Theodore H. Schwartz and Vijay K. Anand
INTRODUCTION
The transsphenoidal approach to the ventral midline skull base was first proposed more than a century ago. However, only recently, following the introduction and development of rigid endoscopes, appropriate microbayonetted instrumentation, and the development of the extended transsphenoidal approaches has the transsphenoidal approach become an accepted standard method of reaching the suprasellar cistern and adjacent intracranial spaces. Several other key developments have led to the widespread acceptance of extended transsphenoidal approaches. The use of neuronavigation has been critical to allow the surgeon real-time updated anatomic information about the location of key anatomic structures. Likewise, the development of techniques to close large openings in the midline skull base endonasally, such as the gasket seal, the button, and the nasoseptal flap, has been critical in keeping the complication rate acceptably low.
Several pioneering groups have published anatomical cadaver studies, case series, and conceptual articles illustrating the possibilities with a purely endonasal endoscopic approach to remove a variety of lesions throughout the cranial base, third ventricle, and subarachnoid cisterns. The endoscopic approaches were heralded for being minimally invasive and enabling the surgeon to approach ventrally located lesions from a ventral trajectory. These approaches avoided the inherent risks of transcranial surgery mainly related to brain retraction and manipulation of neurovascular structures. The endoscopic endonasal transplanum and transtuberculum approach to the suprasellar cistern is the most direct route to the region that avoids manipulation of the optic nerves and carotid artery. The key to the success of this “minimal access” but maximally aggressive approach is in careful case selection. If appropriate cases are selected, then outcomes can be excellent. Likewise, the appreciation, anticipation, and understanding of possible complications and pitfalls ensure the safety of the procedure. We will try to outline these critical factors in this chapter.
HISTORY
A careful history must be performed. Key points to cover include the possibility of visual loss or compromise indicating compression of the chiasm. In addition, limited mobility of the eyes and diplopia may indicate cavernous sinus involvement. Numbness or pain in the face in V1, V2, or V3 distribution may be a key indicator of fifth nerve involvement. Endocrine function should be evaluated as well such as polyuria, polydipsia, enlargement of the hands or feet, temperature intolerance, weight gain and weight loss, or short stature. Headaches can also be important for certain suprasellar lesions such as the Rathke cleft cysts and may indicate pressure within the sella or even early hydrocephalus if the headaches are worse in the morning.
PHYSICAL EXAMINATION
A detailed physical examination should include an examination of the cranial nerves, ophthalmologic evaluation along with visual field testing, assessment of cognitive function, and endoscopic examination of the nasal cavity.
INDICATIONS
· Pituitary adenomas with a significant suprasellar extension. Tumors that extend above the planum sphenoidale (PS) more than 1 cm or have any anterior extension over the planum and in front of the tuberculum may benefit from removal of the tuberculum sellae and a part of the PS. This will allow an extracapsular dissection of the tumor and ensure a complete resection.
· Craniopharyngiomas arise from the stalk and generally lie retrochiasmal in the third ventricle. Some craniopharyngiomas are sellar, and others can extend laterally. For tumors located in the midline and which do not have cysts lateral to the carotid arteries, the extended transplanum approach can afford an excellent corridor for their removal. Often, very little of the planum needs to be removed. The approach will be between the optic chiasm and the normal pituitary gland.
· Rathke cleft cysts also arise from the stalk and are located above the sella. Although some will descend into the sella, others will remain above.
· Chordomas arise in the clivus but may have significant suprasellar extension, which may require a transplanum approach to supplement the transclival approach.
· Meningiomas of the PS and tuberculum sellae and even some small meningiomas of the olfactory groove are amenable to endoscopic, endonasal transplanum resection. It is important to choose these cases carefully. Tumors that extend lateral to the carotid artery may not be suitable.
· Large tumors that cannot be completely resected using an endonasal approach are not always a contraindication. Depending on the age of the patient and the surgical goals, internal decompression or staged resection with an additional transcranial or cranioendoscopic approach may be appropriate.
CONTRAINDICATIONS
· Careful case selection is critical to the success of this minimal access approach. Pathology that extends laterally over the orbits or lateral to or behind the carotid arteries is difficult to remove using even extended endonasal transplanum approaches.
