Aaron M. Cheng and Douglas E. Wood
![]() INDICATIONS/CONTRAINDICATIONS
INDICATIONS/CONTRAINDICATIONS
Airway stenting is used to treat obstructing lesions of the central airways, defined as the trachea, main bronchi, and the bronchus intermedius. Generally, lesions obstructing at the lobar or segmental level are not amenable to stenting due to lack of efficacy or functional benefit. Central airway obstruction can be caused by both benign or malignant etiologies, but the majority of patients suffer from malignancies, primarily direct involvement from bronchogenic carcinoma, or direct invasion from carcinomas of adjacent structures such as the esophagus or thyroid, but occasionally due to metastatic disease involving the airway directly, or indirectly from adjacent mediastinal lymph nodes. Most of these malignant lesions are surgically inoperable due to advanced stage disease and therefore the primary role of airway stenting in malignant disease is usually palliative. Airway stenting is infrequently performed because of surgical ineligibility due to inadequate physiological reserve in patients who might otherwise be candidates for resection. Airway stenting is nearly always combined with other bronchoscopic modalities for treating the endoluminal lesion, which can include (1) core-out and mechanical debridement; (2) airway dilation; (3) resection with different energy sources or cryotherapy; and (4) endoluminal brachytherapy or photodynamic therapy. The modalities chosen to treat the obstruction depend on the anatomy and characteristics of the obstruction, the training and experience of the thoracic surgeon or interventional pulmonologist, and the equipment available; however, lesions causing extrinsic airway compression and mixed lesions with a significant extraluminal component usually require stenting for adequate palliation of symptoms (Fig. 2.1).
Stenting should be considered when there is persistent symptomatic airway narrowing after other interventions such as core-out or dilatation, typically when the diameter of an affected airway remains <50% of normal diameter. Airway stenting should also be considered when there is central airway narrowing and external beam radiation treatment of the central airway is planned, in order to avoid anticipated greater significant airway narrowing that can occur subacutely with radiotherapy. Other indications for airway stenting due to malignant processes include management of airway fistulas.
Airway stenting for central airway obstruction due to benign etiologies should be approached cautiously and generally as a treatment of last resort when other, more definitive, or less invasive techniques are not successful. The most common benign cause of central airway obstruction occurs from postintubation stenosis; typically due to prolonged intubation. Other benign etiologies causing central airway obstruction include infectious and inflammatory processes such as tuberculosis, Wegener’s disease, relapsing polychondritis, fibrosing mediastinitis, and endobronchial papillomatosis. The thoracic surgeon should adhere to the basic principle of treating central airway obstructions caused by benign disorders with curative surgical resection if at all feasible. Patients with benign diseases are usually younger and expected to have longer life expectancies and the complications associated with prolonged airway stenting will often overshadow the palliative benefits. If airway stenting is necessary in patients with benign lesions, fully covered removable stents are required. One increasingly common indication of stenting for benign disease is airway stenoses following sleeve resections and bronchial anastomoses in lung transplant patients.
The primary contraindication for airway stenting is when surgical resection of the disorder causing the airway obstruction is possible. Therefore, it is imperative that an experienced thoracic surgeon evaluates a patient presenting with central airway obstruction before undergoing stenting or other endoscopic treatment. Although endoscopic management such as debridement and dilation may temporarily stabilize and palliate a patient with symptomatic central airway obstruction to evaluate for possible resection, airway stenting preceding a planned resection of the obstructed airway segment can create local inflammation and mucosal damage that may interfere with anastomotic healing following airway resection and reconstruction.
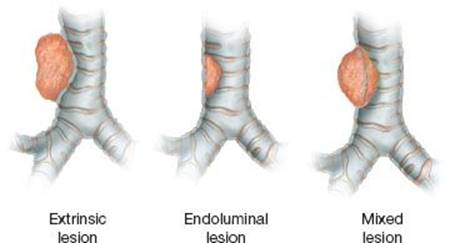
Figure 2.1 Lesions can cause tracheobronchial obstruction by extrinsic compression of the airway, endoluminal obstruction arising from the airway itself, or mixed lesions with both extrinsic and endoluminal obstruction.
