Atul Goel
INTRODUCTION
In skull base surgery, the division between neurosurgical territory and otolaryngology territory is essentially determined by the firm natural compartmentalization that separates the “sterile” and “revered” neurocranium, housing the vital and evolved brain, from the more primitive viscerocranium serving the basic and essential functions of respiration and alimentation.
The demarcation of the borders is created by the cranial base bone and a firm and thick meningeal/dural membrane. The dura has to be understood in its anatomical extension, its functional capacity, and, most importantly, its relationship to the pathology in question. In general, tumors respect this membrane. Basal bone can be thinned, eroded, or destroyed, but the basal membranes remain resistant and intact, demonstrating the biologic superiority of membranes over bones. The membrane may be thinned, may balloon out, or may be buckled, but it is only extremely rarely that it will be transgressed by tumors. Tumors that transgress this membrane are usually malignant in nature and may be present on both sides of the divide. Meningiomas that arise from the membrane can be present on both sides of the membrane and form a discrete group of tumors.
The membrane has to be respected by the surgeon. This understanding forms the basis of deciding the surgical approach. In general, if the tumor is above the dura, a transcranial approach should be adopted, and if the tumor is below it, a nasal or a facial approach is preferred. The recent introduction of a number of vascularized pedicled or free flaps has opened new vistas, permitting surgeons from both specialties to work together and have the freedom to cross this boundary with limited fear. Despite the fact that reconstruction of the skull base can now be done more elaborately and safely, it can never be achieved with the same perfection with which it is naturally designed and configured. For every tumor, there is only one best approach and selection should be based on concepts such as an anatomical and philosophical understanding of the nature of the tumor, its adjoining structures and, most importantly, its relationship to the basal meninges.
There is great potential for the use of an endoscope in the field of skull base surgery. The advantages of endoscopy in the management of extradural lesions in the paranasal sinus, clival region, and medial cavernous sinus are now generally recognized. However, its use after its introduction through the nose for intradural intracranial lesions must take into account various crucial and relevant tumor- and pathology-related issues. Such nasal approaches for intracranial and intradural lesions are controversial to say the least.
Neurosurgical transcranial approaches to a variety of sellar and suprasellar lesions have been developed and refined over decades. Such advances have occurred with contributions from a number of neurosurgeons over many years. Microscopic vision with a possibility of the use of endoscope assistance to look around corners, ease of dissection under controlled conditions, feasibility of drainage of cerebrospinal fluid (CSF) from the cisterns and ventricles, and retraction of the brain for enhancing the exposure are important components of a cranial approach. Moreover, the entire surgery is done under “sterile” conditions, and transgression of the basal divide is not necessary.
HISTORY
Clinical history of the nature and duration of progression of symptoms including prior drug or radiation therapy provides significant information about the physical nature of the tumor and the extent of difficulty of its dissection from adjoining neural and vascular structures. The longer the duration of symptoms and worse the presenting signs, in general, the firmer and more adherent the tumor is expected to be, enhancing the chances of injury to adjoining structures. Every tumor has a specific pattern of clinical presentation. If the history of progression of symptoms is appropriately analyzed and studied, the histologic nature of the tumor can frequently be predicted and the surgery planned and executed accordingly.
PHYSICAL EXAMINATION
Although meticulous neurologic examination of the patient was critically and diagnostically crucial in the era prior to computer-based imaging, its value even in the modern era of high-resolution imaging cannot be overemphasized as not only does it complement the radiology for assessing the diagnoses, but accurate documentation provides for evaluation of postoperative improvement or complications. In addition, it has a critical role in guiding surgery as the extent and nature of neurologic deficits are guiding parameters through the course of surgery.
Basal tumors can cause a wide range of symptoms, from frontal lobe syndromes with cognitive or personality changes, to specific deficits such as vision loss from direct compression or papilledema, to specific cranial neuropathies depending on the location and involvement. All of these functions must be examined closely to determine the extent and severity of their involvement.
INDICATIONS
On most occasions, it is possible for the surgeon to make a working diagnosis regarding the histologic nature of the lesion on the basis of the clinical and radiologic studies. The age and gender of the patient, acuity of the presentation, principal presenting signs, size of the tumor, extent of cranial nerve involvement, nature of carotid artery displacement and/or encasement, imaging characters on computed tomographic (CT) scanning and MRI, and other such features are helpful in estimating the consistency and vascularity of the lesion, site of origin, and direction of its spread. Such information is critically important in guiding the direction of the approach to the lesion and the cavernous sinus, need for carotid artery control, extent of the surgical resection that would be necessary and possible, and prognosticating the ultimate outcome.
With malignant tumors, a history of prior irradiation in the field of surgery makes the tissue more adherent with fragile vessels. Reconstruction may be a difficult and a defining step of surgery. Depending on the histology, surgery on the first attempt often provides the best opportunity for complete tumor resection.
Anatomical Grading of Giant Pituitary Tumors on the Basis of Their Dural Relationship
Most pituitary tumors involving only the sella and cavernous sinus are essentially “extradural” in their location. An extradural or a nasal approach (transsphenoidal) is best suited for such tumors. Even when such tumors become large or giant in size, they respect the dura, remain underneath its confines, and only rarely transgress it.
Giant pituitary tumors (>3 cm) can be divided into four grades based on tumor extension and the relationship of the tumor to the sellar dura and the dura of the cavernous sinus.
Grade I (Fig. 4.1) pituitary tumors are those that are located within the confines of the sella, remain inferior to the superiorly elevated diaphragma sellae, and do not invade the cavernous sinus. The diaphragma sellae is stretched superiorly, sometimes even beyond the corpus callosum, but it covers the entire superior dome of the pituitary tumor. Although the suprasellar extension of the tumor is intracranial, it is “subdiaphragmatic.”