· Invasion of the cavernous sinus is not an absolute contraindication but requires careful preoperative evaluation of surgical goals. The surgeon may elect to enter the cavernous sinus to resect the tumor using a medial approach, understanding the additional risks to the neurovascular contents. Alternatively, the surgeon may opt for an intentional subtotal resection with planned stereotactic radiotherapy postoperatively, depending upon the pathology. Availability of an interventional neuroradiologist is critical in preparation for surgery around the carotid artery.
· Vascular encasement of the A2 branches of the anterior cerebral artery is not an absolute contraindication depending on the goals of the surgery. If microdissection can be performed to remove the tumor from the A2 branches, then surgery can proceed. Leaving residual tumor adherent to vessels may be appropriate.
· Edema within the brain or floor of the third ventricle is not an absolute contraindication. Careful dissection of the posterior margin of the tumor, which may invade the brain, may be required. Likewise, leaving tumor invading into the hypothalamus (craniopharyngioma) may be appropriate to preserve function.
· The differential diagnosis of large macroadenomas may include suprasellar lesions such as hypothalamic hamartomas, large intracranial aneurysms, and germ cell tumors, which may not be suitable for this approach. Such lesions require a very different workup and approach, and meticulous evaluation should be undertaken in appropriate patients to rule out such lesions preoperatively.
PREOPERATIVE PLANNING
· Patients with any visual symptoms or compression of the optic apparatus are referred to a neuro-ophthalmologist for complete neuro-ophthalmologic examination with visual field testing. Comprehensive endocrinologic testing is often indicated to establish baseline hormone production capability and deficiencies. An endocrinology referral may be indicated to establish a relationship for postoperative follow-up and to determine whether an adequate stress response can be produced by the pituitary gland or whether cortisol replacement will be likely.
· Careful examination of the preoperative imaging is essential. Both CT and MRI can provide information. The location of the sphenoid sinus septations and their relative position with respect to the midline and the carotid arteries should be determined. The pneumatization of the sphenoid sinus should be understood, and the expected bony landmarks such as the carotid and optic protuberances can often be established from the preoperative imaging studies. The extent of the opening such as the amount of the ethmoid sinuses and planum that will have to be removed to adequately expose the entire base of the tumor should be determined. Septal perforations and spurs should be identified to establish whether a nasoseptal flap can be raised and from which sides of the septum or whether another type of flap will be required for closure. Invasion of the brain and vascular encasement can be determined from MRI scans.
SURGICAL TECHNIQUE
· Positioning and preparation (Fig. 13.1): Under general anesthesia with the patient in the supine position, antibiotics (2 g Ancef or 1 g vancomycin) and steroids (10 mg dexamethasone) are given. Steroids are held in the case of Cushing's disease. For some large extended intracranial cases, we administer triple antibiotics, for example, vancomycin, a second-generation cephalosporin, and Flagyl. After placement of a urinary catheter and arterial line, a lumbar puncture is typically performed during which 10 mL of cerebrospinal fluid (CSF) is removed sterilely, mixed with 0.2 mL of 10% fluorescein, and reinjected into the CSF. We find the use of intrathecal fluorescein useful in identifying CSF leaks, although this practice is optional and we pretreat with antihistamines prior to administration. In cases of extended skull base approaches for large lesions, a lumbar drain may be left in place to drain CSF postoperatively in order to prevent a CSF leak. For the limited endoscopic transsphenoidal approach, postoperative lumbar drainage is not routine. Cottonoids with 4 mL of 4% cocaine are placed in the nares to vasoconstrict the nasal mucosa. The patient is placed in a Mayfield head holder and pins, with the head elevated above the heart, slightly extended (approximately 10 to 15 degrees), and turned toward the patient's right (approximately 5 degrees) (Fig. 13.2). The Mayfield head holder is used in order to maintain accurate neuronavigation during the case and to steady the head for microdissection in extended skull base procedures. Alternatively, the head may be placed on a horseshoe with a headset or tripod-fixed reference marker for neuronavigation.
· Neuronavigation: Neuronavigation is not required but is highly recommended in endoscopic endonasal surgery. Navigation provides real-time information about the angle of approach through the endonasal corridor and allows the surgeon to tailor the approach to maximize visualization of pathology while minimizing required exposure and manipulation of vital neurovascular structures.
