory dermatomes. Thus, for both disorders, pain is often referred to the neck, jaw, ear, shoulders, arms and scapulae. A history of dysphagia is a strong pointer to oesophageal pathology, but mild dysphagia may not be volunteered when overshadowed by chest pain and concern about cardiac disease.
Causes
1. Ischaemic heart disease — angina pectoris
2. Reflux oesophagitis and hiatus hernia
3. Diffuse oesophageal spasm
4. Musculoskeletal or chest wall pain
5. Less common causes
History
1. Ischaemic heart disease — angina pectoris
The retrosternal chest pain of angina pectoris is usually described as pressing, crushing or constricting and, unfortunately, is often assumed to be indigestion. It is classically related to exercise or exposure to cold and responds to nitrates; it is less commonly related to posture, eating or antacids. Exertional dyspnoea and orthopnoea are features of ischaemic heart disease complicated by congestive cardiac failure. Sometimes cardiac pain may follow a meal or even swallowing and may be relieved by belching, particularly when it is described as indigestion. Angina may sometimes wake the patient from sleep. These symptoms suggest that sometimes true angina is triggered by oesophageal reflux. It is in this group of patients that a definite diagnosis can be difficult to establish.
2. Reflux oesophagitis and hiatus hernia
In the majority of patients with reflux oesophagitis, the cause of pain is readily identified from the typical symptom of postural ‘heartburn’– a retrosternal burning discomfort induced by stooping or lying and relieved by sitting up. Another characteristic symptom of reflux to be sought is acid regurgitation producing a sour metallic taste (water brash) in the mouth, burning lips and, in some cases, pulmonary aspiration and chronic cough. Patients often awaken at night with heartburn and water brash, which are relieved by sitting up. Occasionally regurgitation and belching may occur without heartburn. Atypical features are quite common. Angina-like pain may occur, triggered by swallowing. ECG changes, typical of angina, may be produced by acid instillation or balloon dilatation of the distal oesophagus. In these cases a history of intermittent dysphagia is the conclusive distinguishing feature. The pain of oesophageal disease is made worse by postural change and is temporarily relieved by food and alkali, but it is sometimes also exertion-related and relieved by nitrates. The exertion that worsens the pain is not that of walking; pain follows postural changes of lifting and stooping. The pain can also follow emotional tension and can occur at night after large meals. These atypical symptoms of reflux may be associated with significant oesophageal motility disorders.
3. Diffuse oesophageal spasm
Episodic symptoms are characteristic of this disorder. The pain is retrosternal and varies from slight discomfort to severe spasmodic pain — which can lead to admission with the provisional diagnosis of myocardial infarction. Both angina and the pain of diffuse spasm can radiate to the same areas; both are associated with extrasystoles, tachycardia or vasovagal episodes. In oesophageal pain a long history of episodic dysphagia, to solids or fluids, can be diagnostic. Pain may also be precipitated by swallowing and the recumbent posture, either on stooping or on awakening at night. Weight loss is uncommon.
4. Musculoskeletal or chest wall pain
Minor chest trauma frequently triggers the pain of cardiac neurosis. Generally the pain is fleeting, poorly localised, made worse by movement, but not exacerbated by coughing or deep breathing. In contrast to ischaemic and oesophageal pain, the pain tends to be continuous and is atypical in pattern. Costochondritis (Tietze’s syndrome) is a painful, tender, nonsuppurative inflammation of the third or fourth anterior costochondral junction. After some weeks the process resolves, but may recur. Fractured ribs in alcoholics are occasional causes of chronic chest pain, which can be difficult to diagnose until a chest X-ray is performed. Pain arising from cervical spondylitis is felt in the cervical spine and is usually referred to the shoulder and arm but may closely resemble angina or sometimes induce angina in patients with established coronary artery disease. Bony pain from sternal infiltration by myeloma or leukaemia can occur.
Examination
Physical examination is usually unhelpful. There may be evidence of heart failure and pain on movement or localised tenderness with musculoskeletal disorders.
Diagnostic plan
The diagnosis is mainly based upon a careful history. An obvious clinical diagnosis of heartburn may not demand further investigation. The same applies to angina pectoris — a clinical diagnosis can be made and acted upon, even when tests are negative or equivocal. In many cases where an oesophageal motility disorder is suspected, a trial of treatment may be of diagnostic as well as therapeutic value.
Haematological examination
Anaemia may follow chronic blood loss from oesophagitis, but carcinoma of the right colon should always be excluded in patients with iron deficiency anaemia.
Electrocardiography
Evidence of cardiac ischaemia is sought on ECG. In equivocal cases a stress test to induce ischaemic changes may be performed. ECG features suggesting pericarditis may be seen, with ST segment elevation without reciprocal depression.
Organ imaging and endoscopy
Plain X-ray of the chest and cervical spine may reveal signs of aspiration pneumonitis from reflux, congestive cardiac failure, pericardial calcification, paraoesophageal hernia with retrocardiac fluid level, fractured ribs or cervical spondylitis. Endoscopy and possibly barium swallow is indicated when oesophageal disease is suspected. There may be evidence of reflux, hiatus hernia (Fig 7.40) or oesophageal motility disorders. The barium swallow in diffuse oesophageal spasm is usually abnormal, showing the segmental spasm described as ‘corkscrew’ oesophagus. A small hiatus hernia is often present and an epiphrenic diverticulum may be seen. Ultrasound may be indicated if gallstones are suspected.

Figure 7.40 Oesophagus hiatus hernia
A: sliding hernia — front (A1) and side (A2) views; B: paraoesophageal or rolling hernia. Reflux with oesophagitis occurs in about 50% of sliding hernias but is rare in rolling hernia where the main danger is intrathoracic incarceration and strangulation of the stomach.
Endoscopy is indicated in equivocal cases to exclude oesophageal diseases such as oesophagitis or carcinoma and allow biopsy of any suspicious mucosal changes. Associated gastric or duodenal pathology can also be excluded in like manner during the same examination.
Manometry
Measurement of lower oesophageal sphincter pressure contributes little to the diagnosis and management of patients with gastro-oesophageal reflux. Intra-oesophageal acid perfusion (Bernstein test) may reproduce the pain and can be of help in the diagnosis of acid reflux. In some cases of angina secondary to reflux, a Bernstein test can produce ischaemic changes on the ECG. In the difficult case, continuous perfusion manometry may be useful in the diagnosis of oesophageal motility disorders. In about one-third of cases with diffuse oesophageal spasm, painful non-propulsive high amplitude waves are detected in the lower oesophagus, the lower oesophageal sphincter relaxes normally on swallowing and normal peristalsis is seen in the upper oesophagus. In achalasia, primary peristaltic waves are absent in the majority of cases, together with failure of the lower oesophageal sphincter to relax on swallowing. By this stage in achalasia pain is uncommon and dysphagia is prominent.
24-hour pH monitoring for gastro-oesophageal reflux should be considered as well, especially if there is a suggestive history and other investigations have not been diagnostic.
Treatment plan
1. Ischaemic heart disease — angina pectoris
Initial treatment is directed at the control of risk factors. These include smoking, obesity, hyperlipidaemia, diabetes and hypertension. Anaemia may unmask angina and anaemia should always be sought and treated if present. Initially, patients are instructed in the proper prophylactic use of anginine before exposure to triggering factors, such as cold and exercise. The next step in treatment is the use of a long-acting nitrate and then a beta-blocker. Alternatively, calcium antagonists, such as verapamil, may be used. Angiography is indicated in patients where angina is progressive, is present at rest or is unstable and poorly responsive to treatment and coronary artery interventions such as angioplasty, stenting and bypass.
2. Reflux oesophagitis and hiatus hernia
The management of reflux oesophagitis is similar to that of peptic ulcer, with attention to risk factors and the use of specific treatment with proton pump inhibitors or cytoprotective agents. Combination with metaclopramide may assist in the symptomatic control of heartburn. Measures to reduce reflux are important: these include the avoidance of stooping, sleeping with several pillows at night and weight loss (Box 7.11). A heavy meal should be avoided in the evening. Constipation should be avoided with a high-fibre diet. Chronic bronchitis is aided by stopping smoking and physiotherapy.
Box 7.11
General instructions for reflux oesophagitis with heartburn (hiatus hernia)
Specific medicines and tablets may be prescribed by your doctor. You should also try to control risk factors for oesophagitis:
![]() Avoid stooping and avoid eating a large late evening meal to reduce regurgitation, especially at night.
Avoid stooping and avoid eating a large late evening meal to reduce regurgitation, especially at night.
![]() Sleep head up with two or three pillows at night.
Sleep head up with two or three pillows at night.
![]() Lose weight.
Lose weight.
![]() Avoid constipation, with straining, by consuming a high-fibre diet. A normal bowel habit should be established after breakfast in the morning. Eat regular meals.
Avoid constipation, with straining, by consuming a high-fibre diet. A normal bowel habit should be established after breakfast in the morning. Eat regular meals.
![]() Chocolate, coffee, smoking and alcohol interfere with the ability of the oesophagus to prevent reflux and should be avoided.
Chocolate, coffee, smoking and alcohol interfere with the ability of the oesophagus to prevent reflux and should be avoided.
![]() Avoid using aspirin or aspirin-like drugs. Have regular exercise to improve your fitness and general resistance.
Avoid using aspirin or aspirin-like drugs. Have regular exercise to improve your fitness and general resistance.
Surgery is indicated for severe and intractable symptoms of postural heartburn (Box 7.12), especially when there are complications such as anaemia, stricture and aspiration pneumonitis. Pain is a relatively uncommon indication for surgery, as this problem tends to be transient in relation to the long-term history of the illness and is usually controllable. The most effective anti-reflux surgical procedure used is the Nissen fundoplication (Fig 7.41). The operation is usually performed laparoscopically.
Box 7.12
Indications for anti-reflux oesophageal surgery
![]() Severe symptoms despite six to 12 months of intensive treatment; especially when complicated by anaemia or aspiration pneumonitis.
Severe symptoms despite six to 12 months of intensive treatment; especially when complicated by anaemia or aspiration pneumonitis.
![]() Symptomatic patients with Barrett’s oesophagus (metaplastic oesophageal epithelium).
Symptomatic patients with Barrett’s oesophagus (metaplastic oesophageal epithelium).
![]() Severe work disability.
Severe work disability.
![]() Younger patients with stricture or poorly controlled dysphagia despite dilatation.*
Younger patients with stricture or poorly controlled dysphagia despite dilatation.*
![]() Nonhealing or recurrent oesophageal ulceration, especially with blood loss anaemia.
Nonhealing or recurrent oesophageal ulceration, especially with blood loss anaemia.
* Dysplasia on serial biopsy over six months or intractable stricture, are indications for oesophageal resection surgery.

Figure 7.41 Nissen fundoplication
A: initially the hernia is reduced and the defect in the diaphragmatic hiatus is narrowed with sutures; B: the fundus is wrapped around the distal oesophagus down which has been passed a large bougie (50F); C: the wrapped fundus is sutured in place to produce a valve in the lower oesophagus from the pressure of the surrounding stomach, thus preventing reflux and herniation
3. Diffuse oesophageal spasm
Management of disorders such as diffuse oesophageal spasm can be difficult. Anticholinergic drugs or nitroglycerine may relieve pain, but are often ineffective. Cold foods should be avoided. Foods should be chewed carefully in a relaxed environment. Sedatives may be of value. Control of concurrent disease, especially congestive cardiac failure, often helps to reduce symptoms. In more severe cases, oesophagomyotomy is the treatment of choice.
4. Musculoskeletal or chest wall pain
Proof that a patient is free of cardiac disease will assist in the control of symptoms of cardiac neurosis, especially if litigation is not involved. Musculoskeletal disorders are treated mainly by maintaining mobility, local treatment such as heat and ultrasound and regular analgesics. The pain of cervical spondylitis may also require the wearing of a soft cervical collar for relief of symptoms.
5. Less common causes
Oesophageal pain may occur early in the course of achalasia. Pain occurs spontaneously at night or may be brought on by eating. It tends to disappear with the onset of dysphagia. With advanced disease, retrosternal discomfort again develops and may again be difficult to distinguish from angina pectoris. On occasions carcinoma of the oesophagus can present with retrosternal pain on swallowing, appearing before the onset of dysphagia.
Para-oesophageal hiatus hernia may be associated with intermittent distension, with angina-like pain and flatulence soon after a meal and after sitting for long periods. This may lead to vomiting and collapse, but usually the pain resolves before volvulus and strangulation. Heartburn is uncommon. Incipient strangulation is an indication for urgent surgical repair of the hernia. In most patients, discovery on chest X-ray of an asymptomatic hernia is an indication for surgical repair because of the high risk of eventual strangulation.
Biliary colic and post-cholecystectomy pain can sometimes resemble angina. Quite a few patients have been treated for many years for angina when, in fact, the cause of substernal pain was gallstones and symptoms were subsequently relieved by cholecystectomy.
Relapsing pericarditis is an infrequent cause of episodic retrosternal chest pain. The pain is often related to posture, being worse when lying down and relieved by sitting forward. Occasionally the pain can also be made worse by swallowing and deep inspiration. On examination there is typically a pericardial friction rub, evidence of cardiac tamponade, right heart failure and fever. The features of cardiac tamponade are low blood pressure, raised jugular venous pressure and indistinct heart sounds (Beck’s triad).
7.12 Dysphagia
Dysphagia is the main symptom of oesophageal disease. Its presence (however transient) demands full investigation and a firm diagnosis. The history is of great importance, as physical signs are few. Progressive dysphagia for solid food usually means organic obstruction. Intermittent painful dysphagia for solids and liquids, especially when precipitated by hot or cold liquids, suggests a motility disorder. Associated symptoms will often further clarify the cause of dysphagia. A past history of postural retrosternal pain (‘heartburn’) is the cardinal symptom of reflux oesophagitis. Pain on swallowing (odynophagia) suggests oesophagitis or motility disorder. Continuous pain is a feature of advanced carcinoma with invasion of somatic structures. Oesophageal pain may resemble angina, either because of a similar pattern of referred pain or because distension and acid reflux can induce coronary ischaemia. The other important symptom of oesophageal disease is regurgitation (‘waterbrash’). Regurgitation may lead to nocturnal pulmonary aspiration and chronic aspiration pneumonitis with symptoms of ‘asthma’, pleurisy and dyspnoea. Sticking of food, with coughing and spluttering within one or two seconds of swallowing, implies oropharyngeal dysphagia causing respiratory or nasal aspiration.
Causes
1. Foreign body
2. Reflux oesophagitis and benign stricture
3. Carcinoma of the oesophagus
4. Achalasia and diffuse oesophageal spasm
5. Less common causes
History
1. Foreign body
In most cases the dysphagia of foreign body ingestion is sudden and complete and is followed by persistent salivation and odynophagia localised to the site of obstruction, with radiation of pain into the neck and back.
Sometimes, however, the onset of dysphagia is insidious with vague discomfort. This is seen especially with lodgement of smaller foreign bodies such as fish bones, where dysphagia is due to inflammatory oedema and is associated with increasing pain as the infection spreads, with erosion through the wall of the oesophagus. This presentation is more common in children where the foreign body is usually found to have lodged in the upper oesophagus. Occult foreign bodies can also occur in mentally disturbed patients or the aged and edentulous. In these instances lodgement is usually at the level of the cardia. An underlying disorder of the oesophagus must be excluded in patients who suffer from foreign body obstruction; this being the occasional form of presentation of a malignant or benign stricture or a motility disorder. Foreign body obstruction needs to be distinguished from oesophageal moniliasis or corrosive stricture via barium swallow and endoscopy.
2. Reflux oesophagitis and benign stricture
In mild to moderate reflux oesophagitis, intermittent dysphagia is common, probably due to a combination of inflammatory oedema and spasm. Dysphagia due to spasm may be felt at any level, whereas with fibrous stricture, dysphagia is localised to the lower oesophagus. The increasing dysphagia of benign stricture is commonly preceded by diminution or disappearance of regurgitation and heartburn. If regurgitation does continue, it is not sour because gastric juice is no longer present. In some older patients with benign stricture, acute dysphagia may not be preceded by oesophageal symptoms. Occult persistent bleeding is common with oesophagitis and stricture and may be sufficient to produce a hypochromic anaemia. Haematemesis can occur but is less common than chronic oozing. Dysphagia from reflux oesophagitis must be differentiated from stricture due to neoplasms, achalasia, epiphrenic diverticulum, webs and Schatzki’s ring. Schatzki’s ring is a band-like ring of mucosa at the cardio-oesophageal junction, usually associated with hiatus hernia and probably due to reflux.
3. Carcinoma of the oesophagus
Progressive dysphagia affecting predominantly the elderly patient is the cardinal symptom of carcinoma of the oesophagus. The onset of dysphagia is often preceded by a short period of anorexia, fullness, nausea and sometimes odynophagia. The dysphagia usually starts with the ingestion of solids and then progresses to difficulty swallowing liquids. Constant substernal pain suggests locally advanced disease and is a late symptom. Anorexia, hoarseness, coughing on swallowing and haemoptysis are also signs that extra-oesophageal spread has already occurred. Regurgitation and nocturnal pulmonary aspiration, with cough and dyspnoea secondary to aspiration pneumonitis, can occur. Weight loss is a striking feature of even early carcinoma of the oesophagus; the majority of patients have lost more than 10% of body weight by the time of presentation. With weight loss, patients become lethargic and weak and are commonly anaemic. Haematemesis is unusual. Risk factors include smoking, alcoholism, achalasia, longstanding reflux oesophagitis and Barrett’s oesophagus (ectopic columnar cell metaplasia within the distal oesophagus).
4. Achalasia and diffuse oesophageal spasm
Achalasia of the oesophagus is a neuromuscular disorder in which oesophageal dilatation and hypertrophy occur without organic stenosis. Pathologically, the coordinating myenteric plexus of the oesophagus is destroyed and therefore the cardio-oesophageal sphincter fails to relax on swallowing. Lower sternal dysphagia, in patients from 30 to 60 years of age, is the usual presenting symptom. Early in the disease dysphagia occurs for liquids as well as solids. Food usually gets through despite a contracted cardio-oesophageal sphincter and weight loss is not marked early in the disease. Some patients have a long history of oesophageal pain on swallowing cold liquids, preceding the onset of the dysphagia. Nocturnal aspiration of food is common and may lead to repeated bouts of respiratory tract infection, bronchiectasia and lung abscess and may be the cause of a long history of ‘asthmatic’ attacks.
Diffuse oesophageal spasm is characterised by intense, sustained, nonpropulsive contractions of the body of the oesophagus producing transient episodes of painful dysphagia, usually associated with substernal discomfort and sometimes severe spasmodic pain. Pain on swallowing cold fluids is a characteristic symptom of this condition. Confusion with angina is common when pain is severe; indeed, oesophageal distension may produce true angina with typical ECG changes. Most patients are over 40 years of age. Even though Auerbach’s plexus is normal, this condition may be a variant of achalasia as some cases progress to achalasia. Some cases share the features of the two diseases and are called vigorous achalasia.
Signs
Physical signs are usually normal in patients with dysphagia. Significant weight loss is very suggestive of carcinoma. There may be evidence of metastatic disease. Pharyngeal and laryngeal examination may reveal local lesions. Haematological and systemic examination may reveal anaemia or signs of unusual causes of dysphagia such as Parkinson’s disease, scleroderma, myopathy, goitre and lymphadenopathy.
Diagnostic plan
Diagnosis of the cause of dysphagia is based upon endoscopy, barium swallow and manometric studies. CT scanning is essential for staging cancer of the oesophagus and thus to determine operability.
Haematological examination
Anaemia is a frequent finding with carcinoma but can also accompany oesophagitis. Leucocytosis suggests the possibility of chronic lung infection or foreign body perforation.
Radiology
Plain chest film will detect basal atelectasis, lung abscess secondary to aspiration pneumonitis and secondary malignancy and is an important part of the preoperative assessment of respiratory status. Sometimes a primary disorder causing dysphagia is shown, such as a gas–fluid level in the thoracic inlet due to a pharyngeal pouch, the right mediastinal density of achalasia and the retrocardiac fluid level of paraoesophageal hiatus hernia. Plain X-ray of the neck and thoracic inlet can be helpful in cases of impacted foreign body and intra-thoracic goitre.
The next investigation in patients with dysphagia should be a barium swallow, unless the dysphagia is secondary to a painful condition of the oropharynx in which case direct inspection may reveal the diagnosis. Barium swallow is indicated in all patients before endoscopy. Endoscopy is made safer if the anatomy of the oesophagus is known beforehand — this applies particularly to the patient with unsuspected pharyngeal pouch. Cineradiology is most useful for assessing pharyngo-oesophageal function. Barium swallow may reveal a sharply demarcated stricture with proximal shoulders or a shelf-like appearance, both virtually diagnostic of carcinoma. A hiatus hernia is present in most cases of reflux oesophagitis. Benign stricture often has a fusiform shape, without the shoulders that are so typical of carcinoma. The features of achalasia are shown in Figure 7.42. Narrowing at the cardia in achalasia is seen as a smooth, rat-tail narrowing approximately 5 cm long. The body of the oesophagus is dilated and there is no gastric air bubble. Later in the disease an air–fluid level is seen in a grossly dilated sigmoid-shaped oesophagus. In many patients with diffuse oesophageal spasm, segmental spasm of the oesophagus can be seen, a feature that is often described as ‘corkscrew’ oesophagus. Occasionally an epiphrenic diverticulum is present. Confirmation of a benign or malignant cause for dysphagia always requires subsequent endoscopy and biopsy for histological confirmation.

Figure 7.42 Contrast radiological features of achalasia
A: dilated oesophagus; B: rat-tail narrowing of the distal oesophagus; C: no gastric air bubble. Endoscopy demonstrates no organic stricture.
Endoscopy and biopsy
Endoscopy is both a diagnostic and therapeutic measure when foreign body is the cause of dysphagia. In carcinoma of the oesophagus, a mucosal lesion or possibly an intraluminal mass or stenotic lesion is seen and can be biopsied. Various grades of severity of reflux oesophagitis are recognised on endoscopic examination. Fibrous stricture is classed as the greatest severity. Biopsy of the distal oesophagus is necessary to establish the diagnosis of oesophagitis, to exclude carcinoma, to detect Barrett’s oesophagus (in which extension up from the cardia of columnar cell metaplastic change occurs) and to detect dysplastic epithelial change. The last two findings are both signs of premalignant change. In achalasia, a dilated oesophagus is found on oesophagoscopy. The instrument passes easily through the narrowed distal sphincter, thereby distinguishing achalasia from carcinoma or benign peptic stricture.
Manometry
Continuous perfusion manometry is of most value in the diagnosis of motility disorders. Pressures are measured while slowly withdrawing manometric catheters from the stomach towards the pharynx. In about one-third of patients with diffuse oesophageal spasm, painful nonpropulsive high amplitude waves are detected in the lower oesophagus. In most cases the lower oesophageal sphincter relaxes normally on swallowing. Peristalsis is normal in the upper oesophagus in diffuse spasm — in contrast to achalasia. In achalasia, there is absence of primary peristaltic waves in the majority of patients, together with failure of the lower oesophageal sphincter to relax on swallowing.
Treatment plan
1. Foreign body
Some smooth foreign bodies, such as solid food free of bones, will pass and after barium swallow a few hours should be left before endoscopy. As a rule, no foreign body should be left without relief for more than 24 hours. All sharp-pointed or sharp-edged foreign bodies should be removed as soon as possible — before they become fixed within an acute inflammatory swelling and eventually lead to perforation. A wide variety of forceps can be passed down the endoscope to remove various types of foreign body. Open thoracotomy is occasionally necessary to remove a fixed foreign body.
2. Reflux oesophagitis and benign stricture
Intermittent dysphagia from mild to moderate oesophagitis is treated by dilatation (Fig 7.43) and medical treatment, including metaclopramide and acid-reducing medications (Box 7.13). In about one-third of patients, one dilatation alone is sufficient to control the dysphagia. Frequent recurrence of dysphagia in the younger patient is an indication for surgery — in most cases fundoplication is performed either laparoscopically or at laparotomy. Other indications for surgery are patients (young or old) with aspiration pneumonitis, Barrett’s change or evidence of dysplasia, persistent ulceration and iron deficiency anaemia.

Figure 7.43 Oesophageal dilatation of benign stricture
Oesophageal dilatation of benign stricture is performed using A: the Eder-Puestow (graded size metal olives); or B: Celestin dilators (stepped dilators in two sizes). Initially a flexible guide wire is passed under vision through the stricture into the stomach. The encoscope is then removed, leaving the guide wire in place and the dilators are passed over the wire.
Box 7.13
Management of benign stricture secondary to reflux
![]() Dilatation
Dilatation
![]() Medical measures to control reflux oesophagitis
Medical measures to control reflux oesophagitis
![]() Endoscopic surveillance to exclude Barrett’s change or dysplasia
Endoscopic surveillance to exclude Barrett’s change or dysplasia
![]() Anti-reflux surgery in intractable cases and younger patients
Anti-reflux surgery in intractable cases and younger patients
3. Carcinoma of the oesophagus
The main aim of treatment is to control dysphagia for the life of the patient. This is often best achieved by surgical resection. With adequate preoperative preparation, resection is possible in about 40% of cases. Selection is based upon the stage of the carcinoma and the severity of concurrent disease, particularly chronic obstructive airways disease (Table 7.9).
Table 7.9 Management of carcinoma of the oesophagus
|
Stage and incidence |
Site and extent of disease |
Treatment |
|
Stages I and II (30–40%) |
Local disease |
Oesophagectomy |
|
Confined to oesophagus |
||
|
Stage III (10–20%) |
Advanced local disease |
Chemotherapy or radiotherapy or surgical bypass |
|
Length >10 cm |
||
|
Mediastinal sinus on X-ray |
||
|
Break in oesophageal axis on X-ray |
||
|
Local extra-oesophageal extension on CT scan |
||
|
Stage IV (50%) |
Advanced local disease |
Intubation or laser photocoagulation |
|
Poor general condition |
||
|
Advanced extraoesophageal disease |
||
|
Metastases |
Clinical or radiological evidence of poor general condition or extra-oesophageal disease is an indication for a more conservative approach that varies from surgical bypass, to intubation and laser photocoagulation (Fig 7.44). Preoperative radiotherapy and chemotherapy can lead to a reduction in the stage of the tumour and render a previously inoperable tumour operable. The symptoms of advanced disease (stages III and IV) include anaemia, constant pain, hoarseness, cough on swallowing and haemoptysis. The signs of advanced disease are poor general condition with cachexia, a lesion that is greater than 10 cm in length, a mediastinal sinus from the tumour on X-ray, an abnormal oesophageal axis on barium swallow and extensive extra-oesophageal spread of tumour on CT scan.