FIGURE 4.1 A. T1-weighted coronal MRI showing a giant grade 1 pituitary tumor. The tumor does not enter into the confines of the cavernous sinus. The diaphragma sellae is elevated. B. T1-weighted sagittal view showing the large tumor and the elevated diaphragma sellae.
Grade II (Fig. 4.2) pituitary tumors are those that invade the cavernous sinus. Although there are other parameters that suggest invasion of tumor into the cavernous sinus, encasement of the intracavernous carotid artery is a clear sign of cavernous sinus invasion. The cavernous sinus is frequently bloated full by the tumor, but its dural walls are not transgressed.


FIGURE 4.2 A. T1-weighted MRI showing a grade 2 giant pituitary tumor. The tumor invades the cavernous sinus. The diaphragma sellae is elevated. B. T2-weighted axial view of the scan showing cavernous sinus involvement by a grade 2 pituitary tumor.
Grade III (Fig. 4.3) giant pituitary tumors are those where the roof of the cavernous sinus is elevated superiorly, sometimes to a considerable degree. Such an elevation of the roof of the cavernous sinus is frequent and can be mistaken as frontal or temporal lobe extension. The dura of the roof of the cavernous sinus is thin, and such elevation usually displaces the third cranial nerve. Although it is not possible to convincingly confirm, it does appear that the tumors that elevate the roof of the cavernous sinus are inherently more aggressive and the recurrence rates in such cases are relatively higher. Primary radiotherapy may be considered in such cases.


FIGURE 4.3 A. Pre-contrast coronal MRI showing a grade 3 pituitary tumor. The diaphragma sellae is elevated. The roof of the cavernous sinus is seen to be elevated by the tumor. B. Contrast-enhanced coronal MRI showing a grade 3 pituitary tumor. The diaphragma sellae and the roof of the cavernous sinus are elevated.
Grade IV (Fig. 4.4) pituitary tumors are those that transgress the diaphragma sellae boundary and enter into the subarachnoid spaces of the brain. These tumors encase the arteries of the circle of Willis.


FIGURE 4.4 A. T1-weighted coronal MRI showing grade 4 pituitary tumor. The anterior cerebral arteries are encased by the tumor. B. T2-weighted coronal MRI showing the encasement of both A1 segments and Acom complex of the anterior cerebral artery.
The tumors in grades 1 to 3 are below the diaphragma sellae and the superior wall of the cavernous sinus. As the cavernous sinus is considered to be an extradural entity, these tumors are essentially “extradural” in nature. The tumors in these three grades can and should be resected by an extracranial transnasal route. The exact mode and modality of surgery in grade 4 pituitary tumors is still debated. However, this group of tumors may be operated by a transcranial route. A transcranial route for pituitary tumors is also indicated in cases of functioning pituitary tumors that involve the cavernous sinus and extend lateral to the internal carotid artery. Transcranial surgery is sometimes indicated in cases with tumor recurrence where the cranial component is large and adoption of the nasal route is difficult due to previous operation.
Trigeminal Schwannomas
Three distinct types are middle fossa type (type A), posterior fossa root type (type B) where the tumor is in front of the brainstem, and the dumbbell type with both middle and posterior fossa components (type C) (Figs. 4.5 to 4.9). Depending on the nerve of origin, the extracranial part (type D) of the tumor is classified into orbital, infratemporal, and pterygopalatine fossa components. Middle fossa lesions are located within the confines of the dura in the lateral wall of the cavernous sinus and Meckel's cave and as such termed as “interdural.” Despite the sometimes massive growth of these benign lesions, the confines of the dura are not eroded. The posterior fossa component of the tumor can be “intradural” in nature. However, frequently, even this part of the tumor may be “interdural” in nature. The tumor remains within the confines of dura. The cavernous sinus and internal carotid artery are displaced and a relatively thick wall of dura separates them from the tumor. The extracranial component of the tumor is also surrounded by a layer of “dural” sheath. Despite the frequently large tumor size, the majority of trigeminal nerve fibres are only displaced by the tumor.


FIGURE 4.5 A. Line drawing showing the interdural trigeminal schwannoma in the middle cranial fossa. The trigeminal nerve is displaced on the superior aspect of the tumor. The carotid artery and cavernous sinus are also displaced by the tumor. B. Line drawing showing the dumbbell-shaped trigeminal schwannoma extending both in the middle and posterior cranial fossae. The trigeminal nerve is displaced by the tumor.