· For extended approaches in which a large skull base defect is anticipated, we prepare the thigh to harvest fascia lata. If only adipose tissue is required, such as for repairing a CSF leak after resection of an intrasellar lesion, the abdomen is prepared. We also prepare for harvesting a nasoseptal flap if a large skull base defect is anticipated.

FIGURE 13.1 OR setup. Operating room organization. Ergonomic setup of the equipment is essential as endoscopic procedures require nondirect tools for visualization (i.e., viewing screens and neuronavigation). The patient's head is positioned in the center of the operating room, angled slightly away from anesthesia. We use two projection screens to allow direct visualization by both the main operator and the assistant who stand on either side of the bed from each other. This is essential for extended endoscopic cases to avoid the need for either operator to continuously turn their head to see the projection screen. For a right-handed surgeon, the primary surgeon usually stands on the patient's right side. The navigation system is placed at the head of the bed so both surgeons can view the screen simultaneously.

FIGURE 13.2 OR setup. Photograph of the actual operating room setup taken from the foot of the bed. Note the patient's head is tilted slightly so the vertex is pointed away from the operating surgeon on the patient's right side.
Under 0-degree endoscopic view, the inferior, middle, and superior turbinates as well as the sphenoid ostia are identified bilaterally. The middle and superior turbinates are most often retracted laterally; however, in some patients, the middle turbinate is removed to optimize the exposure. A nasoseptal flap is elevated before the posterior septectomy is performed and covers an area extending from the floor of the nasal cavity under the middle turbinate superiorly to the septum adjacent to the nasal roof (Fig. 13.3). The flap is pedicled on the posterior nasal septal artery and is placed out of the way in the nasopharynx until it is needed for the closure.




FIGURE 13.3 Harvesting the nasoseptal flap. A. The first incision is made from the choana reaching anteriorly just above the palate along the septum. B. A second incision is made superiorly a few millimeters below the cribriform plate and connected with the inferior incision. C. The flap is mobilized, the pedicle is preserved, and the flap is (D) stored in the nasopharynx until needed.
The ostium of the sphenoid sinus is then enlarged to expose the sphenoid sinus, and the posterior third of the nasal septum adjacent to the vomer and maxillary crest is resected with a tissue shaver. The sphenoid sinus rostrum is fully exposed, and the floor and lateral wall of the sphenoid sinus are drilled down to facilitate placement of the nasoseptal flap at the end of the operation. The keel of the sphenoid is completely removed. All sphenoid septa are removed with a drill, and the mucosa of the sphenoid sinus is completely removed. Bleeding is stopped with either warm saline irrigation or Gelfoam. At this point, a 0-degree, 30-cm rigid 4-mm endoscope is introduced through the left nostril and held in place with a flexible scope holder. The carotid protuberance, optic protuberance, and medial and lateral opticocarotid recesses (LOCRs) are identified. The anterior and lateral extent of the sphenoidotomy are verified using intraoperative neuronavigation ensuring that optimal exposure is obtained in all dimensions before proceeding with progressively deeper exposure. Bilateral posterior ethmoidectomies are performed to adequately visualize the most anterior portion of the PS.
The bone over the tuberculum sellae is initially thinned with a high-speed diamond drill under constant irrigation. The opening extends between the medial opticocarotid recesses (MOCRs), and the inferior extent of the exposure can extend down to the clival recess. The opening is extended anteriorly above the level of the diaphragma sellae, and the PS is removed, the anterior extent of which is determined by using image guidance (Fig. 13.4). If necessary, the bone overlying the medial opticocarotid recess is then drilled using a diamond drill and copious irrigation to thin and remove the bone overlying the optic nerve dural sheaths. The optic nerve dura is unroofed bilaterally, and the bone overlying the carotid prominences is thinned with a drill and removed with Kerrison rongeurs (Fig. 13.4). The location of the carotid arteries is verified using a Doppler ultrasound probe. The dura above and below the superior intercavernous sinus is opened, and the sinus is coagulated and cut just medial to the cavernous sinus bilaterally. The diaphragma sellae is then incised and removed with microscissors. This maneuver is performed during the approach for craniopharyngiomas and macroadenomas. For tuberculum meningiomas, the diaphragm, which is often the site of origin of the tumor, is removed at the end of the operation to ensure a complete resection.