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
Imaging
Patients with large airway obstruction are likely to present with dyspnea, stridor or wheezing, cough, and occasionally hemoptysis. Chest radiographs are universally obtained in patients with symptoms from airway obstruction and may be useful if the x-ray demonstrates a tracheobronchial mass or distortion of the tracheobronchial airway column. Also if pneumonia or postobstructive consolidation is evident, antibiotic therapy should be appropriately initiated. Neck and chest CT scans provide much better detail of airway and mediastinal anatomy, particularly with the higher resolution scanners that are readily available now at most facilities. Coronal and sagittal views, as well as the capability of generating three-dimensional (3D) reconstructions, provide excellent detail of airway anatomy that helps identify the location and extent of stenosis prior to bronchoscopy. These images can be useful for planning different therapeutic options, potential hazards, and the likelihood of successful intervention.
Choice of Stent
Stenting airway lesions require a thorough understanding of the various advantages and disadvantages of the available airway endoprostheses. The ideal endoprosthesis is a stent that is (1) easily deployed and adjusted but does not readily migrate; (2) resists compressive forces yet does not erode or breach the native airway mucosa; (3) conforms to the airway contour without kinking or bending; (4) elicits minimal foreign body reaction and prevents tissue ingrowth or granulation; and (5) allows mucociliary clearance to decrease mucous impaction. Unfortunately, the ideal airway endoprosthesis is not presently available. Current airway stents have some of these properties, but none have all.
TABLE 2.1 Common Commercially Available Tracheobronchial Stents
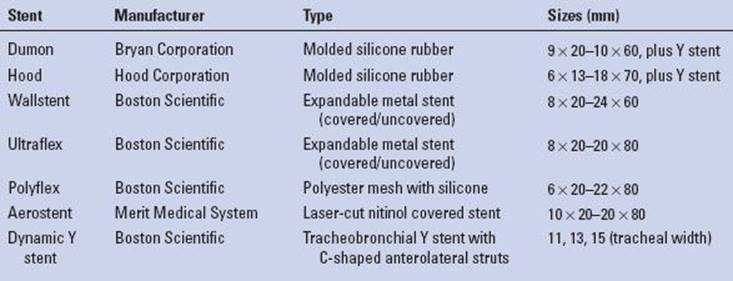
There are basically two main categories of airway stents: Silicone and metal, and selections from both categories should be readily available for deployment in any given clinical scenario. The main advantages of silicone stents are that they are inexpensive to manufacture; once positioned, easy to adjust and remove; and relatively well tolerated with minimal reactivity for granulation formation. These stents come in a variety of different diameters and different lengths (Table 2.1). The reported disadvantages of silicone stents are that they are stiff and more difficult to deploy, requiring general anesthesia, intubation, and skill with rigid bronchoscopy; although this in itself should not be a major limitation in the majority of patients. However, a considerable amount of sheer force may be required to push the stent across a tight stenosis or stricture, which can traumatize the airway. There are times that silicone stents may not generate sufficient radial force for some strictures or extrinsic compression from tumor. They also do not seat well in malacic segments or conform to longer tortuous stenotic segments and thus will commonly migrate from position causing itself airway obstruction. It is important to be aware that silicone stents can ignite with laser treatment, which often is used to treat airway granulation. Two types of molded silicone stents are commercially available, Dumon stents and Hood stents. Both are similar in characteristics, each with different versions of external studs or flanges for airway fixation (Fig. 2.2). The Hood and Dumon stents are essentially cylinderized versions of the Montgomery T-tube, a silicone-molded stent that is useful to consider in select patients who have a tracheostomy stoma or benefit from the additional stabilization of the T-limb, yet have a functional larynx.

Figure 2.2 Molded silicone stents. A: Hood silicone stents and Dumon-style stents are similar. B: Dumon stents have studs.
Expandable bare metal airway stents have a more favorable inner luminal to outer luminal diameter ratio than silicone stents and are easier to deliver, using flexible bronchoscopy with or without fluoroscopy under topical or general anesthesia. Once delivered, these stents are extremely stable and there is low likelihood of dislodgment or migration. However, nearly all stents used today are covered metal stents that have some of the same potential for dislodgment as the solid silicone stents. The second-generation stents (e.g., Wallstent & Ultraflex stent) conform well to airway contours allowing greater adaptability for their use in more tortuous airway anatomy. Expandable metal stents can generate considerable radial force, which may be favored in fibrotic strictures. Uncovered metal stents theoretically allow mucosal neoepithelialization with the reported possibility to resume mucociliary clearance and also allow ventilation of lobar orifices across the open interstices. However, in reality this potential benefit is far overshadowed by tissue ingrowth and granulations through the interstices that frequently occurs due to the foreign body reaction to the stent. This often results in obstruction of the stent itself, yet once embedded in the airway wall the bare metal stents are extremely difficult to remove without causing significant secondary airway damage. Recurrent obstruction from tissue or tumor ingrowth through the metal interstices can require repeated debridement or even repeat stenting within the stent. Expandable stents also can exert considerable radial force and therefore, may erode through the airway into adjacent structures and risk creating a possible life-threatening bronchovascular fistula. As a result of these known shortcomings and complications, the US Food and Drug Administration (FDA) issued a warning that bare metal airway stents should be avoided in the management of benign airway stenosis.