FIGURE 4.6 Trigeminal schwannoma located in the posterior cranial fossa.
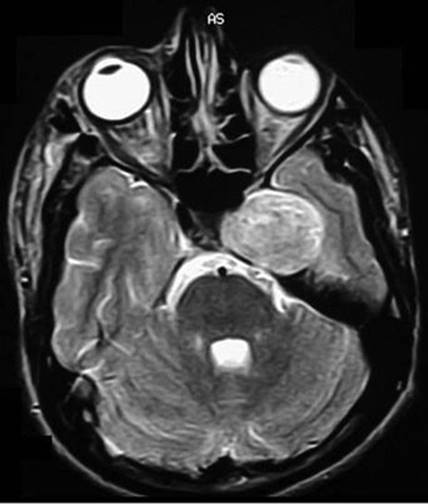
FIGURE 4.7 Trigeminal schwannoma located in the middle fossa.
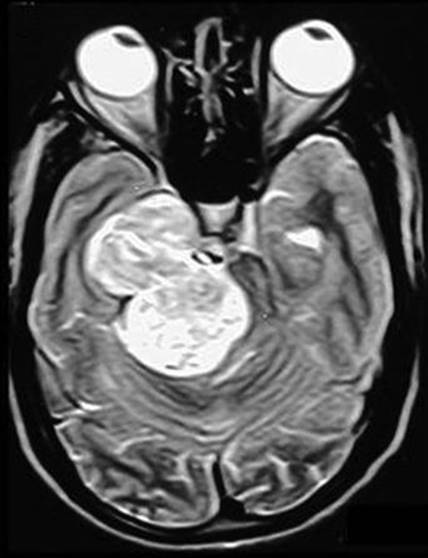
FIGURE 4.8 Large dumbbell-shaped trigeminal schwannoma extending both in the middle and posterior cranial fossae.


FIGURE 4.9 A. T2-weighted sagittal image showing trigeminal schwannoma extending into the extracranial compartment along the mandibular nerve. B. T1-weighted coronal image showing the trigeminal schwannoma extending into the extracranial compartment. The soft tissues are displaced by the tumor.
Chordomas
These tumors destroy bone and displace soft tissue. This character of the tumor distinguishes it from other tumors in the region (Figs. 4.10 and 4.11). Although various authors have noted cavernous sinus invasion by chordomas and chondrosarcomas, it is observed that even in massive tumors the cavernous sinus involvement by these tumors is in the form of displacement rather than invasion. This tumor arises from the clivus or the sphenooccipital synchondrosis and grows eccentrically into the subcavernous sinus region and elevates it on its dome. The functional involvement of the cranial nerves of the region is usually due to stretch. The precavernous and cavernous segments of the carotid artery are displaced anteriorly by the tumor. The tumor is primarily extradural, and actual transgression of the dura is a rare feature. Even in large lesions, a layer of dura separates the tumor from neural and vascular structures, providing a plane of cleavage for surgical resection. The location of the tumor in the extradural space makes an extracranial surgical route a viable surgical option. However, cranial approaches are suitable and safer and are compatible with radical tumor resection in a significant number of these patients. The functional recovery of cranial nerves displaced by these tumors is more remarkable than that following surgery on most other tumors in the region.

FIGURE 4.10 Line drawing showing the extradural chordoma (brown) that displaces the internal carotid artery (red) anteriorly/anterolaterally and the nerves of the cavernous sinus (yellow) superiorly.

FIGURE 4.11 T1-weighted MRI showing the chordoma. The tumor is essentially extradural and displaces the internal carotid artery anteriorly (arrow).
Craniopharyngiomas
Craniopharyngiomas have been treated by craniotomy for decades (Figs. 4.12 and 4.13). However, recently, several groups recommend an endoscopic approach for such tumors. Presence of infradiaphragmatic craniopharyngioma appears to be a clear indication for a nasal approach. Such tumors have a large extension into the sella that is bloated. However, in the presence of supradiaphragmatic craniopharyngiomas and of a small sella, a transcranial approach may be more suitable. Surgery on craniopharyngiomas continues to be debated. The extent of capsular resection, the need to preserve the pituitary stalk, and the indications of radiation treatment are still debated. The dissection of intracranial intradural tumor and of the arteries of the Circle of Willis from the dome of the tumor is simpler and safer by a transcranial approach than by a nasal endoscopic approach.


FIGURE 2.12 A. Sagittal contrast-enhanced T1-weighted MRI showing a craniopharyngioma with enlarged sella. The craniopharyngioma in this case is subdiaphragmatic in nature. B. Coronal post-contrast T1-weighted view showing the craniopharyngioma.

FIGURE 4.13 Sagittal contrast-enhanced T1-weighted view of MRI showing a craniopharyngioma with a small sella. The craniopharyngioma is essentially suprasellar in nature.
Sellar and Suprasellar Meningiomas
Tuberculum sellae meningiomas are relatively common and are a formidable surgical problem (Figs. 4.14 to 4.17). They represent approximately 4% to 10% of all intracranial meningiomas. These tumors arise from the region of the tuberculum sellae, chiasmatic sulcus, limbus sphenoidale, and diaphragma sellae and grow in a subchiasmatic location. As with most other cranial base lesions, tuberculum sellae meningiomas have a relatively innocuous clinical presentation despite their commonly large size. The neurologic, visual, and long-term outcomes are determined by the success of the surgical endeavor. The extent of surgical resection will determine tumor recurrence and regrowth. If they are treated properly and in a timely manner, the results of surgery are gratifying and the long-term outcome is extremely good. Conversely, any surgical mishap can lead to a disastrous outcome. Intradural origin and ease of neurovascular dissection make them well suited for a transcranial approach.

FIGURE 4.14 Contrast-enhanced T1-weighted coronal MRI showing a suprasellar meningioma.


FIGURE 4.15 A. Axial contrast-enhanced T1-weighted MRI of a suprasellar meningioma showing encasement of the internal carotid artery. B. Coronal contrast-enhanced T1-weighted MRI showing a meningioma having encasement of the anterior cerebral artery.


FIGURE 4.16 A. Coronal contrast-enhanced T1-weighted MRI showing a typical tuberculum sellae meningioma. B. Sagittal contrast-enhanced T1-weighted image of MRI showing the meningioma. The pituitary gland can frequently be seen discrete of tumor.


