Paolo Castelnuovo
INTRODUCTION
Large dural defects are usually the result of a major resection of the dural layer for oncologic purposes. After expanding the endonasal approach, theoretically, the dura of the entire ventral skull base, as far as the upper spine, can be resected. Rarely, these defects can be part of a more complex condition related to skull base and cranial malformations. In these latter cases, their presentation is really variable; the most extreme situation is when brain parenchyma can be seen through the oral cavity. Other cases are less clinically evident, especially given the age of the patients, and can become clinically evident after an episode of meningitis.
An important and critical concept needs to be strongly underlined: It is not the size of the defect that makes the reconstruction complex. Rather, it is the location and the definition of the borders of the defect that make the procedure either easy, complex, or sometime impossible. A huge defect in the anterior cranial fossa extending from orbit to orbit and from the planum sphenoidale to the frontal sinuses can be easier to repair than a much smaller defect of the lateral recess of the sphenoid sinus in which the precise borders are hardly identifiable.
HISTORY
With regard to the majority of postresection cases, we must focus on the history of the underlying pathology, but this aspect is addressed in other chapters of this book. Focusing on the topic of congenital malformations, the problem in defining an informative history is related to the young age of the patients. In obvious malformations involving the head and the cranium, no particular difficulty is present in raising doubts about the presence of skull base malformations. In less apparent cases, in which the patient is otherwise normal, a high degree of suspicion is needed. Most of these patients come to attention after an episode of meningitis; rarely are they found incidentally. This is quite obvious because watery secretions in infants, in other aspects normal, are really a common situation. For this reason, delay in diagnosis is the rule and not the exception.
PHYSICAL EXAMINATION
Most of the large dural defects are a consequence of a wide dural resection during removal of a tumor of the anterior cranial fossa. Given the possibility of compromising the availability of fascia lata, previous surgery to the legs must be evaluated. In the significantly rarer cases related to severe malformations of the skull base, a thorough evaluation of the head and cranium is necessary. In some situations, the malformations are so complex that they must be evaluated in a multidisciplinary manner rather than simply with an endoscopic approach. The proper solution should be offered after a careful examination by a team including a neurosurgeon, a maxillofacial surgeon, and a reconstructive surgeon. Notwithstanding this, whenever possible, the endoscopic approach alone should be chosen for the more minor malformations (Fig. 41.1).
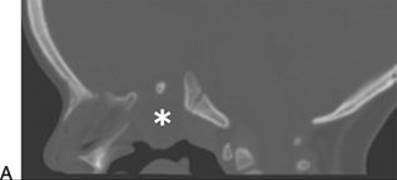




