Dirk De Ridder1 , Jan Moritz Laturnus2 and Karl-Dietrich Sievert3
(1)
Department of Urology, UZ Leuven campus Gasthuisberg, Leuven, Belgium
(2)
Department of Urology, University Hospital Lübeck, Lübeck, Germany
(3)
Department of Urology and Andrology, SALK University Clinic and Parccelsus Private Medical University (PMU), Salzburg, Austria
Dirk De Ridder (Corresponding author)
Email: dirk.deridder@uzleuven.be
Jan Moritz Laturnus
Email: jan.laturnus@uksh.de
Karl-Dietrich Sievert (Corresponding author)
Email: k.sievert@salk.at
Email: kd_sievert@hotmail.com
Management of Vesicovaginal Fistula
Dirk De Ridder4
(4)
University Hospitals Leuven, Leuven, Belgium
Dirk De Ridder
Email: dirk.deridder@uzleuven.be
What Do the Guidelines Say?
The evidence on the treatment of vesicovaginal fistula is limited. While an initial attempt can be made by placing a transurethral catheter for 2–3 weeks in the hope that the fistula will close spontaneously, surgical repair will usually be necessary. There is no evidence that the timing of the repair makes a difference to the chances of successful closure of the fistula. There is no clear advantage of vaginal, abdominal, transvesical, or transperitoneal approaches. Laparoscopic and robotic approaches have been described but without showing any clear advantage over traditional approaches. Interposition grafts can be used, but there is little evidence to support their use. Urethro-vaginal fistula can be complicated by persisting stress incontinence, urethral strictures, or urethral shortening necessitating long-term follow-up. Surgeons involved in fistula surgery should have appropriate training, skills, and experience to select an appropriate procedure for each patient [1, 2].
Clinical Practice
In developed countries, most of the fistulas are seen as complications of gynecological surgery after radiotherapy. In developing countries, obstetric trauma is the main cause. In the developed world the surgical experience is limited to small series of patients using a variety of surgical approaches, while in developing countries fistula surgeons often have operated on more than 10,000 women mostly by a vaginal route. There is a high need to share this vast experience with the urological community in the developed world. A surgeon should choose the approach he or she is most experienced in. There is no advantage of one approach over another.
Clinical Diagnosis
In most cases, the fistula can be seen or felt during clinical examination. The surgeon needs to assess the location and size, number of fistula, amount of scarring, position of the uterus, eventual urethral involvement, and status of in the outpatient clinic the mid compartment and posterior wall. The exact status can be difficult to assess at the outpatient clinic due to pain and discomfort. In many cases, the clinical examination will have to be repeated once the patient is under anesthesia.
Radiation fistula can progress over time. One should allow 6–12 m before a formal repair. Often derivative surgery will be necessary in these cases.
Vaginal Fistula Repair
Gaining Access and Exposure
The position of the patient on the operation table should allow good access to the vagina. Usually the lithotomy position is used, but in some centers and the knee-elbow position is advocated. Traction sutures or Scott retractors as well as adapted specula are necessary for a good surgical exposure. An episiotomy can be necessary. A transurethral catheter, preferably silicone catheter 16–18 French, will be remain in the patient to drain the bladder for at least 2 weeks. The blue dye tests can be repeated in the OR using this catheter, to ensure the correct identification of the fistula and to assess the number of fistulas.
Incision, Dissection, and Mobilization of the Other Fistulas
Several types of vaginal incisions have been described depending on the position of the fistula and accessibility of the vagina.
Latzko Incision
This incision will circumcise the fistula and close the anterior and posterior wall over the fistula in one or two layers. The vaginal wall is then closed separately. This is a nice incision for high vaginal fistulas. The limited reduction in vaginal length will not cause sexual dysfunction.
J-Shaped Incision
An incision in the shape of a J is made with the fistula lying in the top of the curvature of the J. The arms of the J can be directed anteriorly or posteriorly in such a way that ample mobilization of an anterior and posterior flap of the vaginal tissue is possible. The mobilization should be carried out in such a way that a tension-free closure of the fistula becomes feasible (Fig. 11.1).