FIGURE 2.17 A. Axial CT scan showing a calcified suprasellar meningioma. B. Coronal CT scan showing the suprasellar calcification.
Nasopharyngeal Angiofibromas
These lesions are primarily extracranial and extradural (Figs. 4.18 and 4.19). In larger lesions, the basal bones are eroded, thinned out, and elevated superiorly. Dural and cavernous sinus involvement is usually in the form of superior elevation and seldom as invasion. The entire cavernous sinus with its contained neural and vascular structures is pushed superiorly by the tumor. This character of the tumor assists in developing a plane between the tumor and basal dura even with blunt dissection. Although transcranial approaches have been described and become popular, an extracranial approach is the rational surgical approach in a vast majority of these cases.

FIGURE 4.18 Line drawing showing characteristic meningeal relationships of nasopharyngeal angiofibromas. The tumors are not only extradural but are also extracranial in nature.


FIGURE 4.19 A. Contrast-enhanced coronal CT scan showing a nasopharyngeal angiofibroma. The characteristic displacement of basal structures can be seen. The tumor extends on both sides of the pterygoid plates. B. T2-weighted sagittal image showing the nasopharyngeal angiofibroma displacing the basal soft tissue structures.
CONTRAINDICATIONS
In general, identification of the correct indication for surgery is crucial for the overall outcome. Surgery is most often done for affecting resolution of symptoms and seldom for correction of radiologic abnormality. When the conduct of surgery is wrought with potential risks that will be more dangerous than the natural course of the disease process, surgery should be avoided. Some patients are physically unable to tolerate the rigors of a long or complex surgery. Finally, certain malignancies may fare better with nonsurgical treatment. Other than these rare situations, there are no contraindications to a transcranial approach.
PREOPERATIVE PLANNING
The entire surgery should be appropriately planned and executed. The extent of skin incision, bone opening, dural incisions, and strategy for tumor resection and reconstruction must be planned prior to surgery. The better and more effective the preoperative discussion, the better the surgical conduct. The need for preoperative tumor embolization must be evaluated.
With the current sophistication in imaging techniques, surgery is rarely focused on the region, but rather is orchestrated on the basis of the histologic nature of the tumor. Imaging characteristics on CT scanning and MRI demonstrate size of the tumor, extent of cranial nerve involvement, nature of carotid artery displacement and/or encasement, and other such features that are helpful in estimating the consistency and vascularity of the lesion, site of origin, and direction of its spread.
Estimation of Consistency of the Tumor: Tumor consistency usually dictates the extent and ease of surgical excision and ultimate outcome. Estimation of consistency of the tumor on the basis of clinical and radiologic features can guide the planning and execution of the operation. Duration of symptoms and the extent of neural involvement provide useful clues. For example, in cases of meningiomas in the region of the anterior clinoid process, the longer the duration of symptoms and worse the visual impairment, the firmer the tumor consistency. Intensities on T1- and T2-weighted images can also provide a reasonable clue about consistency. Hypointensity on T1-weighted and hyperintensity on the T2-weighted images suggest that the tumor is soft. Softer tumors frequently encase the blood vessels and nerves, while firmer varieties will displace these structures. A softer tumor will encase the artery but may not narrow its lumen, whereas when the lumen is narrowed, the tumor is likely to be firm.
Estimation of Vascularity of the Lesion: The vascularity of the tumor is an important variable that may decide the course of the operation. Although contrast enhancement is not a firm guide, some idea about the degree of vascularity can be gleaned from the extent of enhancement. Presence of punctate or linear flow voids in and around the tumor on MRI predicts increased vascularity of the tumor and the nature, size, direction, and source of the feeding vessels. Firm tumors are typically less vascular. With firm tumors, delayed films may be more useful, as the contrast takes time to enter into and clear from the tumor. Tumors with extensive adjoining edema are usually more vascular.
Estimation of Arachnoidal Plane: Presence or absence of arachnoidal plane around the tumor can usually be seen on a good quality MRI. Rounded or irregular borders of the tumor, tumor bosselations, enhancement of surrounding vessels, and peritumoral edema can provide a clue as to the presence of an arachnoidal plane, which could affect the ability to dissect surrounding structures.
SURGICAL TECHNIQUE
General Considerations in Craniotomy for Sellar/Suprasellar Tumors
Patient Position: Patient and head position and the site and size of the scalp incision should be planned meticulously. The principal operating surgeon should personally observe and approve the patient and head position and mark the incision before prepping and draping is allowed. The patient should preferably be placed in a neutral, supine position, as if “sleeping” comfortably, avoiding stretch on any body part. The operating area should be in the superior most part of the field so that the surgeon can operate in a comfortable sitting position without the need for twisting his or her own body or neck. Considerations of adequate venous drainage, and whenever possible necessary provision for an avenue for lumbar CSF drainage, should be made.
Scalp Incision: The incision should not only be cosmetically acceptable but should also provide an adequate exposure for the entire lesion in one operative field (Figs. 4.20 to 4.22).

FIGURE 4.20 Conventional scalp incision for a frontotemporal craniotomy. The zygomatic bone is shaded to show the area of the osteotomy needed to enhance the basal exposure.

FIGURE 4.21 Relatively small and anterior basal exposure for a pterional craniotomy. The posterior half of the lateral and superior walls of the orbit and the entire lesser wing of the sphenoid bone are removed.