The experience of airway interventionalists and the complications of bare expandable metal stents led to the development of third-generation–covered expandable metal stents. These stents are now widely available and used in the United States and internationally. One version of these stents is covered in the middle portion of the stent but bare at the ends. These are easier to adjust and remove than uncovered stents, but the metal ends of these stents are exposed and can become incorporated in the tracheobronchial mucosa from granulation and tissue overgrowth as well (Fig. 2.3). Yet more modern stents are fully covered, such as the Polyflex or Aero stents. The Polyflex self-expandable stent is composed of a polyester mesh with a silicone coating and does not have uncovered metal edges and therefore share advantages and disadvantages from both silicone and metal airway stents, while the Aero stent consists of a metal nitinol framework covered completely with polyurethane.
Silicone, metal, or covered expandable stents all may offer acceptable palliation in patients with advanced stages of malignancy and central airway obstruction. In this setting, the preferred choice of stent may be determined by the anatomy of the lesion and the experience and preference of the thoracic surgeon, yet today the vast majority of these stents are the latest generation-covered expandable stents. The choice of stent to be used in benign disease is more straightforward as noted above. Expandable metal stents has been discouraged in benign disease due to their permanence and difficulty with repositioning, risk of airway erosion from long-standing deployment and the development of stent-related granulations. However, in some settings, expandable stents may provide the only remedy if silicone stents cannot be well seated due to absence of a fixed stenosis, especially if other options for stent fixation such as carinal Y stent or tracheal T-tube are not better alternatives. Covered self-expandable stents are also the preferred alternatives in these settings (Fig. 2.4).

Figure 2.3 Expandable metal stents can be uncovered (A) or covered (B). Covered stents are easier to remove but tissue and tumor ingrowth can occur at the uncovered ends. (Images courtesy of Boston Scientific.)

Figure 2.4 Fully covered self-expandable tracheobronchial stents are available such as the (A) Polyflex stent (Boston Scientific) and the (B) Aerostent (Merit Medical Systems), which offer excellent inner to outer diameter ratios and are good alternatives to silicone stents. (Images courtesy of Boston Scientific and Merit Medical Systems.)
![]() SURGERY
SURGERY
Although patients undergoing flexible bronchoscopy for evaluation of their airways can be routinely managed by topical anesthesia and sedation, immediate availability to general anesthesia and rigid bronchoscopy is required in patients with critical airway obstruction. The thoracic surgeon who performs airway stenting must be skilled in both flexible and rigid bronchoscopy and adept with either approach to the airway, as even the experienced bronchoscopist skillful in airway therapeutics can find patients with severely compromised airways challenging. The entire procedural team, including the anesthesiologists and nurses, should be familiar with the endoscopic maneuvers and equipment required for airway therapeutics to safely manage a patient with a complicated and threatened airway. It is the authors’ preference to perform airway stenting procedures in the operating room with a dedicated staff of thoracic anesthesiologists and operating room personnel.
Rigid bronchoscopy does require general anesthesia but muscle paralysis is not routinely required. The safest anesthetic induction maintains spontaneous ventilation using both inhaled and short-acting intravenous agents until an airway is controlled by a ventilating rigid bronchoscope. Once the airway has been secured by rigid bronchoscopy and the severity of airway obstruction assessed, muscle paralysis can be administered if necessary.
Positioning
Patients should be placed on an operating room table or bed that allows fluoroscopy to be performed, even if not originally intended. The clinical situation may arise where fluoroscopy may be required or advantageous. Patients are positioned supine and with the head extended in the “sniffing position” used by anesthesiologists for intubation, as this most readily allows exposure and intubation of the ventilating rigid bronchoscope through the larynx. After intubation, it is often beneficial to place a shoulder roll and extend the patient’s neck to facilitate straightening of the oropharyngeal/tracheal axis when performing rigid bronchoscopy.