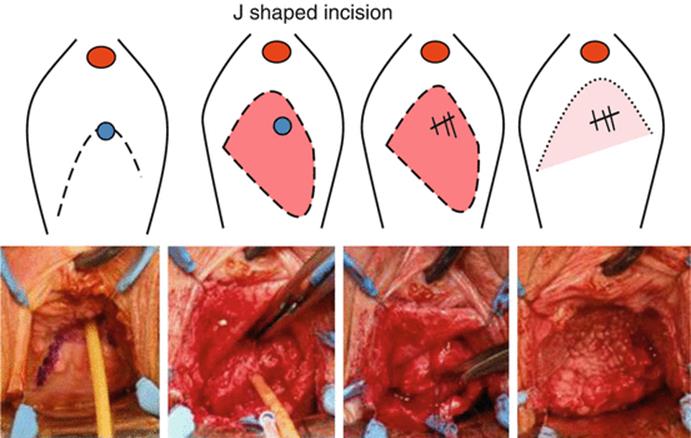
Fig. 11.1
J-shaped incision for small fistula. The tip of the J should encompass the fistula. Flaps can be used to cover the fistula suture line
Inverted Y Incision
This incision is used for larger fistula. The incision starts at the posterior rim of the fistula and can be extended to the lateral walls of the vagina. The circumferential incision is then extended to the midline of the anterior vaginal wall allowing the development of two anterolateral flaps. This incision allows wide mobilization of the fistula tract and even of the urethra. The flaps can be used after the fistula closure to cover the fistula suture line (Fig. 11.2).
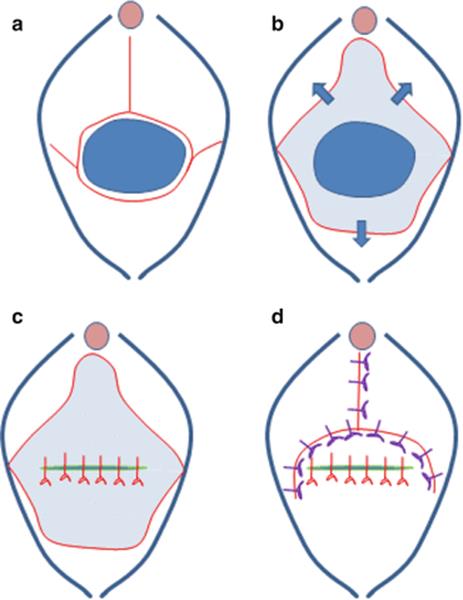
Fig. 11.2
(a) The large fistula is incised at the base. The incisions extend to the lateral vaginal wall and are extended in the midline of the anterior vaginal wall. (b) Dissection of the vaginal wall allows the formation of two anterolateral flaps, and a posterior flap and allows for wide mobilization of the fistula. (c) The fistula is closed in a tension-free way and the repair is watertight. (d) The flaps are used to cover the fistula suture line without creating an overlap between the suture lines
Ensure that the ureters are identified and eventually protected by placing ureteral catheters. If fistulas are located at the bladder neck, proximal urethra, or mid-urethra, make sure to maintain the urethral length as much as possible and think consider adding an autologous sling to prevent postoperative stress incontinence.
Closing of the Fistula
For the obstetric fistula, several consensus meetings have been held on the optimal closure of the fistula. Absorbable sutures 2/0 are recommended. Strong bites in the detrusor and serosal layers are necessary to close the fistula, but there is no need to close the urothelium separately. A single layer of separate sutures of 4–5 mm apart is sufficient to close the fistula. After closure, the bladder should be filled with blue dye to check for watertight closure. Eventually, supporting sutures can be used to stabilize the bladder neck. A Martius flap is optional in most cases [3].