Figure 41.1 The multidisciplinary approach among otolaryngologist, neurosurgeon, maxillofacial surgeons, and reconstructive surgeons allows for proper management of complex cases. A 3-year-old patient with multiple malformations underwent a surgical reconstruction of the cleft lip and palate, combined with the endoscopic endonasal skull base reconstruction of a large meningoencephalocele (asterisk in the figures). A. Preoperative CT scan in sagittal view. B. Preoperative MR scan T2 weighted in sagittal view. C. Preoperative MR scan T1 weighted in coronal view. D. Postoperative MR scan in sagittal view in sequence T2 weighted, showing the skull base reconstruction (white arrow). E. T1 weighted, showing the skull base reconstruction (white arrow).
INDICATIONS
Every case of a dural defect with or without cerebrospinal fluid (CSF) leak must be repaired. Reconstructions strive to recreate a separation of the cranial cavity from the sinonasal cavity, to prevent CSF leak, pneumocephalus, and intracranial infection. Small defects in the skull base can be reconstructed using different types of free grafts with a high rate of success (>95%) (Castelnuovo, 2001). However, larger dural defects should be reconstructed by means of vascularized flaps. Among these, the nasoseptal flap is the preferred choice (Harvey, 2009; Patel, 2010). Personally, I consider every patient a candidate for a nonvascularized reconstruction, unless bone edges are not present. The absence of these bone edges prevents the proper positioning of the inlay grafts, the first intracranial intradural, and the second intracranial extradural (in a sort of “epidural pocket”), and so the risk of postoperative leakage is significantly increased. In this respect, most of the defects of the anterior cranial fossa are amenable to this reconstruction, while for defects of the middle and posterior cranial fossa, the percentage decreases significantly. Anatomically speaking, when dealing with the anterior cranial fossa, the surgeon can extend the intracranial supraorbital dissection without any risks. The same consideration is not appropriate for the middle and posterior cranial fossa. In these cases, the presence of the optic chiasm/pituitary stalk and the abducens nerve makes intracranial dissection very dangerous and not advisable.
CONTRAINDICATIONS
Given the necessity to have bone edges to support the inlay graft (second layer), their absence represents the most important contraindication to a standard multilayer technique with nonvascularized grafts. With regard to the anterior cranial fossa, almost every case can be managed with the nonvascularized graft technique. In this respect, surgeons need to be aware that the roof of the orbit is still present, even in complex cases of congenital malformations or in difficult revision cases, so that it can be used to support the graft. The same considerations are not always true in the middle and posterior cranial fossa, where, after wide resection of bone and dura, really limited edges of bone and dura are present. In some of these cases, we perform a sort of “gasket seal” technique (Lenz, 2008) usually using some type of elastic material such as septal or conchal cartilage to fix the connective tissue inside the dural defect. Furthermore, I am personally convinced that most cases of high-flow CSF leaks can be well managed with this technique if the inlay graft or the gasket seal technique is well conducted. Nevertheless, in some selected cases, especially when dealing with posterior cranial fossa and complex pituitary CSF leaks, I prefer to adopt a hybrid technique in which the multilayer technique is associated with a vascularized flap.
PREOPERATIVE PLANNING
Most of the large dural defects are the result of a resection for sinonasal malignancy. In those cases, resection of the dura is tailored to the needs of the patient and so can be extended from orbit to orbit and from frontal sinuses to planum sphenoidale. Thus, in every case I approach, a large dural defect is anticipated and reconstruction is planned. In this respect, especially when intradural work is anticipated, preoperative imaging is critical in order to evaluate the intracranial relationship of the lesions, mostly with respect to blood vessels (Fig. 41.2). Particular sequences of MR, not limited to anterior cranial fossa but extended to evaluate the entire skull base, are of paramount interest in the detection of suspicious areas, especially in cases of malformations (Fig. 41.3).
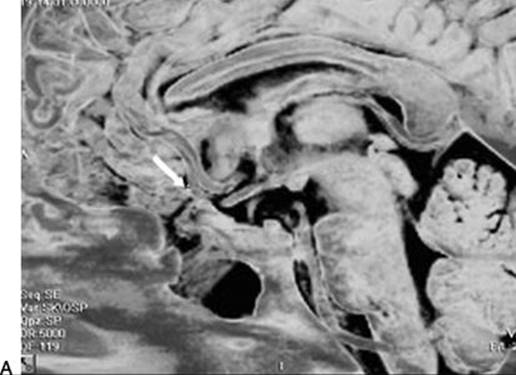

Figure 41.2 Preoperative imaging is critical in order to evaluate the intracranial relationship of the lesions, mostly with respect to blood vessels. A 19-year-old patient previously treated with transcranial surgical approach for posttraumatic pneumocephalus and CSF leak had a history of recurrent meningitis. Before the endoscopic endonasal surgical revision of the skull base reconstruction, an MR scan was performed (A) and angiography (B), which revealed the presence of the medial orbitofrontal artery (MOfA, arrow) inside the scar of the previous surgery, in the same region of the meningoencephalocele. This preoperative finding needs to be carefully evaluated during the surgical procedure, in order to avoid the risk of damaging vascular structures.