FIGURE 4.22 Exposure after bone removal. The orbit, optic nerve, paraclinoid carotid artery, and frontal and temporal lobes are exposed. Surgery in the region of the clinoid process can be carried out with minimal need to retract the brain.
Anatomical Considerations: The frontal and temporal branches of the facial nerve, after their course through the lobes of the parotid gland, traverse superiorly over the anterior aspect of the zygomatic arch. The nerve traverses superficially to the superficial layer of the temporalis and masseteric fascia; by remaining deep to these layers, a subperiosteal dissection over the zygomatic arch provides the best chance of preservation of these tiny nerve branches. Subperiosteal exposure of the zygomatic arch should be in a posterior to anterior direction. It is also advisable to avoid using cautery while exposing the zygomatic arch as the heat can result in compromise of neural function. Retraction of the everted scalp with hooks or stitches needs to be gentle and avoid the region of the course of the nerve. While elevating a bifrontal scalp flap, it is preferable to elevate the flap in a subgaleal plane. The thicker the elevated scalp flap, the better is its vascular nourishment and the chance of preservation of thin nerves within the scalp layers.
The incision for pterional exposure should be made posterior to the superficial temporal artery so that the scalp can receive vascular supply from the artery. In situations where there is no danger of compromise of the scalp circulation, an incision taken anterior to the course of the superficial temporal artery and to its anterior branch and posterior to the course of the facial nerve branches over the zygomatic arch may be suitable. Such an incision may be about 20 mm anterior to the tragus. The preservation of the entire superficial temporal artery can be crucially important in some instances where there is a possibility of operative trauma to the major middle cerebral artery branch and the need for vascular anastomosis is considered.
Direction of Approach to the Tumor: In general, the direction of the approach should be such that the site of origin of the tumor is exposed in the initial phase of the operation rather than the dome of the tumor. This policy holds true not only for meningiomas but also for most other tumors like craniopharyngiomas, pituitary tumors, and chordomas. The dome is the region where the normal neural and vascular tissues are stretched maximally and are liable to be injured. To detach the tumor from the site of origin and debulk it in the direction of the growth provides a better opportunity to preserve the function and integrity of the stretched vessels and nerves.
Anterior Cranial Base Surgical Approaches: Subfrontal approaches are used for midline lesions generally located in the sellar and suprasellar areas. Usually, conventional approaches, wherein the craniotomy is placed above the frontal sinus or including its upper half, are adequate for safe and radical resection of these lesions. Unilateral frontal craniotomy can be centered over the lateral aspect of the orbital rim (Fig. 4.23). Such a lateral exposure avoids opening of a large part of the frontal sinus. Retraction of the frontal lobes is usually safe after drainage of CSF from basal subfrontal, chiasmal, and Sylvian cisterns. After such a maneuver, a large space can be obtained for a comfortable dissection in the sellar region. Extensive manipulation of the frontal sinuses and the supraorbital rims may be avoided and should not be performed as a routine. For some lesions, which involve a prolonged microsurgical dissection, anterior cranial basal approaches can be useful. Basal extension of a conventional frontal craniotomy can be done in various ways.

FIGURE 4.23 Frontal scalp flap elevated anteriorly. Low frontal craniotomy is performed. The area depicted in brown is the basal extension of the frontal craniotomy.
Basal Extension of a Unilateral Frontal Craniotomy: A bicoronal scalp flap for performing basal extension of the conventional frontal craniotomy needs to be slightly longer so that an exposure of the orbital rim may be obtained (Figs. 4.23 and 4.24). If the circulation of the forehead is jeopardized (by procedures like radiation treatment or external carotid artery embolization), the flap should include the superficial temporal artery (by making the incision posterior to it) in the exposure. The superficial temporal vessels must also be preserved in the situation where the lesion is in proximity to the internal carotid or middle cerebral arteries, which could be damaged during the surgery. These vessels may be preserved while taking the incision either anterior or posterior to the trunk of the superficial temporal artery. Dissection in appropriate planes and mobilization of the artery can help to increase the exposure while preserving the artery. The supraorbital nerves and vessels must also be preserved. This can be done by careful subperiosteal dissection in the region of the supraorbital rim. Whenever a foramen is encountered through which these nerves and vessels pass, a small osteotome may be used to open up the foramen. Subperiosteal dissection is continued, and the orbit is exposed, preserving the periorbita. Electric saws can be used to make osteotomies in the superior rim of the orbit. During these maneuvers, the orbital contents and frontal lobes and dura are adequately protected. The roof of the orbit should be cut as posteriorly as possible. One can remove the entire orbital roof lateral to the cribriform plate, or only a partial, supralateral removal may be done, depending on the extent of the exposure needed. The medial osteotomy will often traverse through the frontal air sinus, and adequate precautions need to be taken to seal it at the end of the operation (Figs. 4.25 and 4.26). This medial osteotomy provides a significantly improved basal exposure without disturbing the sense of smell. The globe can be depressed inferiorly to gain additional exposure. The orbital roof can be removed posteriorly up to the optic canal. The optic nerve can be mobilized if truly necessary. This exposure can significantly limit the extent of frontal lobe retraction.