FIGURE 13.4 Bone opening. The bone at the superior aspect of the sella is opened between the MOCR. The tuberculum sellae (TS) and PS are opened with a high-speed drill and Kerrison rongeur depending on the extent of the tumor as determined by navigation and examination of the films. The OCs are opened bilaterally if there is suspicion that the tumor (usually meningiomas) invades into the canal. (ICA, internal carotid artery; LOCR, lateral opticocarotid recess; medial opticocarotid recess; OC, optic canal; PS, planum sphenoidale.)
Large pituitary tumors, meningiomas, and craniopharyngiomas are immediately visualized once the dura is opened. Internal decompression is performed either with two upwardly curved suctions or, if the tumor is firm, with an ultrasonic surgical aspirator, radiofrequency monopolar or ring cautery, or miniaturized microdebrider device as well as with microscissors. Visualization is enhanced with a 30-degree, 30-cm rigid 4-mm endoscope (Karl Storz). Once decompressed, the tumor capsule can be mobilized, and the anterior communicating artery complex and perforators are dissected sharply off of the tumor capsule. Care must be taken to preserve the recurrent artery of Heubner and the subchiasmatic perforating and hypophyseal vessels. The optic nerves and pituitary stalk are identified and dissected off the tumor capsule with preservation of the arachnoid membrane when possible. Avoidance of coagulation and “pulling” are critical to the preservation of vital neurovascular structures while the remaining capsule is removed. The resection bed is examined with a 45-degree, 18-cm rigid 4-mm endoscope to ensure the absence of any residual tumor. Curved suctions, angled micropituitary rongeurs, and dissectors can be used to reach residual pieces of tumor.
Tumor extensions behind the chiasm that extend into the third ventricle are dissected with the view provided by a 0-degree, 30-cm rigid 4-mm endoscope. The solid components are carefully dissected free from the optic chiasm and stalk. Internal decompression facilitates mobilization of the capsule for sharp dissection from the walls of the hypothalamus. It is important to visualize and preserve critical neurovascular structures such as the carotid arteries and hypothalamic and chiasmatic perforators. Once the tumor is partially removed, the location of the stalk may become apparent if it was previously obscured by tumor, and one can safely work on either side of the stalk if the gland is not mobilized. If invasion of the walls of the third ventricle is noted, it may be prudent to leave some wall of the capsule to prevent hypothalamic injury. Once the tumor and cyst wall are completely removed, the resection bed is examined with a 45-degree, 18-cm rigid 4-mm endoscope to ensure the absence of residual tumor. In most cases, the third ventricle ependyma including the foramen of Monro and aqueduct of Sylvius is clearly seen, and contents of the interpeduncular cistern are also apparent (Figs. 13.5 to 13.8).







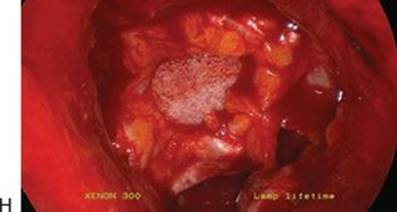
FIGURE 13.5 Surgery to remove a solid craniopharyngioma. A. The preoperative sagittal MRI scan with contrast shows an enhancing mass superior to the pituitary gland. B. After removing the bone and opening the dura, the mass is dissected free circumferentially from surrounding vasculature such as the superior hypophyseal artery. C. The tumor is attached to the bottom of the chiasm. D. Small remnant of tumor seen below chiasm in front of stalk. The basilar artery and posterior cerebral branches can be seen in the distance. E. The tumor has been removed from the undersurface of the chiasm. F. Close-up view of the stalk to the left with the basilar artery, posterior cerebral arteries, superior cerebellar arteries, and third nerves emerging between the two. The mammillary bodies can be seen just above the basilar apex. G.The defect in the skull base with the chiasm behind it. H. Gasket-seal closure of the skull base defect. Note the Medpor buttressing the fascia lata graft.








FIGURE 13.6 Surgery to remove a suprasellar epidermoid. A. The preoperative sagittal MRI scan with contrast shows a mass above the pituitary gland. B. Transtuberculum, transplanum bone opening exposes the intact dura between the optic nerve canals. C. The dura is opened, and edges are cauterized. The green fluorescein–stained CSF is visualized as well as the white tumor. D. The pituitary stalk pushed laterally is seen below the optic nerve with adjacent small bits of tumor remnant. E. Tumor remnants medial to optic nerve. The adjacent carotid artery and A1 segments are easily seen. F. A Comm and both A2 branches are seen emerging from above the chiasm. G. The interhemispheric fissure and adjacent optic nerve. H. Gasket-seal closure of skull base.