Figure 7.44 Palliative treatment of advanced carcinoma of the oesophagus in the poor-risk case
A: endoscopic intubation using an Atkinson or Celestin tube; B: neodymium — Yttrium-Argon laser photocoagulation
Approximately 20% of lesions occur in the upper third, 30% in the middle third and 50% in the lower third of the oesophagus. For squamous carcinomas resection should extend at least 5–10 cm above the tumour, because of the frequency of intramural spread of these tumours. The stomach is the best organ to use for bypass and the procedure of choice for middle and lower third lesions and for adenocarcinoma of the cardia is Ivor Lewis-Tanner oesophagogastrectomy (Fig 7.45). With upper third lesions radiotherapy or laryngo-esophagectomy is indicated.

Figure 7.45 Ivor Lewis-Tanner oesophagogastrectomy for carcinoma of the middle and distal third of oesophagus, and gastric cardia. The stomach is mobilised on the right gastric (1) and right gastroepiploic arteries (2). The left gastric artery (3) and short gastric arteries (4) are divided. After thoracotomy and resection of the tumour (5) the stomach is passed up into the right chest and its fundus (A) anastomosed to the oesophagus. The gastric resection line (B) has been closed primarily. A pyloroplasty aids gastric emptying.
4. Achalasia and diffuse oesophageal spasm
Transthoracic or transabdominal cardiomyotomy (Heller’s procedure) is the treatment of choice in most cases, particularly in those patients with advanced disease or vigorous achalasia, in children and for failed balloon dilatation. Heller’s myotomy is performed by division of the circular muscle over the distal 10 cm of oesophagus. Pneumatic or hydrostatic dilatation procedures are generally reserved for elderly and debilitated patients. Forcible dilatation is carried out by sudden expansion of a pneumatic bag to the limit of a 4-cm outer nylon sheath. Initially, anticholinergic drugs or long-acting nitrates often give some relief for diffuse oesophageal spasm but eventually they become ineffective. Cold foods should be avoided and food should be chewed carefully and eaten in a relaxed environment. Sedatives may be of value. Control of concurrent disease, especially congestive cardiac failure, often helps to reduce the episodic symptoms of pain and dysphagia. In more severe cases a long oesophagomyotomy is the treatment of choice. The extent of the division required is defined by manometry. An oesophageal hiatus hernia may need repair at the same time.
5. Less common causes
The less common causes of dysphagia may be considered in two main groups: motility disorders and organic diseases.
Motility disorders. Cricopharyngeal achalasia with pharyngeal (Zenker’s) pouch (Fig 7.46) is a condition of the elderly that is manifest by cervical dysphagia, more marked for solids than liquids. Cricopharyngeal spasm leads to a proximal pulsion (Zenker’s) diverticulum in the posterior midline area of relative pharyngeal muscle deficiency (Killian’s dehiscence). Treatment is by division of the cricopharyngeus, with resection and repair of the pouch (unless it is smaller than 2 cm in diameter). In addition to dysphagia, the symptoms of pharyngeal pouch include gurgling on swallowing, foul halitosis, regurgitation of undigested food and sometimes aspiration pneumonitis.
Figure 7.46 Cricopharyngeal achalasia with pharyngeal (Zenker’s) pouch
Pharyngeal pouch is a pulsion diverticulum secondary to high pharyngeal pressure above spasm of the cricopharyngeous muscle. A: Lateral view shows the pouch coming from between the cricopharyngeal and thyropharyngeal portions of the inferior constrictor. B: Posterior view shows Killian’s dehiscence, an area of muscle deficiency and weakness through which the pouch develops. The pouch usually extends to the left side of the neck. As it enlarges, the opening comes in line with the oesophageal axis, making endoscopy potentially hazardous. C:Lateral image of contrast study.
In diabetes mellitus, dysphagia is uncommon but, in some patients, motility studies reveal diminished primary peristaltic waves. These may be due to diabetic autonomic neuropathy and can cause dysphagia.
Chagas’ disease is due to infection with Trypanosoma cruzi, a condition found mainly in central South America. The pathology is similar to that of achalasia with destruction of the myenteric plexus, often with more distal gastrointestinal involvement and myocardial disease. The oesophagus, and sometimes the duodenum and colon, becomes grossly dilated. The majority of patients require oesophagomyotomy to control dysphagia.
Scleroderma is associated with atrophy of the smooth muscle of the lower third of the oesophagus and diminished tone in the lower oesophageal sphincter. There is failure of peristalsis and the initial symptoms are from reflux with regurgitation and heartburn. Dysphagia develops at a later stage and is due mainly to benign stricture secondary to reflux oesophagitis. Oesophageal symptoms become manifest after the other features of the disease — such as Raynaud’s phenomenon and skin changes — are well established. The treatment is conservative.
Various neurological disorders are associated with cervical dysphagia, due either to spasticity or hypotonicity of the cricopharyngeal sphincter. Persistent spasticity of the upper sphincter that continues during swallowing is found in multiple sclerosis and after stroke, Parkinsonism, pseudobulbar palsy and poliomyelitis. Hypotonia is found in conditions such as thyrotoxic myopathy, dystrophia myotonica and myaesthenia gravis. The diagnosis can be made by cineradiography during deglutition. Treatment is mainly directed at the underlying condition. In some patients with spasticity cricopharyngeal myotomy may control dysphagia.
Organic diseases. Para-oesophageal hiatus hernia is an unusual cause of dysphagia. In this condition the gastro-oesophageal junction remains at or below the diaphragm, while the fundus of the stomach rolls into the chest through the oesophageal hiatus (Fig 7.47). The symptoms include retrosternal fullness and distension after eating, with a sensation of pressure similar to angina. A retrocardiac fluid level on plain X-ray is diagnostic of the condition. Because of the danger of obstruction and strangulation and the possibility of gastric volvulus, most patients should be treated surgically if their condition permits. Surgical treatment is by repair of the hernia and anterior gastropexy. This can be done laparoscopically in skilled hands and therefore avoid painful thoracic incisions that enhances the postoperative recovery in these often elderly patients.

Figure 7.47 A: Para-oesophageal hiatus hernia. B: Volvulus of the stomach in a paraoesophageal hiatus hernial sac. The stomach has rotated on its long axis (organo-axial volvulus). This produces an acute kink in the distal oesophagus that causes dysphagia, bloating without vomiting, and prevents the passage of a nasogastric tube. An air–fluid level is seen in the chest behind the heart on plain X-ray.
Persistent dysphagia may occasionally follow oesophageal moniliasis, which is usually secondary to prolonged antibiotic treatment. Dense strictures may follow corrosive ingestion and many of these cases require surgical replacement of the oesophagus with colon. First aid treatment — with steroids and copious washing of the oesophagus with water — is essential. The other unusual organic diseases that may present with dysphagia include carcinoma of the left main bronchus, mediastinal node involvement, retrosternal goitre and benign and secondary tumours of the oesophagus.
7.13 Weight loss
Unexplained significant weight loss (involving at least 5% of normal body weight) is a relatively unusual form of presentation as the sole clinical problem. Most diseases associated with weight loss present because of another major problem, although the history may reveal that weight loss was the first suggestion of illness. Weight loss is a symptom that should always be taken seriously. Full investigation will lead to diagnosis of serious disease in many cases. On close questioning, other symptoms may be discovered that often appear insignificant to the patient, but can prove helpful in establishing the cause of weight loss. Two main groups of patients present: those in whom weight loss is due to depression and those with organic disease. The weight loss of dieting (unless concealed) is easily dismissed.
Common causes
1. Depression
2. Abdominal malignancy
3. Diabetes mellitus
4. Thyrotoxicosis
5. Chronic infections
6. Malabsorption
History
1. Depression
Depression is very commonly associated with weight loss (or sometime with weight gain). In many patients the depression is not readily acknowledged and is then said to be masked. The history often reveals that weight loss dates from the loss of someone or something to which the patient was particularly attached. Other characteristic features and psychosomatic symptoms include loss of enthusiasm and drive, tiredness, loss of libido, headache, faintness, amenorrhoea, constipation and insomnia (especially early awakening with a feeling of doom). Attention may be drawn to masking minor gastrointestinal symptoms that resemble irritable colon. Together with the weight loss these are often attributed to cancer, a fear that, however, is rarely acknowledged. Alcoholism is common and may be particularly difficult to recognise. More severe degrees of alcoholism with liver failure may be associated with considerable muscle wasting. The presence of amenorrhoea and (extreme) weight loss in young women suggests anorexia nervosa. In these patients, very low food intake and self-induced vomiting (bulimia) may be resolutely concealed and such denial is delivered with a flat affect. Vague abdominal pain may result from constipation, leading to purgative abuse.
2. Abdominal malignancy
When abdominal malignancy develops insidiously the general symptoms can be similar to those of depression, although they are more severe and progressive. Carcinoma of the stomach or right colon may present with vague dyspepsia and weight loss. The former symptom is often mistakenly considered ‘soft’ and of little significance, although the combination of recent anorexia, altered taste and weight loss is always suggestive of the diagnosis. The weight loss of cancer of the body and tail of the pancreas can be rapidly progressive and profound, with marked debility. There may be vague back pain and constipation or diarrhoea. Occasionally the discovery of diabetes mellitus in a patient with weight loss alerts one to the possibility of cancer of the pancreas. With lymphoma the patient may have noticed swellings in the neck or axilla. The difficult form to diagnose is that of disease confined to the abdomen or chest, presenting as weight loss and a general deterioration in health.
3. Diabetes mellitus
A common presentation of diabetes is mild weight loss in older women. Careful questioning will generally reveal the presence of thirst and nocturia. Vulval pruritus is common. There may also be evidence of urinary tract infection, visual disorder or peripheral vascular insufficiency. In younger patients the condition tends to be more acute, with profound weight loss and the diagnosis is often easier to make.
4. Thyrotoxicosis
Weight loss is an important form of presentation of thyrotoxicosis. The condition is characterised by weight loss with normal or increased appetite, anxiety and irritability and heat intolerance. Menstrual disorders and diarrhoea are also common. When there is minimal neck swelling the diagnosis can be particularly difficult. In the patient who is restless and has a voracious appetite associated with weight loss, thyrotoxicosis should always be considered as a possible cause.
5. Chronic infections
Infections that must be considered particularly are tuberculosis with minimal respiratory symptoms, brucellosis, chronic viral infections (hepatitis, glandular fever and AIDS). Past history may be relevant to establish infectious contact. The presence of fever and lymphadenopathy is suggestive of chronic infection.
6. Malabsorption
Malabsorption is discussed at the end of Chapter 7.21.
Signs
1. Depression
A listless demeanour and mode of communication is suggestive of the diagnosis, especially in the relatively active patient, in contrast with the patient with malignant disease. Before the onset of the depressive illness the patient is often overweight. The history will often reveal a history of loss, poor self-esteem, guilt and an unrealistic perspective of talents and abilities. Minor signs of alcoholism may be found or immature development of secondary sexual characteristics may be noted in young females, suggesting anorexia nervosa.
2. Abdominal malignancy
Apart from cachexia in advanced carcinoma of the stomach and pancreas, there are often no specific physical signs in the patient with weight loss. A local mass may be found or evidence of metastatic disease may be obvious. Diagnosis of lymphoma requires careful assessment of the haemopoietic system, with particular reference to lymphadenopathy.
3. Diabetes mellitus
Physical examination is often normal. There may be hypertension, fundal changes, peripheral vascular disease and pruritus in younger patients.
4. Thyrotoxicosis
The florid physical signs of thyrotoxicosis are a hyperkinetic circulatory state, goitre with bruit and exophthalmos. In the patient for whom diagnostic difficulty exists, it may be virtually impossible on examination to distinguish thyrotoxicosis from anxiety state or anorexia nervosa.
Diagnostic plan
Haematological and biochemical examination
Iron deficiency anaemia and normochromic anaemia are common features of malignancy and suggest a serious organic cause. Raised white cell count can be found with infections or associated with diabetes. A raised serum lipase level should lead to the suspicion of an eating disorder (bulimia) in young females with no other evidence of pancreatitis. Thyroid function tests are indicated if thyrotoxicosis is a possibility.
Urine analysis and culture
Urinary examination for sugar should be routine for all surgical patients. Urinary infection is frequently found with diabetes mellitus and demonstrated on the urinoscopy and culture.
Organ imaging and endoscopy
A chest X-ray is indicated in all patients, particularly if looking for evidence of tuberculosis and mediastinal lymphadenopathy.
Exclusion of cancer is essential. The most useful tests to be performed are gastroscopy, colonoscopy and upper abdominal ultrasound, especially if any symptomatic evidence of gastrointestinal disease exists. Carcinoma of the caecum may also be associated with weight loss and a general deterioration in health. If anaemia is discovered and investigations of the gastrointestinal tract are normal, abdominal CT scan with contrast is indicated in the difficult case.
Serological and microbiological studies will be necessary to investigate for the presence of chronic infections such as AIDS.
It is important in patients with depression to establish to the satisfaction of the patient the link between emotional factors and the current problem and that cancer is not present. This will often produce significant improvement in appetite. If endogenous depression is suspected, particularly if there are suicidal thoughts, then psychiatric opinion must be sought.
Refinements in abdominal imaging make the recommendation of laparoscopy for unexplained weight loss very rare. For diabetes, thyrotoxicosis and specific infections the appropriate medical measures are indicated.
Weight gain
Weight gain is usually due to an increased amount of body fat and does have an impact on the patient’s wellbeing and can have an impact on the outcome of surgical procedures. Causes of weight gain to consider are summarised in Box 7.14.
Box 7.14
Common causes of weight gain
![]() Obesity — excessive food intake, decreased activity
Obesity — excessive food intake, decreased activity
![]() Anxiety and depression
Anxiety and depression
![]() Pregnancy
Pregnancy
![]() Fluid retention (renal, cardiac or hepatic insufficiency)
Fluid retention (renal, cardiac or hepatic insufficiency)
![]() Myxoedema
Myxoedema
![]() Cushing’s syndrome
Cushing’s syndrome
As assessment of obesity that influences the patient’s health and wellbeing should be done by a multidisciplinary team. Some of these patients may be candidates for bariatric surgery.
7.14 Vomiting
Acute transient vomiting is common in gastroenteritis and from many other causes — acute vomiting is not considered in this section. Persistent painless vomiting over many days or weeks is much less common, but is a clinical problem that must be taken very seriously. Two main conditions cause chronic vomiting: functional bowel disease and gastric outlet obstruction. Organic obstruction is suggested by vomiting that is nocturnal or late in the day after meals, of large volume and containing undigested food. Functional vomiting is rarely associated with a significant loss of extracellular fluid or malnutrition. The vomiting of functional bowel disease or psychogenic states affects younger patients, is mainly in the morning, early after meals, of small volume and rarely disabling. Depression is a common accompaniment of functional vomiting.
Anorexia and nausea usually go with chronic vomiting. Anorexia suggests organic disease, particularly when associated with weight loss. Anorexia as a sole symptom is uncommon. Anorexia and nausea, with or without vomiting, are often found as a complication of medication with certain drugs. Occasionally anorexia and vomiting may be the presenting symptoms of gastric and pancreatic carcinoma, renal disease, tuberculosis, diabetes and thyrotoxicosis — a spectrum of causes similar to that for weight loss (Ch 7.13). Nausea is rarely found as an isolated symptom, usually being accompanied sooner or later by vomiting. Vomiting, however, can occur without nausea. The regurgitation and water brash associated with oesophageal disease may be described as vomiting by the patient so an accurate history is important. Heartburn is not necessarily an associated symptom in such patients. Occasionally vomiting without nausea is the main complaint in patients with central nervous system disease or pharyngeal pouch. Gallstones do not cause painless chronic vomiting.
Causes
1. Duodenal ulcer
2. Carcinoma of the pyloric antrum
3. Functional bowel disease and psychogenic vomiting
4. Drug-induced vomiting
5. Less common causes
History
1. Duodenal ulcer
Varying degrees of nausea and vomiting are relatively common in duodenal or prepyloric ulcer (Ch 7.6). The cycles of relapse and repair in peptic ulcer disease may cause obstruction of the gastro-duodenal junction as a result of oedema, muscular spasm and scarring. With marked scarring (usually following a long history of dyspepsia), a change in the pattern of the illness occurs, with decrease in pain. Initially the pain becomes more diffuse and resistant to medical treatment and is more frequently relieved by vomiting. Eventually, in the neglected case, the vomiting becomes painless, of large volume, bile-free, projectile and contains stale food from meals taken hours previously. Vomiting typically occurs late in the day, perhaps after the evening meal. Weakness and weight loss are common. Constipation is frequent because of an inadequate intake, but diarrhoea is found in some patients. A combination of large volumes of regurgitated and vomited fluid at night, associated with diarrhoea, suggests that hypergastrinaemia (Zollinger-Ellison syndrome) may be present. Occasionally, patients may present without vomiting and some may have mental confusion related to prerenal injury. Tetany may also appear with advanced alkalosis. Patients with gastric outlet obstruction rarely present in coma due to the resulting metabolic disturbance.
Gastric ulcer less commonly causes obstruction from an hourglass deformity in the mid-stomach, but occasionally elderly patients with nonobstructing gastric ulcer can present with painless vomiting, probably more because of associated bacterial gastritis. The transient vomiting that is seen with nonobstructing duodenal ulcer may also be due to bacterial gastritis.
2. Carcinoma of the pyloric antrum
Patients with carcinoma of the antrum usually have a much shorter history than is found with duodenal ulcer (Table 7.10). The vomiting is usually preceded by anorexia and weight loss. Vomiting may have a coffee-ground appearance owing to bleeding from the tumour. Pain may be completely absent or a vague postprandial epigastric fullness may be present, which the patient does not identify as pain. Weight loss, anorexia and general deterioration in health are features of the illness.
Table 7.10 Comparison of benign and malignant gastric outlet obstruction
|
Duodenal ulcer |
Carcinoma of the antrum |
|
|
Length of history |
Long |
Short |
|
Pain |
Past history typical of ulcer (70%) |
Pain often absent |
|
Signs of gastric enlargement |
Common |
Unusual |
|
Mass at pylorus |
Very unusual |
Common (50%) |
|
Fluid and electrolyte and nutritional deficiencies |
Common: Na+, Cl+, K+, H+, H2O deficiency |
Weight loss often marked, ECF depletion common |
|
Metabolic alkalosis |
Common |
Rare because of associated achlorhydria |
3. Functional bowel disease and psychogenic vomiting
The diagnosis of functional bowel disease is not just a matter of excluding organic disease. As with the other clinical manifestations of this disease, presentation with vomiting is often triggered by one or more of a heterogeneous group of factors. Recurrent vomiting may be associated with eating certain foods, suggesting the possibility of food allergy. The vomiting tends to be minor in degree and mainly occurs in the morning, but is often diurnal in pattern. There may be a long episodic history but without progression and sometimes starting in childhood. Symptoms of irritable colon (with diarrhoea, bloating and tenesmus) are frequently discovered to be present in the recent past. Psychogenic precipitating factors, such as depression, are often important. Alternatively, depression may be masked, but its presence suggested by insomnia, fatigue, anorexia, loss of interest, loss of libido and mood change. Alcoholism is also common. Occasionally in young women, when the condition is associated with severe weight loss, a diagnosis of anorexia nervosa can be made (Ch 7.13). Another severe, related condition is the superior mesenteric artery syndrome, where this vessel causes obstruction by compressing the duodenum. However, many doubt the existence of this syndrome, unless associated with acute loss of weight following injury. Concealed pregnancy is occasionally a cause of vomiting in female patients.
4. Drug-induced vomiting
Vomiting induced by a drug is characterised by persistent anorexia and nausea, without pain. Sometimes vomiting follows immediately after the start of treatment and sometimes the onset is insidious. An insidious development of toxicity is common in older patients on digitalis treatment, for which there is a narrow margin between therapeutic and toxic levels of the drug. Drugs may induce vomiting for a number of reasons. These include gastric irritation by preparations such as iron, aminophylline and salicylates, and direct stimulation of the chemoreceptor centre by agents like digitalis, opiates, practolol and oestrogens (Box 7.15). Methyldopa and antihistamine preparations also induce vomiting by a central effect. Compounds such as sulfonamides and broad spectrum antibiotics can cause vomiting through both central and peripheral mechanisms.
Box 7.15
Drugs that can cause chronic vomiting
Iron
Aminophylline
Salicylates
Digitalis
Opiates
Practolol
Oestrogens
Methyldopa
Antihistamines
Sulfonamides
Antibiotics
Examination
Functional bowel disease and drug-induced vomiting are rarely associated with abnormal physical signs or evidence of fluid and electrolyte abnormality. Patients with obstructing duodenal ulcer and carcinoma of the pyloric antrum have a varying degree of ECF deficiency and malnutrition. Malnutrition, with a short history, is more a feature of carcinoma of the stomach, marked ECF loss and a long history of duodenal ulcer. Gastric succussion splash, a feature of duodenal ulcer (less common with carcinoma), is best detected several hours after a meal. Occasionally, visible peristalsis will be seen in the epigastrium, with fullness in the left hypochondrium. A pyloric mass is very suggestive of carcinoma. Respiratory problems are common in these patients and are often secondary to aspiration pneumonitis.
The general clinical features of prolonged pyloric stenosis are muscle weakness, paresis, constipation and polydypsia, with a biochemical deficit of hypochloraemic hypokalaemic alkalosis. These metabolic acid–base effects are less common with obstruction due to cancer, but considerable weight loss and fluid and electrolyte losses, can occur with malignant obstruction.
Diagnostic plan
Urinalysis
Protein or sugar in the urine favours the possibility of a gastrointestinal cause of vomiting, but perhaps more commonly indicates the presence of concurrent illness such as renal disease or diabetes.
Haematology
Anaemia suggests the diagnosis of carcinoma. Haemoconcentration is common in duodenal ulcer obstruction.
Biochemistry from urine and blood
The defect that develops in pyloric stenosis is a metabolic hypochloraemic hypokalaemic alkalosis with ECF loss. Measurement of the severity of this disorder determines both the measures taken to correct the disorder and the timing of surgery. Three main stages are usually described (Table 7.11) and depend upon the cause and completeness of gastric outlet obstruction and the delay in presentation.
Table 7.11 Pyloric stenosis: stages of fluid and electrolyte disorders

The hydrogen ion in gastric secretion is obtained from carbonic acid within the parietal cell and is secreted and lost into and from the gastric lumen together with chloride ion, while bicarbonate passes into the extracellular fluid. Vomiting depletes the patient of sodium, potassium and chloride ions; the latter is lost in excess of sodium and potassium as hydrochloric acid. Gastric acid loss causes extracellular alkalosis. Increasing sodium and water deficit stimulates aldosterone secretion. This leads to renal conservation of sodium at the expense of increasing loss in the kidneys of potassium and hydrogen ion, ultimately producing acid urine containing increased levels of hydrogen ion. Glomerular filtration rate may fall sufficiently to produce prerenal injury. Eventually there is a marked deficit of sodium, potassium and chloride ions and of water. Therefore, treatment involves replacement of water and sodium chloride isotonically until the urine output is restored, then potassium chloride replacement can be added.
Radiological investigation
Plain abdominal X-ray may show a large gastric fluid level. CT scanning with oral contrast will often show a dilated stomach and, if the diagnosis is a cancer, provide information about metastatic disease. The causative lesion (benign ulcer or carcinoma) is may be difficult to demonstrate on radiological studies.
Endoscopy can be difficult to interpret but can provide a positive diagnosis of carcinoma on appearance and by biopsy. Both radiology and endoscopy are best performed after emptying the stomach.
Treatment plan
Nasogastric suction
Gastric aspiration enables continuing losses to be estimated and replaced accurately, reduces gastric dilatation, prepares the stomach for surgery and reduces the bacterial content of the stomach. Gastric emptying can also reduce the danger of pulmonary aspiration during early management. A very large amount of gastric acid, of more than 5 L output per day, suggests a more distal obstruction such as upper small bowel obstruction or the Zollinger-Ellison syndrome.
Correction of extracellular fluid loss and hypokalaemic alkalosis
• Extracellular fluid deficit and continuing nasogastric loss is replaced with isotonic sodium chloride. The fluid balance is monitored by serial measurement of haematocrit, serum creatinine and urine output, which is initially monitored hourly. With adequate volume replacement, the stimulus to tubular sodium resorption is diminished and the kidneys can then excrete the excess bicarbonate.
• Potassium replacement is commenced when a satisfactory urine output is demonstrated. The correction of electrolyte balance is monitored by serum electrolytes and osmolality, by the urinary potassium and occasionally by the measurement of urinary bicarbonate.
• If chronic obstruction has produced severe malnutrition, total parenteral nutrition (TPN) should be instituted. The response is assessed by serial body weight and serum albumin.
Surgical intervention
Surgery for duodenal ulcer is generally possible after 72 hours of resuscitation. It may be of value to continue TPN for up to a week before subjecting the patient with carcinomatous obstruction to surgery. During this period of time concurrent disease can be controlled; however, prolonged nasogastric suction and immobilisation may worsen respiratory disease and increase the risk of surgery — thus operation should not be delayed unduly. Bacterial overgrowth is common in the obstructed stomach as there is an increased risk of perioperative infection; a prophylactic broad-spectrum antibiotic is indicated in all cases.
Vagotomy and antrectomy, with a Billroth I anastomosis, is the procedure of choice for obstructed duodenal ulcer, especially in patients where despite gastric aspiration gastric dilatation persists to the time of surgery. A more conservative surgical approach, substituting pyloroplasty for antrectomy, is indicated in the patient in poor general condition and in such a patient postoperative eradication of H. pylori is essential. For carcinoma of the pyloric antrum within 7 cm of the pylorus, distal partial gastrectomy with Billroth II anastomosis will achieve local control of the disease for the life of the patient. For more extensive or more proximal lesions the procedure of choice is total gastrectomy and Roux-en-Y oesophagojejunostomy to ensure complete removal of the lesion and thus avoid anastomotic recurrence (Fig 7.48).