Technique
The initial evaluation of the airway by bronchoscopy is critical to define the nature of obstructing lesion and determine the appropriateness for airway stenting. The initial bronchoscopy should carefully document the airway anatomy. The extent, severity, and complexity of the obstruction must be determined as well as its relation to normal anatomical landmarks, including the distance from the vocal cords, cricoid cartilage, carina, and measurements of the lengths of the lesion and normal airway. Associated findings of granulation tissue, mucosal inflammation, or loss of cartilaginous support are important to detail, as well as the degree of stenosis due to endoluminal pathology versus fibrotic scarring or extrinsic compression.
Although flexible bronchoscopic assessment of obstructing lesions can be performed with small-diameter pediatric bronchoscopes, therapeutic intervention cannot be simultaneously rendered and therefore, it is usually preferable to use an adult bronchoscope with a large working channel (2.8 to 3.2 mm). Not only does this allow adequate suction and clearance of secretions, but also enables the thoracic surgeon to perform other techniques of airway palliation such as balloon dilation, followed by delivery of certain stent devices. The flexible bronchoscope is also a very useful adjunct during rigid bronchoscopy procedures as it allows better angled and distal examination with simultaneous aspiration of blood and/or secretions.
In situations where the etiology of the tracheobronchial stricture is not known or in critical airway conditions, rigid bronchoscopy, as previously stated, is the safest approach to establish a stable airway and secure adequate ventilation. Rigid bronchoscopy can also prepare the airway for stenting by using the bronchoscope as an instrument for mechanical debridement and dilation. The tip of the rigid bronchoscope can be used to “core out” a lesion. When dilation is required, rigid bronchoscopes (3.5 to >10 mm) of varying diameters can be used to serially dilate the obstructed airway segment to an adequate size. Using blunt, rounded-tip rigid bronchoscopes for dilation can decrease the risk of airway perforation or mucosal laceration. Hydrostatic balloon dilation can be subsequently performed to enlarge the airway even more. When assessment or treatment of the distal airway is necessary, the flexible bronchoscope can then be inserted down the barrel of the rigid bronchoscope and advanced peripherally.
The final stage of bronchoscopic preparation is the assessment of the type and size of stent(s) to be deployed. Stent length can be determined by direct measurement of the stenosis length, and the stent diameter is estimated by using the diameter of the bronchoscope as a guide. Radiopaque markers and a ruler to precisely measure the extent of the stenosis are useful aids. Alternatively, commercially designed stent-measuring devices are available that can be used to guide stent selection. For tracheal lesions, the stent length is chosen such that the stent extends usually 1 to 2 cm beyond each end of the stenotic lesion; for bronchial stenosis, the stent usually covers just beyond the proximal and distal ends of the stenosis. If fluoroscopy is to be used for stent deployment, external radiopaque markers can be placed and a guidewire (0.035 to 0.038 in) can be inserted if needed.
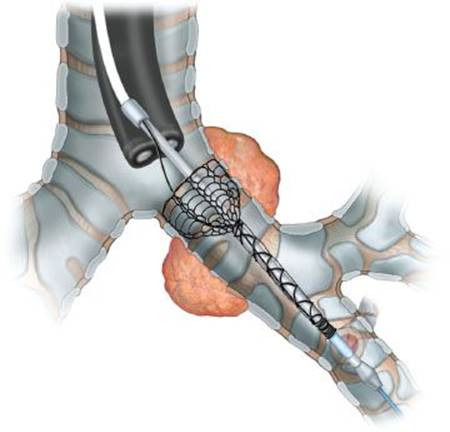
Figure 2.5 Self-expanding stents can be easily deployed over a guidewire using flexible bronchoscopy.
Expandable covered/noncovered metal stents can be deployed by flexible or rigid bronchoscopy. Each specific manufacturer of expandable tracheobronchial stents has specifically designed delivery systems to allow consistent stent deployment. In general, these stents are constrained within a sheath and can be deployed over a wire (Fig. 2.5). Silicone stents are more challenging to deploy through a rigid bronchoscope and variety of different techniques have been described to place these stents.