Figure 41.3 Radiologic preoperative evaluation study (CT scan and MR scan) of all the three cranial fossae and petrous bones leads us to a precise location of the defect, excluding the possibility of multiple sites of malformation. By comparing MR scan in T2-weighted sequence (A,C) with T2 FLAIR sequence (B,D), it is possible to appreciate a left sphenoid CSF leak (white circle in A and B) together with an additional site of CSF leak in the left mastoid region (white circle in C and D).
In these latter cases, CT scans demonstrate their utility allowing a precise and accurate evaluation of the bone structures of the sino–orbito–cranial interface. The preoperative understanding of the surgical field allows an accurate identification of the possible “supporting” structures for the reconstruction.
Of not minor importance is the identification, given our first choice for connective tissue of the patient, of previous surgery in the areas typically as donor sites. I personally prefer fascia lata and the iliotibial tract, so any kind of previous surgical procedures or trauma to the legs is evaluated. Possible connective tissue pathology is ruled out by means of history.
Materials: Some Considerations
My choice for autologous material is based on the scientific demonstration of good integration of the connective tissue, and on long-time otologic experience concerning repair of the tympanic membrane. Rarely, there is some extrusion of the heterologous material even some years after surgery, while no similar problems with autologous materials have emerged. As a technical hint, I advise the use of adipose tissue in small pieces for the reconstruction for two main reasons. The first is related to the role of dead space fillers, and the second one is due to the regenerative power of the adipose tissue cells.
I discourage the use of a bone graft for the reconstruction after resection of skull base malignancies, given the frequent need for postoperative radiotherapy, thus removing the risk of sequestration. Furthermore, I do not advise, in this case for oncologic reasons, the use of the nasoseptal flap for the reconstruction after resection of cancer because it may be involved in the disease process.
Sometimes, it may be possible to use, as the third layer, a free graft of mucoperiosteum from the floor of the nasal fossae that allows rapid and better healing of the surgical field compared to fascia lata. This difference is related to the biologic behavior of the fascia lata. Its direct exposure to air promotes a necrotic phenomenon of the uncovered surface. This favors the presence of abundant crusts for a long period but does not impact the success of the reconstruction.
SURGICAL TECHNIQUE
The technique used is similar regardless of the type of pathology, and it is based on a standardized surgical checklist. First, and probably most importantly, one must expose the defect perfectly. All the bone boundaries, if still present, should be smoothed as much as possible, and every dead space should be removed. In oncologic cases, before opening the dura, it is critical to create an epidural space that allows the placement of an intracranial extradural layer (Fig. 41.4). If the surgeon opens the dura before this step, then the creation of such spaces is technically much more difficult. Obviously, these considerations are valid only for the anterior cranial fossa where it is possible to dissect intracranially without the risk of creating severe damage to critical structures. In this respect, this step is absolutely avoided when dealing with middle and posterior cranial fossa defects. Once the borders of the defect are adequately exposed, the first layer is placed, intracranially and intradurally, as accurately as possible (Fig. 41.5). Usually, the size of this graft exceeds the size of the defect by 30% (Schick, 2003). After the position of the first layer, I place the second one in the pocket between the bony skull base and the dura, intracranially extradurally. A very important consideration in this step is to verify the absence of vacuum spaces. If present, these need to be filled by autologous materials (pieces of fascia, adipose tissue, muscle) in order to keep the grafts in contact with something other than air (Fig. 41.6). The presence of such defects in between the reconstruction is a factor that predisposes to failure. After the placement of the second layer, I use fibrin glue. Fibrin glue should not be placed on the entire surface of the graft but only on the borders of the reconstruction. Then, once the integrity of the reconstruction has been verified, a third layer is placed as overlay, on any remaining bony border. This is compressed a little bit with Tabotamp or Gelfoam. Care is taken in pressing the graft to the remaining bony borders, if any, or to the orbit. In very rare cases, it is also possible to partially fix the graft with sutures. This maneuver is not done to obtain a watertight closure but to guarantee stability of the reconstruction.

Figure 41.4 Crucial for skull base reconstruction of the anterior cranial fossa seems to be the dissection between the dura and the bony boundaries, before opening the dural layer. This procedure allows for the creation of an epidural space that is very useful for the placement of the intracranial extradural layer (white line, dural layer).

Figure 41.5 Sagittal representation of multilayer skull base reconstruction of anterior cranial fossa. The size of the first layer, placed intradurally, has to exceed the size of the defect by 30% at least. The second layer is placed in the pocket between the bony skull base and the dura (epidural space); the third layer is placed extracranially in overlay fashion (white line, dural layer; green, light blue and purple lines, iliotibial tract grafts in multilayer fashion; yellowareas, adipose tissue).

Figure 41.6 During the skull base reconstruction, all the bony boundaries, if still present, should be smoothed as much as possible and every dead space should be removed. After pressing the layer from the center to the borders to remove residual air, some fragment of autologous material (i.e., adipose tissue, muscle) can be placed along the borders, between the second and third layer (white line, dural layer; green, light blue, and purple lines, iliotibial tract grafts in multilayer fashion; yellow areas, adipose tissue).
In the middle and posterior cranial fossa, where bony structures are frequently not available to support the inlay graft, we use a “gasket seal” technique (Fig. 41.7). A large piece of fascia is placed in the dural defect, and the central part of the fascia is pushed intracranially intradurally by means of an elastic material (usually the quadrangular cartilage, when available, or auricular conchal cartilage). If the relationship between the size of the dural defect and the size of the elastic material is acceptable, this procedure is highly effective. Other fascia grafts are usually placed as overlay in order to reinforce the closure. Spongostan and fibrin glue usually complete the reconstruction.