Achieving and Maintaining Continence
Fistulas involving the bladder neck area or mid-urethra have a higher risk of persisting incontinence, despite a successful closure of the fistula. The urethral support mechanisms and the external urethral sphincter might be damaged as a consequence of the fistula, fibrotic process, or fistula repair. The larger the fistula, the more vaginal scarring and the smaller the bladder capacity, the higher the risk for postoperative incontinence. The surgical principles in these cases are the same as for other fistulas, but some additional measures can be taken. The urethral length should be maintained as much as possible. If the urethral length is less than 2.4 cm or if the urethral defect is more than 4 mm, an incontinence procedure should be added. If performed, postoperative incontinence can be reduced by approximately 50 %. Either a sling procedure can be added or a bladder neck suspension can be performed. For the sling procedure, only autologous material should be used. A sling can be created from fibromuscular tissue in the lateral vaginal wall or can be constructed out from the rectus or fascia lata. In the bladder neck suspension, non-absorbable sutures are used to suspend the bladder neck area in a fixed position [4].
Postoperative Care
Bladder drainage should be continued for 10–14 days, in most cases. In smaller fistulas that were easy to repair, shorter catheterization periods might be possible. When draining the bladder, silicone catheters are preferred since these have a larger internal diameter for the same external diameter and are less likely to be blocked by blot clots. A high fluid intake should be maintained to prevent clot formation. There is no need for antibiotics during the catheterization periods. Nursing staff should be trained and supervised adequately to ensure that catheters are not blocked, which could lead to a rupture of the fistula repair.
Abdominal Approach
Abdominal fistula repair can be done open, laparoscopically, or robotically. Extravesical and transvesical techniques have been described. The principles remain the same: identify the fistula, separate the bladder wall from the vaginal wall, and mobilize the fistula well enough to allow tension-free closure. Omentum can be used as interposition material.
Points of Interest
· Most vesicovaginal fistulas can be managed by a vaginal approach, but abdominal, laparoscopic, or robotic repairs are also feasible.
· Wide mobilization of the fistula to allow tension-free and watertight closure is of the utmost importance.
· The timing of fistula surgery can be individualized, except for radiation fistula, where a longer waiting time (6–12 m) is advocated.
Bladder Augmentation
Karl-Dietrich Sievert5 and Jan Moritz Laturnus5
(5)
University of Lübeck, Lübeck, Germany
Karl-Dietrich Sievert (Corresponding author)
Email: kd_sievert@hotmail.com
Jan Moritz Laturnus (Corresponding author)
Email: kd_sievert@hotmail.com
General Introduction
Many patients with small-capacity, high-pressure, or unstable or low compliant bladders will be managed with conservative measures. A small but significant minority of these patients will require surgical intervention, the therapeutic goals of which are to provide urinary storage while preserving renal function, continence, resistance to infection, and convenient voluntary and complete emptying.
Augmentation cystoplasty increases bladder capacity and decreases detrusor overactivity by enlarging the bladder with the addition of a bowel segment and possibly by disrupting the detrusor. Many different surgeons have carried out augmentation for many reasons, using many different types of tissue. The stomach, ileum, cecum, and ascending and sigmoid colon have all been used as tubular or detubularized, simple or complex segments.
The classic, augmentation enterocystoplasty has been performed in patients with neurogenic voiding dysfunction, but it has been shown to be effective for patients with neurogenic and non-neurogenic detrusor overactivity or patients with a reduced bladder compliance/capacity.
What Do the Guidelines Say?
EAU
“Replacing or expanding the bladder by intestine or other passive expandable coverage will reduce bladder compliance and at least reduce the pressure effect of detrusor overactivity. The procedure should be used with caution in patients with neuro-urological symptoms, but may become necessary if all less-invasive treatment methods have failed.”
“In order to treat refractory detrusor overactivity, bladder augmentation is recommended. Detrusor myectomy is an acceptable alternative (LE: 3, GR: A).”
Fourth International Consultation on Incontinence
“Children: For detrusor overactivity/poor compliance, botulinum toxin and bladder augmentation may be performed.”