One described technique places the stent on the outside barrel of the rigid bronchoscope with an endotracheal tube inserted as a sheath over the proximal portion of the scope to serve as a “pusher tube” to keep the stent from displacing proximally on the barrel of the scope during insertion. The entire apparatus—rigid bronchoscope, endotracheal tube, and stent—is inserted. Once the tip of the bronchoscope is inserted beyond the stenosis, the bronchoscope is gradually rotated and withdrawn while the endotracheal tube “pusher” remains fixed in place and pushes the stent into the right location. Grasping forceps are used to adjust the stent’s final desired position. Problems with this technique are associated with the bulk of the entire apparatus; scope, tube, and stent must be carefully inserted past the vocal cords and through the stenotic area.
A second technique uses a universal Dumon-specific bronchoscopy system (Bryan Corporation, Woburn, MA) that can be used to deploy silicone stents (Fig. 2.6). This system uses changeable tubes of various sizes that are used to calibrate and dilate the stenosis. The tubes are able to telescope over each other and can be used to introduce stents of different sizes. Once the appropriate-sized stent has been chosen, the stent is collapsed into the distal end of the introducer tube, which is passed through the bronchoscope to the stenosis. A plunger system is then used to push the stent out of the introducer tube. Again, grasping forceps are used to manipulate the stent into the final position.
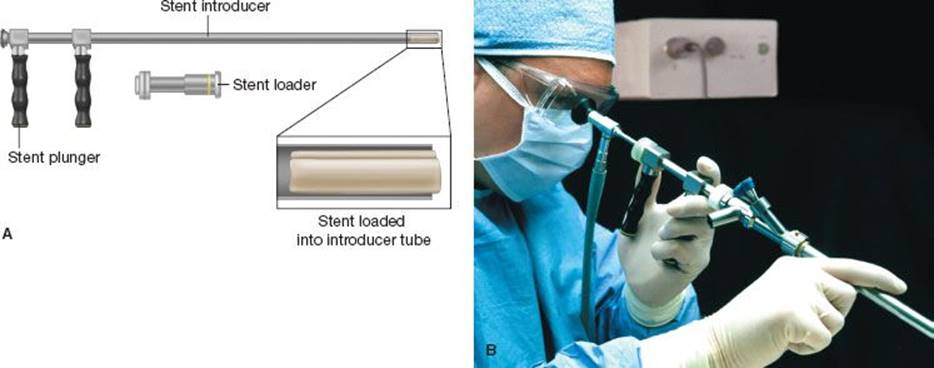
Figure 2.6 A,B: The Bryan–Dumon rigid bronchoscopy system (Bryan Corporation) for deploying silicone stents. (Images courtesy of Bryan Corporation.)

Figure 2.7 Rigid bronchoscopy is performed and the bronchoscope can be used to mechanically debride and core out endoluminal components of the lesion. The scope is inserted beyond the lesion. Once the appropriate silicone stent is determined, it is inserted down the lumen of the bronchoscope with grasping forceps and positioned across the stenosis. The final position of the stent should be positioned as to avoid obstruction of the other airway orifices.
A third technique preferred by the authors does not require specialized equipment and delivers smaller silicone stent through the rigid bronchoscope lumen (Fig. 2.7). A bronchoscope with a smooth inner lumen is used, such as the Storz bronchoscope (Karl Storz Endoscopy-America, El Segundo, CA). Stents up to 14 mm in size can be inserted through the lumen of the bronchoscope, which can be facilitated by lubricating the stent with silicone lubricant. Once the tip of the bronchoscope has been inserted to the stenotic area, grasping forceps are used to push the stent through the bronchoscope and into the appropriate position.
A low-tech technique for larger stent insertion involves first placing the stent through the vocal cords using grasping forceps and then performing rigid bronchoscopy and using the forceps to maneuver the stent into the appropriate position. This technique can be used to place stents larger than 14 mm and carinal Y stents that otherwise will not fit through the lumen of the bronchoscope. Since silicone stents are flexible, this technique allows easy insertion of the stent through the glottis into the proximal airway and then rigid bronchoscopy is used to guide stent placement. This technique has the benefit of minimizing the trauma to vocal cords when placing larger stents (>14 mm) than compared to the first technique described.
Carinal Y stents have their own challenges in stent delivery and seating. One described method of Y-stent placement relies on blind insertion of the stent into the trachea and both bronchial main stem using grasping forceps. The forceps grasp the Y stent at the bifurcation and direct laryngoscopy is performed to insert the forceps and accompanying stent through the vocal cords and into the distal trachea. Once the carina is reached, the stent is released and then rigid bronchoscopy can be performed through the tracheal limb of the Y stent to seat the bronchial stent limbs appropriately.