FIGURE 13.7 Surgery to remove a suprasellar meningioma. A. The preoperative sagittal MRI scan with contrast shows an enhancing mass above the pituitary gland extending along the PS. B. Preoperative coronal MRI with contrast demonstrates the lateral and superior extent of the tumor. C. After removal of the tumor and opening of both OCs within the sphenoid sinus, one can see the optic nerves and chiasm free of tumor. Above the optic chiasm are the lamina terminalis and bilateral A1 branches, A Comm, and bilateral A2 branches. D. When the OCs are opened, lateral slits must be cut in the Medpor before placing the gasket so as not to compress the nerves.




FIGURE 13.8 Surgery to remove a suprasellar solid and cystic giant craniopharyngioma. A. The preoperative sagittal MRI scan with contrast shows an enhancing mass above the pituitary gland extending along the PS and down the clivus in front of the pons. B. The view up into the third ventricle shows the tela choroidea above with residual tumor adherent to the walls of the third ventricle and hypothalamus. C. View up into the roof of the third ventricle shows the foramina of Monro, fornices, and choroid plexus with residual tumor attached to walls of third ventricle. D. Looking through the foramina of Monro into the lateral ventricles from below. The choroid plexus and fornices are more clearly seen.
Multilayered Skull Base Closure
The technique for reconstruction of the skull base starts with an inlay of an adipose tissue graft to fill any dead space and decrease the pooling of CSF. The next step is to create a “gasket-seal” closure. A piece of fascia lata, cut to be larger than the defect by about 1 cm circumferentially, is placed as an onlay, and then, a piece of Medpor, cut to be the same size as the defect, is countersunk into the fascia lata to hold it in place as a rigid buttress. The fascia lata should stick out circumferentially beyond the Medpor. This is then covered with a vascularized nasoseptal flap directly over the gasket-seal construct followed by a final layer of Duraseal to hold everything in place and ensure a watertight closure. No Duraseal is placed between the flap and the gasket-seal construct since it would prevent fibrosis and vascularization of the skull base. If a large sellar opening is performed as would be done to resect a giant macroadenoma, we do not use the gasket-seal technique since the curvature from the bottom of the sella to the front of the planum does not lend itself to a rigid buttress inlay. In this situation, we place adipose tissue in the sella, buttress it with Medpor, and cover it with a nasoseptal flap and Duraseal (Fig. 13.9). We then fill the nasal cavity with Floseal (Baxter, Deerfield, IL) for hemostasis. A small piece of Telfa is finally placed in each nostril overnight to absorb any drainage and is removed in 1 to 2 days. If a lumbar drain is placed intraoperatively, it is typically drained at approximately 5 mL per hour for 24 hours and then clamped and removed in the evening so the patient will lie flat after its removal during the night as they sleep to decrease the risk of spinal headache.
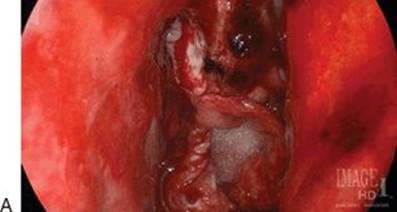


FIGURE 13.9 Placement of the nasoseptal flap. A. The gasket-seal closure in place. B. The nasoseptal flap is placed over the gasket seal. C. Duraseal is used to cover the flap and keep it in place.
POSTOPERATIVE MANAGEMENT
· Patients are closely monitored in the ICU for the first 24 hours.
· Disorders of water balance and anterior pituitary endocrinopathies are by far the most common perioperative events, and their management is the most important aspect of the perioperative care of patients undergoing endonasal, endoscopic transsphenoidal neurosurgery.
· Urine output is monitored, and output greater than 250 mL per hour for 2 consecutive hours with specific gravity less than 1.005 should raise suspicion for diabetes insipidus (DI) and may require treatment with supplemental DDAVP.