“Men: For idiopathic detrusor overactivity (with intractable overactive bladder symptoms) the recommended therapies are bladder augmentation (Grade C) and neuromodulation (Grade B). Botulinum toxin continues to show promise in the treatment of symptomatic detrusor overactivity unresponsive to other therapies.”
“Women: Urgency incontinence (overactive bladder) secondary to idiopathic detrusor overactivity may be treated by neuromodulation (Grade A) or bladder augmentation (Grade C). Botulinum toxin can be used in the treatment of symptomatic detrusor overactivity unresponsive to other therapies (Grade C).”
Clinical Practice
In a large majority of patients with refractory detrusor overactivity or reduced bladder compliance/capacity, augmentation enterocystoplasty is effective, provided that these principles are followed:
1.
2.
3.
4.
Many patients require intermittent self-catheterization after augmentation and need to be prepared to do this prior to consenting to the operation.
Background
Outcome
Augmentation cystoplasty is reported to have a 0–2.7 % mortality. The deaths that were reported were caused by pulmonary embolus, myocardial infarction, and adult respiratory distress syndrome after postoperative pancreatitis. Surgical early complications include a 3–5.7 % rate of adhesive small bowel obstruction requiring operative intervention, a 5–6.4 % incidence of significant wound infection (including dehiscence), and a 0–3 % reoperation rate for bleeding.
Failure of augmentation enterocystoplasty to resolve the underlying lower urinary tract problem was reported in 5–42 % of patients. Failure rates are higher with idiopathic detrusor instability, where long-term symptomatic success was reported in as few as 53–58 % of patients, compared with the 92 % success reported in neuropathic patients. In view of this, bowel augmentation may be the treatment of choice in patients with neuropathic bladders, while autoaugmentation may be the surgical intervention considered first in cases of detrusor instability.
Complications
Typical complications are recurrent infections, stone formation, perforation or possible malignant changes, intestine metabolic abnormality, mucus production, and impaired bowel function.
Recommendations for Follow-Up
Depending on the underlying diagnosis, the follow-up interval may vary between a few months but should not exceed 2 years. The follow-up protocol should consist of a physical examination, regularly performed urine analysis, and a checkup of the upper urinary tract. Also blood gases and folic acid should be checked frequently.
Diagnostics
All patients need to have sufficient intellect to understand the proposed surgery, its pros and cons, and the requirement of lifelong surveillance. They need to be motivated to learn and comply with intermittent catheterization, cope with the physical and emotional stress of what is a significant operation, and (in the case of patients with neuropathy), be prepared for the requirement to perform subsequent procedures to achieve acceptable continence.
First the patient history, with concentration on past and present symptoms and conditions for urinary, bowel, sexual, and neurological functions and a systematic physical examination, is mandatory before further diagnostic tests should be initiated. A history of psychiatric illness should be investigated in all patients, especially those with no neuropathy, as those patients tend should be obtained to have poor outcomes from any surgical intervention. For those with a history of any bladder pathology.
Hypersensitivity in non-neuropathic patients indicates a probably unsuccessful augmentation, especially if intermittent catheterization is required afterward.
Additionally a 24-h bladder diary (recording should be done for three consecutive days) is strongly recommended to evaluate the underlying bladder dysfunction thoroughly. An assessment of the patient’s quality of life (QoL) can be helpful because of the effect on QoL of any therapy used. For example, there is an easy and validated generic tool, known as Visual Analogue Scale (VAS), for symptom bother.
A urodynamic investigation should also be done. This is the only method that can objectively assess the (dys-)function of the lower urinary tract and therefore gives an objective indication to justify an operation. A cystoscopy to measure the bladder capacity (functional and anatomic) and to exclude any bladder malignancies or a subvesical obstruction is also advisable. After a bladder augmentation the upper urinary tract is at risk, so a complete evaluation of both upper and lower urinary tracts is required before treatment. IVU or renal ultrasonography of the upper urinary tract and serum creatinine and radioisotope renal studies provide information about total and differential renal function and the presence of upper tract obstruction.