Figure 7.48 Gastrectomy
B1 = Billroth gastrectomy, B2 = Billroth II (polya) gastrectomy and TG = total gastrectomy and Roux-en-Y oesophagojejunostomy
Based on William, 1998
5. Less common causes
Non-gastrointestinal disease. Patients with renal failure, chronic otitis media in children, diabetes mellitus, thyrotoxicosis and (rarely) intracerebral tumours near the fourth ventricle may present with chronic vomiting. Occasionally, smokers with chronic bronchitis may present with morning ‘vomiting’. These patients have a chronic cough and, with postnasal drip at night, are prone to bouts of coughing and vomiting after the first cigarette in the morning.
Gastrointestinal disease. Post-gastrectomy vomiting may not be readily recognised because it can start many years after surgery. Characteristically it is food-free and is described as bilious vomiting. In most, but not all patients, other post-gastrectomy symptoms are present, including diarrhoea and ‘dumping’. Post-gastrectomy bile vomiting is difficult to treat medically and may require surgery. Upper small bowel obstruction is an important but unusual cause of chronic painless vomiting. There may be little or no pain and minimal signs on the plain X-ray of the abdomen. When suspected, a small bowel enema provides the diagnosis. Occasionally, chronic pancreatitis and carcinoma of the head of the pancreas present with vomiting because of duodenal obstruction. Rare causes include duodenal mucosal diaphragm, bezoars, gastric leiomyoma, retroperitoneal lymphoma and painless femoral hernia. The elderly woman with obscure vomiting may well have a strangulated femoral hernia or, it must be remembered, digitalis toxicity. The hernial orifices must always be examined in all patients presenting with vomiting.
7.15 Jaundice
Jaundice (icterus) is a yellow staining of the body tissues, particularly the skin, sclera and blood vessels, that have a high elastic tissue content.
Jaundice is often detected by the patient or by relatives as yellowing of the sclera or skin. In such instances jaundice is clearly the dominant problem, but even when other symptoms are present jaundice usually remains the focal point of the clinician’s history and physical examination. Associated symptoms (such as biliary pain) help to diagnose the cause of jaundice. Jaundice can be a prognostic sign rather than a diagnostic problem, for example, when seen in patients with advanced cirrhosis of the liver or congestive cardiac failure or in the severely ill, septic patient.
Jaundice is just detectable when the serum bilirubin rises above 40 micromol/L, the normal level being less than 20 micromol/L. Bilirubin is derived from the breakdown of haem within the reticuloendothelial system. Bilirubin is transported to the liver attached to plasma protein. In the liver cell, lipid-soluble bilirubin is conjugated as water-soluble bilirubin glucuronide for excretion in the bile. In the majority of instances in adults, jaundice results from failure to excrete conjugated bilirubin. This may be due to obstruction within the liver or of the extra-hepatic biliary tree, leading to retention of bilirubin, mainly in its conjugated form. In adult patients, rather than categorising the common causes of jaundice as haemolytic, hepatocellular or obstructive, it is best to ask the question: Is the jaundice due to hepatic (medical) or extrahepatic (surgical) disease?
Causes
1. Stone in the bile duct
2. Carcinoma of the head of pancreas
3. Alcoholic liver disease
4. Drug-induced jaundice
5. Viral hepatitis
6. Less common causes
History
1. Stone in the bile duct
Approximately 5–10% of patients with gallstones have stones in the bile duct. About half these are asymptomatic, hence the need for routine operative cholangiography during cholecystectomy. Bile duct stones may present because of biliary pain, jaundice or pancreatitis or a combination of these features, with or without evidence of cholangitis. Characteristically, jaundice due to gallstones occurs intermittently, is of mild to moderate degree, is accompanied by biliary pain and usually resolves within a day or two of admission to hospital. The pain cannot be distinguished from that caused by stones within the gall bladder. Some patients note transient darkening of the urine during an episode of biliary pain, without obvious jaundice. A past history of episodic biliary pain is common. Past biliary surgery suggests the diagnosis of a retained or missed stone in the common bile duct or the possibility that postoperative biliary stricture is the cause of jaundice. Jaundice and pain are often associated with fever. In a few patients suppurative cholangitis is present with high fever and septic shock. The combination of intermittent jaundice, right hypochondrial pain and fever and chills is found in about a third of patients. This syndrome is known as Charcot’s intermittent hepatic fever, a term also used to describe the clinical characteristics of oriental cholangiohepatitis.
2. Carcinoma of the head of the pancreas
Patients with carcinoma of the head of the pancreas classically present with painless, profound and progressive jaundice associated with marked listlessness, anorexia and weight loss. In fact, many patients have dull, deep-seated, nauseating upper abdominal pain that can resemble biliary pain. Radiation of pain through to the back is common and is usually a sign of advanced disease. Fluctuating jaundice is present in about 10% of patients, especially in those with ampullary tumours and cholangiocarcinoma. Ampullary tumours can also produce the sign of an intermittently palpable gall bladder mass. Fever from cholangitis, in patients with jaundice due to carcinoma, is uncommon. Cholangitis is more common with bile duct than with pancreatic tumours. Pruritus due to accumulation of bile salts in the subcutaneous tissues is common with prolonged deep jaundice. It is felt especially in the extremities and is worse in warm weather.
3. Alcoholic liver disease
When alcoholic liver disease must be considered in the differential diagnosis of jaundice, the pathology is predominantly that of alcoholic hepatitis, with fatty infiltration, mild to moderate cirrhosis and usually no evidence of liver failure. Differentiation from surgical causes in these cases can be difficult. A history of a recent alcoholic binge is common. On occasions a cholestatic pattern of jaundice may last for weeks and be associated with pruritus.
4. Drug-induced jaundice
Drugs may produce both a hepatitis-like illness and a predominantly cholestatic picture. Commonly used drugs associated with jaundice are methyltestosterone, phenothiazines, oral hypoglycaemics, methyldopa and oral contraceptives. Pain is common. It is usually continuous and located over the liver. In most instances hepatotoxicity is due to a hypersensitivity reaction and jaundice settles within a few weeks. Chronic active hepatitis is an uncommon sequel.
5. Viral hepatitis
Viral hepatitis occurs usually in patients under 30 years of age, preceded by a prodrome of anorexia, nausea, weight loss and fatigue. Subcostal aching pain over the liver is common. A history of contact with patients with hepatitis, intravenous drug usage or blood transfusion should be sought. Hepatitis may be due to A, B, C or other viral infection. Hepatitis B and C have the worst prognosis, both in the short term and from chronic hepatitis.
Examination
Most patients with stones in the bile duct have no specific or diagnostic physical signs. In a small proportion of patients acute cholecystitis, with a painful gall bladder mass, is present. Sometimes epigastric tenderness points to associated pancreatitis. Fever, leucocytosis and toxaemia of varying degree occur in about one-third of patients; blood culture is often positive in patients with severe toxaemia.
Profound jaundice, with weight loss and pruritus, is suggestive of malignant obstruction. A characteristic, but certainly not an invariable (or even diagnostic) clinical finding in carcinoma of the head of the pancreas, is an enlarged nontender gall bladder. This sign is stated as Courvoisier’s law: in a patient who is jaundiced and has a palpable gall bladder, the cause of jaundice is unlikely to be from stone. Hepatomegaly is uncommon and when present does not necessarily mean metastatic liver disease. Evidence of cholangitis is unusual in patients with malignant jaundice. A pancreatic primary neoplasm is rarely palpable.
Alcoholic hepatitis is usually associated with hepatomegaly, but often signs of liver insufficiency are few. Splenomegaly is an infrequent finding. In drug-induced or viral hepatitis the liver is usually enlarged and tender. Tattoos and evidence of puncture marks associated with drug addiction may be found.
Diagnostic plan
In most cases, a provisional diagnosis can be made from the history and physical examination. The most useful supplementary diagnostic tests are ultrasound and CT scan.
Haematology
Some tests are of value both diagnostically and for preoperative preparation. An elevated white cell count suggests cholangitis and indicates the need to treat sepsis. Clinical or chemical evidence of sepsis suggests that a stone in the bile duct is likely as a cause of jaundice. It is important to take blood cultures before starting antibiotics in patients with cholangitis to detect early the infecting organisms and their antibiotic sensitivities.
Coagulation tests are essential, as jaundiced patients absorb vitamin K poorly and frequently have a prolonged prothrombin time. All patients require vitamin K treatment. A platelet count and thromboplastin time (PTTK) are also necessary.
Biochemistry
Liver function tests are not particularly useful in making a diagnosis. They are measured primarily to give a baseline for monitoring the patient’s progress. The following points are sometimes helpful.
• Bilirubin of greater than 150 micromol/L is very suggestive of malignant jaundice.
• Hepatitis is suggested by a markedly elevated serum transaminase.
• Markedly elevated serum alkaline phosphatase suggests metastatic liver disease or unusual cholestatic diseases such as primary biliary cirrhosis.
• An isolated markedly raised bilirubin with minimal changes in other biochemical tests of liver function suggests prolonged cholestasis from intrahepatic disease.
• It is essential determine the renal function in jaundiced patients and manage their fluid status to avoid ‘hepatorenal syndrome’.
Diagnostic and interventional imaging: ultrasound and CT
Ultrasound and CT scanning (which may provide enhanced images as a CT cholangiogram) are complementary investigations in the diagnosis of the cause of jaundice. Ultrasound is most useful in thin patients, especially to detect gallstones and duct dilatation. Most gallstones are radiolucent radiologically but, with ultrasound, they appear as mobile, shadowing, echogenic foci in a sonolucent gall bladder (Fig 7.49). With real-time ultrasound, sections can be obtained in any plane. Thus, it is possible to follow the dilated ducts to the cause. Ultrasound is also useful in detecting liver metastases and other nonneoplastic diffuse liver diseases (cirrhosis). Unfortunately, ultrasound is operator-dependent and is also limited by obesity, scarring and bowel gas.

Figure 7.49 Sonolucent gall bladder
Most gallstones are radiolucent radiologically but, with ultrasound, they appear as mobile, shadowing, echogenic foci in a sonolucent gall bladder.
From Henry, 2004
The standardised transverse axial sections of CT give better anatomic resolution, capable of better definition of pancreatic masses or liver secondaries. Associated pancreatitis may exaggerate tumour size. Either CT or ultrasound can be used for guiding fine needle aspiration cytology (FNAC), which aids histological diagnosis of pancreatic masses or localised hepatic lesions.
Magnetic resonance cholangiopancreatography (MRCP) is available in some centres and can provide detailed anatomical information to assist with planned resections of malignant obstructive lesions.
Liver biopsy
This is indicated when imaging techniques suggest that the cause of jaundice is intrahepatic. Coagulation disorders must be corrected before liver biopsy. Careful observation after the procedure is necessary to detect postbiopsy bile leak and peritonitis. The assessment of histological changes may be difficult: features looked for are those of fatty change, hepatitis, cirrhosis, intrahepatic or extrahepatic obstruction and malignancy.
ERCP
This is indicated when the diagnosis is in doubt, especially when a less common cause of jaundice, such as bile duct tumour, is suspected. Endoscopic procedures are also often chosen as a form of treatment of obstructing lesions in high-risk patients with relative contraindications to surgery. Endoscopic sphincterotomy for a stone in the bile duct (Fig 7.50) or endoscopic intubation, possibly with stenting, for patients with carcinoma may be indicated.

Figure 7.50 Endoscopic sphincterotomy
ERCP diathermy sphincterotomy before basket removal of a stone or insertion of a stent to bridge an obstruction.
PTC
Percutaneous cholangiography (PTC) is indicated in the case where ERCP has failed to make a diagnosis or was an unsuccessful method of treatment, but rarely for preoperative planning in patients with bile duct tumours, as CT and MRCP fulfills this requirement in a noninvasive way. PTC is more often required in patients with obstruction high in the bilinary tree.
Treatment plan
Treatment aims specifically to relieve obstructive jaundice by the safest and most effective method.
Important aspects of preoperative preparation in the jaundiced patient are:
• prophylactic antibiotics to assist in the control and prevention of sepsis
• treatment with vitamin K to prevent coagulation problems
• maintenance of urine output by precise control of fluid balance, recognising the increased danger of acute renal failure in the jaundiced patient (hepatorenal syndrome).
1. Stone in the bile duct
Spontaneous resolution of the jaundice occurs clinically in the majority of cases of a stone in the common bile duct (Box 7.16). Operation can then be performed as a semi-elective procedure. In the minority of patients whose illness worsens with persisting jaundice — including those with suppurative cholangitis — it is safer to arrange treatment (especially in old and infirm patients) in stages. The first procedure in this group is often ERCP and sphincterotomy to relieve the obstruction, followed, especially in patients with an obstructed cystic duct, by later elective cholecystectomy (subsequent calculus cholecystitis is more likely to occur in the presence of cystic duct obstruction).
Box 7.16
Management of bile duct stone in four clinical situations
Incidental finding at cholecystectomy (operative cholangiography)
Exploration of bile duct
Resolving jaundice
Elective surgery when jaundice resolves:
![]() cholecystectomy, operative cholangiography to determine if stone still present
cholecystectomy, operative cholangiography to determine if stone still present
Persistent jaundice
Staged procedure — especially in old and unfit patient:
![]() ERCP, sphincterotomy and stone removal
ERCP, sphincterotomy and stone removal
![]() later elective cholecystectomy
later elective cholecystectomy
Suppurative cholangitis
Urgent ERCP, sphincterotomy and drainage
Later elective cholecystectomy
Urgent surgery (choledochotomy), if endoscopic sphincterotomy fails
In the younger patient, sphincterotomy — operative or endoscopic — should be avoided if possible and urgent surgical relief of jaundice is required. The operation is usually performed laparoscopically. The gall bladder is removed and an operative cholangiogram is obtained. The number and site of stones is noted on the X-ray. The stones may be able to be removed via the cystic duct or a stent may be placed via the cystic duct into the duodenum to facilitate later endoscopic stone retrieval. Sometimes laparoscopic choledochotomy can be performed when necessary for stone retrieval. If this is not technically feasible, conversion to open choledochotomy may be indicated.
2. Carcinoma of the head of the pancreas
The main principles of treatment are control of obstructive jaundice (by tumour resection if possible) for the life of the patient, with anticipation and treatment of associated symptoms of pain or vomiting from duodenal obstruction. The measures necessary depend on the type and stage of the tumour and the general condition of the patient (Box 7.17). The most appropriate treatment plan should be determined by a multidisciplinary team.
Box 7.17
Management of carcinoma of the head of pancreas
Terminal disease (metastatic): palliative care
Locally advanced disease — poor-risk case: ERCP stent
Locally advanced disease — good-risk case: double bypass
Local disease — good-risk case: Whipple resection (pancreaticoduodenectomy)
Approximately 50% of patients are in good general condition and best palliation is achieved by a double bypass, which consists of gastrojejunostomy and Roux-en-Y choledochojejunostomy (Fig 7.51). These two procedures relieve the jaundice and prevent the subsequent vomiting from duodenal obstruction that will occur in most patients if they live long enough. Pain is also relieved, in most patients, by biliary bypass. Duodenal ulcer can occur after biliary bypass procedures and should be excluded by endoscopy if recurrence of epigastric pain occurs after palliative surgery. Tumour recurrence causing pain is treated by percutaneous splanchnic nerve block or chemotherapy.

Figure 7.51 Palliative double bypass for obstructing carcinoma of the pancreas or distal bile duct
The procedures performed are A: Roux-en-Y choledochojejunostomy to control jaundice and; B: gastrojejunostomy to prevent later symptoms from duodenal obstruction.
In patients with more advanced disease, with severe weight loss and inanition, benefit is best obtained by the endoscopic retrograde insertion of a hollow tube endoprosthesis or stent. In many centres, this is the treatment of choice for all such cases, with operative management reserved for cases where stenting has been unsuccessful or there is concomitant gastric outlet obstruction, which is relatively rare. If this is not possible or fails, an attempt can be made to stent the tumour via the percutaneous transhepatic route. In only about 5–10% of patients is it possible to resect the primary tumour — by Whipple’s procedure of pancreaticoduodenectomy (Fig 7.52). Most resections are performed for ampullary tumours. A pancreatic tumour, small enough so that the portal vein is free of tumour involvement, is an unusual finding. The tumour must usually in such instances be less than 3 cm in diameter and unfortunately most pancreatic cancers present at a later stage. Coeliac axis anaesthetic block for pain control in advanced malignancy may also be useful in short-term palliation.

Figure 7.52 Whipple’s procedure
Whipple’s operation (pancreatoduodenectomy) for resectable carcinoma of the head of the pancreas or ampullary tumours. The nonshaded areas are resected, vagotomy is performed and intestinal continuity is reestablished by A: invaginating the pancreas into the terminal jejunum; B: anastomosing the bile duct to the side of the jejunum; and C: completing a gastrojejunostomy.
Less common causes
A miscellany of less common causes of extrahepatic or intrahepatic obstruction exists, as outlined below.
Extrahepatic obstruction. Metastatic nodes at the porta hepatis or local extension from cancer of the stomach; chronic pancreatitis with or without pseudocyst; acute cholecystitis; carcinoma of the bile duct; benign biliary stricture; stenosis of the ampulla of vater; and oriental cholangiohepatitis.
Acute cholecystitis. Persistent jaundice is uncommon in patients with acute cholecystitis and may indicate an associated bile duct stone or the more severe forms of disease such as acalculus cholecystitis, where there may be gangrene or empyema of the gall bladder. In these patients, jaundice may be contributed to by septicaemia with fever, toxicity and shock. Gangrenous cholecystitis may be insidious in its onset with minimal abdominal signs, especially in the postoperative and posttraumatic patient.
Another mechanism of jaundice in patients with cholecystitis is so called ‘Mirizzi’s syndrome’ where the inflamed gall bladder in combination with an anatomical variant of a short cystic duct causes an inflammatory obstruction of the biliary tree resulting in jaundice.
Carcinoma of the gall bladder. This is seen in older patients and has an extremely poor prognosis. Gallstones are an invariable precursor of gall bladder carcinoma and a very long past history of biliary pain from stones is common.
Malignant tumours of the bile duct (cholangiocarcinoma). These have increased in incidence. They are associated with intense jaundice and pruritus. On investigation, the characteristic findings on ultrasound are a nondilated gall bladder free of stones, with dilated intrahepatic ducts and a normal pancreas. CT scanning confirms that the pancreas is normal, thus suggesting that a bile duct tumour is likely and that ERCP will be necessary to establish the diagnosis. Resection is the best form of treatment, but is rarely possible. Most patients are treated by hepaticojejunostomy, anastomosing a Roux loop of jejunum to the left hepatic duct. ERCP stenting is difficult but is a satisfactory alternative palliation in the more advanced case.
Malignant nodal obstruction at the porta hepatis is also difficult to palliate; stenting is preferably tried first.
Benign biliary stricture. A benign stricture is caused in most instances by surgical trauma. A past history of cholecystectomy suggests stricture as a possible cause of jaundice. The most common basis of this complication of surgery is a short cystic duct, with the bile duct closely applied to the surface of an acutely inflamed gall bladder. The bile duct is mistaken for the cystic duct and a segment is excised. The injury is usually manifest by an early postoperative biliary fistula or biliary peritonitis. With lesser degrees of ischaemic damage to the common bile duct in continuity, a stricture develops within a year or two of operation. A typical patient has episodic pain, fever, chills and mild jaundice within a few weeks or months after cholecystectomy. The stricture is best managed surgically by Roux-en-Y anastomosis between the jejunum and dilated common or left hepatic duct. In some patients ERCP balloon dilatation may be tried, but usually needs to be repeated.
Occasionally jaundice may be due to benign stenosis of the ampulla of Vater and will respond to endoscopic sphincteroplasty.
Chronic pancreatitis. Pancreatitis causes obstruction of the lower bile duct by a combination of inflammatory swelling, fibrosis and compression by a pseudocyst. Occasionally the patient may present with painless jaundice, when it will be difficult to tell whether the obstruction is due to carcinoma or pancreatitis. Differentiation depends on the natural history of each case, CT-guided biopsy, findings at ERCP and, if necessary, at exploration.
Oriental cholangiohepatitis. This is a clinical entity more commonly seen in migrants from Asian to Western countries. The condition is probably a primary disease of the bile ducts; only a small minority have associated gallstones and the gall bladder is usually normal. Chronic recurrent infection often leads to biliary strictures and hepatic abscess formation so, characteristically, the jaundice in these patients is associated with toxicity, fever, chills and biliary pain.
Intrahepatic obstruction
Hydatid disease; secondary carcinoma; chronic active hepatitis; primary biliary cirrhosis; hepatoma; cholangiocarcinoma; and liver abscess.
Hydatid disease. Jaundice is a relatively common complication in patients with hydatid disease. Jaundice follows erosion of bile ductules and entry into the biliary tree of daughter cysts. Visualisation of the biliary tree by CT cholangiography or MRCP should be done if hydatid cyst obstruction is suspected; endoscopic sphincterotomy may temporarily relieve the obstruction. Common bile duct exploration and definitive clearance is usually indicated at subsequent operation, as well as measures to evacuate the cyst, with sterilisation of the primary cavity (Ch 7.9). Bile leaks within the cavity must be found and oversewn before its closure with drainage.
Secondary carcinoma of the liver. Secondaries are much more common than primary tumours. The most common primary sources are from cancers of the stomach, lung, breast and large bowel. The diagnosis of secondary carcinoma of the liver as a cause of jaundice may be difficult, but usually general signs of advanced malignancy coexist with an irregularly enlarged, hard and knobbly liver. In only about 10% of all cases is metastatic disease apparently localised to the liver. When jaundice is present, localised disease is extremely rare and palliative control of pain is usually all that is possible.
Hepatomas and cholangiocarcinomas. These are uncommon outside Asia. Cirrhosis of the liver is often also present. Jaundice is often associated with upper abdominal pain and distension, due to ascites. Hepatoma must be considered when patients with cirrhosis have a sudden deterioration in liver function. Jaundice is almost invariably a sign of inoperability.
7.16 Haematemesis and melaena (upper gastrointestinal haemorrhage)
Acute upper gastrointestinal haemorrhage can present as haematemesis or melaena or both. Haematemesis usually denotes a more rapidly bleeding lesion than when melaena occurs alone. ‘Coffee grounds’ vomitus occurs when blood has been in the stomach for long enough for gastric acid to convert haemoglobin to methaemoglobin; this also indicates a slower rate of bleeding. About a third of patients with upper gastrointestinal haemorrhage present with melaena alone. The black colour of melaena stools is probably due to haematin, the product of the oxidation of haem by intestinal and bacterial enzymes. Melaena can be produced by as little as 50–100 mL of blood loss. With a 1 L loss of blood, melaena usually lasts one to three days and occult bleeding three to eight days. Passage of more than three melaena stools in 24 hours is usually associated with shock and with blood loss approaching 25% of blood volume. Blood pressure may not fall after such a loss, but tachycardia and pallor will be present. Massive bleeding from the stomach or duodenum can lead to red blood being passed from the rectum. These patients are invariably shocked, with severe pallor, hypotension, a thready rapid pulse and loss of 40% or more of the blood volume. Acute rectal haemorrhage of considerable amounts of bright blood, without overt shock, suggests on the other hand that the source of bleeding is the colon (Ch 7.17).
In patients presenting with haematemesis and melaena, immediate assessment and management of shock has priority. Diagnosis must follow resuscitation. In the shocked patient, examination not only reveals the typical peripheral signs, but also central signs of apprehension, anxiety and — in those with massive bleeding — depressed consciousness. Patients in this state who continue to vomit blood are in danger of pulmonary aspiration; endotracheal intubation to protect the airway may be an urgent necessity. The combination of hypoxia from aspiration of gastric content and from hypovolaemia and anaemia in a patient with a diminished cardiac output can be a lethal one and is a common cause of death from upper gastrointestinal haemorrhage. The early detection of concurrent medical illness is also of particular importance; these conditions have a very significant bearing on treatment and on the patient’s likelihood of survival. Equally common is death from further bleeding or from the complications of multiple organ failure thereby induced.
The severity of the initial haemorrhage is the best predictor of the likelihood of further bleeding — especially in older patients with concurrent medical illnesses. The best measure of severity is shock, which depends on the rate and the amount of bleeding and thus determines prognosis. The response to resuscitation of shocked patients gives essential haemodynamic information that assists materially in the timing and safety of surgery. To avoid delay in surgery, a plan of management that recommends early diagnosis (particularly of oesophageal varices) and early operation for bleeding ulcer with shock, after adequate resuscitation, will reduce the mortality of haematemesis and melaena (Box 7.18).
Box 7.18
Principles of management of bleeding ulcer
Accurate resuscitation
Early endoscopic diagnosis
Endoscopic haemostatic techniques if possible
Early surgery in selected high-risk cases
Causes
1. Duodenal ulcer and gastric ulcer
2. Oesophageal varices
3. Haemorrhagic antral gastritis
4. Mallory-Weiss syndrome
5. Less common causes
History
1. Duodenal and gastric ulcer
A past history of periodic epigastric pain, characteristic of peptic ulcer, is present in about 70% of patients. A definite diagnosis of peptic ulcer has been made in at least 50% of patients before presentation with haemorrhage; most have been treated or are being treated with acid-reducing medication. In about 25% of patients there is a previous history of ulcer complications — in most cases, a former episode of bleeding. The bleeding episode is often precipitated by nonsteroidal anti-inflammatory drugs (NSAIDs), especially in gastric ulcer. Most patients with gastric ulcer presenting with bleeding have such a history. The complication of bleeding from a peptic ulcer is at least five times as common in patients over 50 years of age, as it is in those below this age.
2. Oesophageal varices
In approximately three-quarters of patients with varices, portal hypertension is secondary to alcoholic liver disease — with evidence of hepatic failure (Box 7.19). In the remaining quarter of patients no evidence of liver failure is present. Clinical evidence of liver failure does not prove that varices are the cause of bleeding, as quite frequently patients with liver disease bleed from a peptic ulcer. Small varices are an incidental finding in about 10% of patients actively bleeding from another lesion.
Box 7.19
Causes of portal hypertension
Intrahepatic
Cirrhosis, veno-occlusive disease and hepatic vein occlusion
Presinusoidal
Extrahepatic
![]() Splenic or portal vein thrombosis and increased splenic blood flow
Splenic or portal vein thrombosis and increased splenic blood flow
Intrahepatic
![]() Schistosomiasis, congenital fibrosis and portal zone infiltration
Schistosomiasis, congenital fibrosis and portal zone infiltration
3. Haemorrhagic antral gastritis
Two main groups of patients present with bleeding from haemorrhagic gastritis. One has erosive gastritis, the other stress ulceration. Over the past 20 years the incidence of both conditions has fallen significantly.
Erosive gastritis is usually confined to the antrum and the prognosis is much better than stress ulceration. The condition may be triggered by bacterial infection, bile reflux, analgesic ingestion or alcoholism. These patients are referred to hospital with haematemesis; the condition is usually not a complication of serious illness in hospital patients.
Patients with stress ulceration form a heterogeneous collection: multiple acute peptic ulceration develops in association with serious illnesses such as shock, septicaemia, burns and central nervous system tumours or trauma. In many of these patients, superficial ulceration involves the whole stomach. Haemorrhage is the major clinical manifestation of the condition; perforation occurs in a small minority. The duodenum is solely affected in about a third of patients. Most of these patients are already in the hospital when bleeding occurs.
4. Mallory-Weiss syndrome
In this condition, the bleeding is from a 1–4 cm, longitudinal, partial thickness tear, usually in the gastric wall near the oesophagogastric junction. In about half the patients the tear follows a bout of forceful vomiting. Many patients are alcoholics. Typically, the patient first vomits food and gastric contents; this is followed by forceful retching and then bloody vomitus. Oesophageal hiatus hernia with a past history of heartburn is an associated finding in many patients. Occasionally a gastro-oesophageal tear is secondary to other acute intra-abdominal conditions associated with vomiting, such as intestinal obstruction.
Examination
Patients admitted with haematemesis and melaena must be immediately examined for evidence of shock. The possibility of pulmonary aspiration must also be considered. As well as helping to make a diagnosis, the history and physical examination should aim to detect other medical disorders, particularly liver and heart failure and obstructive airways disease. In younger patients with a 10–20% fall in blood volume, the blood pressure is maintained and the patient’s circulatory state is compensated. Decompensation may be induced by sitting up, thus confirming the presence of postural hypotension. As a general rule, when more than 25% of the blood volume is lost, the blood pressure falls to below 100 mmHg and the patient is in frank shock. With more than 40% loss of blood, volume reduction in coronary and cerebral blood flow occur, the patient becomes restless and agitated, with deep sighing respirations and waxy pallor. If bleeding continues, depression of consciousness occurs and the danger of aspiration increases. Approximately one in four patients with bleeding ulcers or varices exhibit frank shock on admission. Shock is much less common (about 5% of patients) in erosive gastritis and the Mallory-Weiss syndrome.
In the majority of patients with upper gastrointestinal haemorrhage, physical signs do not help to make a diagnosis. Perforation with haemorrhage is uncommon, being found in only 2% of cases. Patients with bleeding from oesophageal varices comprise a special high-risk group. Liver function is the best measure of prognosis in these patients and is classified according to a modified Child’s criteria (Table 7.12). Compromised liver function is found in approximately 75% of patients with bleeding varices; these patients will exhibit one or more of the major signs of liver failure. Splenomegaly is found in the same proportion but these comprise a different group, with only partial overlap. Malnutrition, poor hygiene, pulmonary aspiration, pulmonary infection with hypoxia, delirium tremens, purpura secondary to coagulation disorders and coronary artery disease are common factors that contribute to the poor prognosis of these patients.
Table 7.12 Grading of severity of liver disease (modified Child’s criteria — according to Pugh)