· Elevated serum sodium indicates a free water deficit and requires fluid replacement, particularly in patients who are not able to drink to keep up with their fluid needs.
· DVT prophylaxis is important for patients who are immobile.
· Severe headache or mental status change should prompt a CT scan to rule out pneumocephalus.
· Salty taste in the back of the mouth or clear fluid running out of nose when the patient is sitting forward may indicate a CSF leak that requires treatment.
· Spiking fever and neck stiffness may indicate meningitis, which should be diagnosed with a lumbar puncture.
· Fasting AM cortisol off steroids prior to discharge will indicate the need for long-term steroid replacement.
COMPLICATIONS
· Complications can be divided into endocrine (hypopituitarism, DI), vascular (stroke, hemorrhage), CSF leak, infectious (meningitis, abscess), neurologic (visual loss, hemiparesis), nasal (crusting, sinusitis), and medical (DVT, PE, pneumonia, MI).
· Meticulous surgical dissection to preserve the pituitary stalk and supporting vasculature is important to prevent postoperative DI.
· Careful study of neurovascular anatomy on preoperative imaging studies is imperative to prevent intraoperative catastrophic neurovascular injuries.
· Characteristics of the defect, type of tumor pathology, and volume of CSF leak influence the type of skull base reconstruction
· Intrathecal fluorescein can identify subtle CSF leaks and help improve skull base reconstruction, thereby preventing postoperative morbidity.
· Lumbar drainage, bed rest, and stool softeners can successfully treat the majority of postoperative small CSF leaks in the absence of pneumocephalus, assuming a meticulous multilayer closure of the skull base has been performed.
· Infectious complications are rare in endoscopic skull base surgery in the absence of a postoperative CSF leak. These may be reduced with 24 to 48 hours of antibiotics delivered in the perioperative period.
· Mucosal sparing surgery followed by routine and frequent postoperative care (topical nasal therapy, postoperative debridement) can significantly decrease sinonasal complications after endoscopic pituitary and skull base surgery.
RESULTS
The field of endoscopic endonasal skull base surgery has rapidly evolved because of technologic innovations and the development of operative techniques by several innovative groups to reach almost the entire ventral skull base. The endonasal transplanum transtuberculum approach extends the exposure beyond the sella by removing the tuberculum sellae and a portion of the PS to enable surgical access to the suprasellar cistern. As with any new approach, there is a learning curve that must be surmounted to achieve optimal results, and continuing evaluation of new ideas and technology must occur at periodic intervals as these results evolve. In this retrospective series, we have demonstrated, as others have previously, that suprasellar tumors can be safely and completely removed using the endoscopic transplanum/transtuberculum approach. In our series of patients, we have reported low overall complication rates, the evolution of a negligible CSF leak rate (<5%), improved neurologic outcomes, a high extent of resection, and minimal trauma to the surrounding brain parenchyma. The low complication rates represent a substantial improvement compared to the high rates of CSF leaks that were reported early on in the endoscopic skull base experience for suprasellar tumors and justify an important role for the endonasal, endoscopic approach in the management of these tumors.
The direct approach to ventrally located tumors afforded by endoscopic techniques overcomes the difficulties and blind spots of transcranial skull base approaches that expose the tumor from an anterolateral to medial trajectory and inevitably require brain retraction, manipulation of a compressed optic apparatus, and carotid artery dissection to gain access to a tumor. In addition, the endonasal approach is excellent for visualizing and removing tumor extending into the medial optic canal (OC) such as commonly occurs with tuberculum sellae meningiomas. Improved ventral exposure, however, is achieved at the expense of surgical access superior and lateral to the optic nerve and carotid artery, thereby substituting one set of exposure-related operative difficulties with another. Ultimately, the predominant tumor growth pattern and suprasellar/parasellar extension in relation to the optic nerves and carotid arteries dictate the optimal approach or combination of approaches. Appropriate patient selection with careful review of preoperative neurovascular imaging is critical to optimize outcome. In general, tumors with significant lateral extension beyond the carotid bifurcation are difficult to access through an endonasal approach.
PEARLS
· When opening the sphenoid ostium, avoid straying inferolaterally, as this may result in injury to the sphenopalatine artery along its course.
· For very large tumors and for extended approaches, the middle turbinate may be removed to provide additional visualization.
· The micro-Doppler should be used to identify the location of the carotid artery prior to opening the dura.