FIGURE 4.24 Basal frontal craniotomy after the removal of the supraorbital rim using an oscillating saw. The frontal sinus is opened. The orbit is exposed.
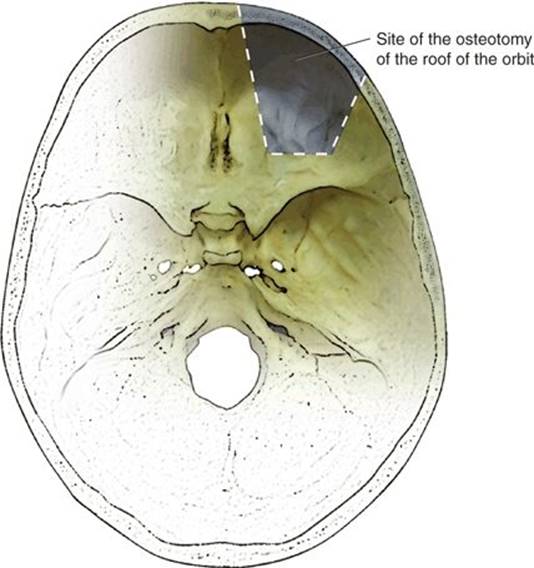
FIGURE 4.25 View of the skull base from above showing the site of the osteotomy of the roof of the orbit.
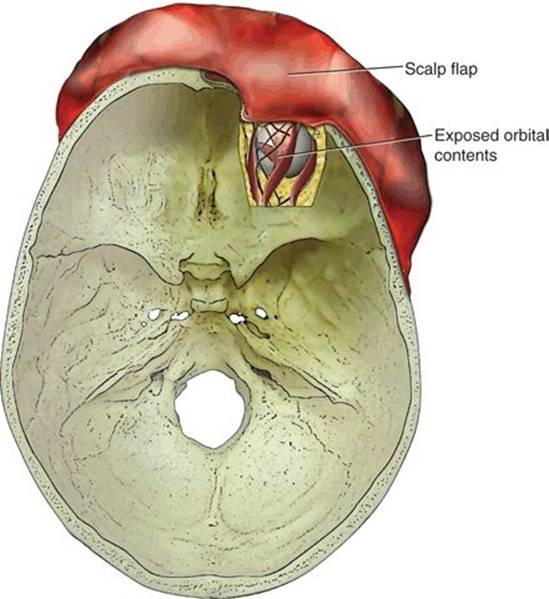
FIGURE 4.26 View of the skull base from above after the roof of the orbit is resected.
Basal Extension of a Bifrontal Craniotomy: Fashioning a supraorbital bar craniotomy extension assists in basal extension of the bifrontal craniotomy (Figs. 4.27 to 4.29). However, the need for such additional exposure is only rarely necessary for resection of basal tumors. The surgical technique for a supraorbital bar osteotomy has been elaborately discussed elsewhere.


FIGURE 4.27 A. Low bifrontal craniotomy. Areas depicted in purple are the supraorbital rims, which will be removed using an oscillating saw. On some occasions, the bifrontal craniotomy can be extended far inferiorly, a procedure that can avoid removal of the supraorbital rims B. Basal bifrontal craniotomy exposure after removal of the supraorbital rims on both sides. The bone in the region of nasion is not removed.

FIGURE 4.28 View of the skull base from above after the orbital roofs are removed on both sides. The area of bone in the region of the cribriform plate is preserved.