Technique
Bladder Augmentation
The objective of augmentation enterocystoplasty is the construction of a low-pressure diverticulum, which will absorb the involuntary contractions and high pressures generated by the abnormal detrusor and increase both the capacity and compliance of the bladder.
Sigmoidocystoplasty and ileocystoplasty have become standard techniques, but there is renewed interest in alternative techniques due to the relatively high morbidity of intestinal cystoplasty.
The ideal ileal segment is 25 cm long and located 25 ± 40 cm from the ileocecal valve, as this produces the least metabolic disturbance. The ileal patch is always detubularized.
A sigmoid segment is generally detubularized as a straight patch or a cup patch and is the usual alternative to the ileum for a straightforward augmentation. The sigmoid has several advantages; its thick muscular wall, large lumen, and abundant mesentery guarantee adequate bladder capacity and maneuverability. The disadvantages of sigmoid cystoplasty are the higher risks of UTI due to the colonic commensal bacteria colonization, more mucus production, and a theoretically higher long-term risk of malignancy.
There are many different techniques described in the literature to avoid inclusion of intestinal mucosa in the urinary tract. Gastrocystoplasty, vesicomyotomy, seromuscular augmentation, and various alloplastic or biodegradable materials or in vitro cultures of autologous urothelium are only some of the possible options. Although encouraging animal and human results have been reported, each technique is associated with its own limitations and disadvantages. While intestinal cystoplasty remains the standard, several alternative techniques show promise, but only gastrocystoplasty, ureterocystoplasty, and seromuscular augmentation should be considered clinically useful.
Bladder Reconstruction
Only in case of bladder diverticula or in certain instances when the native bladder plate is too diseased and cannot undergo augmentation, there is need for bladder reconstruction with resection of the diverticulum or using a neobladder as a substitute.
Point of Interest
· In small children bladder augmentation is commonly performed with a colon/sigmoid segment. In children, colonic anastomoses are less prone to problems.
· Many different tissues and techniques to perform bladder augmentation. Standard: ileum cystoplasty.
Case Study
A boy of 16 years with ataxia telangiectasia and B non-Hodgkin’s lymphoma developed at the age of 13 a severe hemorrhagic cystitis. The cause of the hemorrhagic cystitis was the medication with cyclophosphamide because of an allogenic stem cell transplantation. In addition he had to undergo a ureterocutaneostomy on both sides.
After 3 years the patient a bladder capacity of 50 ml was recorded; the ultrasound study showed both kidneys without any pathologic findings. Normal serum creatinine. No subvesical obstruction was detected in cystoscopy.
A bladder augmentation was planned with reimplantation of both ureters back into the augmented bladder to achieve better quality of life.
Augmentation was performed using an ileal W-shaped augment, of which both proximal lumens of the ileum were kept intact, whereas the rest was detubularized and afterward augmented to the remaining bladder wall. The proximal captured ureters of the former ureterocutaneostomy were implanted in an end-to-side, reflexive technique. (Please see below the figure X-ray documents, and the bilateral remaining ureters, which were not removed in the initial surgery when the ureterocutaneostomy was performed.)
At week eight the boy had a bladder capacity of 500 ml and was able to empty the bladder by voluntary voiding (no need for self-catheterization). The ultrasound study shows both kidneys and a residual free augmented bladder and normal serum creatinine. Normal serum creatinine.