In patients with haemorrhagic gastritis there may also be evidence of liver failure from associated alcoholic liver disease or evidence of uncontrolled sepsis. Sepsis is the common initiating factor for gastrointestinal haemorrhage in critically ill patients.
Diagnostic plan
Haematological assessment
Upon presentation, an urgent of set of specimens is collected for measuring haemoglobin, FBE, clotting and cross-matching for transfusion of blood products. At the same time, blood specimens are sent for serum electrolytes. All of these results are crucial to the patient’s assessment and management but do not necessarily delay the organising and performing of endoscopy in an actively bleeding patient.
Endoscopy
Unless there is a past history of proven ulcer, the history and examination cannot be relied upon to make a diagnosis. Diagnosis is best achieved by urgent fibre-optic endoscopy.
The first aim is to exclude varices as a cause of bleeding. Urgent endoscopy must be performed under carefully controlled circumstances, especially in the shocked patient. Resuscitation must be in progress; facilities for endotracheal intubation must be available to protect the airway of shocked patients who are in danger of aspiration.
Varices are seen on endoscopy as three or four large, tortuous, submucosal, bluish vessels running longitudinally in the distal oesophagus. The precise variceal bleeding point may not always be identified and associated lesions of the stomach and duodenum should be excluded. Bleeding from a duodenal or gastric ulcer is the most common cause and most ulcers can be identified on endoscopy. Endoscopy soon after admission can also give, from the appearance of the ulcer, important prognostic information. Most bleeding duodenal ulcers are located on the posterior wall of the duodenal bulb, where penetration of the gastroduodenal artery can produce massive haemorrhage. A high, lesser curve, gastric ulcer may erode the descending branch of the left gastric artery or when lower down may erode the splenic artery. Stigmata indicating that continued bleeding is likely are active arterial bleeding and a nonbleeding ‘visible vessel’ — which is a small pseudo-aneurysm seen within the ulcer crater. The ulcers of erosive gastritis lie on mucosal folds, are shallow, discrete or confluent lesions that contain a smear of clot and show congestion and oedema but little inflammatory reaction at the margin of the ulcer. In these patients an additional chronic ulcer must be excluded as the main source of bleeding. The short longitudinal tear of the Mallory-Weiss syndrome is seen near the oesophagogastric junction. In about 75% of cases the lesion is confined to the stomach, in 20% it crosses the oesophagogastric junction and in 5% the tear lies entirely within the distal oesophagus.
Failure to make a positive endoscopic diagnosis is uncommon and should point to the need for further endoscopy within a short period. Occasionally a trial of tamponade or urgent surgery may be indicated because of massive haemorrhage in the undiagnosed patient. In these patients it is generally possible for a provisional diagnosis of the source of major bleeding to be made.
Treatment plan
Treatment plan depends upon the prognosis on admission and the cause of the bleeding. Prognosis depends upon the severity of the initial haemorrhage, the presence of active bleeding or a visible vessel on endoscopy and is worse with oesophageal varices and with gastric ulcers.
General measures
• Immediate assessment of the circulatory state and restoration of the blood volume by transfusion. The response is monitored by serial measurement of the pulse rate, blood pressure, haemoglobin and urine output. In the stable patient, transfusion with packed cells is indicated when the haemoglobin falls below 100 g/L.
• The amount and rate of bleeding is assessed by the initial clinical state and the clinical and laboratory response to transfusion; this information assists in the timing of endoscopy and surgery.
• The stable patient is observed for at least three days for evidence of further bleeding. Melaena, tachycardia and sweating are the most reliable signs of recurrent bleeding.
High-risk patients
More intense and more precise resuscitation is indicated in the high-risk, elderly or shocked patient — especially the patient with alcoholic liver disease and varices or with chronic obstructive airways disease. More invasive methods of monitoring are indicated, including the precise correction of hypovolaemia to better preserve vital organ function, with monitoring of central venous pressure to control the rate of fluid replacement. Cautious transfusion is important in the older patient with chronic airways disease to avoid deterioration in pulmonary gas transfer because of pulmonary oedema. In such instances it is often necessary to measure pulmonary artery wedge pressure through a Swan-Ganz catheter, to serially measure arterial blood gases and to perform serial chest X-rays.
1. Duodenal and gastric ulcer
With bleeding from chronic peptic ulcer, early (urgent) endoscopic intervention using local infiltration with an adrenaline-containing solution or sclerosant, or probe therapy is required and if not successful in stopping bleeding or not available, then surgery is indicated for selected high-risk patients: those who either are likely to continue bleeding or have re-bled in hospital. In some instances, an initial trial of medical therapy i.e. intravenous proton pump inhibitor and subsequent H. pylori eradication should be considered.
In summary, surgical treatment for peptic ulcer bleeding is indicated for exsanguinating haemorrhage; patients over 50 years who are shocked on admission; endoscopic findings of visible vessel or active bleeding that cannot be treated endoscopically; and when further bleeding occurs in hospital.
Patients with bleeding duodenal ulcer are best treated by oversewing (Fig 7.53) (as most ulcers are located posteriorly over the gastroduodenal artery), followed by cessation of NSAIDs (where implicated) and empirical H. pylori eradication. Bleeding gastric ulcers may require partial gastrectomy when not settling with medical therapy.

Figure 7.53 Surgery for a bleeding duodenal ulcer
A: the duodenum is opened and the bleeding point is overswen in the base of the ulcer; B: pyloroplasty is completed by closing the longitudinal incision transversely. The operation is completed by performing a total truncal vagotomy.
2. Oesophageal varices
Immediate control of bleeding from oesophageal varices is almost always best achieved by early balloon tamponade (Fig 7.54) provided endoscopic diagnosis is made promptly and before there has been significant blood loss and transfusion demand. Tamponade is safe and effective, provided strict attention is paid to nursing detail and care is taken to avoid pulmonary aspiration. The combination of massive bleeding and disturbed consciousness is an indication for endotracheal intubation and adequate sedation together with tamponade.

Figure 7.54 Ballon tamponade using the quadruple lumen Minnesota tube
The four lumina are A: nasopharyngeal aspiration tube; B: port for the gastric balloon; C: gastric aspiration tube; D: port and pressure gauge for the oesophageal balloon; and E: traction (0.5 kg).
Tamponade is continued for 48 hours. During the period of tamponade, patients are stratified according to the severity of their liver disease into Child’s criteria (A, B or C categories) and general measures to improve vital organ function are instituted (Box 7.20). All patients are treated after balloon tamponade with a course of injection sclerotherapy. Child’s A and B patients may be treated later by semi-elective distal lienorenal (Warren) shunt or portacaval shunt. Poor initial control of bleeding is an indication for urgent portacaval shunt or consideration for transjugular intrahepatic portosystemic shunt (TIPS).
Box 7.20
General management of patients with bleeding varices
![]() Accurate resuscitation; prevention of coagulation disorders with vitamin K and fresh frozen plasma.
Accurate resuscitation; prevention of coagulation disorders with vitamin K and fresh frozen plasma.
![]() Prevention of coma by reduction of nitrogen metabolism in the gut: neomycin and early control of bleeding.
Prevention of coma by reduction of nitrogen metabolism in the gut: neomycin and early control of bleeding.
![]() Liver support by infusion of concentrated glucose and thiamine.
Liver support by infusion of concentrated glucose and thiamine.
![]() Prevention of pulmonary infection, anticipation of gas transfer defects and developing hypoxaemia, monitoring left atrial pressure.
Prevention of pulmonary infection, anticipation of gas transfer defects and developing hypoxaemia, monitoring left atrial pressure.
3. Haemorrhagic antral gastritis
This usually runs a benign course. Eradication of H. pylori is an essential early step in the management but occasionally continued bleeding may require surgery. In patients with more severe bleeding endoscopy should be repeated, as a chronic lesion may have been missed. Vagotomy and antrectomy is the surgical procedure of choice for antral erosions with continuing bleeding but is rarely required. In some patients with portal hypertension, bleeding may occur from lesions with the appearance of erosive gastritis, a condition called hypertensive gastropathy. Surgery in such patients must be directed at the control of portal hypertension if recurrent bleeding is to be stopped. In the critically ill patient with haemorrhagic gastritis (stress ulcer syndrome), the main priority is to control causal factors, particularly sepsis. In those who continue bleeding, total gastrectomy may be necessary if the patient is to have a chance of survival.
4. Mallory-Weiss syndrome
The majority of patients will spontaneously stop bleeding. In about 5% of patients operation is necessary, especially in those with associated alcoholic liver disease. Simple oversewing of the tear is usually sufficient to control bleeding. In high-risk patients angiographic embolisation can be an effective alternative to surgery.
5. Less common causes
A miscellany of less common causes exists: gastric carcinoma; oesophagitis and oesophageal ulceration; recurrent peptic ulceration; Dieulafoy’s syndrome; pancreatitis; haemobilia; gastric leiomyoma; duodenal diverticulum; unusual causes of variceal bleeding — extrahepatic portal venous occlusion, splenic vein thrombosis and the Budd-Chiari syndrome.
Carcinoma of the stomach is a common cause of anaemia, but overt bleeding is unusual. Bleeding may be from an exophytic tumour or from a carcinomatous ulcer. Bleeding ulcer cancer may appear to be benign on urgent endoscopy and even at operation may be difficult to diagnose. When there is any doubt, frozen section should be performed.
The bleeding from oesophagitis is usually minor and self-limiting. Occasionally bleeding occurs from an oesophageal ulcer and requires operation to correct haemorrhage. Some of these ulcers are true gastric ulcers at the neck of a hiatus hernial sac as it passes through the diaphragm or from ulceration in the ectopic columnar epithelium of Barrett’s oesophagus. Bleeding from these lesions may be heavy.
Haemorrhage from a large chronic stomal or recurrent duodenal ulcer is uncommon. Haemorrhage can sometimes result from acute superficial erosions located in the small bowel near the stoma. Histologically, these resemble the Dieulafoy lesion. Vagotomy and oversewing of the bleeding point is usually necessary to control bleeding. Partial gastrectomy is necessary for recurrent duodenal ulcer with further bleeding.
The Dieulafoy lesion may be mistaken for a Mallory-Weiss tear. Pathologically, bleeding develops after erosion of an angiodysplastic arteriole, usually situated high on the lesser curve or posterior fundic area of the stomach. Bleeding is profuse and surgery usually necessary, in contrast to a Mallory-Weiss lesion. This lesion may remain undiagnosed until surgery, when it may be felt rather than seen, as a ‘bristle’-like nodule high on the lesser curve of the stomach.
Haemorrhage in patients with pancreatitis usually arises from chronic peptic ulcer, but direct erosion by pancreatic enzymes of the upper gastrointestinal tract may lead to haemorrhage either from the gastric wall or from secondary haemorrhage within the pancreas. It may be very difficult to find the bleeding point on endoscopy in these patients. Coeliac axis angiography may be necessary in order to both locate the bleeding site and then to attempt embolisation.
Haemobilia usually presents with Quenk’s triad: biliary colic, jaundice and upper gastrointestinal haemorrhage. Most instances occur several weeks after liver trauma. Other causes include primary liver tumour, liver abscess and hepatic artery aneurysm. Diagnosis may be made with CT angiography and treatment is by selective hepatic artery angiography and embolisation.
Gastrointestinal stromal tumours (GISTs, previously called leiomyomas) frequently bleed when they reach a diameter greater than 3 cm. Most are found in the proximal stomach. Diagnosis is by endoscopy and emergency excisional surgery by gastrotomy may be necessary. Larger tumours require wedge gastric excision. Very large tumours may behave in a malignant fashion and more radical gastric resection is necessary.
Portal vein thrombosis commonly causes variceal bleeding in children; recurrent bleeding into adult life can occur. The liver is normal in such patients. Splenomegaly and pancytopenia is common in childhood. Splenectomy is contraindicated in these patients because it sacrifices the splenic vein that may later be required for shunting. Most bleeds in childhood can be controlled by injection sclerotherapy until about the age of 10 years, when the size of the splenic vein allows a successful lienorenal shunt to be performed. Splenic vein thrombosis can result from pancreatitis or trauma. Characteristically the varices are gastric; oesophageal varices are uncommon. Splenectomy cures the condition. The Budd-Chiari syndrome is caused by thrombotic obstruction of the hepatic venous outflow. Presentation is with gross ascites, hepatomegaly, deterioration of liver function and variceal haemorrhage. Diagnosis is by liver scan and liver biopsy. Early side-to-side portacaval shunt, before inferior vena caval obstruction occurs, will relieve the condition. About half the patients have a hypercoagulable state from polycythaemia vera.
7.17 Acute lower gastrointestinal (colonic) haemorrhage
Blood loss occurs from or through the lower gastrointestinal tract in four ways. Bleeding may be occult, presenting as an iron deficiency anaemia (Ch 7.18). Visible bleeding presents as either melaena (invariably from the upper gastrointestinal tract — Ch 7.16); as anorectal bleeding with defaecation (the most common form — Ch 7.19); or as acute lower gastrointestinal (colonic) bleeding. Acute colonic haemorrhage is much less common than the other forms. The distinction between brisk upper gastrointestinal bleeding and acute colonic haemorrhage can be difficult, especially when the patient has not vomited blood. Colonic haemorrhage does not cause melaena, but bright blood (or dark red clots) can be passed per rectum that resembles the bright haemorrhage of severe upper gastrointestinal bleeding. However, if a peptic ulcer is causing loss of bright blood from the rectum, the rate and volume will be such that the patient is virtually always shocked. In contrast, a stable circulation is found in most patients passing bright rectal blood from acute colonic (gastroscopy-negative) haemorrhage. Although often presenting as a clinical emergency, colonic haemorrhage is thus a more benign form of bleeding, which in the majority of patients can be managed conservatively and settles spontaneously. In almost a quarter of the cases with acute colonic haemorrhage a definite cause cannot be established. Bleeding from haemorrhoids (anorectal bleeding — Ch 7.19) can seem profuse and may greatly alarm the patient, but in nearly all cases bleeding from haemorrhoids is limited to defaecation and to the surface of the motion, rarely causes anaemia and should thus not be confused with colonic haemorrhage.
Causes
1. Diverticular disease
2. Colonic angiodysplasia
3. Ischaemic colitis
4. Colonic polyps
5. Carcinoma of the colon
6. Less common causes
History and examination
1. Diverticular disease
The most common reason for acute haemorrhage from colonic diverticula is faecolith erosion of a penetrating arteriole in the neck of an uninflamed diverticulum (Fig 7.55). Haemorrhage is usually unheralded and unexpected, often with no history of preceding bowel symptoms. Colonic diverticula are so common and acute colonic haemorrhage from diverticula so uncommon, that the finding of diverticula is not proof that they constitute the source of bleeding. The patient must be investigated as fully as possible for another source of bleeding. Diverticular disease must similarly not be assumed to be the cause of anorectal defaecatory bleeding (Ch 7.19). Apart from blood seen in the upper rectum at the limits of sigmoidoscopy, physical signs are not helpful in identifying colonic haemorrhage. Some patients will present with associated shock and anaemia.
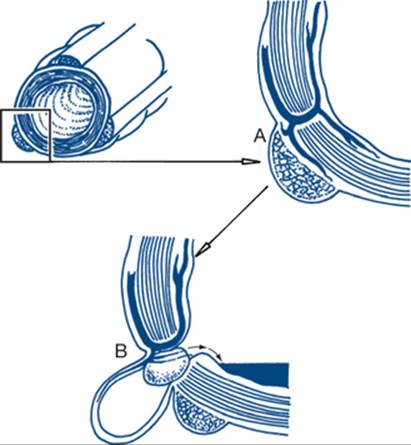
Figure 7.55 Diverticular haemorrhage as a cause of colonic haemorrhage
A: the diverticulum appears through the defect in the inner circular muscle through which passes the vasa recta; the artery is often stretched over the fundus of the diverticulum; B: erosion of the vessel can occur beneath stercoral ulceration produced by a faecolith
2. Colonic angiodysplasia
Colonoscopic detection of angiodysplastic lesions has reduced the number of undiagnosed patients with acute colonic haemorrhage to less than one case in four. Bleeding from angiodysplasia is rare under the age of 50 years. The lesion on histology is a submucosal degenerative vascular ectasia, mainly found in the right colon. Telangiectatic lesions of colonic mucosa seen in younger patients are more commonly a cause of chronic blood loss and iron deficiency anaemia (Ch 7.18). The pathogenesis of the lesion is unknown. Rectal haemorrhage stops spontaneously in most cases, thus allowing time for elective colonoscopy.
3. Ischaemic colitis
Ischaemic colitis usually occurs in patients aged over 50 years and may present as an acute abdomen with gangrenous colitis. The less extensive forms can bleed significantly. Usually there is no significant concurrent illness, although the condition is sometimes associated with malignant disease elsewhere, diabetes mellitus, rheumatoid arthritis or abdominal aortic aneurysm. The common site of involvement is the splenic flexure. In most cases the onset is sudden and without warning. Bleeding is only occasionally severe; abdominal pain is usually present.
4. Colonic polyps
Adenomatous polyps usually present with anorectal bleeding on defaecation. They bleed less predictably and consistently than carcinoma and severe colonic bleeding is unusual. Only about 10% of polyps that bleed present with haemorrhage apart from defaecation. Significant bleeding may occur, however, after colonic polypectomy, especially in patients on anticoagulant treatment.
5. Carcinoma of the colon
Massive haemorrhage from carcinoma of the colon is a quite unusual form of presentation. Anorectal bleeding, altered bowel habit and anaemia are far more common. Sometimes patients with advanced carcinoma or with an anastomotic recurrence of colonic carcinoma can present with severe haemorrhage.
Diagnostic and treatment plans
Resuscitation may be necessary and should precede diagnostic investigations. As is often the case in clinical practice, both diagnosis and treatment proceed simultaneously and in this situation, assessment of the cardiovascular and haematological (FBE, clotting and cross-matching) are essential initial steps.
Barium enema is no longer the preferred first investigation. Barium in the colon interferes with the interpretation of angiographic and radionuclide studies. When a possibility exists that the upper gastrointestinal tract may be the source of the bleeding (in patients without haematemesis) gastroscopy is necessary to be sure that the stomach and duodenum are normal.
Investigation and treatment depends upon which of the two main patterns of bleeding are found.
When the bleeding stops promptly
The first investigation should in this instance be colonoscopy. Sometimes it is possible to identify a diverticulum that is the site of bleeding. Angiodysplasia may be seen as a macular or plaque-like blush of superficial capillary vessels that blanch on pressure. The lesions may be single or sometimes multiple and extensive. If endoscopy fails to identify a lesion a small bowel contrast enema should be the next investigation, in order to exclude a small bowel lesion such as a GIST. Angiography may also demonstrate vascular small bowel tumours. Capsule endoscopy may be used to examine the small bowel mucosa. If no diagnosis is made after full investigation, the usual practice is to observe the patient for evidence of recurrent bleeding and reinvestigate, if possible, during the acute phase of bleeding. Endoscopic control of haemorrhage may be possible, for example, by polypectomy or coagulation of vascular lesions such as angiodysplasia (although surgery is probably the safest method of treatment for angiodysplasia).
With continued bleeding
Ideally, colonoscopy should still be the first investigation. Whether this is possible depends on the severity and pattern of blood loss. Interpretation of colonoscopic findings is often difficult or impossible because of the continued severe bleeding. When colonoscopy fails to identify the lesion or is not performed because of progressive bleeding, emergency angiography or radionuclide scanning is the preferred investigation. Angiography will identify an actively bleeding site in most instances. An alternative noninvasive investigation is radionuclide scanning using labelled red cells. Because this test poorly localises the source of bleeding, it is best used as a screening investigation to establish that active bleeding is present and thus increase the chance of successful angiography.
Angiography can also be used as treatment — by embolisation or infusion of a vasoconstrictor such as vasopressin. The vascular catheter can be left in situ, or removed after injection of methylene blue, in order to define the area in which the bleeding lesion is to be sought when laparotomy is necessary for definitive treatment.
In most patients with acute colonic haemorrhage the bleeding stops spontaneously. As a general rule, laparotomy is only indicated for persistent bleeding, that is, when the rate requires more than four units of transfusion within 24 hours, with shock or after a total of 10 units of blood has been transfused. In the absence of angiographic localisation it can be difficult, if not impossible, to localise the site of colonic bleeding at the time of laparotomy. In this situation, on-table endoscopy, colonoscopy or enteroscopy may be performed to identify the site of bleeding prior to any resection occurring.
There may be a strong suspicion that sigmoid diverticular disease is the source of bleeding. However, because the right colon is as frequent a source of bleeding as the left colon, subtotal colectomy and ileorectal anastomosis is the surgical procedure of choice for nonlocalised bleeding. The emergency operative rate required for acute colonic haemorrhage is, fortunately, low — about 10%. Localised lesions such as ischaemic colitis will also require excision.
6. Less common causes
A miscellany of less common causes exists: spontaneous lower bowel haemorrhage can complicate oral anticoagulant treatment. The complication is often triggered by synergistic medication or colonoscopic polypectomy done without reversal of anticoagulants. Both ulcerative colitis and Crohn’s disease are occasionally complicated by severe rectal haemorrhage. Rectal haemorrhage is rarely the first form of presentation of inflammatory bowel disease. Sometimes the fistulous tracts of Crohn’s disease between the small bowel or a fistula from the small bowel to the colon can bleed massively. Solitary ulcers in the rectum, or sometimes the colon, occasionally cause severe rectal haemorrhage. The rectal ulcer is usually situated on the anterior rectal wall, 4–10 cm from the anal verge. The condition probably shares the aetiological factors associated with rectal prolapse and puborectalis spasm. Massive haemorrhage may occur from radiation enteritis of the lower gastrointestinal tract, which may develop up to 20 years after radiotherapy, usually for gynaecological or prostate cancer. Right colonic superficial ulceration is sometimes seen with viral infections in immunosuppressed patients and after renal transplantation. These patients can present with acute or subacute rectal haemorrhage. On occasions, anastomotic recurrence after resection of cancer of the colon, or fistulas secondary to intraperitoneal recurrence of tumour, present with rectal haemorrhage. Acute pancreatitis, pancreatic pseudocyst and arterial aneurysm are rare causes of colonic bleeding following erosion into the gastrointestinal tract. Small bowel tumours, particularly GISTs, may present with massive rectal haemorrhage but these lesions present more commonly with iron deficiency anaemia. Another small bowel source of apparent colonic bleeding, especially in younger patients (under 30 years), is Meckel’s diverticulum, which may be identified with a radionuclide labelled red cell scan.
7.18 Iron deficiency anaemia
Patients with chronic intestinal blood loss commonly present because they are discovered to have a hypochromic, microcytic, iron deficiency anaemia. In this distinct group of patients there is neither macroscopic gastrointestinal blood loss nor any clinical evidence that might point to a source of the bleeding. The classic cause is carcinoma of the caecum that is notoriously silent in its presentation. If a caecal carcinoma is not found, a gastric cancer should be suspected next. Other causes represent unusual forms of presentation of common conditions, for example, peptic ulcer or oesophagitis. Patients present because they feel vaguely unwell or because of symptoms of anaemia. Symptoms suggestive of mild heart failure or angina may be secondary to the anaemia. Sometimes occult intestinal blood loss is diagnosed because of a positive screening test for faecal occult blood. This test is of low specificity and many healthy patients have been subjected to unnecessary investigations for a positive faecal occult blood finding. In the elderly patient with iron deficiency anaemia a number of possible causes of occult blood loss often exist, without a single defined diagnosis. This group of patients without specific diagnosis makes up about 25% of the cases presenting with iron deficiency anaemia.
Distinction of iron deficiency anaemia from other types of anaemia is rarely difficult. Haemoglobinopathies, the anaemia of chronic disease and sideroblastic anaemias are rarer causes of a hypochromic microcytic anaemia. The anaemia of chronic disease can be the most difficult condition to distinguish from chronic blood loss anaemia. The serum iron is decreased in both conditions. Serum ferritin levels are usually decreased in iron deficiency and raised in chronic disease. Where doubt exists, total iron body stores must be assessed by marrow examination.
Low intake and absorption of iron is a rare cause of iron deficiency anaemia. Deficient iron intake should not be assumed to be the cause of anaemia without excluding blood loss. Malnutrition may, however, be a contributing factor in elderly patients — in varying combination with such conditions as reflux oesophagitis, antral erosions and haemorrhoids. Diverticular disease or reflux oesophagitis must not be assumed to be a cause of occult blood loss and anaemia. Intestinal causes of malabsorption, such as coeliac disease, are nearly always associated with symptoms such as diarrhoea and thus present with that problem (Ch 7.21). Women with menorrhagia frequently become anaemic but present with menorrhagia — the anaemia is secondary. When doubt still exists whether iron deficiency anaemia is secondary to intestinal blood loss, it is best to quantify gastrointestinal loss by chromium labelled red cell studies.
Common causes
1. Carcinoma of the caecum
2. Carcinoma of the stomach
3. Idiopathic group
4. Reflux oesophagitis with oesophageal ulceration
5. Silent duodenal ulcer
History and examination
A full systems review should be performed although significant symptoms and signs are unusual. Some features may appear to be insignificant to the patient but can suggest a possible cause, such as regurgitation of gastric contents. A history of NSAID ingestion is important, as may be a history of poor nutrition.
In many patients with carcinoma of the caecum and some with carcinoma of the stomach, mild anorexia and vague dyspepsia may be associated with the onset of the illness. Anorexia and weight loss and a bitter metallic taste are prominent features with silent carcinoma of the stomach presenting with anaemia. Reflux oesophagitis rarely causes anaemia, unless ulceration is deep and chronic. Silent duodenal ulcer with chronic blood loss can occasionally present with anaemia.
Features of arthritis or other painful chronic diseases may be present on examination when NSAID ingestion is a factor in the production of anaemia. In the idiopathic group other chronic gastrointestinal disorders such as mild oesophagitis, antral erosions, haemorrhoids, colonic polyps and angiodysplasia of the colon may contribute to the anaemia but no definitive cause is found despite full investigation.
Diagnostic plan
The diagnostic plan is based upon exclusion of caecal and gastric carcinoma.
Gastroduodenoscopy
Upper gastrointestinal endoscopy should include examination of the second part of the duodenum. Lesions in the upper gastrointestinal tract that may present with anaemia are carcinoma of the stomach, duodenal ulcer and occasionally GISTs. When no obvious cause of bleeding is found in the oesophagus, stomach or duodenum, a small bowel biopsy from the beginning of the third part of the duodenum should be taken. A normal biopsy from this site effectively rules out those mucosal causes of malabsorption that can contribute to a hypochromic anaemia. Acute gastric and duodenal erosions or mild oesophagitis without ulceration may be seen, but alone, these lesions rarely cause chronic anaemia. Their presence should not delay or prevent further essential investigation, especially to exclude carcinoma of the right colon.
Colonoscopy
While barium enema will diagnose most obstructive lesions causing altered bowel habit and abdominal pain, colonoscopy is the optimal investigation for the patient with chronic intestinal blood loss — particularly for detection of polyps and other mucosal lesions. Angiodysplasia is also a relatively common cause of chronic lower intestinal blood loss that cannot be identified by contrast studies. These blotchy red submucosal areas of vascular ectasia are usually confined to the caecum and can be recognised at colonoscopy, provided that the bowel is free from blood and faecal material. For a colonoscopy to be complete the ileocaecal junction must be seen and, if possible, it is also desirable that the colonoscopic examinations have included the last 20 cm of ileum.
If both gastroscopy and colonoscopy are negative, two questions need to be asked: Were the endoscopic examinations adequate; and Is there any doubt that the anaemia is due to intestinal blood loss? In most patients it is worth repeating the endoscopic investigations before proceeding to a study of the small bowel by small bowel enema or a capsule endoscopy (pill cam) examination. The latter investigation furnishes mucosal detail about the small bowel and is useful in investigating those patients with apparent gastrointestinal bleeding but with negative gastroscopy and colonoscopy.
Small bowel enema
Instillation of contrast media from a long tube positioned in the proximal jejunum gives films of much better quality than the pictures obtained from conventional barium meal and follow-through examination. Occasionally lymphoma, previously unsuspected Crohn’s disease, Meckel’s diverticulum, small intestinal polyp(s) or a GIST of the small bowel may be discovered on small bowel enema.
Radionuclide scan and angiography
If the above investigations are negative, radionuclide scan may be of value. Scanning is of particular value in those patients with lesions that have bled significantly, but intermittently, rather than those with chronic blood loss, because scans can be performed over the hours following the test to show a steady accumulation of radio-active material in one site of origin. Angiography requires a minimum rate of blood loss of 0.5 mL per minute to reveal the site of bleeding. Angiography, like scanning, is therefore unlikely to detect the site of chronic intestinal loss. The right colon is a common site of bleeding and so should receive maximum attention during the study, but triple vessel (coeliac, inferior mesenteric and superior mesenteric) angiography is indicated in most cases. Sometimes abnormal vascular patterns of the small bowel may be identified, such as arteriovenous malformation, hereditary haemorrhagic telangiectasia, angiodysplasia, Meckel’s diverticulum, vascular leiomyoma GIST, carcinoid tumour and, rarely, small bowel carcinoma.
Abdominal CT scan with IV contrast
This may pick up some of the less common extramucosal lesions or those of the small bowel, but should only be considered after the preceding luminal investigations have been completed. It should be performed if laparotomy is being considered.
Diagnostic laparotomy
Diagnostic laparotomy must be considered if a lesion is identified on angiography. The surgeon can be directed to the lesion either by leaving the catheter in situ along the feeding vessel or by injecting methylene blue to localise in a vascular malformation. The chance of finding a lesion at laparotomy when the above tests are negative is slight. Before diagnostic laparotomy the whole situation should be carefully reviewed. This means not only confirming that intestinal blood loss is definitely present, but being sure that endoscopy was performed close to the time when bleeding occurred. All major investigations should be performed on at least two separate occasions before laparotomy.
Treatment plan
In most instances the patient is started on ascorbic acid and oral iron as soon as iron deficiency anaemia is detected. This may remain the only form of treatment for the undiagnosed patients and for the elderly patient with several possible causes and will control the anaemia in most of these cases.
The bowel should be adequately prepared before operation and facilities for intra-operative endoscopy should be available. Intra-operatively, the bowel should be examined carefully from oesophagus to pelvis. Areas where visualisation is difficult for the endoscopist should receive special attention, particularly the fundus of the stomach for GISTs, the third and fourth parts of the duodenum, the ileocaecal region and the hepatic and splenic flexures of the colon and for other intra-abdominal lesions that may bleed into the gut — such as pancreatitis, splenic artery aneurysms, invasive tumours of other organs or retroperitoneal recurrence of previously resected carcinomas.
Patients with anaemia due to carcinoma of the stomach are best treated by gastric resection.
In most patients requiring surgery, the cause of anaemia will have been found to be carcinoma of the caecum. Principles of management include:
• The preoperative preparation of the patient, including control of concurrent illness and transfusion of packed red cells should be conducted to correct the anaemia. At least two days should elapse between transfusion and surgery so that oxygen delivery is optimal from transfused red cells.
• The colon is prepared by preoperative fasting and bowel preparation; prophylactic antibiotics are given with the premedication.
• The procedure of choice is right hemicolectomy with primary anastomosis (Fig 7.56).