FIGURE 2.29 A. The basal exposure after removal of the supraorbital rim that includes nasion and the basal bones with the cribriform plate. B. Lateral view of the exposure.
Preservation of Olfaction in Anterior Cranial Basal Exposure: Unilateral frontal craniotomy and resection of the supraorbital bar following a slight lateral corridor to the planum sphenoidale without elevation of the dura in the region of the cribriform plate can allow preservation of olfaction and provide adequate exposure to the sphenoid sinus, sella, and superior and midclivus in an extradural perspective. Intradural extensions of the lesion can be dealt with after opening the dura and elevating the frontal lobe off the orbital roof. Such a limited approach to midline lesions is satisfactory in most cases for large parasellar lesions where transsphenoidal approaches are considered to be unsuitable. Bilateral exposure leaving both cribriform plates undisturbed can be performed if needed. A larger exposure may be required for malignant tumors where radical en bloc excisions are indicated.
Keyhole Approaches: Advances in microsurgical techniques have led to reduction in the extent of surgical incisions and the successful application of keyhole approaches. These approaches should be employed only if they do not compromise the extent of tumor removal or patient safety with respect to neurovascular control. For lesions in the sellar and parasellar areas where a pterional approach may be selected, extensive temporal or frontal lobe exposure may be unnecessary. If so, the incision can be moved anteriorly toward the lesser sphenoid wing or even to the eyebrow. The basal extension of the approach can be obtained by removing the entire lesser wing and part of the roof and lateral wall of the orbit (Fig. 4.22). This keyhole approach can provide a basal view to the lesion and also avoids unnecessary and avoidable scalp incision or bone removal.
Considerations in Cranial Approach to Sellar/Suprasellar Meningiomas
In the majority of patients with suprasellar meningiomas, a basal unifrontal craniotomy is performed on the side of worse vision (Figs. 4.14 to 4.17). The patient is placed in the supine position with the head extended. The craniotomy extends to the orbital roof inferiorly. A more lateral basal frontal approach is usually necessary and avoids the frontal sinus though the sinus should be opened when needed to achieve adequate basal exposure. The frontal lobe is dissected from the orbital roof, and all attempts are made to preserve the olfactory tract. The medial part of the Sylvian fissure is opened to drain CSF and relax the brain. The tumor is then exposed, and the optic nerves and internal carotid arteries are identified whenever possible. The tumor is progressively disconnected from the site of dural attachment. Space for basal dissection and coagulation is obtained by tumor debulking, avoiding the optic nerves, anterior cerebral arteries, anterior communicating artery, and pituitary stalk. The optic nerves and anterior cerebral arteries should be identified early in the course of dissection. Radical removal of all tumors is attempted, but if the dissection of tumor from the anterior cerebral arteries and internal carotid artery entails undue risk of damage, that portion of the tumor is left behind. Some surgeons prefer to open the optic canal early in the operation to resect the portion that extended into it. However, we prefer to open the canal after the majority of the suprasellar tumor is resected. This provides the ability to observe the canalicular extension directly and allows more space for drilling the canal under direct vision. The site of tumor attachment to the dura is widely coagulated, and the part of the dura grossly involved by tumor is resected. No special attempt is made to remove the bony hyperostosis unless it interferes with visualization. Various authors have advocated resection of the involved dura and bone in patients with meningiomas. However, the possibility of CSF leakage and consequent problems led us to adopt a relatively conservative approach. The skull base is reconstructed, and the open frontal sinuses are separated from the intracranial space with vascularized pericranial and galeal flaps.
The degree of surgical exposure in each patient in our suprasellar meningioma series did not correlate with the extent of basal tumor involvement. Frontobasal craniotomy on the side of the more severe visual deficit was appropriate even for more extensive tumors. There was no need to section the anterior part of the superior sagittal sinus or perform a dissection in the interhemispheric region in any patient. A unifrontal approach is superior to pterional exposure in that it provides wide, symmetric exposure and direct access to both internal carotid arteries. It has the advantage over a bifrontal exposure of avoiding handling of and potential damage to the contralateral olfactory tract.
The difficulty in surgically excising a tuberculum sellae meningioma stems from its relationship to the optic nerves and chiasm and to the anterior cerebral and internal carotid arteries and their perforators, which are frequently encased and/or displaced. The success of surgical resection hinges on the preservation of these vessels. Despite encasement of the internal carotid artery seen on MRI, it is often observed that tumor lobulations engulf the artery, and the dissection is relatively simple. All of our patients who worsened after the operation had intraoperative damage to the anterior cerebral artery or its large perforator branch, making early identification of the anterior cerebral artery critical. The arterial supply of these tumors usually comes from the ethmoidal arteries. Rarely, small branches of the anterior cerebral artery can also contribute to the vascular supply. Also, the arterial wall in the proximity of the tumor can be thinned out and can even have an aneurysmal dilation. In case of damage, an immediate anastomosis should be carried out, though this procedure is difficult given the nature and size of the involved artery.
Considerations in Surgery for Trigeminal Schwannomas
The dura is relatively separate from the gasserian ganglion in the region of Meckel's cave, and normally, there is a large subarachnoid space in the region. Anterior to the gasserian ganglion, the dura is adherent to the divisions of the nerve and then continuous with their epineurium. Adhesion of the dura is more firm to the first division than the second and third. Schwannomas usually arise in the region of the gasserian ganglion, and the growing tumor can be accommodated within the confines of Meckel's cave, which becomes distended. Anterior extension along the mandibular branch into the infratemporal fossa is rare, probably due to natural adhesions of the dura and the nerve. The intracavernous carotid artery is displaced medially and the petrous carotid artery inferiorly by the tumor. As a result, working within the dural boundaries is relatively safe and effective. There is seldom any need to have proximal arterial control for tumor excision in these cases. In addition, the outer dural sheath of the gasserian ganglion can be opened without exposing the temporal lobe. Therefore, trigeminal schwannomas can be resected using a relatively small exposure if the dural wall and its relationship to the tumor is understood and respected.