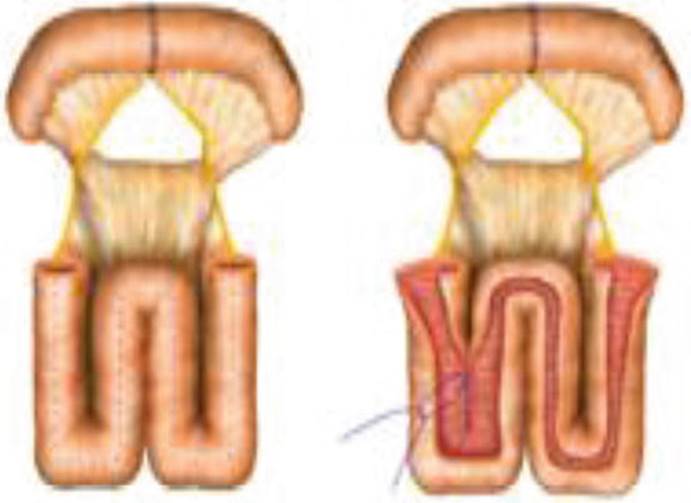
Figure 1a: Terminal Ilium positioned in a w shape which will be opened longitudinal to become the augment of the bladder (Figure 1b)
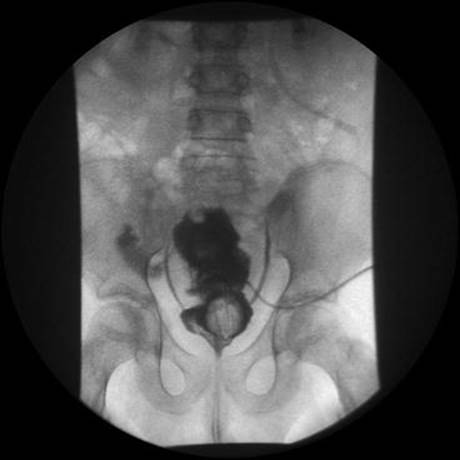
Figure 2: Xrays of the abdomen after augmentation
Different to this picture both ends (lumen) of 4–5 cm were kept intact to have the ureters of each side implanted in a side-to-end fashion.
The rest of the W was detubelarized and shaped to become the new dome of the remaining bladder.
References
1.
De Ridder D, Abrams P, De Vries C, Elneil S, Emasu A, Esegbona G, et al. Fistula. In: Abrams P, Cardozo L, Khoury S, Wein A, editors. Incontinence. 5th ed. Paris: EAU-ICUD; 2013. p. 1527–79.
2.
Abrams P, Andersson KE, Birder L, Brubaker L, Cardozo L, Chapple C, et al. Fourth International Consultation on Incontinence Recommendations of the International Scientific Committee: evaluation and treatment of urinary incontinence, pelvic organ prolapse, and fecal incontinence. Neurourol Urodyn [Internet]. 22 Dec 2009 ed. 2010;29(1):213–40. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20025020.
3.
Browning A. Obstetric fistula: clinical considerations in the creation of a new urethra and the management of a subsequent pregnancy. Int J Gynecol Obstet. 2007;99:S94–7.CrossRef
4.
Waaldijk K. Prevention of post IIAa fistula repair incontinence: a prospective study in 845 consecutive patients. Nairobi: International Society Obstetrical Fistula Surgeons; 2009.
Further Reading
Abrams P, Andersson KE, van AC V, Heesakers JP, et al. Fourth International Consultation on Incontinence. Recommendations of the International Scientific Committee: evaluation and treatment of urinary incontinence, pelvic organ prolapse, and fecal incontinence. Neurourol Urodyn. 2010;29:213–40.CrossRefPubMed
EAU Guidelines 2015. http://www.uroweb.org.
Stein R, Schröder A, Thüroff JW. Bladder augmentation and urinary diversion in patients with neurogenic bladder: non-surgical considerations. J Pediatr Urol. 2012;8(2):145–52.CrossRefPubMed
Vainrib M, Reyblat P, Ginsberg DA. Differences in urodynamic study variables in adult patients with neurogenic bladder and myelomeningocele before and after augmentation enterocystoplasty. Neurourol Urodyn. 2013;32(3):250–3.CrossRefPubMed
Veenboer PW, Nadorp S, de Jong TP, et al. Enterocystoplasty vs detrusorectomy: outcome in the adult with spina bifida. J Urol. 2013;189(3):1066–70.CrossRefPubMed