Figure 7.56 Right hemicolectomy
1: superior mesenteric artery; 2: middle colic artery; 3: right colic artery; 4: ileo-colic artery. Dotted line represents line of resection.
Uncommon causes such as GIST of the stomach and small bowel, extrinsic invasion of the small and large bowel by tumour, anastomotic recurrence, Meckel’s diverticulum, lymphoma, carcinoid tumours and carcinoma of the small bowel are treated surgically in most cases. Vascular lesions such as angiodysplasia, teliangectasia and arteriovenous malformations may be best treated by endoscopic sclerotherapy, diathermy or argon laser photocoagulation, especially if they are multiple, aided by intra-operative endoscopy if necessary.
7.19 Bleeding with defaecation (anorectal bleeding)
Bleeding associated with defaecation is a very common problem. Blood loss from such anorectal bleeding is usually mild or moderate and almost always confined to defaecation (Fig 7.57). Anorectal bleeding from haemorrhoids often appears profuse to the patient, but is hardly ever haemodynamically significant and only occasionally causes anaemia. If hypochromic anaemia is found on the blood film in patients with rectal bleeding, malignant disease of the large bowel must be excluded by investigation of the whole colon with colonoscopy.

Figure 7.57 Anorectal bleeding
A: blood coming from the rectum is persistent, associated with tenesmus, and due to polyps, cancer or proctitis; B: bleeding from haemorrhoids in the anal canal is periodic, painless, profuse and bright red; C: bleeding from the anal verge is small in amount, bright red, seen only on the toilet paper and due to fissure or skin tag. 1: anorectal ring; 2: dentate line; 3: anal verge
The combination of haemorrhoids and an associated carcinoma of the rectum or colon is common and is a notorious trap for the diagnostician. Therefore, a thorough anorectal examination is necessary in all patients presenting with rectal bleeding. This should include an examination of the whole colon in patients over 40 years of age, especially when the rectal bleeding is of recent onset, even though the bleeding may be typical of haemorrhoids. A recent alteration in bowel habit in association with rectal bleeding or the passage of blood mixed throughout the stool is always an indication for colonoscopy. Occasionally, rectal bleeding may continue intermittently after defaecation or cause spotting of underpants between bowel actions. This suggests anal pathology, particularly such conditions as advanced haemorrhoids, chronic anal fissure or perianal fistula. Of clinical importance in the assessment of the problem of anorectal bleeding is the fact that diverticular disease is rarely a cause of this problem and should never be assumed to be the cause until other lesions have been convincingly excluded. While diverticular disease rarely causes anorectal bleeding with defaecation, it is a common cause of acute colonic haemorrhage (Ch 7.17).
Common causes
1. Haemorrhoids
2. Carcinoma of rectum and sigmoid colon
3. Adenomatous polyps
4. Anal fissure
5. Localised proctitis
6. Less common causes
History
1. Haemorrhoids
In most patients with bleeding haemorrhoids, the pattern of the bleeding is typical. Bleeding tends to be at the end of defaecation, bright red and is seen on the toilet paper or may drip or squirt into the toilet bowl. Bleeding is clinically painless although eventually, with persistent prolapse of haemorrhoids, moderate discomfort is common. The history also reveals a pattern of remissions and exacerbations, with gradual change in the symptoms over several years. Initially bleeding is only intermittent but, as prolapse develops, the bleeding tends to diminish and there is an increase in mucous discharge, anal discomfort, pruritus and tenesmus. At first the prolapsing haemorrhoids return spontaneously after defaecation; later they can only be returned manually; eventually they prolapse at times other than on defaecation; later they can be returned only with difficulty. These patients do not complain of constipation in the sense of irregular or infrequent bowel actions, but often there is a long history of a sense of incomplete evacuation and straining. The dietary history often reveals a low fibre intake; breakfast often has not been eaten for many years. In these patients the gastrocolic reflex is thus both weakened and neglected and defaecation habitually occurs in the evening. Excess straining increases the venous pressure and the engorged inferior haemorrhoidal vascular cushions eventually prolapse as haemorrhoids and bleed when the venous return is obstructed by the anal sphincter.
2. Carcinoma of the rectum and sigmoid colon
Anorectal bleeding is the most common form of presentation of distal large bowel cancer. Characteristically, bleeding from carcinoma of the rectum is of recent onset in a patient over 40 years of age. The bleeding may be red or dark, depending upon location of the tumour, is mixed with the motion and persists without remission. A feature of more distal tumours is a recent history of increasing tenesmus (a sense of incomplete evacuation). Tenesmus of severe and recent onset strongly suggests that anorectal bleeding is from a carcinoma, rather than from haemorrhoids. When tenesmus is marked, painful urgency and straining is associated with the frequent passage of blood and slime. This combination of frequency and difficulty of defaecation is not seen with haemorrhoids but may occur with proctitis and the solitary rectal ulcer syndrome, as well as with carcinoma. General deterioration in health and weight loss are commonly associated features in carcinoma of the large bowel.
3. Adenomatous polyps
When polyps bleed they tend to do so intermittently without change in bowel habit, in contrast with the everyday bleeding and tenesmus associated with carcinoma. Rectal bleeding is by far the most frequent presenting complaint of polyps. The blood is bright or dark red, depending upon the location of the polyp. Occasionally, intussuscepting polypoid tumours can produce abdominal discomfort, but irritative symptoms in patients with polyps are usually because of associated diverticular disease. Large benign tumours of the rectum may sometimes bleed and produce tenesmus, constipation or increased frequency of bowel action — especially when the benign tumour is a villous adenoma. In these cases copious amounts of mucus can be evacuated per rectum; presentation is then with ‘diarrhoea’ (Ch 7.21).
4. Anal fissure
Bleeding from anal fissure is usually accompanied by severe pain on defaecation (Ch 7.22) and presentation is anal pain, together with bleeding on defaecation. The loss of a small quantity of bright red blood, usually noted on the toilet paper, alarms the patient enough to see a doctor. Pain is occasionally not a dominant feature of the illness or can be delayed until well after defaecation. Fissure can also occur in association with carcinoma, probably because of tenesmus-induced straining.
5. Localised proctitis
The bleeding of localised distal proctitis tends to be darker and mixed with the motion. This condition is often associated with constipation and significant, sometimes severe, tenesmus. The disease is subject to remissions and exacerbations. Patients with ulcerative colitis sometimes recognise blood in motions from proctitis as their first symptom but this is soon followed in nearly all cases by diarrhoea with mucus and pus as the whole colon become involved, contrasting with the syndrome of localised granular proctitis that does not usually progress to diffuse ulcerative colitis.
Examination
On general examination, in most cases of rectal bleeding there are no clinical signs that contribute to the diagnosis. Abdominal examination is usually negative. Secondaries in the liver from bowel cancer may be palpable — about 15% of patients with carcinoma of the colorectum have clinical evidence of metastatic disease. In all patients with rectal bleeding anorectal examination and sigmoidoscopy are necessary and will establish the diagnosis in many cases. Routine examination is as follows.
• Anal inspection for fissure, external haemorrhoids and internal haemorrhoids revealed by straining and associated perianal conditions. Anal fissures are commonly posterior and only the lower margin of fissure may be visible, because of sphincter spasm and pain.
• Digital rectal examination can examine the mucosa and surrounding tissues up to 10 cm from the anal verge. Painful sphincter spasm is marked in acute anal fissure and rectal examination is often impossible. This may preclude further investigation, but is an indication for examination under anaesthesia. On digital palpation the elevated, indurated and ulcerated lesion of a carcinoma may be felt within the rectum. Blood on the glove after the examination suggests colorectal ulceration.
• Proctoscopy is necessary to detect the presence of first- and second-degree haemorrhoids, which can be seen but not felt. Inspection during straining can demonstrate prolapse of second-degree piles. Haemorrhoids are usually classified as first, second, third or fourth degree (Fig 7.58). Internal and external haemorrhoids represent two parts of the same pathological process — internal haemorrhoids being derived from the inferior haemorrhoidal plexus and external haemorrhoids from the external haemorrhoidal plexus. They are separated by a groove that corresponds to an area of squamous mucosa closely attached to the internal sphincter, just distal to the dentate line about 1.5 cm above the anal verge. Low rectal carcinomas may be seen on proctoscopy, as will fissure, unless painful anal spasm prevents the examination.

Figure 7.58 Classification of haemorrhoids into four degrees based on the severity of the disease
First-degree haemorrhoids presents with bleeding; second-degree haemorrhoids prolapse but return spontaneously; third-degree must be manually returned; and fourth-degree are permanently prolapsed.
Diagnostic plan
Sigmoidoscopy. All patients with rectal bleeding should routinely be examined by rigid or flexible sigmoidoscopy. More of the distal colon can be examined on flexible fibre-optic sigmoidoscopy and therefore more polyps and carcinomas can be discovered. This examination requires bowel preparation and should be performed by an expert.
Colonoscopy is indicated in selected patients with rectal bleeding. Many young patients with a typical history of haemorrhoids and a good response to treatment will require no further investigation than sigmoidoscopy. Colonoscopy is a superior investigation to barium enema in the evaluation of the patient with undiagnosed rectal bleeding, because polyps are better detected and can be removed at colonoscopy. If polyps are detected on flexible sigmoidoscopy, better preparation of the bowel is necessary before later removal at colonoscopy by hot biopsy forceps or diathermy snare. Colonoscopy is also necessary before operation for carcinoma in order to detect synchronous lesions.
Imaging. Patients diagnosed with rectal carcinoma will usually require endorectal ultrasound and CT scanning to assist with staging the disease.
Treatment plan
1. Haemorrhoids
A long history of straining and a diet low in fibre are features. An increase in dietary fibre intake can abolish straining and induce a remission of symptoms in most early cases of haemorrhoids. Thus, first- and second-degree haemorrhoids can usually be managed by simple local measures and dietary advice. Particular attention should be paid to eating unrefined cereal breakfast, together with supplementing the diet with either bran or commercially available hydrophilic bulking agents. Suppositories and rectal ointments have little place in the management of haemorrhoids. Sometimes second-degree haemorrhoids may need additional treatment by injection or rubber band ligation of the internal component (Fig 7.59). Haemorrhoidectomy is the procedure of choice for third- and fourth-degree haemorrhoids although, in poor-risk surgical patients, rubber band ligation may provide good relief of symptoms. The most effective surgical procedure is excision and low ligation of the primary haemorrhoids at 3, 7 and 11 o’clock. Postoperative pain control is important and can usually be achieved by intra-operative caudal or local anaesthesia, anal dilatation and the liberal postoperative use of narcotic analgesics and bland aperients.

Figure 7.59 Barron band ligation of internal hemorrhoids
Working through a proctoscope a rubber band (A) is dislodged from the cylinder to encircle the neck of an internal haemorrhoid (B).
2. Carcinoma of the rectum and sigmoid colon
As with other gastrointestinal tumours, surgical resection is the basis of treatment. Surgery is the only form of treatment that, in a majority of cases, can control local disease for the life of the patient. Preoperative pelvic and abdominal CT scanning can detect the presence of ureteric involvement; many patients are assessed and receive at this stage preoperative chemoradiotherapy. Colonoscopy is done before operation to exclude synchronous lesions. About one-third of cases have associated polyps and 5% have a synchronous carcinoma.
Preoperative preparation aims to control concurrent disease as much as is possible in the time available before surgery, to correct iron deficiency anaemia and to minimise the chance of surgical infection secondary to contamination using mechanical bowel cleansing and prophylactic antibiotics. The patient should be prepared for a stoma, which may be a covering ileostomy, and its site marked preoperatively. The choice of operative procedure depends upon the height of the carcinoma above the anal verge. The rectum is usually divided into three parts: the lower third from 0–7 cm, the middle third from 7–12 cm and upper third from 12–18 cm. As a general rule, abdominoperineal resection of the rectum, with permanent sigmoid colostomy is necessary for lower third lesions, although after CT and endorectal ultrasound assessment, a number of patients will be suitable for an ultra-low anterior resection. For upper- third lesions an anterior resection can nearly always be performed — that is, resection of the tumour through an anterior abdominal laparotomy, with reanastomosis of the bowel (Fig 7.60). Other factors influence the choice of procedure for middle-third lesions — including the extent of the tumour, the differentiation of the tumour and the general condition of the patient. In women, more anterior resections or other sphincter-saving procedures can be performed because of better pelvic exposure. The introduction of end-to-end stapling devices has also increased the number of anterior resections and thus sphincter-saving procedures that can be performed. In most cases, the anastomosis after mid, low or ultra-low rectal resection is ‘protected’ by a temporary loop ileostomy.

Figure 7.60 Operative procedures performed for carcinoma of the distal large bowel
A: anterior resection with primary anastomosis for middle- and upper-third rectal lesions; B: abdominoperineal resection with end sigmoid colostomy for distal-third rectal carcinoma. 1: inferior mesenteric artery; 2: left colic branch; 3: sigmoid branch; 4: superior rectal artery
Prognosis depends upon the site, extent, differentiation and stage (Dukes’ classification) of the tumour. All patients require follow-up colonoscopy, at first after two years from operation, and thereafter every three to four years to remove any new polyps and prevent the development of new tumours. Patients who have had one cancer of the colorectum removed represent the main high-risk group for new tumours. In about 40% of cases recurrent polyps will require removal.
3. Adenomatous polyps
Adenomatous (colonic) polyps are readily removed colonoscopically by electrocautery snaring or hot biopsy forcep removal, if they are small (Fig 7.61). Larger villous tumours have a higher malignant potential and require complete excision, either by transanal local resection or segmental bowel resection. The problem of the pedunculated polyp with a focus of neoplastic change is an indication for formal bowel resection if the tumour has involved the pedicle of the polyp.