After the stent has been placed, thorough bronchoscopic evaluation should be performed immediately to evaluate stent position. Fine-tuning of stent position is done with the grasping forceps. It is important that the stent does not occlude adjacent bronchial orifices or incompletely cover the tumor or stenosis, as this will result in inadequate palliation and complications. Often the most difficult site for airway stenting is the right main bronchus due to its short length and the risk of covering the right upper lobe orifice. The silicone stent can be obliquely cut such that the lateral end of the stent is shorter than the medial aspect to allow patency to the right upper lobe orifice. Modifications also have been described when stenting the bronchus intermedius to avoid occlusion of the adjacent segmental bronchial orifices. In some complex cases of central airway obstruction, more than one stent may need to be inserted to achieve acceptable palliation and relief of symptoms (Fig. 2.8).

Figure 2.8 Complex airway lesions may require deployment of multiple stents to achieve acceptable symptom palliation. This drawing depicts the use of two silicone stents to restore right main bronchus and bronchus intermedius patency, while avoiding occlusion of the right upper lobe orifice.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
It is important to obtain a chest x-ray in the postanesthesia care unit to establish a baseline radiograph. There are no studies that support specific poststent medical interventions for decreasing stent complications and usually each institution develops its own practice policy. Many thoracic surgeons will routinely endorse saline or mucomyst nebulization immediately after stent placement in an effort to reduce the encrustations that can accumulate within the stent. In addition, the patient should remain well hydrated to decrease the thickness of tracheobronchial secretions, although this may be challenging without intravenous fluid administration in cachectic patients with advanced stages of cancer. Routine prophylactic antibiotics are not required after stent placement; however, if postobstructive infection is present, this should be treated with an appropriate course of tailored antibiotics. Finally, a role for routine steroid administration is not indicated unless glottic edema occurs, usually secondary to trauma from stent placement.
Some centers will schedule routine bronchoscopic evaluation usually 2 to 3 weeks after stent placement, but this should be planned very selectively, as routine “surveillance” bronchoscopy adds significant inconvenience and cost to both the patient and to society. Clinical symptoms provide the main indications for performing follow-up bronchoscopy, and this is valid regardless of the time frame of symptom presentation, whether immediate poststent placement in the recovery unit or several years after stent placement following a prolonged period of airway stability and symptom palliation. Bronchoscopy is obviously urgently required in situations of acute respiratory decompensation, but it is also performed if there is unexplained worsening of a patient’s respiratory status or when there is lack of anticipated symptom relief following the stenting procedure. As a general rule, any patient with an indwelling stent who develops new respiratory symptoms should undergo bronchoscopy to evaluate for appropriate stent position and patency as well as for disease progression within the airway, both of which may be amenable to therapeutic bronchoscopy.
Stent obstruction from inspissated secretions usually can be managed by flexible or rigid bronchoscopy to irrigate and aspirate the thickened secretions or mechanically debride the hardened secretions. Silicone stents can also be temporarily removed, cleaned, and then reinserted by rigid bronchoscopy. Granulation tissue or tumor progression infiltrating through the interstices of bare metal stents or the bare ends of coated expandable stent can be successfully managed by mechanical debridement with or without additional laser therapy.
TABLE 2.2 Stent-related Complications

In some cases, follow-up bronchoscopy is useful when it is known that the stent is not optimally positioned within the airway, or when the distal airway anatomy could not be adequately evaluated during the initial stenting. In the former situation, follow-up bronchoscopy may allow early stent revision for better seating. In the latter case, follow-up bronchoscopy after airway stent palliation may afford a more thorough evaluation of the distal airways, such as after treatment of postobstructive pneumonia. Finally, in situations where airway stenting may no longer be required for symptom palliation (e.g., after adequate endobronchial tumor response to systemic or radiotherapy), follow-up bronchoscopy to evaluate for possible stent removal is favored.