The extracranial portion of the tumor follows the divisions of the trigeminal nerve, and despite the fact that some of the extensions of the tumor can become large, the tumor remains within the confines of the perineural layer (extension of the dural layer) of the divisions. These tumors never encase any major blood vessel or cranial nerve in the infratemporal fossa or in the orbit. As a result, a “reverse skull base approach” that involves a limited “transcranial” extradural avenue can be used to resect the majority of tumors. The surgical strategy is to perform a small basal temporal craniotomy, resect the floor of the middle cranial fossa, and expose both the middle cranial fossa and the infratemporal fossa components in the same exposure. Depending on the nature and extension of the tumor, the basal temporal craniotomy can incorporate drilling of the root of the zygoma, roof of the mandibular condyle, and roof of the external ear canal and partial mastoidectomy up to the mastoid antrum as needed. As the temporalis muscle and the other muscles of the infratemporal fossa are atrophied in most cases, their retraction is relatively straightforward. The temporalis muscle can be retracted anteriorly, posteriorly, and inferiorly or be split into two parts for exposure. Addition of a zygomatic osteotomy provides significant additional space for muscle retraction. When there is orbital involvement, a lateral orbitotomy is performed and the bone over the lateral aspect of the superior orbital fissure is widely resected to expose both the orbital and middle fossa components of the tumor in the same surgical field. Dural incision is then made in the outer wall of the tumor and the tumor debulked taking care not to violate the deep layer of dura, which forms a firm protective layer over the carotid artery and cranial nerves. The tumor does not involve all the fibers of the nerve. Some fibers are invariably spared and can usually be preserved. Working within the tumor, using blunt dissection with the help of suction or Cavitron ultrasonic surgical aspirator (CUSA), and avoiding coagulation as much as possible can avoid injury to these fibers.
After resection of both the middle fossa and the extracranial components of the tumor, wide exposure of the posterior cranial fossa portion is also possible through this exposure. Lateral and superior dissection provides exposure of the tumor, temporal lobe, brainstem, and petrous carotid artery simultaneously in the surgical field allowing dissection of the tumor from these structures under direct vision and control. Moreover, these structures are closer in the surgical field when compared to most other available surgical approaches.
This type of transcranial approach is suitable to resect schwannomas and many other parasellar tumors. Intraoperative lumbar drainage is used to relax the brain, and its superior retraction provides additional exposure to the infratemporal fossa and even any orbital component of the tumor. Maintaining and working outside the middle fossa dura adds protection to the temporal lobe. The strategy of retraction of the brain to expose an extracranial tumor, although against the principles of skull base surgery, in this case can provide an easy, quick, and wide access to the entire tumor. In anterior transfacial and anterolateral approaches, such an elaborate and simultaneous exposure may not be possible.
POSTOPERATIVE MANAGEMENT
Patients are observed in the ICU in the initial postoperative period. Anticonvulsants are generally used prophylactically for any transcranial approach. The dressing needs to be kept dry until the sutures are removed. Appropriate antibiotic prophylaxis based on personal preferences and experience is used. If cerebral edema and neural manipulation are concerns, perioperative steroids can be used.
COMPLICATIONS
All the potential avoidable complications are a result of either inappropriate planning or less than optimal performance of the surgical procedure. The initial complication is when poor selection of a surgical candidate is made. When selecting a patient for surgery, the only consideration is the need of the patient. Although the physical presence of the tumor is sometimes a consideration, surgery is generally done for symptoms.
For ultimate avoidance of complications, the entire operation, including reconstruction, has to be planned carefully to be successfully executed.
Dealing with Air Sinuses: The paranasal sinuses are practically sterile but potentially infective spaces as they may contain commensal organisms. The sinuses ultimately communicate with the nasal cavity, and opening the sinuses during intracranial surgery provides communication between the intracranial compartment and the nose. Opening of these sinuses should be avoided as much as possible, but the frontal sinuses are frequently opened during basal frontal exposures. When this occurs, it is critical that the communication with the sinus be adequately sealed. The frontal sinus communicates with the nose through the frontonasal duct, which can be packed with a small bone strut. If the duct is sealed, residual mucosa in the excluded sinus must be removed as it can lead to collection of secretions and formation of a mucocele or a pyocele and can invite ascending infection. Ideally, the sinus should be packed with a vascularized pedicled flap obtained either from the pericranium or galea.
Stroke and visual compromise remain the major complications associated with suprasellar or parasellar surgery. Visual compromise can result from optic nerve injury, hypoperfusion, or cranial nerve manipulation. Stroke is usually avoidable, but the risk depends on the adherence of the tumor to the involved artery.
RESULTS
The results of craniotomic approaches to the supra- and parasellar regions are well described in the literature and remain the gold standard. Maintenance of these results requires careful selection of indication and surgical route and care to conduct safe and effective surgery. The expected result of surgery depends on the type of tumor in question. In general, surgery for pituitary and other suprasellar tumors results in excellent symptomatic recovery. Radical resection is the aim for each tumor, but this may need to be tempered, and patient safety should always be the highest priority.
Cranial approaches to suprasellar tumors need to be tailored to anatomical and physical characteristics of the tumor that vary with the pathology. Understanding of the growth pattern of the tumor and its relationship with the meninges forms the basis of approach selection and guides the surgery.
PEARLS
· The basal meningeal divide determines the direction of surgical approach; tumors above the divide need a transcranial approach, and those below the divide need a transnasal or facial approach. The endoscope is an invaluable tool for nasal approaches to skull base lesions.
· Pituitary tumors have a well-defined dural relationship. Even massive pituitary tumors can be approached by a transnasal route if the dural relationships of these tumors are adequately understood.
· In the middle cranial fossa, trigeminal schwannomas are located within the dural confines of the lateral wall of the cavernous sinus or are interdural in location. The surgical exposure can be minimized if the dural relationship is appropriately understood.
· Chordomas grow by destruction of bone and by displacement of soft tissues that include cranial nerves and blood vessels.
· Nasopharyngeal angiofibromas are generally extradural and extracranial tumors.
· Suprasellar meningiomas are above the meningeal divide and therefore best removed by transcranial surgery.
· Infradiaphragmatic craniopharyngiomas are best suited for the endoscopic endonasal approach, whereas all others are above the meningeal divide.
PITFALLS
· The course of the frontal branch of the facial nerve should be understood and carefully protected during frontotemporal exposure to avoid palsy.
· Knowledge of and preservation of perforating branches at the base of the skull is critical for avoiding vision loss and other neurologic injury.
· Arterial tumor involvement should be identified pre- and intraoperatively to avoid injury. When thinning of the artery is present, this should be identified at the time of surgery and repaired if possible.
· The paranasal sinuses must be properly sealed and excluded mucosa removed to avoid mucocele, CSF leak, or infection.
INSTRUMENTS TO HAVE AVAILABLE
· Standard microneurosurgical tools
· High-speed drill
· Surgical microscope
SUGGESTED READING
Goel A. Infratemporal fossa interdural approach for trigeminal neurinoma. Acta Neurochir (Wien) 1995;136:99–102.
Goel A. The extradural approach to lesions involving cavernous sinus. Br J Neurosurg 1997;11(2):134–138.
Goel A, Muzumdar D, Desai K. Tuberculum sellae meningioma: a report on management on the basis of a surgical experience with 70 cases. Neurosurgery 2002;51:1358–1364.
Goel A, Nadkarni T, Muzumdar D, et al. Giant pituitary tumors: a study based on surgical treatment of 118 cases. Surg Neurol 2004;61(5):436–445.
Kothari M, Goel A. Maternalizing the meninges: a pregnant Arabic legacy. Neurol India 2006;54(4):345–346.