Figure 7.61 Diathermy snare colonic polypectomy
A: the snare is tightened on the neck of the polyp; B: current is applied while the polyp is continuousy moved to avoid a local full thickness burn; C: the resected polyp is sucked against the tip of the colonoscope for removal
4. Anal fissure
Treatment of this condition is considered in Chapter 7.22.
5. Localised proctitis
Treatment of this condition is considered in Chapter 7.22.
6. Less common causes
There are few uncommon causes of the problem of anorectal bleeding. Sometimes anorectal bleeding may be the major form of presentation in patients with solitary rectal ulcer, angiodysplasia, radiation damage to the large bowel or ileocaecal Crohn’s disease, but bleeding is usually more obviously mixed with faeces or as larger volume, darker blood clots (Ch 7.17). Sometimes patients with perianal fistula present with a bloodstained perianal discharge (Ch 7.27), which may be mistaken for anorectal bleeding.
7.20 Altered bowel habit (constipation)
The major consideration in patients who present with altered bowel habit is to exclude carcinoma of the colon. Patients with carcinoma of the rectum and rectosigmoid usually present with frank bleeding and those with carcinoma of the right colon, with anaemia from occult loss. These three chronic forms of presentation (altered bowel habit, bleeding and anaemia) occur in 75% of patients with carcinoma of the large bowel. In the remaining 25%, 20% present with subacute or acute large bowel obstruction and 5% with perforation and peritonitis (Fig 7.62).

Figure 7.62 Forms of presentation of large bowel cancer
A: right colon cancer usually presents with anaemia, often associated with mild dyspepsia or a mass; B: left colon cancer presents with altered bowel habit associated with varying degrees of pain from partial obstruction; C: cancer of the sigmoid colon sometimes presents with an acute abdomen from large bowel obstruction or perforation; D: distal large bowel and rectal cancers usually present with rectal bleeding associated with varying degrees of hypogastric pain and tenesmus
Altered bowel habit is a portmanteau term for a group of symptoms affecting bowel habit and function, often associated with lower abdominal discomfort and sometimes with a general deterioration in health. The usual symptom is increasing constipation, sometimes increased frequency of defaecation (Ch 7.21) or sometimes alternating constipation and diarrhoea. Constipation implies irregular, infrequent bowel actions. Tenesmus is an uncomfortable sense of incomplete evacuation; patients may return several times to the toilet. Tenesmus of mild degree is a ubiquitous symptom of rectal dysfunction and functional bowel disease (Fig 7.63). Severe tenesmus (a sense of incomplete evacuation leading to frequent and painful straining) is a highly significant symptom; carcinoma of the rectum is the likely diagnosis. Tenesmus is frequently an associated symptom helping to establish the clinical diagnosis. Occasionally, patients with painful anorectal disease (Ch 7.22) present because of increasing constipation, which dominates the clinical picture. A careful history reveals pain as the major problem. Dieting, particularly the form that excludes high residue foods, can also lead to constipation. Dieting is usually readily detected and easily excluded as a cause. Common factors contributing to constipation include lack of fibre, lack of exercise, debility, depression, dehydration and certain drugs.

Figure 7.63 Common causes of tenesmus
A: haemorrhoids; B: carcinoma of the rectum; C: rectal prolapse; D: faecal impaction; E: inflammatory bowel disease
Causes
1. Carcinoma of the left colon
2. Functional bowel disease and diverticular disease
3. Psychiatric disorders
4. Drug-induced constipation
5. Less common causes
History
1. Carcinoma of the left colon
Because of the smaller lumen and semisolid faecal content, carcinomas of the transverse and left colon usually present as a change in bowel habit because of partial obstruction; this may be associated with deterioration in general health if chronic blood loss has produced anaemia. Change in bowel habit is insidious in onset, usually has been present for several months and is typically noted for the first time in a patient over 40 years of age. Increasing constipation leads to more and more aperients being taken. Occasionally diarrhoea occurs or constipation alternates with diarrhoea every few days. These symptoms can be secondary to faecal impaction above the tumour. Sometimes patients complain of ‘dyspepsia’ with distension and loud borborygmi, especially after meals. The symptoms tend to be relieved by the passage of flatus, favouring a lower rather than upper gastrointestinal cause of the dyspepsia.
2. Functional bowel disease and diverticular disease
Spastic colon is one common form of colonic motility disorder or functional bowel disease characterised by lower abdominal discomfort and constipation. The diarrhoeal form of functional bowel disease is generally called irritable colon (Ch 7.21). Symptoms of spastic colon can closely resemble those of carcinoma. However, the altered bowel habit is usually nonprogressive and of much longer duration than in patients with cancer. Windy distension and discomfort can be severe in spastic colon, a symptom that is partially relieved by passing flatus or by defaecation. The stool is often described as ‘rabbit pellet’ or narrow and ‘ribbon-like’. Anorexia, weight loss and rectal bleeding are uncommon.
Features of diverticular disease resemble those of spastic colon, but patients with diverticular disease are older, most being over 50 years of age. Diverticulitis may produce extrinsic compression and obstructive symptoms as a result of repeated and/or chronic inflammatory episodes resulting in cicatrisation of the colonic wall and resultant narrowing (Fig 7.64).

Figure 7.64 Diverticulitis
Partial obstruction of the sigmoid colon by diverticulitis and a peridiverticular phlegmon. Obstructive symptoms are produced by extrinsic compression.
3. Psychiatric disorders
Constipation is common with psychiatric disorders; especially when the illness is associated with inactivity and is treated by drugs with constipating side effects. Depression is a common disorder and constipation a common complication of depression. When constipation is severe it may be emphasised by the patient, to the virtual exclusion of other symptoms more obviously characteristic of the depression itself. The patient’s general demeanour and affect often suggest the diagnosis and indicate the need for a search for possible causes of depression. Constipation is also common in patients with anorexia nervosa in which case low food intake may be combined with vomiting and these patients frequently also take diuretics and purgatives. While it is unusual for constipation to be the dominant feature of the illness, it may occasionally be the presenting symptom. Often the underlying anorexia is vigorously denied and a careful history and high clinical index of suspicion are essential to make the diagnosis. Addictive purgation can also be difficult to detect. In these patients a history of constipation is often triggered by low fibre intake and typified by delayed colonic transit. Longstanding purgative abuse may eventually lead to presentation with intractable constipation and electrolyte depletion. Sometimes gross colonic dilatation may occur — in the chronic case this is called cathartic colon; in the acute case, idiopathic large bowel obstruction or faecal impaction (Ch 7.8).
4. Drug-induced constipation
The drugs that most commonly cause constipation are the hypnotic sedatives and antidepressants. A drug history must be taken in all patients with constipation, looking especially for diuretics (especially when potassium supplements have not been given), iron compounds, antacids (including aluminium and calcium compounds), anticonvulsants (especially phenytoin), calcium antagonists such as verapamil and ion-binding agents such as resonium. Codeine is a commonly used constipating opiate. Other opiates can lead to presentation with constipation in patients who may deny any history (Box 7.21). Sometimes drug-induced constipation is precipitated by prolonged hospitalisation, presenting later during the recovery phase.
Box 7.21
Drugs that cause constipation
Hypnotic sedatives
Antidepressants
Diuretics (without potassium supplement)
Iron compounds
Opiates (codeine)
Antacids
Anticonvulsants (phenytoin)
Calcium antagonists (verapamil)
Ion-binding agents (resonium)
Examination
A carcinoma of the left colon with partial obstruction can rarely be palpated abdominally, because obstructive symptoms often occur relatively early in the disease. A diverticular phlegmon may be palpable, so a left iliac fossa mass may, paradoxically, be more suggestive of benign disease. A tender palpable left colon is often felt in both functional bowel disease and diverticular disease. Faecal loading of the colon may be present. Rectal examination is often normal, but associated minor anorectal disorders are quite common. These associated disorders do not detract from the need for full investigation to exclude an underlying carcinoma.
Diagnostic plan
Haematology
This will detect iron deficiency anaemia secondary to chronic blood loss from carcinoma of the colon. Leucocytosis indicates the possibility of an inflammatory cause of altered bowel habit such as diverticulitis.
Sigmoidoscopy
The rectum and lower sigmoid colon are routinely inspected, usually with a rigid 25–30 cm sigmoidoscope. Most conditions causing altered bowel habit are beyond the reach of the rigid sigmoidoscope. Melanosis coli is an important sign indicating a chronic intake (perhaps denied) of anthraquinone purgatives.
Colonoscopy
This is the usual investigation of colon symptoms. In patients with functional bowel symptoms, psychiatric disorders and possible drug-induced constipation the main value of colonoscopy is to exclude carcinoma.
Colonoscopy can distinguish carcinoma from diverticular disease but, if the colonoscope cannot be passed through a stricture of the bowel, then contrast radiology may be required. If no mucosal lesion is identified, then colon transit studies and defaecography should be considered for the investigation of slow transit constipation/megacolon and evacuation disorders.
Occasionally, if after all investigations no lesion is found, the patient should then have a detailed review of their diet.
Treatment plan
General measures used in the treatment of constipation are indicated in Box 7.22.
Box 7.22
Common therapeutic agents used in the management of constipation
Hydrophilic bulking agents
Bran
Mucilagenous seed coats
Mucilagenous gums
Irritant purgatives
Senna — a plant glycoside
Suppositories
Bisacodyl — a polyphenolic peristaltic stimulant
Enemas
Saline bowel washout
Olive oil or hypertonic phosphate retention enema
1. Carcinoma of the left colon
After careful preparation of the bowel, the treatment of carcinoma of the colon consists of wide surgical excision of the primary lesion (Fig 7.65) and its regional lymphatic drainage area. The primary tumour should be resected even if metastases are present, since relief of obstruction and bleeding gives the best palliation. If a safe anastomosis cannot be performed to the surgeon’s satisfaction, a protective temporary loop ileostomy may be necessary. Specifically, a covering stoma is indicated when exposure is difficult because of obesity, when there is faecal contamination during surgery, when the bowel has been inadequately prepared preoperatively or when the anastomosis is technically difficult.

Figure 7.65 Segmental colectomy
A: transverse colectomy for carcinoma of the transverse colon with division of the left main branch of the middle colic artery; B: left hemicolectomy for carcinoma of the descending colon, with ligation of the left colic artery
Preoperative preparation includes mechanical bowel cleansing and total colonoscopy to exclude synchronous neoplasms, control of concurrent medical illness, preoperative transfusion of packed cells for significant anaemia, prophylactic antibiotics and preoperative contrast CT scan to exclude ureteric obstruction in sigmoid carcinoma and identify possible metastatic disease.
2. Functional bowel disease and diverticular disease
Surgery is not required for functional bowel disease or for uncomplicated diverticular disease. Many cases respond to a high-fibre diet (see below). Where there is obstruction due to peridiverticulitis, surgical resection may be necessary.
Patients with proven pelvic floor dysfunction may require more detailed neurological assessment, possible training in biofeedback techniques with a specialist physiotherapist or consideration for surgical repair of the pelvic floor, especially if there is associated rectal prolapse.
3. Psychiatric disorders
In patients with psychiatric and drug-induced constipation, treatment for the underlying condition is necessary, together with active rehabilitation and a high-fibre diet. In many cases a good response is obtained with dietary measures. Older patients often prove intractable to dietary treatment and the cocktail of aperients must be continued. In some patients with severe slow transit constipation and cathartic colon, colectomy and ileorectal anastomosis must be considered.
4. Drug-induced constipation
Refer to the discussion of psychiatric disorders above.
5. Less common causes
An exacerbation of distal colitis may be associated with proximal faecal stasis and can present with a short history of constipation. The patient is usually unwell and may be feverish and toxic. Occasionally Hirschsprung’s diseasepresents in early adult life. These patients often have a short aganglionic segment in the rectum, which is best detected manometrically and by biopsy and treated by myotomy of that segment. Chagas’ disease is a cause of megacolon due to infection by Trypanosoma cruzii, which until recently has been confined to South America. Megacolon results from toxic destruction of submucosal and myenteric plexuses and is manifest by severe constipation of gradual onset. Other symptoms include congestive cardiac failure. Mega-oesophagus also occurs and is identical to achalasia. Hypothyroidismmay cause severe constipation of insidious onset and may be associated with megacolon. Constipation with megacolon is a late manifestation in patients with scleroderma.
High-fibre diet
Fibre is found naturally in plant foods and is one name given to the skeletal or supporting structure of plants; others are roughage or residue. Fibre is present in all parts of the plant: leaves, flowers, seeds, fruits, stems, roots, pith, husk, bulbs and tubers. Modern food processing removes much of the fibre; for example, when a cereal such as wheat is milled to make white flour, the outer covering of husk or bran containing most of the fibre is removed. Most of the fibre eaten is not digested or absorbed into the body but remains in the gut, where its main action is to stimulate intestinal propulsion (peristalsis) and finally to form most of the faecal residue excreted in the stool. Fibre is necessary for the normal function and health of the whole intestine, not only for excretion but for normal transit at all levels, beginning with chewing and swallowing. Lack of fibre (particularly at breakfast) is an important risk factor for gastrointestinal disease. The proper intake of fibre can be calibrated by increasing fibre in the diet gradually, until a soft motion is passed at least once a day in the morning. It must be stressed that fibre is not the only important dietary factor. Normal diet should be balanced and retain an adequate intake of animal protein (including fish), unless this clashes with other medical dietary advice, for example, for cholesterol reduction.
Fibre contains few available calories, in contrast to refined food, and is filling, so an adequate intake is also of importance in the maintenance of normal body weight. A high-fibre diet will not lead to an increase in weight, unless sugar is added. In general, fibre should be increased at breakfast and lunch and the evening meal should be of the usual pattern, with added vegetables.
7.21 Diarrhoea
The patient presenting with chronic diarrhoea has increased frequency of stools with increased fluid content. Increased volume can be due to either a secretory or an osmotic disturbance of intestinal function. Acute diarrhoea is much more common than chronic diarrhoea (persisting for more than two weeks). Acute diarrhoea is not considered in this section in detail, but the appearance of blood and pus in the stool in patients with acute diarrhoea makes it essential to examine the stool, the anorectum and the large bowel. Patients with severe tenesmus have pseudodiarrhoea, with a frequent call to stool manifest by painful straining, but the volume lost is small. These patients usually present with associated symptoms of rectal bleeding or alternating constipation and diarrhoea. Spurious diarrhoea, often with faecal incontinence, is also commonly found in patients with faecal impaction from chronic constipation. In most patients, chronic diarrhoea is due to colorectal disease. Small bowel disease and pancreatic malabsorption are less common causes of chronic diarrhoea. The bulky, pale, difficult-to-flush stool of steatorrhoea malabsorption is an even more unusual cause of chronic diarrhoea. Irritable colon is a frequent cause of chronic diarrhoea.
Causes
1. Functional bowel disease
2. Inflammatory bowel disease
3. Carcinoma of the colon
4. Less common causes
History
Two clinical features deserve special attention when taking a history from a patient with chronic diarrhoea: the recognition of blood in the stool and the symptoms of systemic illness (Table 7.13). When both are present, inflammatory bowel disease is probably the cause; when both are absent, a diagnosis of functional bowel disease, malabsorption, laxative abuse or chronic parasitic infection is more likely. Systemic illness without blood suggests inflammatory bowel disease, small bowel or pancreatic malabsorption, chronic parasitic infestation or carcinoma of the colon. Blood without systemic illness suggests distal colitis, carcinoma or polyp or a minor anorectal disorder. Local anal lesions are often associated with chronic diarrhoea. The patient may complain of the intermittent passage of bright red blood seen on the toilet paper; blood mixed with the stool is of even more clinical significance. A clinical diagnosis of irritable colon should not be assumed without adequate investigation to exclude more serious causes. Frank blood mixed with diarrhoeal motion is found in ulcerative colitis, amoebiasis, shigella and campylobacter infections, ischaemic colitis and radiation enteritis. Blood may not be recognised by the patient, particularly if the stool is a rusty colour rather than being frankly bloodstained, as may occur in ulcerative colitis.
Table 7.13 Diarrhoea: clinical features assisting the diagnosis
|
Blood in the stool |
Systemic illness |
|
|
Inflammatory bowel disease |
Yes |
Yes |
|
Functional bowel disease |
No |
No |
|
Inflammatory bowel disease |
No |
Yes |
|
Distal colitis |
Yes |
No |
Laxative abuse is quite a common cause of chronic diarrhoea and should always be considered, especially when hypokalaemia is discovered on investigation. Hypokalaemia is also seen in cases of profuse mucous diarrhoea from villous adenoma. Hypokalaemia is a rare finding in irritable colon, although it may occur coincidently in patients on diuretic treatment without potassium replacement.
1. Functional bowel disease
The form of functional bowel disease that presents with chronic intermittent diarrhoea is called irritable colon (Box 7.23). The diagnosis is suggested by a long history of intermittent diarrhoea, often triggered by particular environmental factors. These include food sensitivities (especially to wheat, nuts, chocolate, citrus fruit and occasionally milk and milk products), broad- spectrum antibiotics, alcohol and travel. Excessive alcohol consumption is a common cause of watery diarrhoea. Anxiety is also a common precipitating factor, which is in turn worsened by fear that cancer is the cause of the symptom. The symptoms of early diverticular disease can be identical with those of the irritable colon syndrome. In practice, when a patient over 50 years presents with the symptoms of irritable colon, diverticulosis of the colon is often found on subsequent investigation. Diverticular disease will thus become the diagnosis, although in these older patients more care must be taken to exclude carcinoma.
Box 7.23
Cardinal features of the syndrome of functional bowel disease
![]() Abdominal discomfort or pain with bloating
Abdominal discomfort or pain with bloating
![]() Partial relief of abdominal pain by defaecation
Partial relief of abdominal pain by defaecation
![]() Past or present altered bowel habit (constipation — spastic colon or diarrhoea — irritable colon)
Past or present altered bowel habit (constipation — spastic colon or diarrhoea — irritable colon)
![]() Mild tenesmus
Mild tenesmus
![]() Long history, intermittent and nonprogressive symptoms
Long history, intermittent and nonprogressive symptoms
In contrast to the diarrhoea of organic disease, the symptoms of irritable colon rarely disturb the patient at night and symptoms seldom become worse over months to years. No anorexia or weight loss is present, in fact usually quite the contrary. The diarrhoea is worse in the early morning and the patient has to hurry to the toilet on arising (morning rush). For the remainder of the day, few symptoms are experienced. Lower abdominal discomfort, borborygmi and bloating, are characteristic of the condition. The discomfort is partially relieved by defaecation and is associated with mild tenesmus. A long history of constipation is common, both before and between the bouts of chronic diarrhoea. Systemic signs are absent. Rectal bleeding, unless from a minor anorectal disorder, is not found with irritable colon or diverticular disease.
2. Inflammatory bowel disease
There are two forms of inflammatory bowel disease: (mucosal) ulcerative colitis and (transmural) Crohn’s disease. Symptomatically, ulcerative colitis and Crohn’s disease can be very similar. It may be impossible from the history to differentiate between the two and histological differentiation may be required (Table 7.14). In most instances the onset is in young adult life and in both conditions a slight rise in incidence again occurs in patients over 50 years of age. In contrast to irritable colon, the diarrhoea is progressive and nocturnal and systemic illness occurs with weakness, anorexia and weight loss. Other systemic manifestations — including fever, arthritis, uveitis, pyoderma gangrenosum, erythema nodosum and hepatitis — occur with equal frequency in the two forms of inflammatory bowel disease. Gross bleeding, with episodic diarrhoea and cramping lower abdominal pain, is more suggestive of ulcerative colitis. Perianal disease is a feature of Crohn’s disease and may precede bowel symptoms; perianal disease is infrequent with ulcerative colitis.
Table 7.14 Pathological features of inflammatory bowel disease
|
Ulcerative colitis |
Crohn’s disease |
|
|
Distribution |
Colon |
Segmented ‘skip’ lesions |
|
Macroscopic findings |
Mucosal granularity with contact bleeding |
Transmural involvement |
|
Microscopic findings |
Mucosal inflammation and submucosal abscesses |
Transmural fissured inflammation |
In ulcerative colitis the onset of the first attack is often gradual, with weeks or months of increasing diarrhoea. Sometimes the earliest symptom is bright bleeding without diarrhoea. Blood mixed with the loose motion produces a rusty appearance to the stool, rather than the appearance of frank blood. A substantial minority of patients have fulminant disease with an acute onset to the disease and become seriously ill within days or weeks. In Crohn’s disease, the onset is also gradual, often with symptoms for more than a year before the disease is recognised. Obvious blood loss is uncommon in Crohn’s disease, which can be particularly difficult to distinguish from irritable colon in its early stages, but progressive weight loss and worsening colicky abdominal pain usually occur. While ulcerative colitis is always confined to the colon, Crohn’s disease involves three main sites: the small bowel, the ileocaecal region and the remaining colon. In addition to the symptom of diarrhoea (which is common to all types), in small bowel Crohn’s disease, obstruction tends to dominate the clinical picture; in the colon, perianal disease, deterioration in general health and toxicity with weight loss are more prominent symptoms. In patients with ileocaecal Crohn’s disease, both obstruction and toxicity coexist with diarrhoea.
3. Carcinoma of the colon
In most patients with carcinoma of the large bowel, presentation is with iron deficiency anaemia (the right colon), altered bowel habit with constipation (transverse and left colon) or anorectal bleeding on defaecation (sigmoid colon and rectum). In a proportion of cases (particularly those with faecal impaction and in proximal lesions with partial obstruction), presentation may be with profuse diarrhoea of relatively recent onset. There is usually no past history of episodic diarrhoea unless it is as a complication of chronic ulcerative colitis. However, colonic cancer may develop in patients with longstanding symptomatic diverticular disease. The diarrhoea in patients with cancer is nocturnal and progressive. A steady general deterioration in health occurs, rather than the acute and florid toxicity seen in patients with inflammatory bowel disease. Cancer is also often associated with evidence of obstruction, with increasing lower abdominal discomfort and distension.
Examination
Inspection of the abdomen may reveal scars, suggesting the possibility of a surgical cause of malabsorptive diarrhoea. The type of surgery performed must be sought from past records. A palpable, tender sigmoid colon is commonly detectable in irritable colon and diverticulosis. Abdominal tenderness and guarding is often found in ulcerative colitis and a tender mass suggests Crohn’s disease. In the majority of patients with early presentation of inflammatory bowel disease, abdominal examination is normal. Most obstructing carcinomas of the colon are impalpable. Liver metastases may be felt. The indentable masses of faecal impaction may be palpable. Multiple firm masses may also be due to Crohn’s skip lesions of the small bowel. Extracolonic manifestations of inflammatory bowel disease may be present and include erythema nodosum, pyoderma gangrenosum, conjunctivitis or uveitis, polyarthritis and evidence of liver disease. Except for the liver disease of sclerosing cholangitis, the manifestations are related to disease activity.
Rectal examination — anoproctoscopy
Rectal inspection and proctoscopic examination may reveal the diffuse granular friable mucosa with contact bleeding of ulcerative colitis. Patchy involvement of the rectum is seen in about half the patients with Crohn’s disease presenting with diarrhoea. In Crohn’s disease perianal disease is also common, with induration of the perianal skin, indolent undermining bluish ulceration, fistula formation and fissuring. Perianal abscess is common. Sigmoidoscopy confirms the above findings and rectal biopsy should be performed to establish the diagnosis. Biopsy of the rectal mucosa may show granulomas that are diagnostic of Crohn’s colitis, even though the mucosa appears normal. Diffuse rectal changes may also be due to Crohn’s disease, shigellosis, salmonellosis, amoebiasis and antibiotic colitis. Pseudomembranous colitis secondary to antibiotic treatment appears as raised yellow membranous plaques scattered over the mucosa. Patchy mucosal changes may also occur in amoebiasis. The blister-like gas cysts of pneumatosis cystoides intestinalis are unmistakable. The dusky black appearance of melanosis coli, secondary to anthroquinone laxatives, suggests that laxative abuse may be the cause of chronic diarrhoea.
Diagnostic plan
A sample of stool may be brought by the patient for examination. As well as clarifying the character of the diarrhoea, stool examination can be of potential diagnostic importance.
In many cases a provisional clinical diagnosis of irritable colon can be made and treatment commenced. Further investigation is necessary for persisting symptoms.
Blood examination. The FBE and the ESR or CRP is normal in functional bowel disease. Anaemia and leucocytosis suggest inflammatory bowel disease. A raised eosinophil count may indicate parasitic disease. Iron deficiency anaemia suggests malignancy. Serum electrolytes may show hypokalaemia, suggesting that laxative abuse or (rarely) villous adenoma must be excluded as the cause of the diarrhoea.
Stool examination — microscopy and culture. The stool is examined immediately (as a wet film or concentrated deposit) for cysts, ova and parasites — especially Giardia and Entamoeba. It is essential that specific pathogens are eliminated as the cause of diarrhoea for patients in whom ulcerative colitis and Crohn’s disease is suspected. Special culture methods may be necessary for Shigella, Salmonella, Campylobacter and Yersinia. The finding of Clostridium difficule toxin in the stools assists in the diagnosis of pseudomembranous colitis.
Radiology. Plain abdominal X-ray in patients with acute ulcerative colitis may show the dilatation of fulminant colitis (Fig 7.66). Monitoring changes in the diameter of the bowel and mucosal detail on plain X-ray is a valuable method of following the progress of patients with inflammatory bowel disease. Plain abdominal examination in patients with malabsorption may reveal pancreatic calcification in chronic pancreatitis or multiple fluid levels in obstructed and dilated bowel.