· Carefully examine the preoperative CT and CTA or MRI to understand the relationship between the septations in the sphenoid sinus and the carotid artery. CTA navigation can be useful intraoperatively.
· Use a diamond rather than a cutting burr to remove bone over the dura and carotid artery.
· For extended approaches, it is important not to make the opening in the bone too small. Once you are confident in your ability to close large skull base defects, larger openings in the bone are possible. Navigation is useful to determine the required size of the opening to expose the extent of the tumor.
· Frequent internal decompression and extracapsular dissection permit the surgeon to avoid blindly pulling the tumor into the surgical field.
· For extended transsphenoidal approaches where there is significant risk of CSF leak, more elaborate methods of closure are necessary. We recommend either the “gasket-seal” closure using a fascia lata/vomeric bone or rigid buttress with a vascularized nasoseptal flap. Intradural fat can decrease the dead space but can make postoperative imaging of residual tumor challenging requiring the use of fat saturation imaging.
· Visual identification of the pituitary and stalk is important, as accidental injury to these structures must be avoided. Devascularization of the stalk can cause hypopituitarism, and the superior hypophyseal arteries should be identified and preserved if possible.
· Hemostasis of venous bleeding is most effectively performed with hemostatic agents and gentle pressure.
PITFALLS
· If the bone opening is too small, the surgeon is forced to pull the pathology into the field of view, which risks injury to vessels attached to the posterior aspect of the tumor.
· The venous plexus in the clival dura can be extensive. Slow and meticulous opening with careful hemostasis is imperative to ensure the success of this approach.
· If injury to the carotid artery occurs, the sphenoid sinus should be packed quickly and a Foley catheter inflated to maintain pressure. The patient should be taken intubated for emergent endovascular assessment and treatment.
· If reoperation is undertaken to close a CSF leak, it is useful to identify the source of the leak prior to surgery. This can be done with intrathecal iohexol and a CT or intraoperatively with intrathecal fluorescein.
INSTRUMENTS TO HAVE AVAILABLE
· Endoscopic endonasal surgery is best performed with a set of instruments completely different from those used for standard transcranial microsurgical approaches.
· Whereas bayoneted instruments are necessary in microsurgical techniques to maintain the surgeon's hands outside the view of the microscope, long straight instruments and pistol grips are better for endoscopic approaches. Specially designed bayoneted instruments can be used.
· While monopolar cautery is favored during the approach for mucosal bleeding, as with open transcranial surgery, bipolar coagulation is used on the dura and intracranial structures.
· A tissue shaver or microdebrider is useful for resection of intranasal pathology, whereas intracranial pathology requires more precision such as an ultrasonic aspirator, radiofrequency device, or gentle bimanual suction.
· A micro-Doppler probe is particularly useful in identifying vascular structures.
· A range of endoscopes including 18′ and 30′ scopes with 0-, 30-, and 45-degree lenses should be available. It is also important to ensure that all endoscopic visualization equipment is working prior to the case.
· High-definition cameras and wide screen large displays substantially help the surgeon visualize the normal and abnormal sellar and suprasellar structures.
· A sheath around the scope can be used to irrigate and clean the lens during the operation to minimize the need for repeated removal and introduction of the scope.
· Finally, a scope holder is often useful to maintain a fixed, steady field of view during aspects of the case in which mobile visualization is not required.
SUGGESTED READING
Kassam A, Snyderman CH, Mintz A, et al. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus 2005;19:E3.
de Divitiis E, Cavallo LM, Cappabianca P, et al. Extended endoscopic endonasal transsphenoidal approach for the removal of suprasellar tumors: Part 2. Neurosurgery 2007;60:46–58; discussion 58–49.
Laufer I, Anand VK, Schwartz TH. Endoscopic, endonasal extended transsphenoidal, transplanum transtuberculum approach for resection of suprasellar lesions. J Neurosurg 2007;106:400–406.
Leng LZ, Brown S, Anand VK, et al. “Gasket-seal” watertight closure in minimal-access endoscopic cranial base surgery. Neurosurgery 2008;62:ONSE342–ONSE343; discussion ONSE343.
Schwartz TH, Fraser JF, Brown S, et al. Endoscopic cranial base surgery: classification of operative approaches. Neurosurgery 2008;62(5):991–1002.