![]() COMPLICATIONS
COMPLICATIONS
Well-positioned stents are remarkably well tolerated; however, specific complications associated with airway stents are related to the location and type of stent deployed and the individual’s tolerance to have foreign body prosthesis in the airway. Table 2.2 lists the common stent-related complications. The most common complications associated with airway stents are those that cause airway obstruction, including stent displacement, dried secretions within the stent, and granuloma tissue formation or tumor ingrowth causing reobstructive symptoms. As already mentioned, airway stents that are occluded by impacted mucous secretions can usually be cleared by rinsing and aspirating through a flexible bronchoscope. However, sometimes rigid bronchoscopy is required to remove silicone stents to clean it out and then subsequently be replaced. Likewise, stent migration or displacement usually requires rigid bronchoscopy for repositioning and granuloma or tumor growth causing stent obstruction can be mechanically debrided by rigid bronchoscopy or by a variety of ablative energy sources guided by bronchoscopy.
![]() RESULTS
RESULTS
Published studies of stenting for central airway obstructions report excellent short-term outcomes, with greater than 90% of the patients reporting satisfactory results. Like most procedural outcomes, good results are dependent on appropriate patient selection and proper airway preparation. Usually the more distal the extent of stenosis or obstruction the less likely that stenting will be able to achieve successful symptom palliation. Long-term outcomes with airway stenting are mostly dependent upon underlying pathology and the type of stent deployed. In our institutional experience, we previously reported overall palliation of central airway obstruction in 97.8% of patients, without known procedure-related mortality or subsequent mortality related to stent complications. However, 39% of the patients required repeat endoscopy to revise the stent or to manage obstructing secretions. Overall, silicone stents deployed in the airway are successful in 90% of patients with the caveat that 10% to 40% of the patients will require further endoscopy or stent revision.
Covered expandable metal stents are increasingly being used and have been reported to have similar success rates (>90% of patients) as silicone stents, but no direct comparison between expandable stents and silicone stents has been studied. As already mentioned, stent migration is uncommon with covered metal stents but granulation tissue can occur at the uncovered ends of these stents and require repeat endoscopic intervention. Fatigue fracture of metal stents has been described and removal of incorporated stent wires in uncovered metal stents can be difficult, tedious, and occasionally require open surgical extraction.
![]() CONCLUSIONS
CONCLUSIONS
Management of the patient with central airway obstruction requires thorough consideration of the surgical and endoscopic options. For patients with both benign and malignant diseases, definitive surgical resection and reconstruction is preferred. However, when this is not feasible, endoscopic options can provide substantial palliation of symptoms. Tracheobronchial stenting is often employed as part of a combined endoscopic strategy to relieve obstructive central airway symptoms and can be performed safely and effectively with appropriate decision making and execution. The thoracic surgeon must be knowledgeable of and skillful with the different stenting options available; he or she should carefully weigh the potential short- and long-term benefits and complications of airway stenting for each individual case. The decision to perform airway stenting depends upon the patient’s clinical status, the underlying disease, natural history, and the patient’s airway anatomy. The choice of stent(s) to be deployed should also be based upon these considerations.
Recommended References and Readings
Bolliger CT. Airway stents. Seminar in Respiratory and Critical Care Medicine. 1997;18(6):563–570.
Breen DP, Dutau H. On-site customization of silicone stents: Towards optimal palliation of complex airway conditions. Respiration. 2009;77:447–453.
Cavaliere S, Venuta F, Foccoli P, et al. Endoscopic treatment of malignant airway obstructions in 2,008 patients. Chest. 1996; 110(6):1536–1542.
Conacher ID. Anaesthesia and tracheobronchial stenting for central airway obstruction in adults. Br J Anaesth. 2003;90:367–374.
Gaissert HA, Grillo HC, Wright CD, et al. Complication of benign tracheobronchial strictures by self-expanding metal stents. J Thorac Cardiovasc Surg. 2003;126(3):744–747.
Sonett JR, Keenan RJ, Ferson PF, et al. Endobronchial management of benign, malignant, and lung transplantation airway stenoses. Ann Thorac Surg. 1995;59:1417–1422.
Stephens KE Jr, Wood DE. Bronchoscopic management of central airway obstruction. J Thorac Cardiovasc Surg. 2000:119(2):289–296.
Wood DE. Airway stenting. Chest Surg Clin N Am. 2001:11(4):841–860.
Wood DE, Liu YH, Vallières E, et al. Airway stenting for malignant and benign tracheobronchial stenosis. Ann Thorac Surg. 2003;76:167–174.
Wood DE. Tracheal and bronchial stenting. In: Grillo HC, ed. Surgery of the Trachea and Bronchi. Hamilton, ON: BC Decker Inc; 2004:763–790.