Figure 7.66 Diagrammatic representation of toxic megacolon
There is dilatation, gas in the bowel wall and loss of haustra in the transverse colon.
Barium enema, which is used less commonly in patients with ulcerative colitis, is better left until after the acute attack has settled. Mucosal granularity or frank ulceration with pseudopolyp formation may be seen in cases with more advanced disease. In Crohn’s disease the changes are segmental and patchy, with a cobblestone or thumb print mucosal appearance with deep mucosal fissuring or ‘rose thorn’ ulceration and eventually fibrosis and stricture in chronic disease (Table 7.15). Small bowel enema may assist in the diagnosis of small bowel diarrhoea, by showing flocculation and clumping of barium, blind loops or small bowel stricturing or diverticula.
Table 7.15 Radiological features of inflammatory bowel disease
|
Ulcerative colitis |
Crohn’s disease |
|
|
Distribution |
Continuous, confined to colon |
Segmental asymmetrical involvement (skip lesions, string sign) |
|
Mucosal appearance |
Granularity |
Cobblestone mucosal fissuring (‘rose thorn’) |
|
Stricture |
Uncommon; suspect malignancy |
Common |
|
Fistulas |
Rare |
Common |
Colonoscopy. The chief value of flexible sigmoidoscopy or colonoscopy is the detection of local obstructing lesions such as carcinoma and biopsy of mucosal lesions to confirm the diagnosis. Colonoscopy is indicated in patients in whom frank rectal bleeding is mixed with the motion, but should not be performed during an attack of acute colitis. Colonoscopy is particularly useful for the long-term follow-up of patients with ulcerative colitis — to detect malignant or premalignant change in colonic epithelium.
Special investigations. Investigations are often indicated when there is doubt about the diagnosis. The combination of raised faecal fat and reduced folate levels suggests that diarrhoea is caused by a small bowel mucosal lesion and is an indication for small bowel biopsy. Small bowel biopsy is essential for the histological diagnosis of coeliac disease and giardiasis. Increased faecal fat and normal folate levels suggest pancreatic disease and the need for investigation by CT scan, ERCP and serum gastrin estimation. A small bowel enema is the best currently available method of demonstrating small bowel anatomy. Crohn’s disease, lymphoma, carcinoma or other causes of small bowel malabsorption, such as the blind loop syndrome, may be detected by this investigation.
Treatment plan
Antibiotics, lactose and gluten-free diets, together with standard methods of management for the irritable colon syndrome, may be justified as a therapeutic trial before proceeding with further investigations. A trial of metronidazole may be warranted in patients with diarrhoea in whom giardiasis is suspected, even if protozoa cannot be identified.
1. Functional bowel disease
The basis of long-term management of irritable colon is the gradual introduction of increased fibre in the diet (Ch 7.20). In order to achieve the best response to dietary change, it should be stressed that breakfast is the most important meal and that fibre intake must be increased at that time, as well as throughout the day. Although the clinical diagnosis is often obvious, it is essential to exclude cancer and to reassure the patient. Colonoscopy is therefore required in many instances and especially in patients aged over 50 years. Most patients will benefit in the short term from the administration of a sedative and an antidiarrhoeal agent such as aluminium hydroxide. Some patients have specific food sensitivities. The specific foods that cause symptoms can be identified and thus avoided, while still increasing the food residue intake. The use of a bulking agent or hydrophilic colloid may be a useful supplement to dietary measures to establish early symptomatic improvement and to improve patient compliance. If depression is a significant factor, appropriate treatment may induce a remission of symptoms.
2. Inflammatory bowel disease
In ulcerative colitis, the goal of conservative therapy is to terminate an acute attack as rapidly as possible and to prevent relapse. Patients with a mild attack can be treated as outpatients with topical corticosteroids, self-administered as a nocturnal retention enema. Mesalazine is also begun and reduces the incidence of relapse in the long term. If topical corticosteroids fail to control the symptoms within two weeks, the patient should be admitted to hospital for more intensive treatment.
Patients with a severe attack of colitis need admission to hospital. The management plan depends upon the severity of the illness and is usually orchestrated by a multidisciplinary team. Patients with the most severe and fulminant disease can have a high mortality and very careful management is necessary. Initially the patient is resuscitated; intravenous corticosteroids are commenced. In most patients with chronic diarrhoea from ulcerative colitis, significant potassium loss occurs in the stool, causing a marked reduction in total body potassium levels.
The major sign of fulminant disease is colonic dilatation on plain X-ray of the abdomen, called toxic megacolon; these patients are considered for urgent surgery. Anticholinergic drugs used to control diarrhoea must be stopped because of the danger that toxic megacolon may be induced thereby. Parenteral nutrition is indicated during acute attacks to correct protein deficiency (as well as for fluid and electrolyte replacement) and to maintain nutrition during a period of gastrointestinal rest. Blood transfusion of packed cells is often necessary in anaemic patients.
The patient with established perforation and peritonitis is prepared for urgent surgery. Because of the immediate danger of perforation, toxic megacolon is also an indication for urgent surgery. In about 60% of cases of acute nonfulminant disease, clinical remission occurs within five days of presentation, at which time oral feeding and oral corticosteroids are started. Failure to improve clinically by this time is another indication for emergency colectomy. Persistent toxicity, abdominal tenderness and colonic dilatation on X-ray are the main criteria used to establish a failed response to conservative treatment. Surgery is also indicated if steroids cannot be withdrawn after six weeks of intensive treatment (Box 7.24).
Box 7.24
Indications for surgery in ulcerative colitis
Acute disease
Failed medical treatment
Toxic megacolon and perforation
Colonic haemorrhage
Chronic disease
Intractable symptoms
Extra-intestinal manifestations
Precancerous dysplasia, cancer
Total proctocolectomy with permanent ileostomy is the standard emergency surgical procedure. Elective surgery is indicated when there are chronic disabling symptoms, poor nutrition and growth failure in children. In younger patients ileoanal anastomosis with formation of a pouch may be considered (Fig 7.67). Because of the increased risk of carcinoma after 10 years of the disease, patients should receive regular colonoscopic follow-up with multiple biopsies. If dysplasia is found on colonic biopsy on several occasions separated by six months, elective colectomy is indicated.

Figure 7.67 Surgical procedures used for ulcerative colitis
A: protocolectomy and Brooke ileostomy; used mainly for emergency surgery and for older patients; B: ilio-anal J-pouch with preservation of anal sphincters, used mainly as an elective operation in younger patients; contraindicated in Crohn’s disease
Crohn’s disease may also present with fulminant disease. The more common mild attack is treated by rest, low residue, lactose-free, high-protein diet with vitamin and mineral supplements. Malnourished and obstructed patients may benefit from total or supplementary parenteral nutrition. Corticosteroids are of questionable value. Urgent proctocolectomy with ileostomy is indicated in some patients with fulminant colonic disease and electively in resistant patients with extensive colonic involvement — especially when associated with severe perianal disease. In about half the patients with small bowel and ileocaecal Crohn’s disease, surgical resection is necessary for persistent obstruction. Internal fistula, abscess and perianal disease are other indications for surgery (Box 7.25).
Box 7.25
Indications for surgery in crohn’s disease
Failure of medical treatment
Acute or chronic obstruction
Fistula or abscess
Perianal disease (especially with fistula)
Haemorrhage
3. Carcinoma of the colon
Patients with carcinoma of the colon who present with diarrhoea often present special technical difficulties during surgery, due to faecal loading above the tumour. Preoperative measures to empty the bowel may be unsuccessful and intraoperative washout of the colon may be necessary. A proximal colostomy is often required after resection.
4. Less common causes
Colonic diarrhoea
Laxative abuse is an important cause of colonic diarrhoea. This may be difficult to detect when the patient denies purgative intake. Some patients may have associated muscle weakness because of potassium loss. Laxative abuse is common in patients with anorexia nervosa, both to lose weight and for the constipation induced by low food intake and self-induced vomiting. Anal stenosis is commonly found on rectal examination and the melanosis coli seen on sigmoidoscopy may be the only clue to the diagnosis. Chronic laxative abuse with longstanding biochemical disturbances and structural damage to the colon can lead to gross colonic dilatation and ileus (‘cathartic colon’). The colonic damage of cathartic colon is irreversible and there is little alternative in treatment but to continue the patient’s laxative intake. Several other drugs may cause chronic diarrhoea (Box 7.26).
Box 7.26
Drugs that cause chronic diarrhoea
Digitalis
Laxative abuse
Alkalis
Antibiotics
Folic acid antagonists
Indomethacin
Large villous adenomas of the rectum may present with profuse mucous diarrhoea sufficient to produce dehydration and lethargy and weakness from hypokalaemia. Low anterior resection or colo-anal anastomosis is often necessary for adequate removal. In most patients ischaemic colitis follows an episode of localised venous thrombosis. Chronic diarrhoea follows an acute episode of bleeding and pain, especially in patients in whom acute ischaemia is followed by fibrosis, stricture and ulceration. Barium enema shows a stenosed segment, usually in the region of the splenic flexure. Resection may be necessary for obstruction. Pseudomembranous colitis occurs in debilitated patients and usually follows a mild course of diarrhoea for a few weeks. Sigmoidoscopy reveals an erythematous mucosa covered with patchy yellow fibrinous plaques. The diagnosis is made by the detection of Clostridium difficiletoxin in the stool. Treatment is with vancomycin combined with measures to control the diarrhoea. Pneumatosis coli is an unusual condition of unknown cause in which submucosal gas cysts form in the bowel wall. Associated chronic obstructive airways disease is common. Symptomatically there is an intermittent history of diarrhoea, with mucus and sometimes blood. Treatment is symptomatic. Eventually permanent resolution of symptoms occurs.
Malabsorption and small bowel diarrhoea
There are many causes of malabsorptive diarrhoea that can be classified as being due to a structural defect, a mucosal lesion, infection or extragastrointestinal disease. The major symptoms of malabsorption are watery diarrhoea, abdominal discomfort, anaemia and weakness. Steatorrhoea is not common. The structural defects of significance are pancreatic insufficiency, small bowel resection, radiation enteritis, small bowel malignancy and the postsurgical blind loop syndrome with bacterial overgrowth. The important mucosal lesions causing chronic diarrhoea are coeliac disease and hypolactasia. In coeliac disease the diarrhoea is severe and often associated with weight loss and fluid and electrolyte loss. Glossitis and stomatitis are striking features. The diagnosis is usually made after the exclusion of other causes and is based upon the severity and persistence of the symptoms. This eventually leads to small bowel biopsy that shows the subtotal villous atrophy that is characteristic of coeliac disease. Treatment by gluten-free diet produces a complete remission of symptoms and mucosal recovery. The infections that can lead to chronic small bowel diarrhoea are Entamoeba, Giardia, Strongyloides, Campylobacter, Yersinia and Mycobacterium. These infections are especially seen in the immunocompromised host. Thyrotoxicosis is the most common extragastrointestinal disease that can present with chronic diarrhoea. Diabetes can present with diarrhoea and with nocturnal faecal incontinence.
7.22 Acute anal pain
The columnar mucosa of the rectum and upper anal canal is insensitive to ordinary painful stimuli. Therefore, ulceration or other lesions at this level are relatively painless. In contrast, lesions below the dentate line, which has somatic innervation, are accompanied by pain that is often severe and can usually be accurately localised. In most patients presenting with anal pain, the history is short and the pain severe and progressive (often described as painful piles). In many cases a lump is present; pain, lump, fever and toxicity are the most important features to consider when making a diagnosis. Poorly localised, deep-seated rectal and perineal pain can usually be distinguished from anal pain. Pelvic inflammatory disease, prostatitis and functional bowel disease with rectosigmoid spasm are conditions that may initially be considered in differential diagnosis but can usually be excluded as causes of acute anal pain. Pilonidal abscess is an important and common cause of pain in this area, but pain is localised posterior to and above the anus in the natal cleft, extending to the buttock.
Causes (see Fig 7.68)
1. Perianal haematoma
2. Anal fissure
3. Complicated haemorrhoids
4. Perianal abscess
5. Less common causes

Figure 7.68 Common causes of acute anal pain
A: perianal haematoma; B: anal fissure; C: strangulated haemorrhoids; D: perianal abscess
History
1. Perianal haematoma
In most patients the history is of sudden pain and a lump appearing after straining. Milligan described the lesion as the ‘five day, painful, self-curing pile’. Sometimes when pain is not marked, the patient with perianal haematoma may present because of the lump alone (Ch 7.24). Pathologically the condition is a clotted haematoma affecting an external haemorrhoidal vein.
2. Anal fissure
Anal fissure presents as severe pain on defaecation, usually developing after a period of constipation and tenesmus or occasionally following a bout of acute diarrhoea. The change in bowel habit leading to fissure is frequently precipitated by dietary change, constipating drugs, immobilisation because of ill-health, dehydration and poor bowel habits. Fissure is common in shift workers and travellers, and after childbirth. The symptoms usually develop gradually as a burning or aching pain on defaecation, which then persists. Sometimes the pain is delayed, not starting until an hour or so after defaecation, then lasting for hours and sometimes radiating down the back of both legs. The patient enters a vicious circle with increasing pain, fear of defaecation and increasing constipation. Most patients present within a week or two of the onset of pain, but sometimes do not present until after months or even years of persistent discomfort on defaecation. Self-treatment with various medicaments is common. Presentation in these cases may be because of small amounts of bright red anorectal bleeding, seen as spotting on the toilet paper (Ch 7.19).
3. Complicated haemorrhoids
Most patients with acutely thrombosed haemorrhoids present with a recent history of painful irreducible prolapse. The condition is very distressing and the patient usually takes to bed. In nearly all cases, a past history of prolapsing or bleeding haemorrhoids is present. The haemorrhoids gradually become more difficult to return until suddenly acute strangulation and irreducibility occur. In most patients strangulation is a complication of third-degree haemorrhoids. While the symptoms associated with third-degree haemorrhoids may be diminished by increasing the fibre intake, the incidence of the complication of strangulation is probably not reduced in the long term by dietary changes. Fourth-degree haemorrhoids that are permanently prolapsed can undergo strangulation, but this is unusual. Some patients present with acute strangulation of haemorrhoids with no previous history. In these cases the underlying pathology may be anterior rectal descent or early mucosal prolapse.
4. Perianal abscess
Patients with perianal abscess present with a short history of severe, constant, throbbing perianal pain and swelling associated with fever and toxicity. In the less common intermuscular (formerly called submucosal) form of perianal abscess, with upward spread between internal and external sphincter, there is no external evidence of the abscess (Fig 7.69). Examination under anaesthesia is often necessary for diagnosis. Chronicity is more common in patients with intermuscular spread from anal gland infection. Other, more unusual causes of recurrent or chronic perianal abscess formation include carcinoma, Crohn’s disease, foreign body perforation of the rectum and tuberculosis. Pilonidal abscess, Bartholin’s gland infection, infected sebaceous cyst, furuncles and suppurative hydradenitis are subcutaneous infections that may extend to produce a perianal abscess. Only occasionally are these confused with the primary form of anal gland infection.

Figure 7.69 Anatomical location of perianal abscesses
A: perianal; B: intermuscular; C: supra-levator
Examination
Perianal haematoma appears as a well-defined, tender, bluish lump, close to but clearly separated from the anus. In the neglected case the overlying skin can ulcerate, exposing a black clot. Sometimes the haematoma is deeper and is difficult to distinguish from a perianal abscess, as the overlying skin is oedematous and a diffuse rather than localised swelling; it may occupy up to one half of the anal verge.
In the majority of patients anal fissure is situated in the midline posteriorly, involving the lower third of the anus as an elliptical ulcer extending from the dentate line to the anal verge (Fig 7.70). A sentinel skin tag may be present. Digital examination and sigmoidoscopy are difficult because of painful anal sphincter spasm. Examination often needs to be performed under anaesthesia. The anal fissures of Crohn’s disease have a different appearance, being indurated, oedematous, bluish in colour and often multiple and often not in the midline.

Figure 7.70 Natural history of anal fissure
A: acute fissure; B: chronic fissure — weeks later; C: chronic fissure — months later. 1: hypertrophied anal papilla; 2: sentinal pile; 3: subcutaneous fistula
A strangulated internal haemorrhoid appears as a dusky or black and sometimes ulcerated lump, contiguous with and prolapsing from the anus. Adequate digital and proctologic examinations are usually not possible and the haemorrhoids can rarely be reduced manually.
The two main forms of abscess are perianal and intermuscular. In the more common perianal form there is a brawny, inflammatory swelling adjacent to the anus, the patient can point to the maximum point of discomfort and there is associated fever and tachycardia. Diagnosis of perianal infection is obvious. Fluctuation is not usually detectable because the pus, which is invariably present, is deep-seated and confined. An intermuscular abscess usually points into the rectum and does not present towards the skin. Diagnosis is less obvious. Careful palpation, proctoscopy and sigmoidoscopy (sometimes under anaesthesia) are necessary in order to make a diagnosis.
Diagnostic plan
Diagnosis is usually made on the history and physical examination. Rectal examination digitally and by sigmoidoscopy is necessary in all cases but may need to be performed under anaesthesia or be delayed. Both fissure and perianal haematoma may be induced by the tenesmus of carcinoma of the rectum. Performing an examination under anaesthesia is essential when the spasm associated with fissure precludes normal assessment and also to make a diagnosis of intermuscular abscess. Urinalysis is essential to exclude diabetes mellitus. The presence of diabetes or alcoholism in association with anorectal suppuration can sometimes be associated with potentially lethal, spreading anaerobic infection. Early exploration under general anaesthesia is mandatory.
Treatment plan
Most of the conditions presenting with acute anal pain are associated with longstanding faecal stasis. Correction of colonic dysfunction with a high-fibre diet is important if recurrence is to be prevented.
1. Perianal haematoma
Most superficial small perianal haematomas will settle down in several days. The only treatment required is reassurance, a local cooling lotion and analgesia. Incision may be necessary for the very painful lesion; however, this method of treatment is unsatisfactory for deeper perianal haematomas, which are not as painful. The majority of these deeper lesions settle within 10–14 days. Little is gained by incising the lesion, but sometimes excision of the lump with local anaesthesia will provide immediate relief if pain is severe.
2. Anal fissure
The transverse fibres of the internal sphincter can be seen to form the base of the ulcer, a sentinel pile is common and, in longstanding cases, a subcutaneous fistula is seen at the anal verge with fibrosis and anal stenosis. Most patients require a short admission to hospital. Lateral sphincterotomy is indicated in the majority of patients. Recently fissures have been successfully treated by botulinum toxin (Botox) injection that acts by relieving muscular spasm.
3. Complicated haemorrhoids
In patients with a past history of internal haemorrhoids, early surgery with local ligation and excision of the thrombosed haemorrhoid is indicated, unless a significant degree of local sepsis exists or the patient is a very poor anaesthetic risk. Even if the haemorrhoids reduce in hospital, it is usually best to perform haemorrhoidectomy before discharge. In patients in whom strangulation appears to be the first form of presentation of internal haemorrhoids, and in those patients presenting late with gross necrosis, treatment should be conservative: rest, analgesia and a local anaesthetic ointment. Haemorrhoidectomy is usually required at a later date. Fourth-degree haemorrhoids with persistent discomfort are also best treated by haemorrhoidectomy.
4. Perianal abscess
Suppuration is the rule in perianal abscess, despite lack of fluctuation. Prompt surgical drainage by a cruciate incision over the point of maximum induration is the primary method of treatment. Antibiotics are not indicated unless the patient is septicaemic or because of concurrent medical illness.
With intermuscular abscesses, incision and drainage into the anal canal is performed together with internal sphincterotomy below the abscess.
As most perianal abscesses arise from the anal glands, there is potentially an internal communication to an anal crypt. This internal communication into the crypt of Morgagni can rarely be defined when draining an abscess; such an internal communication should not be overzealously sought under general anaesthesia, because of the danger of creating a false passage. The incidence of subsequent development of a perianal fistula connecting the skin to anal crypt can be reduced by careful postoperative dressing of the abscess cavity. It is not practical in most cases to continue dressing in hospital; unsupervised dressing by the patient is a more economic and effective approach. A silastic elastomer foam dressing pad can be very helpful. Subsequently, if necessary, at a second stage, a persisting perianal fistula can be laid open (Ch 7.27).
5. Less common causes
Carcinoma of the anus usually presents with a lump, but frequently carcinoma is painful and this symptom leads to presentation. Treatment is usually with chemoradiotherapy (Nigro regimen), with abdominal resection being reserved for incomplete response or recurrence. Painful herpetic ulceration occurs especially in patients being treated with cytotoxic agents. Coccydynia is a poorly defined condition that presents with pain and soreness over the coccyx, sometimes initiated by trauma. The pain may occasionally be reproduced by manipulation of the coccyx. Associated functional symptoms are common. Conservative measures are indicated. Steroid injections can be tried if conservative treatments with analgesics are ineffective. Surgical excision should not be performed. Proctalgia fugax means fleeting rectal pain. The pain from this condition varies in severity from mild discomfort to a very severe pain. The attacks usually start in early adult life, lasting from a few minutes to half an hour and often wake the patient at night. This condition is probably a form of functional bowel disease and should be treated as such. The solitary rectal ulcer syndrome can also present with pain, but usually presents as the sensation of a rectal lump causing obstructed defaecation. The pelvic and perineal pain of pelvic inflammatory disease (Ch 7.7) and of prostatitis may sometimes, particularly when acute, be localised to the anus, but does not usually give problems in the differential diagnosis of acute anal pain.
An anal foreign body can cause severe pain if the mucosa is breached and presentation then may be as a perianal abscess. Intraluminal foreign bodies due to exotic sexual practices are usually obvious on examination, although their presence may not be mentioned in the history. Their variety is endless and their extraction, which may require general anaesthesia, can tax the dexterity and resourcefulness of the surgeon and require obstetrical forceps or other special tools or techniques.
7.23 Anal pruritus
Lesions that tend to produce chronic itch are skin tags, anal condylomas, anal fistula and, rarely, Bowen’s disease (intradermal carcinoma). Haemorrhoids are often associated with itch, but are rarely the principal cause. Pruritus vulvae may be confused with anal pruritus (pruritus ani). The two conditions coexist in a number of patients, most commonly in post-menopausal women. Anal pruritus can be part of generalised skin conditions such as psoriasis or seborrhoeic dermatitis. In many patients with anal pruritus, no specific cause for the condition is discovered.
Common causes
1. Defaecatory habits and local hygiene
2. Local anorectal conditions
3. Generalised skin or systemic disorders
Inadequate dietary fibre intake, poor defaecatory habits and obsessive or poor hygiene can lead to pruritus. The neglect or absence of the normal gastrocolic reflex after breakfast, diminished colonic propulsion, difficulty initiating rectal emptying with a history of straining at defaecation and mucosal prolapse, are common features of the history. Sometimes poor hygiene is important, but more commonly an excessively scrupulous attention to cleanliness perpetuates the condition. In some cases alcoholism appears to be a dominant factor — perhaps because of the frequency of intermittent diarrhoea. Other important factors are the macerating effect of heat, moisture and friction, producing intertrigo. Tight-fitting nylon underwear and obesity may further contribute and are risk factors that often need attention. Contact dermatitis is sometimes a factor. This may result from local anaesthetic and antihistamine creams and ointments, alkaline soaps (the skin of the anus is mildly acid) and suppositories. Diabetes mellitus must always be excluded. The itch in these patients may be due to perianal candidiasis. Oral antibiotics such as tetracycline can often initiate itch. Pinworm or threadworm infestations also cause itching.
History and examination
Pruritus is worse at night, during exercise and during hot weather. The history should be evaluated for the common risk factors — especially oral or topical medications, diet and defaecatory function, cleaning methods and clothing worn (Box 7.27). The skin changes may be minimal or extreme, varying from localised oedema of the anal folds, skin pallor and small superficial linear ulcers, to maceration, coarse white dullness of the skin, leucoplakia or lichenification. Superficial skin changes tend to be of two sorts: a moist macerated perineum and a dry scaling one. Histologically there is often hyperkeratosis with nonspecific inflammatory changes of the dermis. Full anorectal examination is necessary. Hypertrophied anal papillae are frequently found and anal spasm of mild degree is common.
Box 7.27
Risk factors for pruritus ani
Diabetes mellitus and candidiasis
Inadequate fibre intake
Over-scrupulous hygiene
Intertrigo
Contact dermatitis
Diagnostic plan
1. Full anorectal examination for local causes
2. Urinalysis for sugar to detect diabetes mellitus
3. Scrapings and microscopy to detect yeasts, fungi or parasites
4. Stool examination for parasites such as enterobius vermicularis (pinworm)
Treatment plan
An instruction form for the patient is useful (Box 7.28).
Box 7.28
Management of anal irritation
Diet
The amount of fibre (roughage) in the diet must be sufficient to abolish straining at stool (see high-fibre diet). An adequate breakfast is essential so that a normal bowel habit in the morning is established. Weight reduction will reduce sweating and friction and thus discomfort. A high-fibre diet will not increase weight if no sugar is added and if the other rules for weight reduction are followed (reduced fat intake).
Hygiene
The area must be kept clean and dry. Excess rubbing or scrubbing must be avoided. Gently wash after defaecation, make sure no soap is left, then dab dry or ideally use a hair dryer. Wear cotton, not nylon underclothing. Use a bland soap.
Local treatment
Ointments, creams and irritating soaps must not be used. If scratching while asleep is a problem, wearing cotton gloves may help. If the skin is broken, a soothing lotion such as calamine or a 50% solution of Castellani’s paint (magenta) may be applied after washing.
Measures to improve defaecatory habits
Measures include dietary correction with increased dietary fibre, weight reduction and attention to the need for defaecation in the morning by restoring a normal gastrocolic reflex after breakfast, sometimes with the assistance of a glycerine suppository. Diabetes will need to be controlled.
Local toilet hygiene
Attention to hygiene and the avoidance of the macerating effects of heat, moisture and friction, are important. Over-scrupulous efforts to gain cleanliness must be avoided. Scrubbing is replaced by a soft, moistened tissue used to clean the perianal area, then dabbing with a cloth or using a hair dryer to dry, aided if required by a bland powder to diminish friction. Sedatives may be necessary for a short period. Wearing gloves at night sometimes helps by reducing scratching. Clothing should be loose; nylon underpants should not be worn.
Remove irritants
Agents that may trigger contact dermatitis must not be used. No soap should be applied to the perianal area; self-medication with local anaesthetic, antihistamine and steroid ointments and creams must be stopped; powder should be used lightly. Oral antihistamines may help to control itch at night and reduce scratching.
Encourage healing
When the skin is broken, local treatment is applied each day after carefully and gently cleaning and drying the area. Local applications include 50% Castellani’s paint in water (magenta), 0.5% silver nitrate in water and calomine lotion. Sometimes a water-soluble corticosteroid cream at an acid pH, if applied three to four times per day, can give relief in acutely painful cases.
Surgery
Surgical treatment of associated local anorectal lesions may help, but surgery is rarely curative by itself.
Despite all measures, the condition can prove intractable.
7.24 Anorectal lump
Patients with anal lump often present because they fear cancer. This fear is usually groundless so far as the presenting lump is concerned — anal cancer is uncommon and haemorrhoids are common. Some patients with constipation present because of a sensation of a lump in the rectum that obstructs defaecation. This form of tenesmus is known as anismus and is usually secondary to spasm of puborectalis muscle. Longstanding dystonia of the puborectalis may lead to rectal prolapse.
The relationship of anorectal lump to defaecation is of diagnostic importance. A lump arising from the anal canal or rectum tends to appear intermittently upon defaecation and reduce afterwards. A persistent lump may be due to a permanently prolapsed lesion arising from the rectum or anal canal. A lump arising from perianal tissues is also present all the time and does not prolapse further. Common prolapsing lesions include second- and third-degree haemorrhoids, hypertrophied anal papilla, polyps and rectal prolapse. Common persisting lumps include skin tags, fourth-degree haemorrhoids and condylomata (Fig 7.71).