FIGURE 41.7 Skull base reconstruction technique adopted for the middle or posterior cranial fossa defects reconstruction, called “grandma’s jam cap” (according to the Naples’ group) or “gasket seal” (according to the New York group) (white line, dural layer; purple line, large piece of fascia placed on the dural defect; green layer, cartilage or other elastic material; light blue layer, Hadad flap placed in an overlay fashion).
Small technical differences are present when dealing with patients with congenital malformations. There is the need to use larger grafts because these cases usually occur in children and it is important to think of further development. Of course in these cases, it is of paramount importance, especially due to the unfamiliar anatomy, to follow the critical structures in order to create accurate borders for the reconstruction. The role of the orbit should not be underestimated. As additional advice, to reduce the bulging of the brain parenchyma within the nose, a careful and delicate bipolar coagulation of the mass usually permits its reduction and removal to the level of the skull base.
Regardless of the kind of reconstruction performed, it is important to clean the fascia lata accurately from the detached fibers and adipose tissue once taken from the leg, in order to make it more manageable. If this is not done, during the placement of the grafts, the surgeon can have nasty surprises: The graft can remain attached by means of these fibers to the instruments and removed with it. The iliotibial tract (Fig. 41.8) is thicker and allows easier placement of the grafts during the reconstruction, while fascia lata is thinner and sometimes not as easy to place.


FIGURE 41.8 Fascia lata (FL) (A) and iliotibial tract (ITT) (B) employed as a graft for the skull base reconstruction.
I do not advise the use of any supporting structures for the reconstruction, such as balloons. I put only a limited amount of Oxicell on the borders of the reconstruction in addition to fibrin glue. This demonstrates our philosophy of not producing a counterpressure on the reconstruction, but only acting on the borders of the reconstruction. In this respect, I pack the nose only at the level of the floor of the nasal fossae, thus leaving an empty, ventilated space below the reconstruction. This allows better healing of the surgical field, and it is in contrast with what I did in our first years when I completely filled the nasal cavity, which resulted in delayed recovery of the mucosa.
POSTOPERATIVE MANAGEMENT
Serial careful endoscopic evaluations are done in the early postoperative period in order to check for any signs of CSF leakage. Prophylactic therapy (antibiotics, antihistamines, and laxatives) is given for 7 to 10 days. An early (<24 hours) postoperative CT scan is performed in order to evaluate the presence of pneumocephalus. Nasal packing is usually removed on the 2nd postoperative day under endoscopic vision, and if no sign of CSF leak is visible, the patient is allowed to remain in a sitting position. Major crusts have to be removed, unless underlying tissues are bleeding. In that case, they should be left in place and allowed to heal naturally. It should be understood that the presence of crusts acts in favor of superinfections. Patients are asked not to blow their nose for at least 1 month and not to fly in an airplane until the sinus ostia are well cleared and ventilated. During the follow-up, patients are seen as necessary. For the first few months, patients are asked to avoid excessive physical activity. Usually, within a few months, complete mucosalization of the graft is visible.
COMPLICATIONS
Complications are usually related to the lack of integration of the grafts. CSF leaks are uncommon, occurring in less than 8% of cases. When the reconstruction is “wet,” or in very rare cases of CSF tears, I usually prefer a conservative procedure based mostly on lumbar drainage (for 5 days) and endoscopic reinforcement of the reconstruction. In the case of progressive pneumocephalus, the lumbar drainage must absolutely be avoided, while revision surgery is indicated (Fig. 41.9). If the leak is more evident, an early second return to the operating room is needed in order to reexplore the surgical field. In most of these cases, I did not observe a failure of the whole reconstruction, and the leakage is usually related to a circumscribed defect.