Figure 7.71 Anal lumps
A: prolapsing lumps; 1: second- and third-degree haemorrhoids; 2: rectal polyp; 3: rectal prolapse; 4: hypertrophied anal papilla B: persistent lumps; 1: condylomata; 2: skin tag; 3: carcinoma of the anal canal; 4: fourth-degree haemorrhoids; 5: perianal haematoma; 6: perianal fistula; 7: perianal abscess
Common causes
1. Haemorrhoids
2. Anismus and rectal prolapse
3. Skin tags and hypertrophied anal papilla
4. Condylomata acuminata (viral warts)
5. Carcinoma of the anus
History and examination
1. Haemorrhoids
Lumps due to internal haemorrhoids (second- and third-degree) usually appear and bleed on straining and defaecation and are spontaneously or manually reducible. Haemorrhoids presenting with a persistent anal lump include advanced fourth-degree haemorrhoids, which are prolapsed most of the time. The diagnosis of haemorrhoids is established on inspection and at proctoscopy. Occasionally hypertrophy and fibrosis can occur in a second- or third-degree internal haemorrhoid from repeated intussusception, producing a fibrous pseudopolyp. Malignant change rarely develops in the haemorrhoidal mucosa, leading to a prolapsing early carcinoma of the rectum being mistaken for a benign haemorrhoid on cursory examination. Sigmoidoscopy should be performed as a routine in all cases to exclude an associated higher rectal carcinoma. Any evidence of altered bowel habit demands full colonic examination.
2. Anismus and rectal prolapse
A common complaint in patients with longstanding history of constipation is the sensation of an obstructing lump in the rectum (anismus). This symptom is probably due to spasm of the puborectalis muscle; no abnormal signs are evident but mucosal prolapse can follow. There are two main groups of patients with constipation — those with delayed colonic transit and those with anismus. In patients with anismus the anal sphincter remains contracted despite straining so that defaecation becomes ineffective. Symptoms can mimic anal fissure or rectal carcinoma. In longstanding cases, mucosal prolapse, partial and complete rectal wall prolapse and the solitary rectal ulcer syndrome can develop (Fig 7.72). Solitary rectal ulcer develops at the apex of the anterior descending mucosal fold at about 5–10 cm from the anus. The histological findings on biopsy are described as proctitis cystica profunda.

Figure 7.72 Rectal prolapse
1a: puborectalis spasm (anismus) with ineffective straining; 1b: treatment by division of the puborectalis muscle; 2: anteriror rectal wall descent with solitary rectal ulcer from abrasion of the apex; 3a: rectal prolapse; 3b: rectal prolapse treated by rectopexy (Ripstein repair)
Rectal prolapse on defaecatory straining is usually a dramatic symptom; a history of ineffective straining at stool is commonly found on questioning. Prolapse may involve just the rectal mucosa — in part or the whole of its circumference. This can resemble prolapsing haemorrhoids; however, mucosal prolapse is initially anterior and later completely circumferential. Mucosal prolapse, in either instance, does not comprise a trio of focal lumps localised to the anatomical position of the haemorrhoidal vascular cushions (at 3, 7 and 11 o’clock) as occurs with prolapsed third- or fouth-degree haemorrhoids.
Prolapse of the complete rectum is usually unmistakable. The redoubled unfolded layers of the prolapse form a direct extension of the anal verge and are very different from the appearance and feel of a prolapsed polyp or carcinoma or apex of an intussuscipiens. Prolapse occurs in two main patient groups:
• in young children, it relates to problems of toilet training and usually does not persist into adult life
• in elderly patients (usually women), it is most often associated with lax sphincters and diminished anal tone. In most cases incontinence does not persist after repair of the prolapse so sphincter laxity is possibly secondary to prolapse rather than its cause.
3. Skin tags and hypertrophied anal papilla
Skin tags may arise from thrombosed external piles that have undergone fibrosis. Alternatively, tags develop from hypertrophied anal papillae that are found at the lower end of the anal columns at the dentate line. These lesions probably occur because of repeated minor defaecatory trauma. Diagnosis is obvious on inspection and at proctoscopy.
4. Condylomata acuminata (viral warts)
Anal warts are venereal lesions of the perianal skin due to a viral infection. They may also be present on penile and vulval skin. They appear as papillomatous lesions that coalesce and spread. A small fixed focal point of attachment remains, a fact that allows local treatment by diathermy or excision with preservation of skin and rapid healing.
5. Carcinoma of the anus
These uncommon tumours originate in the true skin of the anal margin or the modified skin of the lower anal canal. They have the characteristic appearance of squamous carcinoma elsewhere in the body and spread to involve the inguinal nodes. The usual presentation is with an anal lump and discomfort. Pain is rarely a prominent symptom. Occasionally carcinoma of the anus presents with a groin node. The differential diagnosis includes condylomata, anal fissure, severe pruritus ani, Crohn’s disease and herpetic ulcer. Biopsy is necessary to confirm the diagnosis.
Diagnostic plan
A careful history is the initial basis of a diagnosis, which is usually confirmed on inspection and proctosigmoidoscopy. Malignant disease must be excluded. Biopsy is necessary when ulceration is present and with prolapsing fibrous polyps and adenomatous polyps. Manometric studies and defaecation proctography may assist in the diagnosis of obstructed defaecation.
Treatment plan
1. Haemorrhoids
Advanced haemorrhoids are treated by haemorrhoidectomy. Fibrous polyps and adenomatous polyps require snare excision biopsy.
2. Anismus and rectal prolapse
While anismus may be triggered by lack of dietary fibre, patients with the symptom of obstructed defaecation do not respond (or might indeed become worse) by simply increasing dietary fibre — in contrast to beneficial effects of fibre in other types of functional bowel disease. The disorder of defaecatory function may be primary or the pathology may have advanced to a degree that correction of colonic propulsion with fibre is unavailing. Because anismus is not associated with slowed colonic transit (the other major cause of constipation), colonic resection fails to control the problem because the defect is located in the pelvic floor. Obstructed defaecation is best managed by using the gastrocolic reflex to assist the retraining of bowel function. Breakfast, morning exercise and regular suppositories may assist in the restoration of normal morning defaecatory habits and thus control both straining and obstructed defaecation.
Rectal prolapse is an indication for surgery. There are many operations for prolapse but resection of the prolapsed rectum or operations to ‘sling’ the rectum in the pelvis are most favoured.
3. Skin tags and hypertrophied anal papilla
These can be locally excised.
4. Condylomata acuminata (viral warts)
These can also be locally excised.
5. Carcinoma of the anus
In most patients with carcinoma of the anus, initial chemoradiotherapy is the mainstay of treatment and, rarely, abdominoperineal excision with permanent colostomy may be necessary. Later excision of inguinal nodes is indicated if they remain enlarged despite resolution of perianal sepsis. This procedure gives worthwhile palliation, avoiding malignant infiltration in the groin with leg oedema and local ulceration.
7.25 Anal discharge and incontinence
Anorectal incontinence can be defined as the involuntary passage of faeces, flatus, mucus or pus from the anus. The patient may or may not be aware of incontinence or of poor control of sphincter competence. The voluntary control of the passage of flatus and faeces is a complex process, depending upon the integrity of the sensory innervation of the rectum and the anorectal musculature. The muscles involved are both the voluntary external sphincter muscle group and the involuntary portions of the levator and internal sphincter muscles. Incontinence may therefore occur with diseases that interfere with sensation or with the function of the anorectal muscles. Mucosal apposition of the anal verge and stool consistency and urgency are also important factors.
A minor degree of faecal soiling is quite a common symptom. Often this results from poor hygiene in individuals with a deeply set anus. The symptom is also common after a sudden increase in fibre intake. Nocturnal incontinence occasionally occurs in diabetics with autonomic neuropathy, which can also be associated with nocturnal diarrhoea. Persistent anal or perianal discharge may be confused with incontinence but, in these patients, control is normal and faecal soiling of the clothing is not found. Most cases of mucus discharge are secondary to advanced haemorrhoids or pruritus ani (Fig 7.73). In some instances continuous mucus discharge presents as spurious diarrhoea when secondary to faecal impaction. A fistula (especially rectovaginal fistula with faecal soiling) is an uncommon but important cause of incontinence. A purulent discharge without faecal incontinence is suggestive of perianal fistula.

Figure 7.73 Causes of anal or perianal discharge
A: advanced haemorrhoids; B: advanced carcinoma of the rectum (or villous adenoma); C: faecal impaction; D: pruritus ani; E: perianal fistula
Functional conditions, including obstructed defaecation and anterior rectal prolapse, are not associated with primary incontinence. However, longstanding extreme straining may lead to sphincter atrophy and secondary incontinence owing to eventual rectal prolapse. Colorectal disorders such as inflammatory bowel disease, faecal impaction, laxative abuse, mucus-secreting rectal tumours and irritable bowel syndrome can lead to poor control and urgency incontinence, despite the fact that the sphincter mechanism is normal. However, these patients usually present with urgent diarrhoea (Ch 7.21). Crohn’s disease with a contracted rectum may cause incontinence; associated bowel symptoms and signs usually make the diagnosis obvious.
Common causes
Figure 7.74 depicts the common causes of anorectal incontinence.
1. Sphincteric injury: traumatic and postoperative incontinence
2. Neurogenic (true) incontinence and idiopathic incontinence
3. Contracted or obstructed rectum
4. Fistula — perianal and rectovaginal

Figure 7.74 Anorectal incontinence
A: neurogenic and idiopathic incontinence; B: incontinence associated with rectal prolapse; C: traumatic incontinence; D: incontinence secondary to rectal Crohn’s disease; E: rectovaginal fistula (pseudo-incontinence)
History and examination
1. Sphincteric injury: traumatic and postoperative incontinence
In traumatic or postoperative incontinence, the external sphincter muscle is disrupted and has healed with scar formation of the external sphincter muscle. The defect can be felt; the site is often seen as an area of flatness or scarring with loss at one point of the corrugations and folding produced by an intact corrugator cutis ani muscle. Partial incontinence sometimes develops after internal sphincterotomy or anal dilatation for fissure. Sometimes incontinence is due to a localised keyhole defect following posterior internal sphincterotomy. More serious degrees of incontinence may be seen after falls astride or penetrating wounds, obstetric injury and fistula surgery — from damage to the puborectalis muscle or the anorectal ring. Digital examination, as well as excluding neoplasia, allows evaluation of the sphincter tone, the ability to contract the rectal muscles and rectal capacity and distensibility.
2. Neurogenic (true) incontinence and idopathic anorectal incontinence
In neurogenic incontinence the sphincters are atonic, with laxity of the anal canal, loss of tactile sensation, inability to contract muscles at will and absence of anal reflexes. Spinal cord and cauda equina lesions can cause anal paralysis and incontinence. Idiopathic faecal incontinence of the elderly may be due to a stretch injury of the perineal or pudendal nerves as a result of longstanding perineal and rectal descent. Secondary neuropathy may result from repeated straining at stool, rectal prolapse or childbirth. These patients commonly have a long history of straining at stool. Perineal or anterior rectal descent may be seen on straining and a lax and patulous anus is found on digital examination. Loss of the normal anorectal angle is a characteristic feature seen on sigmoidoscopic examination and on barium enema.
3. Contracted or obstructed rectum
Loss of rectal distensibility may produce incontinence. The conditions that can cause this form of incontinence are proctitis and Crohn’s disease, rectal neoplasm (with invasion of the sphincters), the descending perineum syndrome (occasionally with benign rectal ulcer), rectal prolapse and, rarely, scleroderma. The descending perineum syndrome and prolapse can cause incontinence both because the rectal capacity is reduced and because secondary neuropathy may occur. Simple repair of the prolapse will cure associated incontinence in most patients.
4. Fistula — perianal and rectovaginal
Most rectovaginal fistulas result from obstetric or surgical trauma, inflammatory bowel disease, cervical cancer and radiotherapy. There is vaginal incontinence of faeces or flatus. The site of the fistula can usually be seen and probed through to the rectum on vaginal examination.
The main complaint in patients with perianal fistula is intermittent or constant purulent anal discharge. Usually a perianal abscess has discharged spontaneously or has been surgically drained. Continence is normal. The diagnosis is soon made by the finding of a nodule of perianal granulation tissue that extrudes pus. An indurated subcutaneous track from this may be felt, extending towards the anus.
Diagnostic plan
In most patients a careful history and physical examination are sufficient to assess the cause and severity of the condition and to decide upon a plan of management. Barium studies will assist in the evaluation of rectal anatomy and function and the site and cause of rectovaginal fistula. Examination under anaesthesia may be necessary to evaluate fistulas fully.
In difficult cases electromyography, anorectal manometry and defaecography can assist in diagnosis and management. Electromyography or endoanal ultrasound may be necessary to precisely localise the site of damaged sphincter muscles before repair and sometimes MRI scanning can be useful.
Treatment plan
Minor degrees of idiopathic incontinence can usually be treated conservatively. A biofeedback approach linking defaecation in the morning with breakfast may be helpful. Bulking agents and suppositories can assist the aims of treatment, which are to encourage defaecation at the normal time and to avoid the prolonged straining at stool associated with perineal or anterior rectal descent. For more severe disease — especially with neurogenic incontinence — low residue diet, codeine and daily enemas with a plug to retain the irrigating fluid may be sufficient to control symptoms. Physiotherapists specifically trained in incontinence treatment should be involved in management, especially before any surgical intervention is contemplated. Electrical stimulation through an anal plug may assist in control; more severe cases usually require colostomy. For severe idiopathic incontinence with loss of the normal anorectal angle, operation is required, with a posterior anal repair of the external sphincter (Parks’ repair). Direct repair of the area of past injury to the external sphincter can successfully control traumatic incontinence. Scar tissue is preserved to preserve blood and nerve supply. The scar is folded and sutured by an overlapping technique. Sacral nerve stimulation is a technique that is used in some specialist centres for the management of difficult cases.
Before repair of traumatic rectovaginal fistulas, bowel preparation is necessary. The procedure is not performed until the inflammatory oedema has settled. In some fistulas, especially those following radiation, a diverting ileostomy should be performed some months before the repair. In patients with Crohn’s fistula, direct repair usually fails and severe symptoms usually lead to proctocolectomy and ileostomy. Ileostomy and laser photocoagulation to a malignancy can provide the best palliation for fistula secondary to cervical carcinoma.
7.26 Pneumaturia
Pneumaturia is an uncommon symptom that is virtually diagnostic of colovesical fistula. Occasionally, intractable urinary tract infection is the presenting symptom of such a fistula.
Common causes
1. Colonic diverticulitis
2. Carcinoma of colon
About 50% of fistulas are due to diverticulitis (Fig 7.75), but this complication occurs in less than 5% of all patients with this condition. Carcinoma of the colon is the cause in about 30% of cases. Other causes include bladder cancer, Crohn’s disease, radiation injury and trauma.

Figure 7.75 Vesico-colic fistula
The fistula is usually short, arising from a sigmoid peridiverticular abscess which has discharged into the bladder.
History and examination
Apart from the pneumaturia these patients may be asymptomatic and in good general health, the fistula of diverticulitis or carcinoma being seldom sudden in onset or associated with significant abdominal symptoms. Direct questioning may reveal mild changes in bowel function.
No abnormality is found on physical examination abdominally or rectally in most cases.
Diagnosis
Leucocytosis is uncommon. Urinalysis usually reveals infection and faecaluria. Plain X-ray may show air in the bladder. Contrast enema often as part of pelvic CT scanning demonstrates the fistula in less than 10% of cases but a cancer, Crohn’s disease and diverticular disease will be demonstrated if present. Cystoscopy often shows a localised point of oedema and reddening at the posterior–superior aspect of the bladder, but the fistula is usually not seen. Cystography and methylene blue instillation are also not usually diagnostic. In most cases the diagnosis is made upon the history and findings on barium enema.
Treatment plan
At operation severe acute diverticulitis and peritonitis is an exceptional finding. In most cases the fistula is short and the sigmoid colon is closely and densely attached to the dome of the bladder. There may or may not be an abscess in the region of the fistula. In some patients with Crohn’s disease a long fistulous tract may be present. The colon can usually be easily detached from the bladder by blunt dissection. With cancer, a disc of bladder wall should be excised in continuity with the colon and the defect re-sutured. Urinary catheter drainage is provided for seven to 10 days after surgery. The colonic lesion is best managed by resection and primary anastomosis. A covering ileostomy is indicated when there is technical difficulty because of obesity, when there is a significant local abscess and in patients with concurrent medical illness. Hartmann’s procedure should be avoided if possible, because of the difficulty of secondary surgical anastomosis between colon and rectal stump.
7.27 Perianal fistula and sinus
Perianal fistula or sinus presents with persistent seropurulent discharge from a perianal nodule of granulation tissue, which extrudes pus on pressure. Careful probing may be necessary to distinguish between sinus and fistula. In contrast to incontinence (Ch 7.25), most patients with anal fistula have control of defaecation and the discharge is not faeculent. Certain types of fistula can be associated with faeculent discharge and varying degrees of incontinence. Such severe fistulas include high fistulas extending above the anorectal ring, Crohn’s perianal fistulas and the complex and multiple perianal fistulas seen sometimes in alcoholics, where urgency and diarrhoea may also contribute to incontinence.
A serous generalised discharge is characteristic of the weeping dermatitis of anal pruritus. Mucus discharge, together with prolapse, is typical of advanced haemorrhoids. Such discharges are easily distinguished from fistula (Fig 7.76).

Figure 7.76 Perianal discharge observed in left lateral position
1: pruritus ani with exudation; 2: haemorrhoids; 3: perianal fistula; 4: furunculitis with chronic sinus formation
Causes
1. Perianal fistula
2. Less common causes
History and examination
1. Perianal fistula
Perianal fistula produces a persistent or intermittent seropurulent discharge and a previous history of perianal abscess is common (Fig 7.77).

Figure 7.77 Perianal abscess leading to perianal fistula
1: anal gland; 2: anal gland abscess; 3a,b: fistulous tract left after drainage; 4: established fistula
Pain is not a feature of an established fistula. On examination, the fistulous opening will be seen as a small pouting area of granulation tissue exuding pus on pressure. Probing usually reveals that when the external opening lies anterior to the anal ring the tract is direct but, when the external opening is posterior the tract is curved, opening internally in the posterior midline (Goodsall’s rule, Figure 7.78).

Figure 7.78 Goodsall’s rule
A: anterior fistulas open directly ito an adjacent end crypt; B: posterior fistulas usually take a curved path to open into a midline posterior crypt; C: a horseshoe fistula may also have an anterior component
Anal fistulas are classified according to their anatomical location: as anorectal, transsphincteric, intersphincteric and subcutaneous (Fig 7.79). A high anorectal fistula is often the result of careless probing under general anaesthesia, with formation of a false passage, and may present subsequently with a faeculent discharge. An indolent oedematous appearance, with multiple fistulas, suggests Crohn’s disease. In an occasional patient with Crohn’s disease, the perianal fistula or abscess is the form of presentation of the condition. In these patients investigation of the colon with radiological or colonoscopic studies is essential and biopsy of the tract may establish the diagnosis. Rectal biopsy may also reveal diagnostic granulomas, even when the mucosa is macroscopically normal.

Figure 7.79 Anal fistulas
1: anorectal (supraphincteric); 2: intermuscular; 3: high transsphincteric; 4: low transsphincteric; 5: subcutaneous (superficial), with anal fissure
2. Less common causes
Chronic folliculitis (furunculosis) or infected sebaceous cysts can resemble perianal fistula until probing (and sometimes contrast sinugram) establishes that the lesion is a sinus: a blind-ending tract, with no internal communication. Examination under anaesthesia may be necessary to be sure there is no internal opening.
Diagnostic plan
Diagnosis is established by probing and occasionally fistulography, under general anaesthesia if necessary. Biopsy may be necessary when chronic specific infection or malignancy is suspected. Sigmoidoscopy is indicated in all cases and colonoscopy whenever a possibility of associated colonic disease exists. Barium enema is the best method of delineating colovaginal fistula.
Treatment plan
The majority of patients with sinuses and fistulas have chronic faecal stasis. Normal defaecatory habits should be restored with the aid of a high-fibre diet. Diabetes must be excluded in all cases, especially in patients with furunculosis.
1. Perianal fistula
The treatment of the common low perianal fistula is either by simply laying open the fistula or by fistulectomy. The tract is delineated with a probe passing from the skin to the internal opening in the anal crypt (Fig 7.80). High anorectal fistulas above the sphincters should not be laid open in one stage, for this will result in incontinence. In these instances, abscesses are first drained. Subsequently a seton may be passed and tied loosely with the aim of producing fibrosis and obliteration of the tract. Tissue glue may be used to obliterate the fistula tract. Alternatively, the internal opening may be covered by a mucosal advancement flap, leaving the external opening to heal by secondary intention.

Figure 7.80 Probing a fistula at operation
A, B: the tract is gently probed to find the internal opening; C: the fistula is laid open by deroofing over the probe
The principles of treatment of ordinary fistulas are: the primary (internal) opening must be found and excised; all tracts must be identified; and the tract must be unroofed or excised through its whole length. A wound must be created so that drainage is adequate and dressings applied so that there is healing from the base to the surface. Recurrence is likely when the primary opening in the anal crypt is missed or a false passage created higher in the anorectum, collateral tracts are not laid open, the operation is tentative and inadequate because of the fear of incontinence or careless dressing of the wound fails to ensure healing from the base of the tract to the skin surface. Sometimes careless or injudicious fistulectomy can lead to damage of the sphincter sufficient to produce incontinence or recurrence in a more severe form, such as high anorectal fistula. This in turn increases the danger of incontinence from treatment. In high fistulas (i.e. those extending above the sphincters), a seton is often used by the colorectal surgeon to treat this variant and minimise the possibility of causing permanent sphincter injury and subsequent incontinence.
Fistulectomy should not be performed in the presence of Crohn’s disease. Treatment of patients with Crohn’s is confined to control of sepsis by adequate drainage of abscesses.
2. Less common causes
Infected sebaceous cysts and furunculosis can produce a somewhat similar picture to fistula, with a history of recurrent infection and discharge. These conditions require drainage. Excision of the residual fibrotic sebaceous cyst is necessary later during a quiescent phase. Hexachlorophene soap is used in furunculosis to control overgrowth of skin bacteria. Astringent antiperspirants and deodorants must be avoided and nylon underwear should not be worn.
Other less common causes of sinuses and fistulas include infected Bartholin’s cyst (a gland normally secreting into the posterolateral vagina), which may sometimes discharge in the region of the anus. Subcutaneous lodgement of a foreign body may cause a persistent sinus. The common types of foreign body sinus follow rectal injury from fish bones, sclerosant injection of haemorrhoids and external penetrating injuries especially with wooden objects. Hydradenitis suppurativa is a disease of the apocrine sweat glands, which is characterised by multiple deep perianal sinuses. Pilonidal sinus may occasionally track towards the perianal area and thus resemble perianal fistula. The direction of the tract on probing, together with the presence of a hair-containing cavity over the sacrum, confirms the diagnosis. Early carcinoma of the anus can present as perianal discharge.