FIGURE 41.9 A patient with a left ethmoidal squamous cell carcinoma T3N0M0, with contrast enhancement T1-weighted MR scan in coronal view (A) and sagittal view (B) who underwent an endoscopic resection with transnasal craniectomy (ERTC). The postoperative CT scan in coronal view (C) and sagittal view (D) reveals a large and progressive pneumocephalus (asterisk in the figures); a strong indication for early revision surgery. The CT scan in coronal view (E) and sagittal view (F) performed 5 days after the endoscopic endonasal revision of the skull base reconstruction (white arrow) confirms that the complication has resolved.
I strongly call for early revision surgery if the amount of the leakage is significant. In this respect, I have reported a subdural hematoma caused by an excessive leakage. In our series, I have had no meningitis in patients with CSF leaks, probably due to the antibiotic therapy. Nevertheless, this is a critical problem, and the presence of CSF leaks should not be underestimated and calls for early intervention.
Rarely, complications can also be related to technical errors or most commonly to incorrect preoperative planning. I have had a vascular injury of the medial fronto-orbital artery during revision surgery that required transcranial management. In that specific case, the position of the frontopolar artery was not correctly evaluated in the preoperative images.
Although in our series no major postoperative bleeding has been described, probably due to accurate management of the nasal vessels, I underline how this complication can represent a significant problem, especially in the early weeks after surgery.
The presence of crusting is not to be considered as a complication, representing the natural way nature heals.
RESULTS
I reported an overall failure rate of about 8% in a series of more than 100 cases of reconstruction of large skull base defects for different conditions, most of these regarding the anterior cranial fossa. However, if we analyze the data according to the time course, it is understood that the rate of CSF leaks in the last 5 years is less than 2%. This is due mostly to the standardization of the technique and to the refinement of the surgical details and secondarily to the learning curve of the surgeons.
By selecting my evaluation of middle and posterior cranial fossa reconstructions, I observed a similar rate of recurrence. This suggests that, if the technique is well applied and the patient selection well conducted, an avascular reconstruction is possible and effective also in these areas (Fig. 41.10).









Figure 41.10 Endoscopic endonasal skull base reconstruction after resection of sinonasal malignancies encroaching on the anterior skull base. Contrast enhancement T1-weighted MR scan in coronal (A) and sagittal (B) view of an intestinal- type adenocarcinoma (ITAC) with intracranial extension (T4bN0M0), marked in the figures with black asterisk, that underwent an endoscopic resection with transnasal craniectomy (ERTC). Postoperative T2-weighted MR scan in coronal (C) and sagittal (D) view, showing the skull base reconstruction (white arrows). 3D-VIBE sequence MR scan in coronal (E) and sagittal (F) view of the left olfactory esthesioneuroblastoma (T4bN0M0), marked in the figures with black asterisk, that underwent an endoscopic resection with transnasal craniectomy (ERTC). Postoperative 3D-VIBE sequence MR scan in coronal (G) and sagittal (H) view, showing the skull base reconstruction (white arrows).
PEARLS
· Whenever possible, autologous materials are preferred due to good biologic integration.
· Creation of an epidural space in order to place the second layer (intracranial extradural). This maneuver is better performed before opening the dura.
· Placement of the grafts as accurately as possible, 30% size larger than the dural defect. Fill all the dead spaces with autologous materials (adipose tissue, muscle, fascia).
· Accurate post-op management with CT scan and serial endoscopic evaluations is critical for good healing.
PITFALLS
· Revision surgery calls for careful examination in order to accurately plan the surgical procedure and identify possible risky points.
· Optimal exposure of the surgical defect with clear identification of the borders and smoothing of the surrounding bone is essential.
INSTRUMENTS TO HAVE AVAILABLE
· Double curved forceps
· Angled positioners (with teeth on the tip)
· Angled instruments with spatula tips are useful in performing extradural pockets
SUGGESTED READING
Castelnuovo P, Mauri S, Locatelli D, et al. Endoscopic repair of cerebrospinal fluid rhinorrhea: learning from our failures. Am J Rhinol 2001;15(5):333–342.
Schick B, Wolf G, Romeike BF, et al. Dural cell culture. A new approach to study duraplasty. Cells Tissues Organs 2003;173(3):129–137.
Lenz LZ, Brown S, Anand V, et al. “Gasket-seal” watertight closure in minimal-access endoscopic cranial base surgery. Neurosurgery 2008;62(5 Suppl 2):ONSE342–ONSE343.
Harvey RJ, Nogueira JF Jr, Schlosser RJ, et al. Closure of large skull base defects after endoscopic transnasal craniotomy. J Neurosurg 2009;111:371–379.
Patel MR, Stadler ME, Snyderman CH, et al. How to choose? Endoscopic skull base reconstructive options and limitations. Skull Base 2010;20:397–404.